Abstract
Background
Vestibular migraine affects 1% of the general population, and 30%–50% of all migraine patients describe occasionally associated vertigo or dizziness. We aimed to identify brain regions altered in vestibular migraine in order to evaluate the connection between migraine and the vestibular system.
Methods
Seventeen patients with definite vestibular migraine were compared to 17 controls using magnetic resonance imaging-based voxel-based morphometry.
Results
We found grey matter (GM) volume reduction in the superior, inferior and middle (MT/V5) temporal gyrus as well as in the mid. cingulate, dorsolateral prefontal, insula, parietal and occipital cortex.
A negative correlation of disease duration and GM volume was observed in areas associated with pain and vestibular processing. Moreover, there was a negative correlation between headache severity and prefrontal cortex volume.
Conclusion
Alterations identified in vestibular migraine resemble those previously described for migraine, but also extend to areas involved in multisensory vestibular control and central vestibular compensation possibly representing the pathoanatomic connection between migraine and the vestibular system.
Keywords
Introduction
About 30% to 50% of all patients with migraine describe vertigo or dizziness associated with their migraine at least occasionally (1,2). In population-based studies the lifetime prevalence of migraine and vertigo in the general population of Western industrial nations is approximately 16% and 7%, respectively (3,4). In the general population the lifetime prevalence of vestibular migraine (VM) was 1%, with a one-year prevalence of 0.9% (5). The most common terms to describe the combination of migraine and vestibular symptoms are VM (6), migranous vertigo, migraine-associated vertigo, vertiginous migraine, migraine-associated balance disturbance and benign paroxysmal vertigo. The International Headache Society (IHS) and the Bárány Society (International Society for Neuro-Otology) created a consensus document with diagnostic criteria for vestibular migraine to clearly diagnose and compare patient populations in a standardized manner (7,8) and it was added to the appendix of the revised version of the International Classification of Headache Disorders (ICHD-3 beta) (9).
The exact pathophysiological mechanisms of VM remain unclear. Most theories concerning the pathogenesis of VM resemble those of typical migraine without offering a hypothesis explaining the complex processes to its full extent (10,11). The neuroanatomically evident tight inter-neural connections between the vestibular system and nociceptive brain stem areas, i.e. the dorsal raphe nuclei, the locus coeruleus and the lateral tegmentum, as well as the connections between inferior, medial and lateral vestibular nuclei and the caudal trigeminal nucleus, strongly indicate an interaction between the nociceptive and vestibular system at brain stem level (10). However, spatial orientation and perception of movement require processing of vestibular information at a cortical level. Multimodal sensory cortical areas that integrate vestibular, visual and somatosensory signals were identified in animal experiments (12–14) and were partly reconfirmed to exist in humans by functional imaging (positron-emission tomography, functional magnetic resonance imaging) (15–17). Structural imaging showed grey matter (GM) changes in peripheral vestibular disorders related to central vestibular compensation in regard to cortical adaptation mechanisms of the vestibular system (18–20). Higher level cortical areas were also described to be structurally altered in migraine (21–27). Exactly which cortical and subcortical brain regions are involved in VM is not known and imaging data on VM specifically are unavailable. We therefore aimed to identify structural brain changes associated with VM in order to demonstrate involvement of not only somatosensory-, pain- and migraine-related structural changes but also changes associated with central vestibular control, dizziness and vertigo.
Methods
Subjects and clinical data
Seventeen patients (14 women) with VM were recruited prospectively from a tertiary vertigo and dizziness centre between May 2012 and January 2013. All patients fulfilled the diagnostic criteria of the IHS (ICHD-3 beta, appendix) and the Bárány Society for definite VM (7,9). Additional neurological and neuro-otologic examination was unremarkable in all patients. No peripheral vestibular dysfunction was found in videonystagmography (VNG) recordings with caloric testing.
Five of these patients were on a migraine prophylactic drug (e.g. beta blockers). Most (10) of the patients used nonsteroidal analgetics for attack treatment, while four regularly used triptans. Two of the patients investigated did not take any medication regularly.
Patients were compared to 17 age- and gender-matched healthy controls. Exclusion criteria were any other primary headache disorder or chronic pain condition, unilateral or bilateral vestibulopathy as assessed by VNG, previous vestibular neuritis, Ménière’s disease, secondary somatoform vertigo, ischemic or haemorrhagic stroke and other severe somatic or psychiatric illness. Demographic data were assessed in a face-to-face interview with a standardised questionnaire including questions from the German version of the Center for Epidemiologic Studies Depression Scale (CES-D) called Allgemeine Depressionsskala (ADS).
Standard protocol approvals, registrations, and patient consents
All participants gave their written informed consent according to the Declaration of Helsinki prior to study inclusion. The local ethics committee of the University of Duisburg-Essen approved the study protocol.
Voxel-based morphometry (VBM)-data acquisition
Imaging was performed on a Siemens Avanto scanner operating at 1.5 T using a standard eight-channel birdcage head coil. T1-weighted magnetic resonance imaging three-dimensional (3D) datasets were obtained using magnetisation prepared rapid acquisition gradient echo (MP-Rage) sequence (repetition time (TR)/echo time (TE)/inversion time (TI) = 2400 ms/3.53 ms/1200 ms, flip angle = 8 degrees, field of view = 256 mm, 160 slices, voxel size 1 × 1 × 1 mm3). The same scanner and the same scanning protocol were used for all patients and healthy controls.
Processing of structural data
VBM is a whole-brain technique capable of revealing subtle, specific changes in GM by applying voxelwise statistics within the context of Gaussian random fields (28). It has been cross-validated with region-of-interest (ROI) measurements and functional data in several studies. Data processing and analysis was performed with SPM8 (http://www.fil.ion.ucl.ac.uk/) including New Segment and Diffeomorphic Anatomical Registration Through Exponentiated Lie Algebra (DARTEL) using Matlab (Matlab 7.6, MathWorks, Natick, MA, USA) (29,30). Pre-processing involved spatial normalisation into the Montreal Neurological Institute (MNI) template space, GM segmentation, spatial smoothing, and modulation in order to adjust for volume changes during spatial normalisation. The spatially normalised and modulated GM partitions were smoothed with an isotropic Gaussian kernel of 10 mm full width at half maximum (FWHM).
Statistical analysis
Statistical analysis tested GM volume differences between VM patients and healthy controls using a two-sample t-test. To control for age- and gender-related GM changes, both were implemented in our statistical model as covariates. To avoid any unintentional bias by a priori hypotheses in the primary analysis, a whole brain analysis was performed. To correct for multiple comparisons, ROI analysis was performed based on anatomic structures previously described to be altered in migraine or affected in vestibular disorders (i.e. cingulate cortex, precentral cortex, thalamus, insula, amygdala, dorsolateral prefrontal cortex (PFC), middle, superior and inferior temporal gyrus, MT/V5, precuneus, superior parietal lobule, supramarginal gyrus, and posterior parietal cortex (18,19,24,25,27,31). The ROIs were adapted from the anatomical automatic labelling (AAL) ROI library (32). Correction for multiple comparisons was performed with family-wise error correction (FWE) p < 0.05 within the predefined ROI. No further additional extent threshold was applied.
Correlation analysis was performed for attack frequency, disease duration, mean pain intensity on a verbal/numeric rating scale (VRS; 0 = no pain, 10 = worst imaginable pain) and ADS/CES-D using SPM8. Age and gender were implemented in those as additional covariates. The primary analysis was performed for the whole brain uncorrected at a threshold of p < 0.001 and FWE corrected for multiple comparisons at a threshold of p < 0.05. The same ROI analysis as described above was applied with FWE correction at a threshold of p < 0.05.
Results
Demographics and patient characteristics.
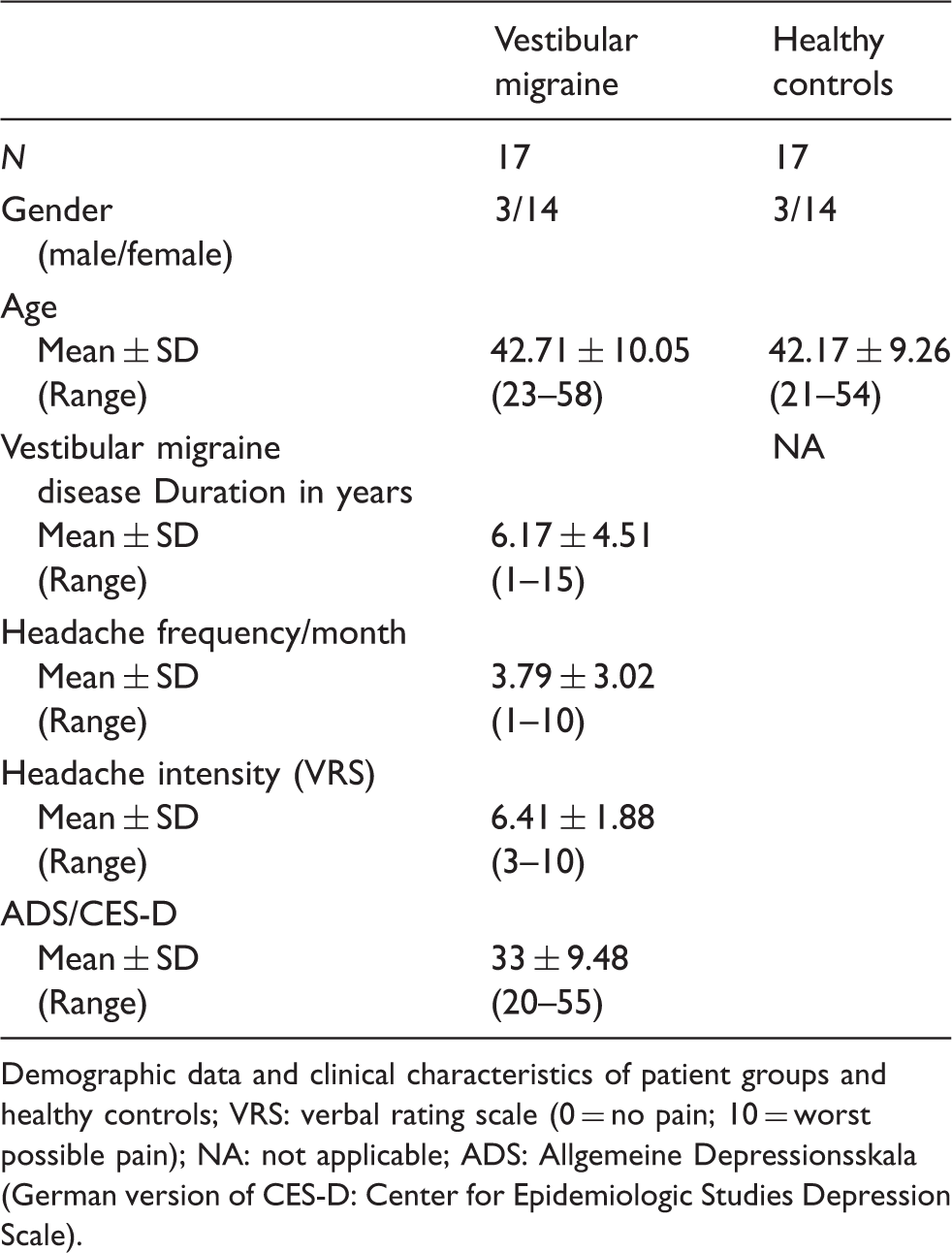
Demographic data and clinical characteristics of patient groups and healthy controls; VRS: verbal rating scale (0 = no pain; 10 = worst possible pain); NA: not applicable; ADS: Allgemeine Depressionsskala (German version of CES-D: Center for Epidemiologic Studies Depression Scale).
Cross-sectional analysis revealed regional differences in GM volume between healthy controls and VM patients. In VM patients a GM volume decrease was identified bilaterally in the inferior temporal gyrus, the cingulate cortex and the posterior insula. Further areas with GM reduction are the left-sided superior temporal gyrus, MT/V5 (mid. temporal gyrus), supramarginal gyrus and superior parietal lobulus as well as the right sides of the dorsolateral PFC and inferior occipital gyrus/V4 (Figure 1 and Table 2).
Structural brain changes related to vestibular migraine (VM vs. health controls (HCs)). Statistical parametric maps showing grey matter volume decrease within the trigeminal pain-processing and vestibular network at defined Montreal Neurological Institute (MNI) coordinates. Grey matter changes are shown superimposed on a standard T1 image. Right side of the image is the right side of the brain. All images are shown with a threshold of p < 0.001 uncorrected; the colour bar displays t values. Row 1 showing sagittal slices with given x-coordinates: 48: right inferior temporal gyrus; 35: right inferior occipital gyrus/V4, right posterior Insular, right dorsolateral prefrontal cortex; 7: right mid. cingulate cortex; −32: left superior parietal lobulus, left posterior insular; −39: left mid. temporal gyrus (MT/V5), left inferior temporal gyrus, left posterior insular −48: left supramarginal gyrus, left inferior temporal gyrus; −64; left superior temporal gyrus. Row 2 showing axial slices with given z-coordinates: 50: right dorsolateral prefrontal cortex; 33: bilateral mid. cingulate cortex; 16: left supramarginal gyrus, bilateral posterior Insular; −8: left mid. temporal gyrus (MT/V5); −25: bilateral inferior temporal gyrus. Regions of reduced grey matter volume in patients with vestibular migraine compared to healthy controls. Regions with grey matter (GM) reduction in comparison to healthy controls. MNI: Montreal Neurological Institute; MNI coordinates in three planes x, y, z in mm; all regions at a threshold p < 0.001 uncorrected; kE: number of voxels per cluster; WB: whole brain uncorrected p value; ROI: region of interest analysis using family-wise error correction at p < 0.05; NA: not applicable; n.s.: not significant.

No increase in regional GM volume was detected at a threshold of p < 0.001 uncorrected for the whole brain.
Regression analysis testing for a correlation between attack frequency or ADS/CES-D and GM volume produced no significant results. There was a marked and widespread negative correlation of disease duration with GM-volume reduction in pain and vestibular processing areas illustrated in Figure 2 and Table 3. Furthermore, the PFC showed GM reduction as mean individual headache severity increased (Figure 2 and Table 3).
Correlation of grey matter (GM) volume decrease with (a) duration of disease (showing sagittal slices with given x-coordinates: x = 57: superior and mid. temporal gyrus (right); x = 30: amygdala (right); x = 13: thalamus (right), anterior cingulate cortex (right); x = −3: middle cingulate cortex (left), posterior anterior cingulate cortex (left); x = –17: thalamus (left), superior parietal lobulus (left)) and (b) headache intensity (VRS: verbal rating scale) (showing sagittal slices with given x-coordinates: x = 36: middle frontal gyrus (right), inferior occipital gyrus/V4(right); x = −32: middle frontal gyrus (left); x = –46: precentral gyrus (left), insula (left), inferior temporal gyrus (left)). GM changes are shown superimposed on a standard T1 image. Right side of the localising image is the right side of the brain. All images are shown with a threshold of p < 0.001 uncorrected. Regions with reduced grey matter in correlation to disease duration and headache intensity. Regions with grey matter (GM) reduction in correlation analysis with (a) disease duration and (b) headache intensity (VRS: verbal rating scale). MNI: Montreal Neurological Institute; MNI coordinates in three planes x, y, z in mm; all regions at a threshold p < 0.001 uncorrected; kE: number of voxels per cluster; WB: whole brain uncorrected p value; ROI: region of interest analysis using family-wise error correction at p < 0.05; NA: not applicable; n.s.: not significant.

Discussion
GM volume reduction was found in brain regions previously described to show structural alterations in patients with migraine. In addition, we found regions that are generally associated with higher-level multisensory vestibular function emphasising the extensive overlap of migraine-related central circuits with central vestibular pathways beyond the vestibular nuclei, other brain stem structures and the cerebellum. These brain areas may represent higher order cortical vestibular processing centres involved in the pathophysiology of VM and thus influencing dizziness, vertigo, disequilibrium and ataxia that often accompany migraine attacks either as premonitory symptoms, in the form of aura, or as autonomic symptoms similar to nausea.
One striking region in this regard appears to be the temporal lobe, which is altered in headaches (25,33,34) as well as in central vestibular processing (18,31). Certainly the exact function in both symptoms/conditions is far from understood. Multiple areas within the temporal lobe were altered in VM. The superior temporal gyrus (STG) is consistently described in different studies investigating central vestibular processing and function. Its specific role remains under discussion, but it seems to be involved in the secondary processing of coordination of the eyes and head as well as spatial orientation (19). Different studies on structural brain changes following vestibular neuritis described GM decrease in the STG similar to what we found in VM (18,19). This was interpreted as part of compensatory mechanisms in regard to central vestibular compensation of the dysfunctional vestibular organ and subsequent cortical plasticity of secondary vestibular processing centres. The STG is closely connected to the multisensory parieto-insula cortex, which may explain the involvement in our patients and other pain/headache conditions.
Alterations in the middle temporal gyrus (MTG) further underline the connection between the vestibular system and migraine as the lateral temporal lobe plays a role in both conditions (25,35,36), even though, similarly to STG, this may not be specific for either of the two systems (i.e. vestibular and trigeminal pain or migraine premonitory).
The MTG appears to be functionally relevant for vestibular compensatory mechanisms and is strongly interconnected with other multisensory cortical areas forming a multisensory-integrative network (36). Repeated VM attacks over time that seem to lead to an alteration of pain and vestibular processing structures may provide an explanation as to why VM patients interictally often show central vestibular function abnormalities such as gaze-evoked nystagmus or saccadic eye movements and are more sensitive to movement perception outside and during migraine attacks (37,38). Nystagmus and saccadic eye movement are generally considered to be infratentorial problems, but cortical control of vestibular function is not well understood and the alterations identified may be an indirect correlate in terms of altered top-down modulation.
The MTG and adjacent STG together with the lateral aspect of the inferior parietal cortex represent the temporal perisylvian vestibular cortex that is particularly sensitive for dizziness (39). During and between VM attacks the temporal cortex metabolism is activated, supporting our structural findings (31).
Interestingly both regions (STG and MTG) show a more pronounced decrease with the duration of the disease in correlation analysis suggesting a temporal relationship between migraine and vestibular symptoms that may intensify or worsen over time in parallel to the structural changes identified with VBM. As the changes are not related to attack frequency or pain severity they may represent the brain’s attempt to compensate for the frequent vestibular symptoms in a similar fashion as described for peripheral vestibular disorders. In contrast, the inferior temporal gyrus is not altered with disease duration but with headache intensity.
Many regions we found are multi-integrative, secondary processing areas known to be involved in evaluation, integration and anticipation of pain with a strong association with mood and anxiety. Correlation analysis revealed that GM decrease in the prefrontal brain areas correlates with increasing subjective pain intensity on a VRS in VM. The PFC is part of a network involved in affective and cognitive evaluation of pain (40). Thus, these alterations could reflect the affective modulation of pain and vestibular symptoms in VM, which are often perceived much as worse in combination by the affected patient than any of the two alone. As for its role in pain processing and interoceptive awareness, it may contribute to a higher vulnerability for catastrophising and misattribution.
The correlation of disease duration and GM decrease showed alterations in both classical pain-processing areas as well as multisensory cortical regions. Some of the regions identified in the correlation analysis were not altered in group comparison (e.g. anterior cingulate cortex and bilateral thalamus), which may be explained by the wide range of disease duration in the investigated cohort (one to 15 years). Nevertheless, the correlation suggests that these changes are most likely the consequence of frequent sensory and vestibular input over time in regard to cortical plasticity.
Unfortunately, VBM has considerable methodological limitations. The VBM algorithm identifies variations of GM concentration per voxel as well as changes in classification of individual voxels, e.g. from white to GM (41). Several underlying pathophysiological mechanisms have been proposed to reflect GM alterations measured by VBM, such as increase/decrease in cell size, neural or glial cell genesis/degeneration, and changes in blood flow or interstitial fluid (42,43). It remains unclear whether GM alterations are caused by irreversible mechanisms such as neuronal degeneration/apoptosis or by fast adjusting, reversible neuronal processes such as dendrite spine and synapse turnover (44) or whether they simply reflect changes in extracellular space or microvascular volume. Only direct histopathological analysis can unequivocally disclose the underlying cytoarchitecture and pathoanatomical relevance associated with GM changes detected by VBM.
In summary, we were able to depict GM volume decrease in patients with VM in brain regions characteristic for pain processing, as well as multisensory vestibular processing and central vestibular compensation. Whether these changes are a consequence of the disorder or whether pre-existent or acquired alterations of these regions make patients more susceptible to the development of VM or vestibular symptoms in migraine respectively remains unclear. Most likely the observed alterations reflect adaptation mechanisms related to cortical plasticity in patients with VM.
Clinical implications
Brain regions were identified that relate to migraine as well as the vestibular system. Vestibular migraine (VM) shows brain changes similar to those reported in central vestibular compensation following peripheral vestibular dysfunction. Widespread volume decrease in pain and vestibular processing areas including bilateral thalamus and cingulate cortex parallel increasing disease duration. Predominant frontal cortex and insula volume decrease as mean headache severity increases, reflecting a strong affective component in VM.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
Dr Obermann has received scientific support and/or honoraria from Genzyme, Biogen Idec, Novartis, Sanofi-Aventis, Pfizer, and Teva. He has received research grants from Electrocore, Allergan, and the German Ministry for Education and Research (BMBF).
Mr Wurthmann, Mr Schulte Steinberg, Dr Naegel and Dr Theysohn have nothing to declare.
Prof Dr Diener has received honoraria for participation in clinical trials, contributions to advisory boards or lectures from Addex Pharma, Allergan, Almirall, AstraZeneca, Bayer Vital, Berlin Chemie, Coherex Medical, CoLucid, Böhringer Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Grünenthal, Janssen-Cilag, Lilly, La Roche, 3 M Medica, Minster, MSD, Novartis, Johnson & Johnson, Pierre Fabre, Pfizer, Schaper and Brümmer, SanofiAventis, and Weber & Weber; and has received research support from Allergan, Almirall, AstraZeneca, Bayer, Galaxo-Smith-Kline, Janssen-Cilag, and Pfizer.
Author contributions
Dr Obermann: study design, acquisition of data, statistical analyses, interpretation of data, drafting the manuscript.
Mr Wurthmann: acquisition of data, interpretation of data, statistical analyses, revising the manuscript.
Mr Schulte Steinberg: acquisition of data, interpretation of data, revising the manuscript.
Dr Naegel: acquisition of data, statistical analysis, interpretation of data, revising the manuscript.
Dr Theysohn: interpretation of data, revising the manuscript.
Prof Dr Diener: study design, revising the manuscript.
