Abstract
Background
The human in-vivo functional somatotopy of the three branches of the trigeminal (V1, V2, V3) and greater occipital nerve in brainstem and also in thalamus and insula is still not well understood.
Methods
After preregistration (clinicaltrials.gov: NCT03999060), we mapped the functional representations of this trigemino-cervical complex non-invasively in 87 humans using high-resolution protocols for functional magnetic resonance imaging during painful electrical stimulation in two separate experiments. The imaging protocol and analysis was optimized for the lower brainstem and upper spinal cord, to identify activation of the spinal trigeminal nuclei. The stimulation protocol involved four electrodes which were positioned on the left side according to the three branches of the trigeminal nerve and the greater occipital nerve. The stimulation site was randomized and each site was repeated 10 times per session. The participants partook in three sessions resulting in 30 trials per stimulation site.
Results
We show a large overlap of peripheral dermatomes on brainstem representations and a somatotopic arrangement of the three branches of the trigeminal nerve along the perioral-periauricular axis and for the greater occipital nerve in brainstem below pons, as well as in thalamus, insula and cerebellum. The co-localization of greater occipital nerve with V1 along the lower part of brainstem is of particular interest since some headache patients profit from an anesthetic block of the greater occipital nerve.
Conclusion
Our data provide anatomical evidence for a functional inter-inhibitory network between the trigeminal branches and greater occipital nerve in healthy humans as postulated in animal work. We further show that functional trigeminal representations intermingle perioral and periauricular facial dermatomes with individual branches of the trigeminal nerve in an onion shaped manner and overlap in a typical within-body-part somatotopic arrangement.
Trial registration: clinicaltrials.gov: NCT03999060
Keywords
Introduction
The trigeminal nerve with its three branches (V1, V2, V3, see Figure 1A) is the anatomical source for primary headache and facial pain syndromes. Although the anatomical location of the spinal trigeminal nucleus (STN) is principally known, the functional representations of all three branches on brainstem level (1–6), at least below the pons (7), is unknown in humans. Particularly, the exact anatomical correlation between the nuclei of the fifth cranial nerve and the greater occipital nerve (GON), the so-called trigemino-cervical complex (TCC), has not been shown, although the evidence of its existence is strong (8). The latter is clinically important: primary headache syndromes such as migraine and cluster headache involve mainly the first trigeminal branch (9), while trigeminal neuralgia mainly affects V2 and V3 (10). Pharmacologically blocking the GON reduces the number of headache days in certain primary headaches like cluster headache (11–16), trigeminal neuralgia (17) and probably also migraine (18–25). This raises the question of the central processing of trigemino-nociceptive pain and indicates that the central organization of the heads’ somatotopy is more complex than assumed.
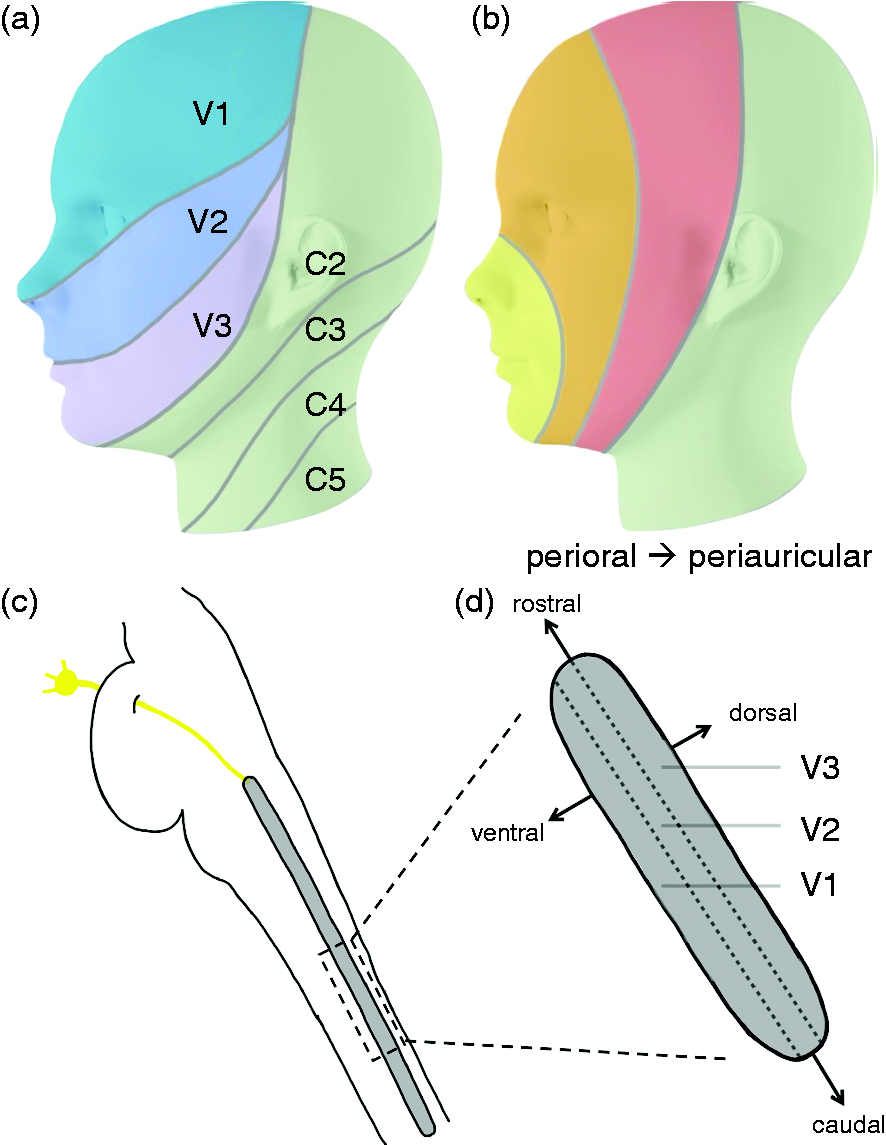
Innervation of the face. Innervation by the three peripheral trigeminal branches (a) and the central onion model somatotopy (b) with three layers representing facial dermatomes from perioral to periauricular. The spinal trigeminal nucleus (c) is the first relay of the trigeminal nerve in the brainstem. While the three trigeminal branches are represented as columns within the spinal trigeminal nucleus, the facial dermatomes are probably organized such that perioral is represented more rostral while periauricular dermatomes are represented more caudal (d).
Animal (26–28) and recently also human data (29) suggest a convergence of V1 and GON input (30) in the TCC at the level of C2, where the GON enters the brainstem, which is thought to explain the functional interrelation of the two systems (Figure 1a). Based on animal data (1–6) it has further been suggested that the representation of the trigeminal branches in the STN follow a somatotopic arrangement which follows an onion-shaped segmentation (7,31) reaching from perioral (front, nose) to periauricular areas of the face (ear, back of the head) (Figure 1b). According to this theory, the perioral dermatomes in the STN are represented more rostrally, whereas the periauricular dermatomes are placed more caudally. This somatotopy of rostral versus caudal arrangement in the brainstem is thought to be intermingled with the three trigeminal branches (7,31). It is interesting that although a somatotopic arrangement for example of the limbs in the somatosensory and motoric cortices has clearly been shown in the homunculus, it has also been shown that the central representation within one limb exhibits wide overlaps of the representations of its individual dermatomes (e.g. for the individual digits) in the motor cortex but also partly in the somatosensory areas (32–35). Within-limb representations also seem to overlap in the spinal cord at least in some nuclei while others show a more distinct somatotopy (36–41).
To investigate the functional representations on brainstem level of the individual dermatomes of the head we recorded advanced high-resolution functional magnetic resonance imaging (fMRI) (42,43) simultaneously to painful, electric stimulation of rostral V1, V2, V3, and the GON. Since the template for the Montreal Neurological Institute (MNI) space only reaches as low as z = −70 (∼1 cm below the cerebellum), previous studies (7,44) might have missed important nuclei involved in trigemino-cervical nociception. Hence, we specifically acquire data with a lower anatomical boundary of C2/3 (Figure 1a). Of specific interest is the co-localization of the GON with the first branch of the trigeminal nerve (V1) and the connectivity to the hypothalamus (45) as a possible modulator/generator of migraine attacks (46,47). In a second step, we aimed to disentangle the representations of the trigeminal branches according to their rostral versus caudal positions along the perioral-periauricular axis of the facial dermatomes. We aimed to provide evidence of an onion-shaped representation intermingled with the brainstems’ representation of the individual branches of the trigeminal nerve in the lower brainstem.
Materials and methods
Study population and experimental design
Results from a pilot study with 25 healthy participants using the same paradigm on repetitive, randomized, peripheral, painful electrical stimulation of the rostral part of the three trigeminal branches (V1R, V2R, V3R) and the GON were used to generate a hypothesis, i.e. primary and secondary outcomes, and estimate effect. The actual study, built on these preliminary results, was consequently preregistered (clinicaltrials.gov: NCT03999060). We then investigated a new and independent group of 38 participants from which two had to be excluded due to technical problems. This left 36 participants for the final analysis (age 29.17 ± 10.27 years [mean ± SD], 22 females). In a second experiment with complementary locations for the painful electrical trigeminal stimulation in perioral as well as periauricular dermatomes of the face, we included 26 independent participants (age 24.42 ± 4.11 years [mean ± SD], 14 females). The participants were screened according to the following exclusion criteria: pain and/or headache disorders (people reporting infrequent episodic tension-type headache (TTH) were accepted, as long as they had less than 12 episodes a year), first-generation family member with primary headache disorder, intake of weak analgesics more than 14 days a month, skin lesions or other dermatological illnesses in the applied areas (neck and face region), men with full beard, neurological, psychiatric or other chronic disorders, whiplash or other damages to the cervical spine, pregnancy or breast feeding, misuse of alcohol or drugs, inability to speak or understand German. The study was approved by the local ethics committee (Ethikkommission der Ärztekammer Hamburg, PV5490) and all participants gave written, informed consent.
Experimental design: Electrode positioning and equipment
Electrical stimulation was applied using a MR-compatible Digitimer DS7A Current Stimulator (Digitimer Ltd., Welwyn Garden City, UK), which was coupled to four WASP electrodes (Specialty Developments, Bexley, UK) via a D188 Remote Electrode Selector (Digitimer Ltd., Welwyn Garden City, UK) and custom-built MR-compatible cables. The cables were built using a MR-safety tested design (48) to prevent tissue damage due to currents induced by electro-magnetic wave coupling. The four electrodes were positioned on the left side according to the three branches of the trigeminal nerve and the GON (Figure 1a). The GON was located by palpation according to validated procedures (49,50) and the electrode was positioned immediately above. Rostral V1 (V1R) was stimulated by means of an electrode placed on a vertical line between the medial and lateral quarter of the face corresponding to the middle of the eyebrow and approximately 1 cm above. The rostral V2 (V2R) was stimulated 1 cm lateral of the same vertical line on the level of a horizontal line through the inferior part of left ala of the nose. Rostral V3 (V3R) was stimulated along the same vertical line approximately 1 cm caudal from the corner of the mouth. Figure 2a demonstrates the location of the electrodes in the first experiment. In the second experiment, we changed the locations of two of the four stimulating electrodes to additionally cover the caudal parts of the first and third trigeminal branch (V1C and V3C, see Figure 2b). The stimulating electrode in V1C was attached 2 cm lateral to the left as well as 2 cm rostral from the point at the half of the distance between nasion and inion. Stimulation at V3C was located 1 cm rostral to the tragus.

Experimental Design. Sketch of the localization of the four electrodes (a) in the first Experiment and (b) their expected representation in the STN. (c) Rearranged stimulation electrode positions of the second experiment and (d) their expected representations in the STN and (e) The timeline of the experiments. EDT, electrical detection threshold; VAS, Visual Analogue Scale; V1, ophthalmic division of the trigeminal nerve; V2, maxillar division of the trigeminal nerve, V3, mandibular division of the trigeminal nerve; GON, greater occipital nerve; V1R/V2R/V3R/GON, (rostral) stimulation sites in first experiment, V1c/V3c, (caudal) stimulation sites in second experiment.
Experimental design: Stimulation paradigm
After fixing the electrodes, the subjects were moved into the scanner and the electrical detection thresholds (EDTs) at all electrode sites were determined by means of the QUEST-procedure (51). The final current was set to 10 times the EDT of the V3R electrode for both experiments but was not allowed to exceed 5 mA nor a pain rating above 50 (on a numeric rating scale from 0 to 100) for a single pulse. The actual stimulation consisted of a small train of three pulses separated by 100 ms each with 400 V and 2 ms duration. Each stimulus was followed by a break of 3 s (jittered between 2 and 4 seconds); then a pain intensity rating on a visual analogue scale (VAS) with levels between 0 and 100 using a button box with the right hand and then another break before the next trial. The inter-trial interval was set to 15 s (jittered between 12 and 18 s). The stimulation site was randomized and each site was repeated 10 times per session. The participants partook in three sessions resulting in 30 trials per stimulation site during approximately 30 minutes of fMRI scanning. The experimental design is shown in Figure 1c.
MR data acquisition and processing
All MR data was recorded with a Siemens 3T PRISMA scanner (Siemens, Erlangen) using a 64-channel head coil. During the actual experiment we recorded three sessions with 230 images each using an echo-planer imaging (EPI) protocol (repetition time 2.93 s, echo time 33 ms, 1.3 × 1.3 × 2.0 mm3 spatial resolution, GRAPPA acceleration, flip angle 80°, 72 slices with a multiband factor of 2, FOV 215 mm, no gap, flow rephasing) with a field of view covering the brainstem as low as C2/3, cerebellum, midbrain and the insula cortices. In each session the first five images were removed to avoid scanner saturation effect. Afterwards we recorded fieldmaps (repetition time 0.792 s, echo times 5.51 and 7.97 ms, 3 × 3 × 2 mm3 spatial resolution, flip angle 20°, 72 slices, FOV 222 mm, no gap) covering the same volume as the EPIs to attenuate inhomogeneities of the magnetic field. Pulse and breathing were recorded simultaneously to attenuate extra-cerebral (i.e. cardio-vascular) artifacts. Afterwards we acquired high resolution (1 mm3) anatomical images (MPRAGE, repetition time 2.3 s, echo time 2.98 ms, flip angle 9°, 240 slices, FOV 256 mm).
All fMRI data were first filtered using the spatially adaptive non-local means filter (52) implemented in the CAT12 toolbox. The fMRI data was then corrected for movements and for distortions of the homogeneity of the magnetic field (fieldmaps) using the realign and unwarp algorithm as implemented in SPM 12. Additionally, slice time correction was performed using the onsets of the single slices as suited for our multiband protocol. We then calculated a subject-wise general linear model (GLM) including condition-wise onsets of each stimulus as stick functions, which were then convolved with a hemodynamic response function (HRF). The button box responses as well as the onset and duration of the VAS were modelled as regressors of no interest. Additional regressors of no interest were included to correct for (uncorrelated) movement, cardiovascular influence, using the algorithms proposed by Deckers and colleagues (53), and changes in the spinal fluid extracted from the fourth ventricle. The co-registered structural images were segmented with the unified segmentation approach algorithm (54) implemented in SPM12 but using the templates provided by Blaiotta et al. (55), which are optimized for the brainstem and spinal cord, to gain deformation fields used to warp the contrast images of the subject-wise GLM into MNI space. Each step was controlled by visual inspection. We further calculated a group template and gray and white matter masks from the warped structural images.
Primary and secondary outcomes
Our primary outcome was that using functional neuroimaging and a standardized trigemino-nociceptive input, we would be able to observe a somatotopical arrangement of the trigeminal areas in the brainstem below the pons at a statistical threshold of T-value 3. To test our preregistered primary and secondary outcomes we calculated repeated measure ANOVAs as implemented in SPM12 for the four stimulated regions for both cohorts of the first experiment separately. As described in the preregistration, we calculated small volume corrections in the second cohort at MNI coordinates and T-value thresholds derived from the first cohort. For the brainstem below the pons, these are a T-value >3 and a sphere with a search radius of 10 mm around the MNI coordinates [−1,−48,−75] for V1, around [−4,−47,−69] for V2, around [−1,−46,−66] for V3, and around [−1,−45,−64] for GON. The same approach was used at a statistical threshold of T-value >4 to test a functional, somatotopical arrangement in the thalamus (V1 [11,−5,6]; V2 [13,−11,5], V3 [12,−10,5], GON [15,−12,−7]), and the insula (V1 [38,−15,8], V2 [37,−8,−5], V3 [39,2,−11], GON [38,−12,−3]). As secondary outcome we hypothesized a somatotopy in the cerebellum which was tested with a Family Wise Error (FWE) corrected threshold of p < 0.05. For visualization purposes we created masks for the individual regions of interest using the WFU PickAtlas (56) (Online Supplemental Figure 1).
Functional representations of the trigeminal branches and the GON within the lower brainstem
To investigate the functional somatotopy of the rostral part of the three branches of the trigeminal nerve and the GON in the brainstem we calculated a repeated measure ANOVA with all data from both studies (i.e. n = 61 subjects). Since the co-localization of V1 and GON was of special interest, we further conducted a conjunction analysis between them. We performed differential contrasts between the three trigeminal branches (i.e. V1 vs V2, V1 vs V3, V2 vs V3). The statistical threshold for the results was set to T-values higher than 3. We additionally assigned each voxel surviving aforementioned threshold to the stimulation site (dermatome) which, within this voxel, had the highest T-value in order to present not only the overlaps but also the somatotopic arrangements of the individual trigeminal branches and the GON within the brainstem.
Functional connectivity for the co-localization of V1 and GON
Because the co-localization of V1 and GON carries a particular significance in primary headache syndromes, and since the hypothalamus plays a specific role in generating migraine attacks (46,47), we investigated the functional connectivity between the individual clusters of their conjunction and the hypothalamus with a psychophysiological interaction (PPI) (57). Since we had four different stimulations, and the current implementation of PPI is only capable of analyzing a contrast between two conditions, we used a generalized approach for this analysis (58). We report results at a statistical threshold of p < 0.05 (FWE-corrected) using a small volume correction within a sphere of 6 mm radius around previously reported coordinates (45–47) ([0, 2, −6] mm xyz in MNI space) of the hypothalamus.
Somatotopy of perioral versus periauricular facial dermatomes of the trigeminal branches
To reveal the functional somatotopy of the trigeminal branches according to their perioral or periauricular position we calculated a repeated measure ANOVA for the 26 subjects included in the second experiment. We further analyzed differential contrast between the stimulated sites. The statistical threshold for the main effects was set to FWE-corrected p < 0.05 and for the differential contrast to T-values higher than three both exceeding a minimal cluster extent of five voxels. Results are presented for activations in the lower brainstem.
Data availability statement
European and German data protection rules as well as the hospitals data protection officer prevent us from uploading raw data to a public repository. Nevertheless, unconditional access to anonymized data and access to the code are available to qualified investigators on request to the corresponding author.
Results
Behavioral data
The ratings of electrical stimuli at the sites of all four electrode positions were significantly painful in the first experiment (p < 0.0125 Bonferroni corrected; average VAS: 56.4 +/− 20.3 at V1R, 56.4 +/− 19.7 at V2R, 48.8 +/− 19.9 at V3R, 55.6 +/− 22.8 at GON) as well as the second experiment (p < 0.0125 Bonferroni corrected; average VAS: 55.7 +/− 24.5 at V1C, 56.6 +/− 14.8 at V1R, 49.2 +/− 12.2 at V3R, 34.6 +/− 20.7 at V3C).
Primary and secondary outcomes
All preregistered primary and secondary outcomes regarding the functional representations of the three trigeminal branches and the GON in lower and upper brainstem, insula, thalamus and cerebellum were significant (Table 1). The representations of the trigeminal branches in thalamus, insula and cerebellum are presented in Figure 3 using data from all participants (n = 61) of the first experiment. The representations of the individual trigeminal branches overlap greatly on brainstem level and less in thalamic and insular cortex, which is typical for within-body-part representations as opposed to a more distinct somatotopy between body parts.
Functional representations of the trigeminal branches (V1-3) and the GON in brainstem, diencephalon, cerebellum and cortex. These results were defined as primary and secondary outcome (n = 36, df = 105, minimal cluster extent 10 voxel) in the preregistration (clinicaltrials.gov: NCT03999060). V, 5th cranial nerve/trigeminal nerve; FEW, family wise error rate; GON, Greater occipital nerve.

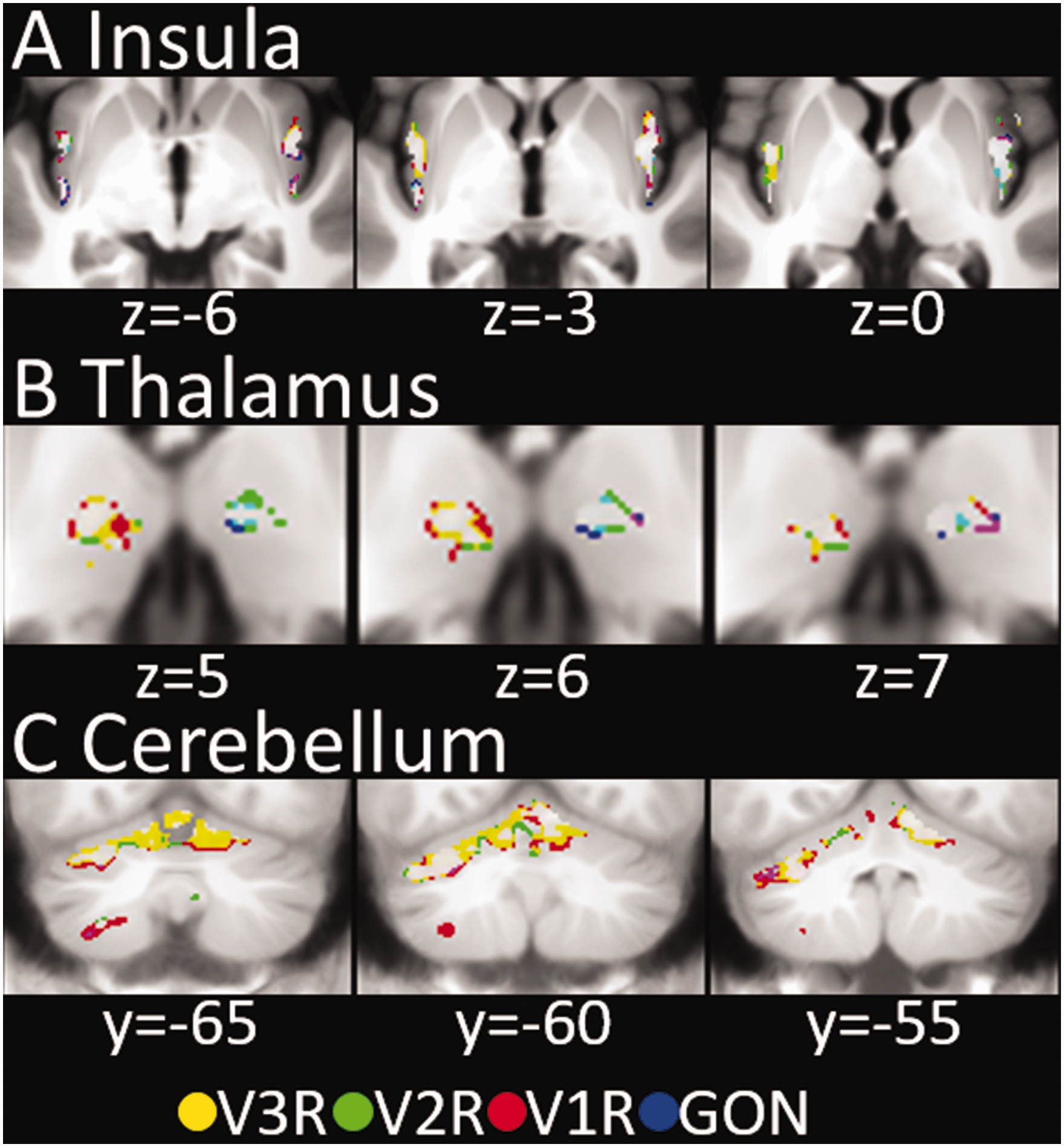
Functional Representations of the trigeminal branches (V1, V2, V3) and the GON in insula, thalamus and cerebellum. Overlapping activity is coded in the resulting mixed colors (e.g. purple for the conjunction of V1 and GON). Overlap of all four locations is presented in white. GON, greater occipital nerve; V1R/V2R/V3R/GON, stimulation sites in first experiment.
Functional representations in the lower brainstem
The functional somatotopic arrangement in the brainstem below the pons revealed several clusters for each of the three trigeminal branches as well as for the GON. The trigeminal branches are represented at up to six locations along the spinal trigeminal nucleus, namely: at the level of the pons (around MNI z = −50), below the pons (around MNI z = −56), at the lower end of the cerebellum (around MNI z = −66), at C1 (around MNI z = −78), at C2 (around MNI z = −84), and between C2 and C3 (around MNI z = −94). The GON is represented at five different levels within the lower brainstem. A co-localization and thereby possible locations for interaction between V1 and GON is present at five levels below the brainstem. We summarized these results in Table 2, Online Supplemental Table 1 and Figure 4. The differential contrast between the individual branches of the trigeminus as well as for the GON did not show any significant results at the chosen statistical threshold (Figure 2b). This is probably due to the onion-shaped central representation of the trigeminal nerve at the brainstem level, which is why we conducted the second experiment testing perioral versus periauricular facial dermatomes (see below).
Somatotopy in the brainstem below the pons (n = 61, df = 180, T > 3, minimal cluster extent 10 voxel), sorted along the z-axis.

also significant at an FWE-corrected threshold of p < 0.05.
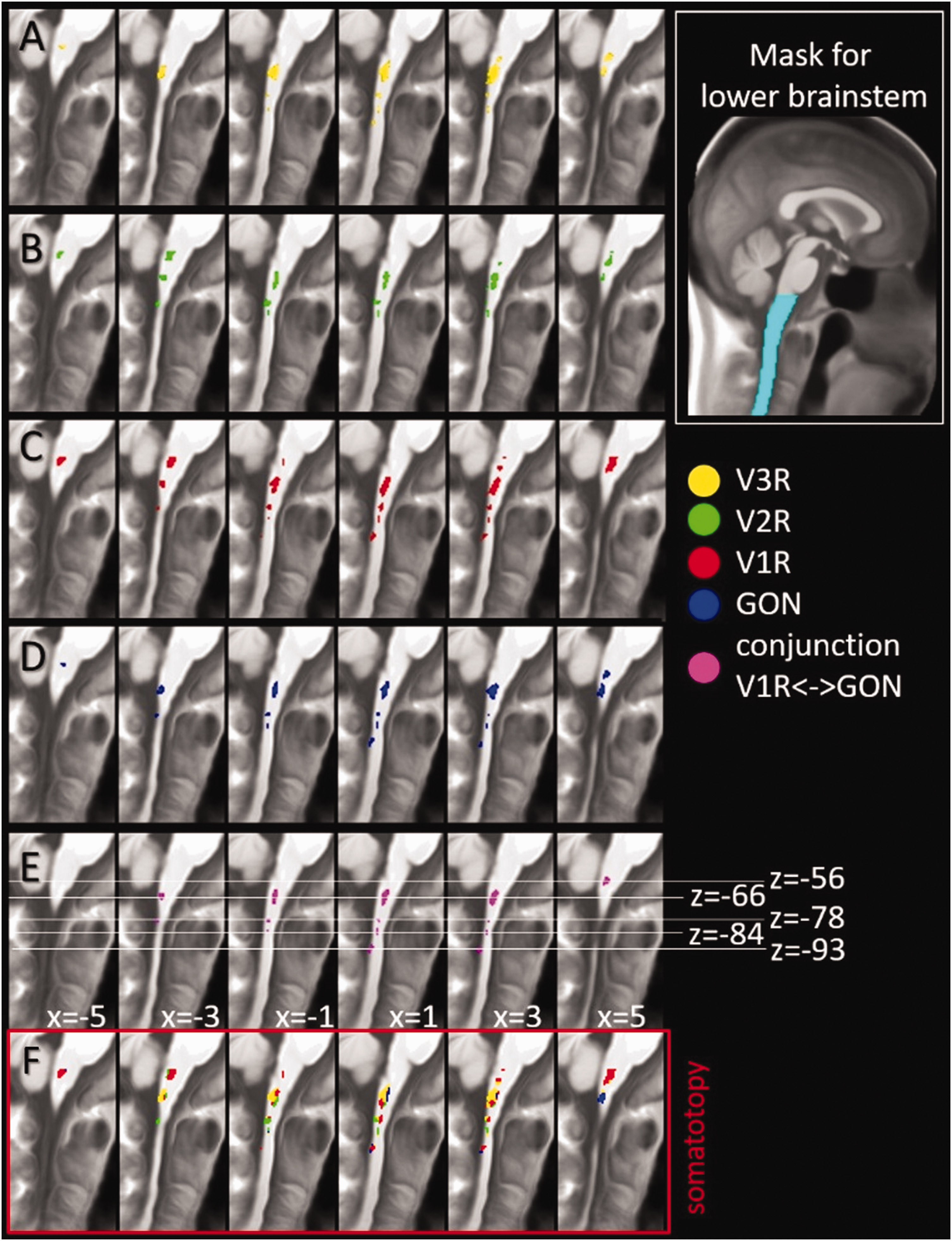
Functional Representations of the trigeminal branches (V1, V2, V3), the GON and the co-localization of V1 and GON in the lower brainstem. A-D) Representations of V1, V2, V3 and GON. E) Common representation, i.e. conjunction, of V1 and GON. F) Somatotopical arrangement retrieved by assigning each voxel to the stimulation site with the highest activity per dermatome. Results are presented for a statistical threshold of T > 3 (n = 61, df = 180) and a minimum cluster extent of >10 voxel. GON, greater occipital nerve; V1R/V2R/V3R/GON, stimulation sites in first experiment.
The analysis where we assigned each voxel surviving the aforementioned threshold to the stimulation site (dermatome) which, within this voxel, had the highest T-value (Figure 4f) showed a clear somatotopic representation of the individual trigeminal branches at different heights of the brainstem/spinal cord.
Functional connectivity between the hypothalamus and the conjunction of V1 and GON
Using the five co-localized clusters of V1 and GON as seeds for a functional connectivity analysis, we found a stronger connectivity (p = 0.027 (FWE-small volume corrected), T = 3.85) between the cluster directly below the pons (MNI coordinate z = −56) for V1, but not between the GON and the hypothalamus (MNI coordinates: [−4, 0, −9]). We found no significant results for the opposite contrast (GON>V1R) nor for the other four clusters which showed a conjunction between V1R and GON (Table 2) within the lower brainstem (Figure 5).
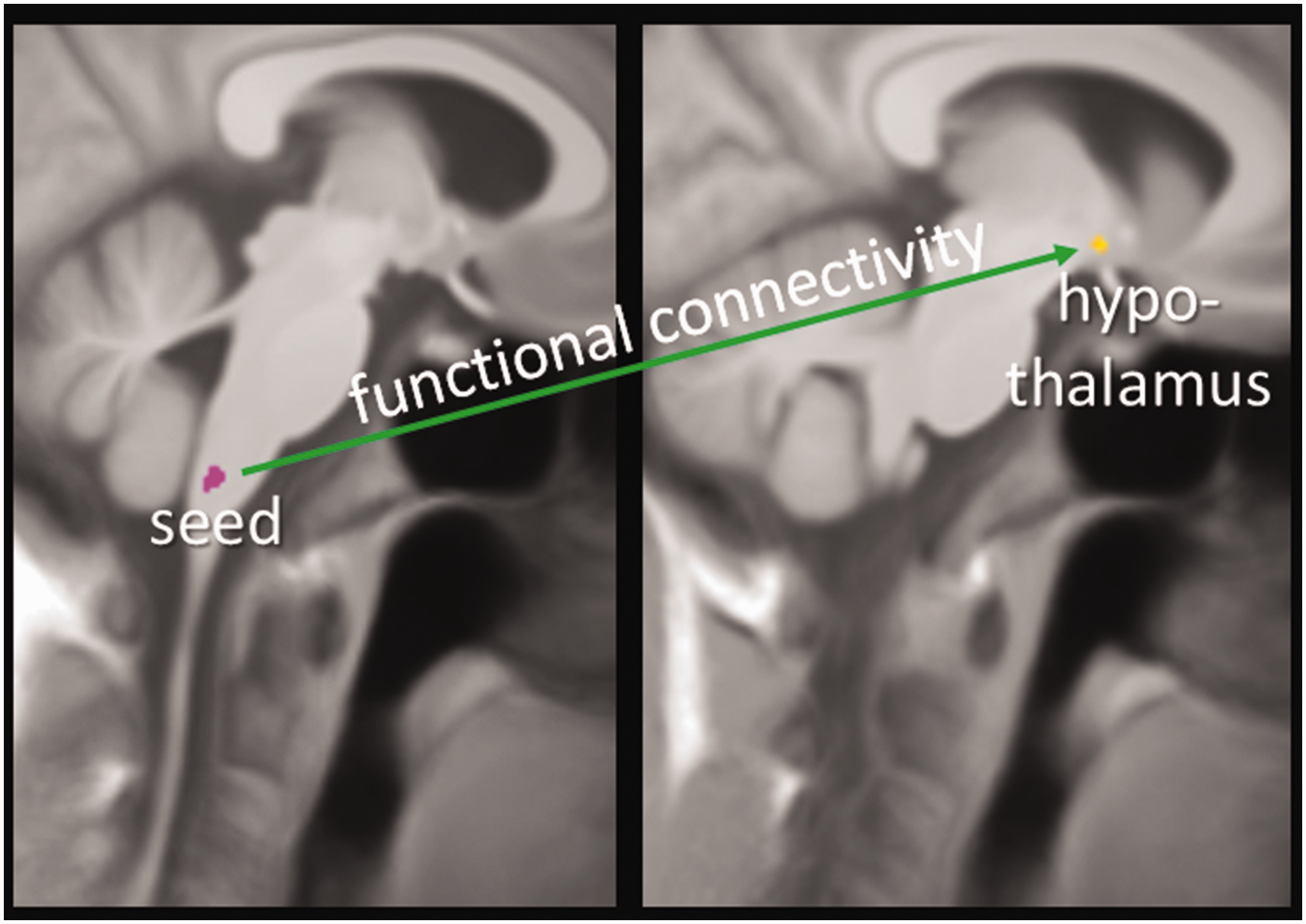
Functional connectivity. Stronger functional connectivity for V1R (stimulation site ophthalmic trigeminal branch first experiment [n = 61]) than for GON (stimulation site greater occipital nerve first experiment).
Functional representations of perioral versus periauricular facial dermatomes of the trigeminal branches
The main effect of the second experiment, testing perioral versus periauricular facial dermatomes along V1 and V3 revealed more rostral representations of the perioral and more caudal representation of the periauricular facial dermatomes along the first as well as the third trigeminal branch (Figure 6a, Table 3, Online Supplemental table 2) at the level of C1. We found differential effects (V1 vs. V3) directly below the pons (for V3C vs V3R, and V3C vs V1R), at the level of C2 (V1C vs V3C) and between C2 and C3 (V1R vs V3R) (Figure 6).
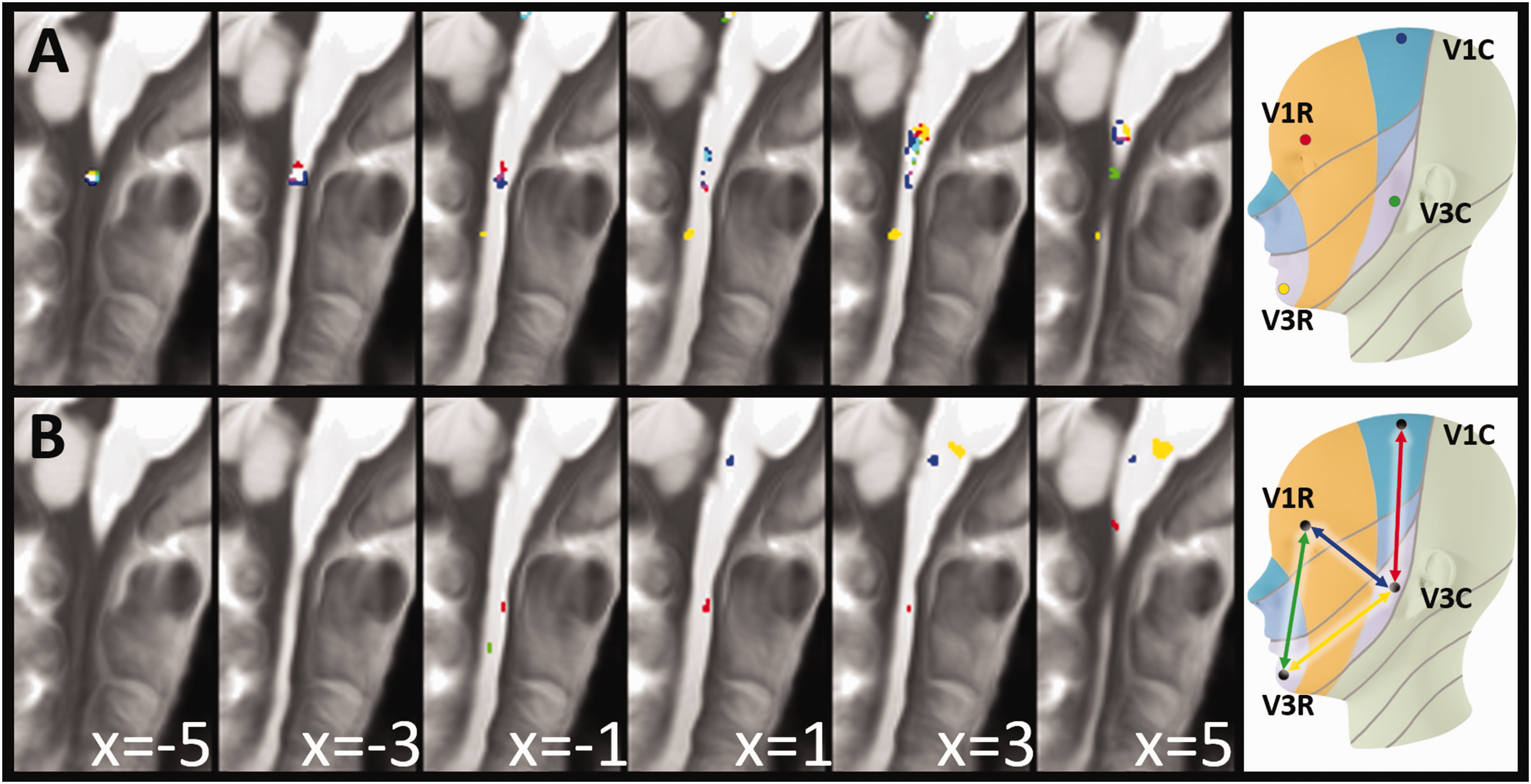
Somatotopy of perioral versus periauricular facial dermatomes. Functional representation of the first and third trigeminal branch in the lower brainstem. (a) Main effects of the stimulation (FWE-corrected p < 0.05, minimum cluster extent of 5 voxel) and (b) significant differential contrasts (T > 3, minimum cluster extent of 5 voxel).
Somatotopy of perioral versus periauricular facial dermatomes of the trigeminal branches in the brainstem below the pons.

Discussion
The aim of this study was to map the functional representation of the three trigeminal branches in the cerebellum, thalamus, insula, and brainstem and to pinpoint a functional co-localization of the trigeminal nuclei with the nucleus of the GON in the lower parts of the brainstem. The STN is the first relay station of the trigeminal nerve for sensory and nociceptive input. We found that this macro-anatomically quite long and thin structure of the lower brainstem holds functional representations of the trigeminal branches at six different levels, where three of them are below the default templates of human fMRI analysis. Our findings are limited by the variance of the individuals’ anatomy in brainstem and spinal cord (for the imaging data) as well as the face and back of the head (for stimulation electrode positioning).
Evidence of a functional relationship between the sensory systems of the face and occipital regions was recently revisited with behavioral data (59) and the question arises where in the brainstem such a functional connection could take place. So far evidence from neuroimaging has pointed towards areas within the brainstem at levels as low as the C2 (44). In our study, we identified possible sites of interaction at five different brainstem levels where representations of the GON are co-localized with the trigeminal nerve. Of particular interest is the level directly below the pons, since this is the only area where the functional connectivity between the first trigeminal branch and the GON to the hypothalamus differs. It will be interesting to investigate whether the functional connectivity is different between patients where the migraine attack starts in the neck/back of the head versus those where the pain starts in the forehead. It is also of interest whether and where in these five anatomical brainstem areas the functional connectivity between the occipital and the trigeminal system changes during the migraine phases or indeed following clinical intervention, e.g. GON-block as well as a possible predictor for efficacy. Nevertheless, since the occipital nerve enters the brainstem and the TCC at the level of C2 and at this same level crosses to the contralateral side from where the nociceptive signal is transmitted to higher processing centers, the most likely functional modulatory effect between both systems is at the level of C2 (27,29,30,60).
The central representation of the three trigeminal branches In the spinal trigeminal nucleus intermingles in an onion-shaped manner with the peripheral facial dermatomes along the perioral-periauricular axis in such a way that, at least at the level of C1, perioral dermatomes are presented more rostrally than periauricular dermatomes. While no differences between the perioral sites of the three trigeminal branches could be shown in the first experiment, an optimization of the stimulation sites to presumably more distant representations in the STN revealed differences between the trigeminal branches as well as along the perioral-periauricular axis. This supports the findings of a central onion-shaped representation of facial dermatomes in humans (7).
In conclusion, our study provides evidence for multiple central representations of the three trigeminal branches as low as C2/C3 and in the brainstem, the cerebellum and the thalamus in humans. Thereby, the individual branches in the STN seem to follow an onion-shaped representation of the facial dermatomes where perioral dermatomes are located more rostrally. Furthermore, we have identified several sites of action for a GON blockage along the STN, where functional activity of V1 and GON are co-localized.
Article highlights
The findings reported in this article suggest a functional inter-inhibitory network between the trigeminal branches and greater occipital nerve in healthy humans. Functional trigeminal representations intermingle perioral and periauricular facial dermatomes with individual branches of the trigeminal nerve in an onion shaped manner and overlap in a typical within-body-part somatotopic arrangement.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231174862 - Supplemental material for Functional brainstem representations of the human trigeminal cervical complex
Supplemental material, sj-pdf-1-cep-10.1177_03331024231174862 for Functional brainstem representations of the human trigeminal cervical complex by Jan Mehnert, Hauke Basedau, Lisa-Marie Sturm, Trine Nielsen, Rigmor Højland Jensen and Arne May in Cephalalgia
Footnotes
Author Roles
JM analyzed the data and wrote the first version of the manuscript; HB analyzed the data; L-MS collected and analyzed the data, TN collected and analyzed the data, RHJ supervised the experiments and interpreted the data; AM designed and supervised the study, interpreted the data and wrote the manuscript. All authors reviewed the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – SFB 936 – 178316478 – A5 to AM
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
