Abstract
Background
Vestibular migraine is considered the most common cause of recurrent vertigo for which specific treatments are missing. Monoclonal antibodies against calcitonin gene-related peptide,, are effective in preventing migraine. Since CGRP is also detected in human cochlear and vestibular organs it may also play a role in vestibular physiology.
Methods
This is a prospective observational cohort study, aiming at evaluating the efficacy of erenumab, fremanezumab or galcanezumab for the treatment of fifty vestibular migraine patients. We assessed mean monthly days with headache and dizziness/vestibular symptoms, pain intensity and migraine-related clinical burden occurring for 18 months.
Results
Response to treatment was excellent as 45 (90%) patients had at least a 50% reduction in vertigo frequency, 43 (86%) had at least a 50% reduction in headache frequency, and 40 (80%) a MIDAS reduction of at least 50%. Overall, 39 (78%) patients had a concomitant reduction of all three parameters. Mean monthly days with dizziness/vestibular symptoms showed an overall significant decrease from a mean of 10.3 ± 1.9 at baseline to 0.8 ± 0.3 days, difference 9.5 (CI 95% 3.6, 15.4; p < 0.001) after twelve months.
Conclusion
We show that anti-CGRP mAbs may be effective in the treatment of Vestibular Migraine. Their use should be encouraged early in the disease course to allow for a better symptom control and quality of life improvement.
Keywords
Introduction
Migraine and dizziness commonly occur together in the general population. The estimated prevalence of dizziness ranges from 1% to 15% while migraine prevalence is 12% (1–2). Several studies have shown the existence of an epidemiological association between the two conditions that is higher than expected by chance. Dizziness is more common in migraine patients compared with those suffering from other primary headache subtypes, conversely, migraine is found in up to 38% of patients with recurrent dizziness (3–5). These data strongly suggest the existence of a pathogenetic link between migraine and vestibular symptoms. This prompted the recognition of Vestibular Migraine (VM) whose diagnostic criteria have been included in the International Classification of Headache Disorders III (6).
VM is considered the most common cause of recurrent vertigo, with a prevalence of 2.7% in adults although its pathogenesis remains unknown (7). Reciprocal connections link the brainstem vestibular nuclei with brain structures involved in modulation of trigeminal nuclei activity and might explain the simultaneous presentation of migraine and vestibular symptoms (8). The trigeminal nucleus is connected to the contralateral thalamus, which in turn sends projections to the temporal, parietal, insular, and cingulate cortical regions. Nociceptive brainstem centers such as nucleus raphe magnus, periaqueductal grey and hypothalamic areas are also connected with the trigeminovascular system (TVS) and vestibular nuclei. These connections can modulate neural activity within either TVS or vestibular system.
Interestingly, the calcitonin gene-related peptide (CGRP), a powerful vasodilating neuropeptide of the TVS, is also expressed in the vestibular system and could be involved in VM pathophysiology by promoting neurogenic inflammation (9).
Current medical treatments for migraine prevention in VM patients consist of antihypertensive, antidepressant, and antiepileptic medications whose effectiveness in VM treatment is not supported by randomized controlled trials. Beta-adrenergic inhibitors, calcium channel blockers and anti-epileptic medications have been reported as helpful in migraine prophylaxis, but their role in VM treatment is not fully understood (10). Flunarizine and cinnarizine are partially effective in decreasing the severity and frequency of vertigo attacks in VM patients (11).
Monoclonal antibodies against CGRP (anti-CGRP MAb) are a new class of drugs that block the CGRP receptor or CGRP itself. Erenumab, fremanezumab, and galcanezumab, are suitable and safe in preventing episodic migraine and chronic migraine (12). Since CGRP is also detected in human cochlear and vestibular organs it may also play a role in vestibular physiology and be effective in VM (13).
We hypothesize that anti-CGRP MAbs could be effective in the treatment of VM, with similar results as seen in episodic or chronic migraine. Since anti-CGRP MAbs are currently used to treat migraine patients fulfilling prescription criteria, many of these patients also meet the diagnostic criteria for VM. The aim of our study was to evaluate the efficacy of anti-CGRP MAb for the treatment of VM using real-world data.
Methods
This was a prospective observational cohort study evaluating the efficacy of erenumab, fremanezumab and galcanezumab for the treatment of patients with chronic migraine who meet the criteria for VM. The study was approved at our local ethics committee and all patients gave written informed consent before any procedure linked to the study.
We identified 50 migraine patients at the Headache Center of University Federico II of Naples for whom the decision to start an anti-CGRP MAb therapy had already been taken. Patients had been treated between November 2020 and June 2021. During this timeframe, treatment choice between the three MAbs was casual as no criteria exist for the choice of one MAb for a single patient. This applies to the general population of migraine patients treated at our center, but no stratification occurred for VM.
Inclusion criteria for the present study were: diagnosis of VM fulfilling the International Headache Society Appendix criteria; age >18 years; history of ≥3 failed treatments with validated migraine preventatives at a standard dose for at least two months. Alternatively, preventive treatments had to be contraindicated. Exclusion criteria were: diagnosis of benign paroxysmal positional vertigo (BPPV), or even the absence of a previous screening for BPPV with an otolaryngologist; diagnosis of Meniere’s disease; diagnosis of other neurological diseases (prior cerebrovascular disease, multiple sclerosis, cerebellar disorders).
We collected data for a maximum follow-up of 18 months. Demographics, detailed medical history, and the presence of comorbidities were recorded at baseline. During a one-month run-in period and for the study duration, patients are routinely asked to complete a headache and vertigo diary. The patient's identity was known only to the treating physician. During data collection, privacy was guaranteed by assigning a pseudonym to each patient.
Erenumab (monthly dose 140 mg), fremanezumab (monthly dose: 225 mg) or galcanezumab (at initial dose of 240 mg and subsequent monthly doses of 120 mg) were administered according to manufacturer’s recommendations. Patients were allowed to take their current preventive therapies if the dose had been stable for at least three months before starting anti-CGRP MAb treatment.
Mean monthly days with headache and dizziness occurring during the run-in period, as well as during anti-CGRP MAb treatment, were evaluated by reviewing standardized paper patient headache diaries at baseline and every three months during follow-up visits. We asked patients to record the presence of vestibular symptoms on a daily basis (defined as any type of internal or external vertigo or dizziness) headache, headache duration, headache-related symptoms (photophobia, phonophobia, aura, nausea, vomiting, motion sensitivity), pain intensity using a 0–10 analogic scale (0 no pain, 1–3 mild, 4–7 moderate, 8–10 severe pain) and the use of acute medication to treat headache (14).
We assessed the migraine-related clinical burden with the Migraine Disability Assessment (MIDAS) at baseline and every three months for 12 months (15).
The primary outcome of this study was the reduction of mean monthly days with vertigo and/or dizziness after 12 months of continuous treatment with MAbs. The secondary outcomes were the reduction of mean monthly days with headache, MIDAS score, and the rate of adverse events.
Since 25 patients completed 18 months of follow up, we additionally included their results in the analysis, as a proxy of longer-term treatment.
Statistical analysis
This is the primary analysis of these data. A descriptive analysis is provided for baseline variables, this included frequency and percentage (for categorical variables), or mean and standard deviation (for continuous variables). We analyzed headache and vertigo days reduction using a General Linear Model for repeated measures (GLM-RM), where headache and vertigo days were set as the dependent variables (repeated measures). We tested gender, type of migraine, number of failed therapies, and concomitant medications, as factors and covariates. The GLM-RM incorporates a two-way analysis of variance. Since sphericity was not met, we used the Greenhouse-Geisser correction. Difference between monoclonal antibodies was assessed with a linear regression model with the maximum reduction in vertigo days as the dependent variable and type of monoclonal antibody as the independent variable correcting for baseline number of vertigo days. The same was done for migraine days, correcting for baseline migraine days, and MIDAS, correcting for baseline MIDAS. Headache and vertigo days are to be intended per month. Significance was set two sided at p < 0.05. Statistical analysis was performed with SPSS (IBM SPSS version 23).
Results
Fifty patients were eligible and included for the study. Five patients discontinued treatment due to inefficacy: three of them were treated with fremanezumab, one with galcanezumab and one with erenumab (two patients discontinued treatment after three months of follow up, two patients after six months and one patient after nine months). We lost data of one patient after six months of follow-up.
Demographic and baseline headache characteristics of patients are reported in Table 1.
Demographic and baseline headache characteristics of patients.

Most patients were female (78%), with a mean age of 45.0 ± 13.3 years. The average time since onset of migraine was 18.7 ± 10.9 years. Two or three or more previously failed treatment were reported in 36% of patients (18/50) or in 32% (16/50) respectively. Eighteen patients (36%) started anti CGRP MAb as add-on therapy due to a reported partial efficacy of ongoing treatment. Among the cohort 25 (50%) patients received Fremanezumab, 18 (36%) Galcanezumab and 7 (14%) Erenumab.
Response to treatment was excellent as 45 patients (90%) had at least a 50% reduction in mean monthly dizziness/vestibular symptom days, 43 patients (86%) had at least a 50% reduction in headache frequency, and 40 patients (80%) a MIDAS reduction of at least 50%, after 12 months of treatment. Overall, 39 (78%) of all treated patients had a concomitant reduction of all three parameters.
Mean monthly days with dizziness/vestibular symptoms showed an overall significant decrease (F = 21.0; p < 0.001; Figure 1a) from a mean of 10.3 ± 1.9 at baseline to 6.4 ± 1.4 days (difference 3.9; CI 95% 1.3, 6.4; p < 0.001) after three months (F = 24.3; p < 0.001), to 3.8 ± 1.0 days (difference 6.5; CI 95% 1.9, 11.1; p < 0.001) after six months days (F = 21.4; p < 0.001), to 1.0 ± 0.5 days (difference 9.3; CI 95% 3.3, 15.3; p < 0.001) after nine months (F = 25.8; p = 0.012), to 0.8 ± 0.3 days (difference 9.5; CI 95% 3.6, 15.4; p < 0.001) after twelve months (F = 27.6; p < 0.001) and to 0.7 ± 0.2 days (difference 9.6; CI 95% 3.6, 15.6; p < 0.001) after eighteen months. Twenty patients had 0 vertigo days at the end of the observation period and only 2 patients had more than 5 days per month.
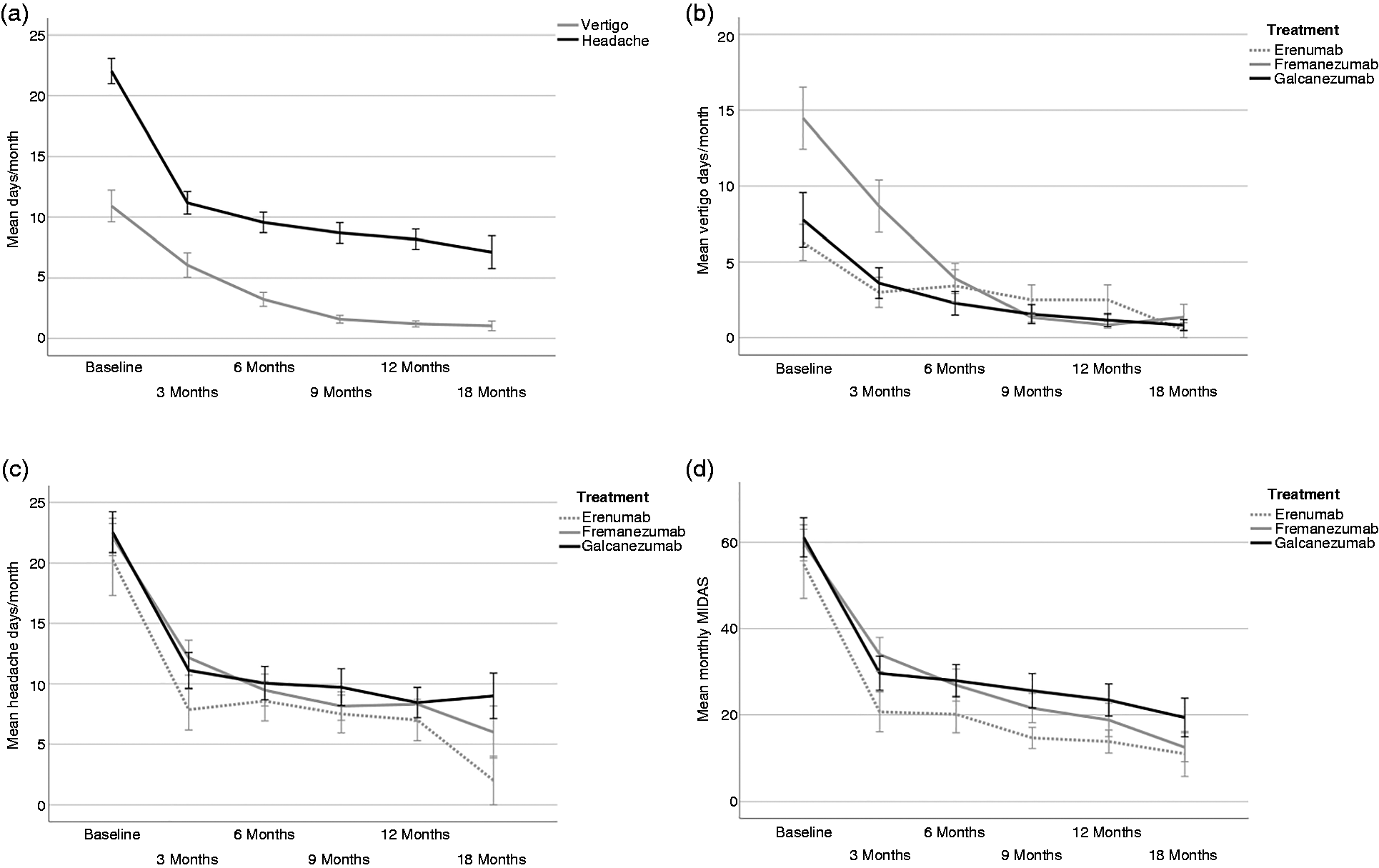
Mean monthly days with vertigo symptoms and mean monthly days with headache (a). Comparison between Erenumab, Fremanezumab and Galcanezumab in terms of mean monthly days with vertigo symptoms (b), headache frequency (c) and MIDAS score (d). Values are mean ± standard deviation (SD).
No significant differences were found in efficacy between three different anti CGRP Mab (B = 0.322; CI 95% −0.50, 1.10; p = 0.434; Figure 1(b).
Migraine monthly frequency showed an overall significant decrease (F = 46.1; p < 0.001; Figure 1a), from a mean of 20.9 ± 1.6 at baseline, to 9.3 ± 1.0 days (difference 11.6; CI 95% 8.1, 15.2; F = 114.1; p < 0.001) after three months, to 8.2 ± 1.0 days (difference 12.7; CI 95% 8.6, 16.8; F = 104.5; p < 0.001) after six months, to 7.5 ± 1.2 days (difference 13.4; CI 95% 8.9, 18.0; F = 93.8; p < 0.001) after nine months, to 7.1 ± 1.1 days (difference 13.8; CI 95% 9.3, 18.3; F = 101.2; p < 0.001) after 12 months and to 6.4 ± 1.2 days (difference 14.5; CI 95% 8.8, 20.2; F = 69.6; p < 0.001) after 18 months.
No significant differences were found in efficacy between three different anti CGRP Mab (B = 0.100; CI 95% −2.0, 2.2; p = 0.925; Figure 1(c).
Monthly MIDAS scores showed an overall significant decrease (F = 33.0; p < 0.001) from a mean of 52.8 ± 5.0 at baseline to 23.7 ± 3.3 (difference 29.1; CI 95% 17.7, 40.6; F = 70.1; p < 0.001) after three months, to 20.7 ± 3.2 (difference 32.1; CI 95% 18.9, 45.4; F = 64.0; p < 0.001) after six months, to 18.4 ± 3.5 (difference 34.4; CI 95% 19.6, 49.4; F = 58.0; p < 0.001) after nine months, to 16.7 ± 3.3 (difference 36.1; CI 95% 21.0, 51.3; F = 61.8; p < 0.001) after 12 months, and to 14.3 ± 3.2 (difference 38.5; CI 95% 21.2, 55.9; F = 53.5; p < 0.001) after 18 months.
No significant differences were found in efficacy between three different anti CGRP Mab (B = 0.709; CI 95% −0.50, 0.93; p = 0.648; Figure 1(d).
Discussion
To date there are no prospective studies aimed at specifically demonstrating the efficacy of anti-CGRP MAbs on VM. In a recent retrospective study on a smaller series of VM patients treated with either erenumab, fremanezumab, or galcanezumab, a trend towards the improvement in intensity of vestibular symptoms was retrospectively assessed using a self-reported scale (16). However, the limited number of patients did not allow for a statistical analysis, and conclusions were limited.
VM is an unmet clinical need as specific treatments are missing, and the disease burden is incredibly high. This is supported by the mean baseline value of the MIDAS score of 52.8 in our patient population, indicating severe disability. In this scenario, the absence of treatment guidelines make it difficult for the clinician to find an adequate treatment aimed at reducing vertigo and migraine days/month, at reducing drug abuse, and at increasing quality of life.
Since CGRP receptors are expressed in the vestibular system of the inner ear, there is the possibility that CGRP is causatively involved in vestibular symptoms associated with migraine. Our data indirectly support this hypothesis, as treatment with anti-CGRP MAbs reduced vertigo days, with many patients showing a complete remission. This is a novel finding and encourages early treatment with MAbs in VM. Clinical trials including VM patients with vertigo days as inclusion criteria and primary endpoint could clarify this point.
Our strongest finding is that almost 80% of our patients had at least a 50% simultaneous reduction in migraine days, vertigo days, and MIDAS score. Efficacy of MAbs on migraine days was higher than previously reported in clinical trials, with more than 80% reduction in migraine days and 80% in MIDAS score.
In conclusion, our data suggests that treatment of VM with anti-CGRP MAbs is effective and should be started early in the disease course.
One limitation of this study is the low number of included patients that did not allow for conclusive data on the direct comparison between different MAbs and for a deferential timing of their efficacy. Also, the relatively small sample of participants limits the strength of our conclusions. It should be noted that this is a prospective observational study, and therefore it may be biased due to it not being randomized and not having a controlled design. The use of diagnostic headache diaries does have some limitations because the patient's general acceptance is still limited, and some subjects may not be able to fill them in properly. We also suggest that future studies should stratify patients both on number of migraine and vertigo days, and Mab choice.
Clinical implications
Our findings have important clinical implications given that specific treatments for migraine prevention in Vestibular Migraine patients are missing, and the disease burden is incredibly high. This is the first prospective study aimed at specifically demonstrating the efficacy of anti-CGRP MAbs on VM. Our data suggests that treatment of VM with anti-CGRP MAbs is effective and should be started early in the disease course. This novel finding is of health relevance for clinical papers.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CVR received personal compensation from Sanofi Genzyme and Merck Serono. MS, SB, AM and AS declare that there is no conflict of interest.
FS received public speaking honoraria from Alexion, Argenx, Biogen, Mylan, Novartis, Roche, Sanofi, Teva; he also received compensation from Advisory Boards and consultation fees from Alexion, Almirall, Argenx, Avexis, Biogen, Forward Pharma, Lexeo Therapeutics, Merk, Novartis, Novatek, Roche, Sanofi, Takeda. RDS received personal compensation from Lilly for oral presentations (2020–2021).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
