Abstract
Background
This systematic review focuses on chronic migraine patients with medication overuse headache using, respectively, topiramate, botulinum toxin type A, and human monoclonal antibodies targeting calcitonin gene-related peptide or its receptor.
Methods
A systematic search was conducted in the databases CENTRAL, MEDLINE, Embase and Web of Science until May 2022. We included randomized controlled trials reporting the outcomes of change in monthly headache/migraine days, ≥50% response rates and change in medication overuse status. Studies were excluded if response rates were not reported. Risk of bias assessment was performed using the Cochrane RoB2 tool. The quality of evidence for outcomes across included studies was evaluated according to the five factors outlined in Cochrane GRADE approach.
Findings
The initial search resulted in 1599 records. Following screening, 10 studies met our inclusion criteria, while seven studies with sufficient data were included in the meta-analysis. Studies assessing Botulinum toxin type A included 1139 patients and showed a mean reduction in headache frequency by 1.92 days per month compared to placebo (−1.92; 95% CI −2.68 to −1.16). Studies assessing human monoclonal antibodies included 1982 patients, and showed significant positive effect compared to placebo for all measured outcomes. The overall odds ratio for the ≥50% response rate was 2.90 (95% CI, 2.23 to 3.78). No significant difference was observed in the frequency of adverse effect for both Botulinum toxin type A and low dose of human monoclonal antibodies compared to placebo. There is currently insufficient evidence to determine the impact of topiramate in chronic migraine patients with medication overuse headache.
Interpretation
Botulinum toxin type A and human monoclonal antibodies targeting calcitonin gene-related peptide receptor were beneficial in reducing monthly migraine days and ≥50% response rate, but uncertainties remained for Botulinum toxin type A regarding response rate. The effect size for human monoclonal antibodies was greater with relatively lower drop-out rate. High-quality randomized trials are required to evaluate the effect of topiramate in chronic migraine patients with medication overuse headache.
Introduction
Migraine is a chronic neurological disorder, affecting one billion people worldwide (1,2). Recently, migraine was ranked as the second largest contributor to the disability-adjusted life-years (DALYs), and is the most burdensome in productive age groups (3,4). The symptoms have substantial impact debilitating the individual’s quality of life, family relationship, loss of productivity, and economic cost (5). Individuals fulfilling the criteria of chronic migraine (CM) suffer from headache for ≥15 days per month for ≥3 months, in comparison, at least eight days a month fulfills the diagnostic criteria for migraine (6). Worldwide, 0.5%–2% of the general population have CM (7,8). The overuse of acute medication may facilitate the transition from CM to medication overuse headache (MOH), and approximately half of the individuals with CM have MOH (7). Steiner et al. (9) reported that CM was the sixth highest cause of disability and became the third leading cause of disability in the presence of MOH. Moreover, a recent systematic review argued that CM and MOH sufferers had a lower health-related quality of life than episodic headache sufferers (10).
MOH largely affect individuals aged 30 to 50 years and is more common among women than men (11,12). The mean prevalence in the general population in the industrialized countries is 1–2% (13). The Eurolight project demonstrated that MOH is one of the expensive headache disorders to the individual as well as to the health care system (14,15). MOH has been defined as headache occurring ≥15 days per month in individuals with existing primary headache and developing as a consequence of regular overuse (use on ≥10 or ≥15 days/month, depending on the medication of acute medication for >3 months that significantly worsen pre-existing headache disorder) (6).
The treatment strategies for MOH patients have changed during the last decades. Previously, there was a general belief that patients with MOH need to discontinue overused medications before they respond to preventive medication (16). Recently, however, several randomized trials have shown that MOH patients may respond positively to early introduction of preventive treatment (16,17). Over the past few decades, treatment with topiramate, botulinum toxin type A (BoNTA), and human monoclonal antibodies (mAbs) targeting calcitonin gene-related peptide (CGRP) or its receptor (CGRPr) have proven effective in patients with CM, but uncertainties remain for MOH patients. Given a huge disability and hardship associated with CM and MOH, evidence to ameliorate the clinical outcomes in this population is inadequate. Thus, this systematic review and meta-analysis aims to describe and evaluate the relative effects of preventive treatment of topiramate, BoNTA, and CGRP-antibodies in CM patients with MOH.
Methods
The present systematic review was conducted according to the recommendations of Preferred Reporting Items for Systematic reviews and Meta Analyses (PRISMA) statement (18), as well as the Cochrane Handbook for Systematic Reviews of Interventions (19). The review protocol was published in Cephalalgia Reports (20).
Eligibility criteria
Included studies were required to be placebo-controlled randomized trials examining the effect of topiramate, BoNTA, or mAbs targeting CGRP or its receptor (CGRPr) on an adult population (≥18 years of age) diagnosed with the combination of CM and MOH. The mABs targeting CGRP or CGRPr include eptinezumab, erenumab, fremanezumab and galcanezumab. All available dosing regimens were included regardless of the formal approval for the respective intervention drugs. At the protocol phase, we anticipated to include individuals with CM and MOH defined according to ICHD criteria (ICHD-2 or ICHD-3 or ICHD-3 beta) (6,21,22). However, the majority of the studies did not fulfill ICHD criteria for MOH definition with regard to the duration of overuse for at least three months. Thus, MOH population in this review included both short- and long-term evaluation for overuse medications. In conditions when studies included multiple population or multiple interventions, data that fulfils the eligibility criteria for this review study were extracted for evidence synthesis. Studies were required to report primary outcomes that included headache/migraine frequency, ≥50% response rate (proportion of participants reporting a reduction in migraine or headache frequency by at least 50% from baseline), changing status from MOH to no MOH or episodic migraine without MOH. The secondary outcome variables collected were frequency of acute medications intake (analgesics, triptans and other type of pain killers), adverse events, levels of disability and quality of life. Studies were excluded if response rates were not reported, or if outcomes comparing preventive drugs were reported. Details on exclusion and inclusion criteria is described in detail in the published protocol (20).
Information sources
For this systematic review, the trained information specialists (SAP) developed detailed search strategies for each different database. The search included thesaurus- and free-text terms for the main concept MOH combined with prophylactic treatment in the form of either topiramate, BoNTA or mABs. The search was adapted and run in the bibliometric databases MEDLINE, CENTRAL, Embase, Web of Science and the registers ClinicalTrials.gov and ICTRP. All resulting records were collected in EndNote, where duplicates were removed prior to screening. The detailed description of the search strategies used in the present review and a detail example for search in Ovid MEDLINE is provided in online Supplementary Material 1–2.
Study selection
Two reviewers (SG and KH) screened the titles and abstracts of the studies individually according to the inclusion and exclusion criteria. Studies accepted during abstract screening were retrieved for full text review, two reviewers (SG and KH) screened these independently. Study selection process is shown in Figure 1. Disagreements between the reviewers were resolved through discussion. Placebo-controlled randomized controlled trials (RCTs) reporting primary outcomes for topiramate, BoNTA and mABs were included in the quantitative synthesis. The reviewers were not blinded to study investigators identification, journal of publication, or study results at any stage of the review.
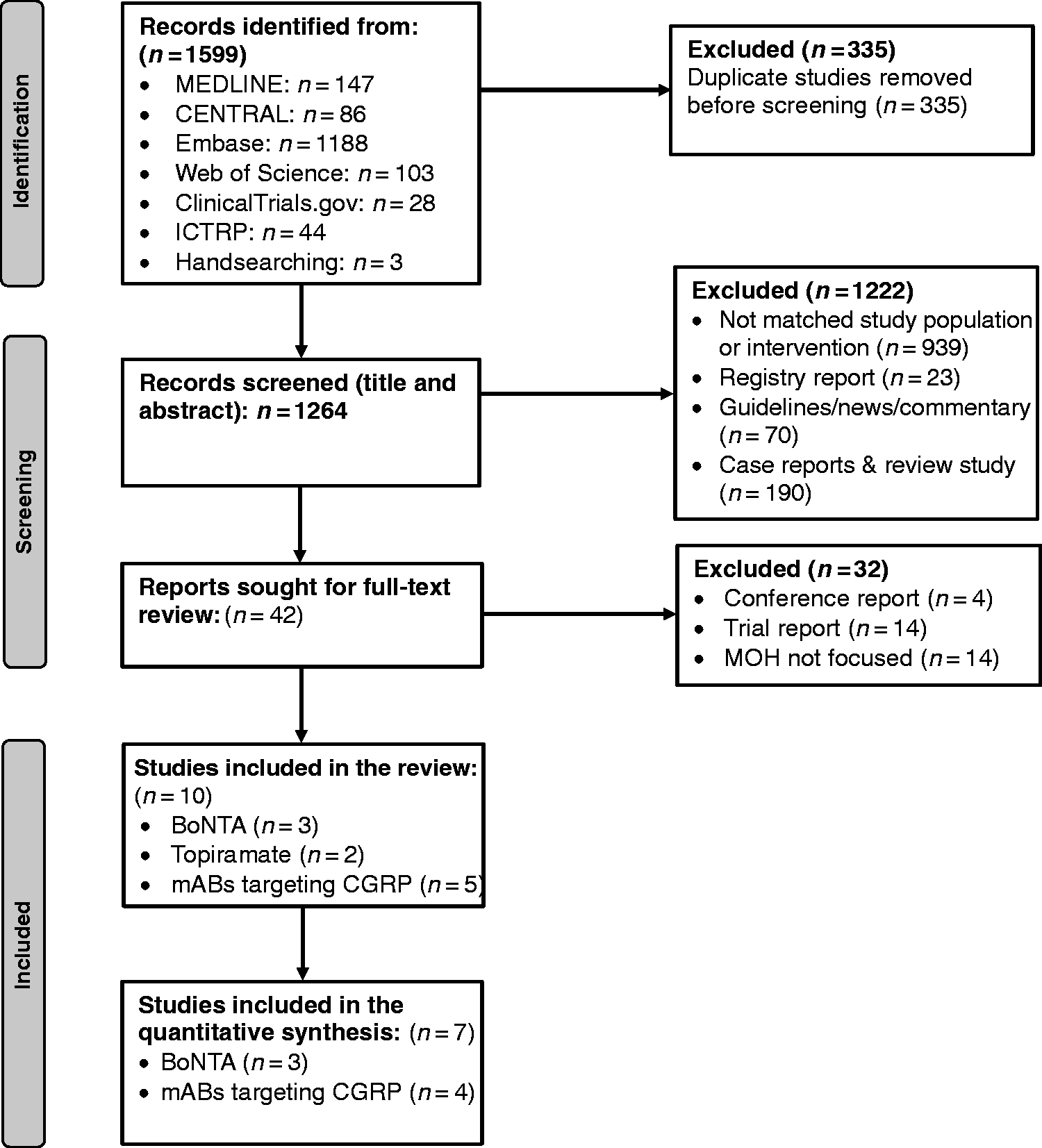
PRISMA flow diagram of study selection process.
Data collection process
Two authors (SG and KH) independently extracted data from the included studies in the purpose-build pre-tested forms. Information extracted included study title, authors, publication date, journal published, methodology (study design, study setting, date of study, number of study centers, withdrawals, intervention, duration of study/intervention, inclusion/exclusion criteria), intervention description (frequency, intensity, duration, modes of intervention), sample size (number randomized, loss to follow-up), population characteristics, efficacy outcomes with time points, adverse events, and potential bias. Disagreements between the authors were resolved through discussions.
At the protocol phase, we expected that the study period would vary (e.g., number of headache/migraine days per 12 weeks vs 24 weeks), and the results would be reported for different time points. Thus, we sought to standardize the unit of time-points over which headache/migraine frequency was measured at 12 weeks wherever possible.
We anticipated that the outcomes measured on a continuous scale (e.g., headache/migraine frequency) would be reported in several ways (e.g., mean baseline/endline, and or change mean headache days). We preferred to report change from baseline to post-treatment in each group with mean difference (MD) and standard deviation (SD) wherever feasible, assuming the baseline data would be balanced due to the study design (RCTs). It was expected that many trials would report different efficacy measurements including means with or without SDs. In missing cases, we either asked the primary authors or SDs were calculated based on available information (SD = SE*√N).
For dichotomous outcomes (success/failure), we expected that the response rate would either be reported as ≥50% reduction in headache/migraine frequency or ≥30% reduction in headache/migraine frequency. Based on clinical significance and our primary outcome to be measured, we choose to report ≥50% response rate. Likewise, change in medication overuse (MO) to no MO was investigated as another dichotomous outcome. In such cases, we extracted data for each treatment arm, the number with success and the number with failure and the total number of patients included in the analysis.
Risk of bias assessment
Two review authors (SG and KH) independently assessed risk of bias using the Cochrane Risk of bias in randomized trials (RoB2) tool (23). We assessed risk of bias for each outcome included in the quantitative synthesis using the five domains of Cochrane RoB2 tool (23). This includes bias arising from: randomization process, deviation from intended interventions, missing outcome data, measurement of the outcome, selection of the reported result, and other risk of bias. For each domain, a series of signaling questions with answers (yes, probably yes, no, probably no, no information) determine the risk of bias as “low risk”, “some concerns” and “high risk”.
Certainty of evidence
The quality of evidence for outcomes across included studies were rated according to the five factors outlined in Cochrane GRADE approach: risk of bias (study design and limitations), heterogeneity or consistency of effects/results, directness (generalizability), precision (sufficient data), and publication bias (reporting of the results across all studies that measure that particular outcome) (24). Four levels of certainty (high, moderate, low and very low) were provided for each outcome. The certainty of evidence was assessed by two reviewers (SG and KH) independently using GRADEpro tool (25). Disagreements between the authors were resolved through discussions. The quality ratings started at high because of RCTs and were downgraded by one level for each of the factors not met, to a maximum of three levels for all factors. We justified all decisions to downgrade the quality of studies in the footnotes.
Analysis
The summary estimates were extracted where available for all the primary outcomes and consolidated by the lead author (SG). Aggregated data from the included studies were transferred to STATA and meta-analysis were performed using STATA 17.1 (StataCorp LP, College Station Texas, USA). Meta-analysis was done for the outcomes whose data was provided from at least three studies. The primary outcomes for the efficacy analysis in this study were mean change in headache/migraine frequency, ≥50% response rate, and changing MO status to no MO. According to the available data, we reported change in monthly headache days and ≥50% response rate for BoNTA studies. For mABs studies, change in monthly migraine days, ≥50% response rate and change in MO status was reported. As an effect measure, we choose to report MD and SDs for change in headache/migraine frequency from baseline, and odds ratios (ORs) for ≥50% response rates and for changing MO status. All studies reported mean change in headache frequency from baseline and SE/SD, as well as baseline and end-line mean frequency with SD. SD for mean change was calculated if required. MD for change in headache/migraine frequency from baseline, and ORs for achieving ≥50% response rate and changing MO status were calculated and pooled across studies using random effects meta-analysis (26). Odds ratio is a robust measure that minimizes discrepancies in study designs such as, duration of study, duration of treatment, and inclusion criteria. The model’s confidence interval (CI) was defined as 95%. Bigger box in the forest plot indicates higher weighting, and weighting was performed with inverse variance method. Heterogeneity across studies was assessed using I2 statistics, I2 >50% is evaluated as considerable heterogeneity (27). Effect sizes were demonstrated through forest plots. Weight for each study were calculated by the inverse variance of their effect estimates.
Results
A total of 1599 records were identified through database, trial registry and manual search (Figure 1). The search was conducted until May 2022. After removing duplicates, 1264 unique records remained for title and abstract screening. After the initial screening, 42 studies were identified for full-text screening. Four relevant ongoing RCTs (1 BoNTA and 3 mABs) were identified, but no response was received from the authors or the sponsors upon contact attempts by e-mail. By restricting the studies to MOH population, 10 RCTs were finally identified for inclusion in the systematic review (Figure 1).
Characteristics of included studies
The 10 studies reported data from nine unique studies. Of these, three compared different doses of botulinum toxin type A (100–195 units) with placebo (28–30), four studies compared different doses of mAbs targeting CGRP(r) (70–1125 units) with placebo (31–34), and two compared topiramate (25 mg with titration to100 mg per day) with placebo (35,36). All trials were based in the Europe and North America, while few trials for mABs also included sites in South America and Asia. The date of publication ranged from 2011 to 2019 for BoNTA trials, 2019 to 2021 for mABs, and 2006 to 2007 for topiramate. Seven of the 10 included trials (three BoNTA and four mABs) with sufficient data were included in the meta-analysis (Figure 1). Three studies were included only in the narrative synthesis, one mABs study reporting changing status of acute headache medication intake (37), and two studies reporting efficacy of topiramate (change in monthly migraine days and response rate) (35,36).
The characteristics of the included studies are summarized in Tables 1–2. All included studies were a parallel-arm double-blinded study with placebo as a control group. A total of 3121 participants were included in the quantitative synthesis, 36.5% in trials investigating the effects of BoNTA and 63.5% in mAbs targeting CGRPr. The mean age of patients varied between 41 to 49 years, and 83.5% were female. The double-blind treatment period (DBTP) of the included trials varied from 12-24 weeks. With the exception of one study evaluating mABs (32), all included trials followed patients after DBTP for a period between eight weeks to 36 weeks. Two studies evaluating BoNTA (28,30), and three mABs targeting CGRPr (32–34) trials had defined MOH based on four weeks baseline period, while others have defined MOH for at least three months’ evaluation (29,31).
Characteristics of included studies assessing botulinum toxin type A for the prevention of chronic migraine with medication overuse headache.

aDropout rates implies withdrawal with any reason in the treatment group, unless otherwise noted.
bResembles to subjects terminating due to treatment-emergent adverse events.
cIncludes PREEMPT 1 and PREEMPT 2 studies (PREEMPT 1 = 56 sites & PREEMPT = 66 sites).
dPlacebo group received 17.5 units of botulinum toxin type A in the frontal area.
DBTP: double-blind treatment phase; HD: headache days.
Characteristics of included studies assessing human monoclonal antibodies targeting calcitonin gene-related peptide or its receptor for the prevention of chronic migraine with medication overuse headache.

aDropout rates implies withdrawal with any reason in the treatment group, unless otherwise noted.
bSubjects terminating due to the treatment emergent adverse events.
cThe reported data is from Silberstein et al., 2017 that included only chronic migraine but the study population is same (38).
DBTP: double-blind treatment phase; MD: migraine days.
The most common outcomes measured in the included studies were headache/migraine frequency (28–35), and ≥50% response rate (28–34), measured via headache diary. A study evaluating eptinezumab has also reported ≥75% response rate (31). Changing MO status to no MO was measured in all studies evaluating the efficiency of mABs (31–34), and in two studies evaluating BoNTA (28,30). A study evaluating BoNTA had also reported changing CM status to EM, and a success to withdraw from medication (28). Change in acute headache medication intake was measured in two BoNTA studies (29,30), and in all four mABs (32–34). However, one mABs study evaluating this outcome following eptinezumab treatment was reported in a separate report (37). All studies evaluating BoNTA, mABs, and topiramate provided data for adverse events, however, studies evaluating fremanezumab and galcanezumab presented adverse events in the separate report (38,39). Quality of life was measured in five studies (using SF-36 and MSQoL) (28,30,32,33,35), and assessment of disability was reported in six studies (using HIT-6 and MIDAS) (28–30,32,33,35).
Effects of interventions
Botulinum toxin type A versus placebo
Studies evaluating the effectiveness of BoNTA were heterogenous. Doses varied from 100 units to 195 units. Of the total three studies, individuals in one study had 16 intramuscular injections (29), and the other two had 31 injection sites with fixed-dose across seven specific head/neck muscle areas (28,30). To prevent unblinding, a placebo group in the study by Pijpers et al. (28) received 17.5 units of BoNTA in the forehead. Use of any prophylactic medication within four weeks before the start of baseline, and previous exposure to any BoNTA serotype was an exclusion criteria in all three studies. Subjects in Sandrini et al. (29) went through eight days of withdrawal therapy before a treatment with BoNTA, Pijpers et al. (28) instructed participants to withdraw any acute medications intake during the treatment period, while Silberstein et al. (29) provided no instructions regarding change in acute medication intake. The later study included opioid users and were carefully examined during the study.
A total of 1139 participants reported changes in monthly headache days and were included in the random-effect meta-analysis. All studies assessing the effect of BoNTA showed positive results regarding change in headache frequency from baseline compared to placebo (Figure 2). The combined effect size was also significant with MD of −1.92 (95% CI, −2.68 to −1.16). For achieving ≥50% response rate (≥50% reduction in headache days from baseline), two studies showed significant positive results (Figure 3). However, the overall combined effect of BoNTA studies achieving ≥50% response rate was non-significant (OR 1.56, 95% CI, 0.42 to 5.75). Statistically significant heterogeneity (I2) was identified for this outcome. Using GRADE, we downgraded the certainty of evidence by two levels for the outcome “monthly headache days” due to concerns over high risk of bias and imprecision (Table 3). For outcome “response rate”, we judged the certainty of evidence to be “very low” due to serious to very serious concerns regarding risk of bias, inconsistence, and imprecision.

Forest plot of comparison between botulinum toxin type A (BoNTA) versus placebo. Change in monthly headache days from baseline to post-treatment among patients with chronic migraine and medication overuse headache.
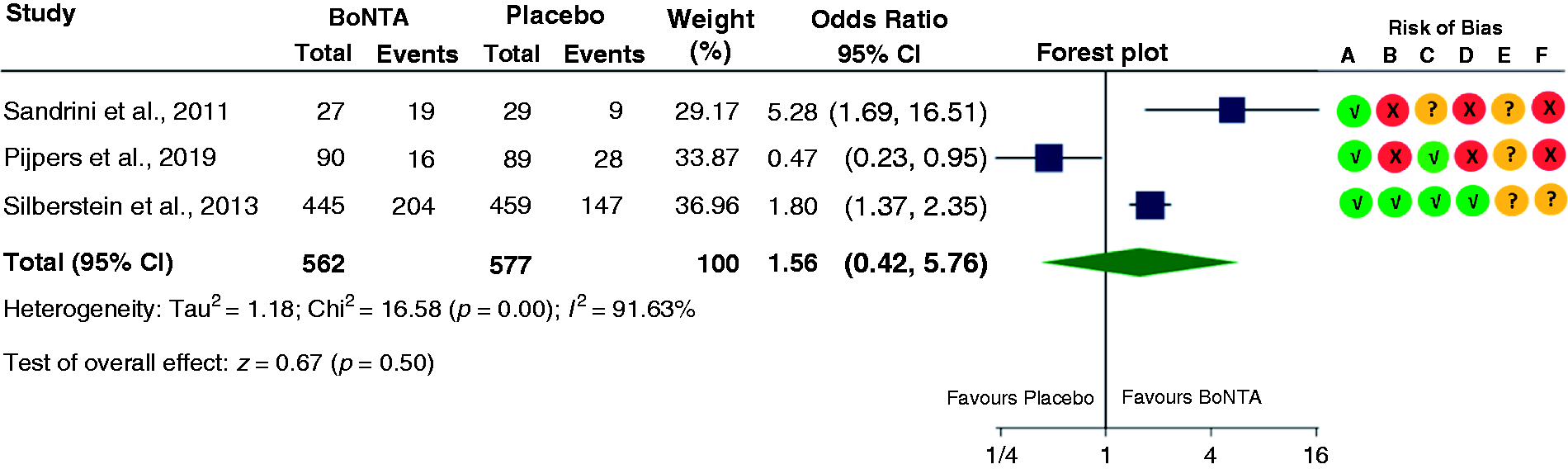
Forest plot of comparison between botulinum toxin type A (BoNTA) versus placebo. ≥50% reduction from baseline in headache days among patients with chronic migraine and medication overuse headache.
Summary of findings for the main comparison. BoNTA or mABs targeting CGRP(r) compared to placebo for migraine prevention in adults with CM and MOH.

GRADE Working Group grades of evidence.
bInconsistent directions of effect and considerable heterogeneity (I2 = 92%). Estimates were imprecise including overall estimate with wide confidence interval. Thus, the certainty of evidence was downgraded by 1 level.
cIndirect MOH population, MOH in three studies were defined with short duration for four weeks during baseline period. The certainty of evidence was downgraded by 1 level (downgraded by 2 levels in the outcome “change in medication overuse status”).
The overall adverse event (AE) rate and drop-out rate varied from 28.5% to 56.3% and 2.9% and 7.4% respectively. No significant differences in the frequency of AEs in BoNTA group were observed compared to placebo (Figure 4). The most common AEs reported were pain in injection site, hematoma at injection site and muscular weakness.
mABs targeting CGRP versus placebo
All four included studies evaluating mABs for the prevention of CM with MOH had three treatment arms (two arms with varying doses of intervention) and a placebo. Three studies had monthly subcutaneous injections, whereas the other had injections at day 0 and on week 12 (31). Dosing varied from 70 units to 1125 units. All studies excluded individuals who underwent treatment with BoNTA for at least four months before entering the baseline period. Three mABs studies allowed to take oral concomitant preventive medications (topiramate or propranolol) during the trial if use and dose were stable for two to three months (31,33,34). One of these studies allowed 30% of the patients to use additional preventive medication (33). The other study evaluating erenumab prohibited any preventive medications during the study and two months before the start of baseline (32). Patients were allowed to take acute headache medication as needed throughout the trial, but use of opioid and barbiturates was limited to no more than three to four days per month (31–34).
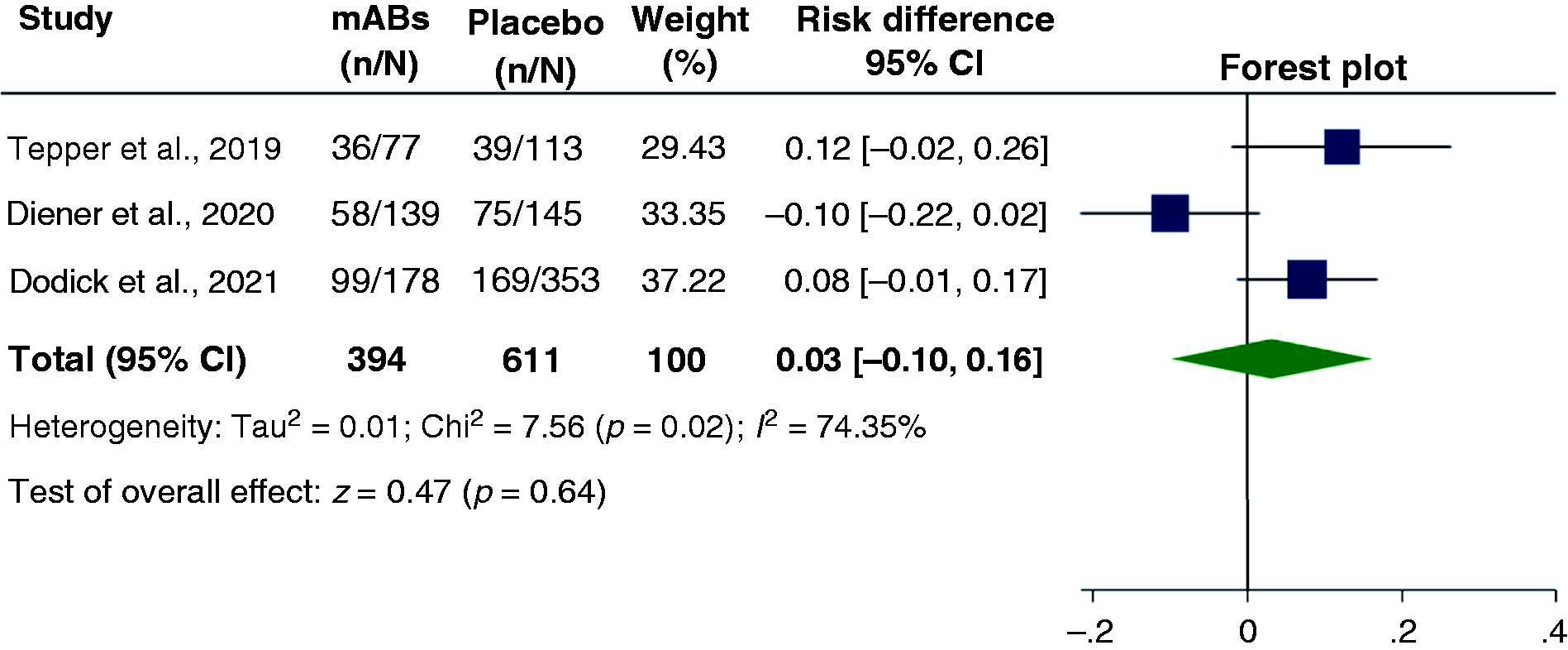
The outcomes change in monthly migraine days and ≥50% response rate (≥50% reduction in migraine days from baseline) was reported in all four mABs studies that included 1982 participants. Three studies reporting the outcome “change in MO status” was included in the meta-analysis with 1726 participants (31,33,34). Although all studies have two active intervention groups with varying doses of mABs, in the meta-analysis we included intervention group with higher doses only. All studies assessing mABs with a varying dose of 140 mg to 1125 mg showed significant positive effect compared to placebo. The combined effect size for mABs studies were −2.68 (95% CI, −3.46 to −1.91), 2.91 (95% CI, 2.23 to 3.78) and 2.09 (95% CI, 1.64 to 2.67) respectively for outcomes “change in monthly migraine days”, “≥50% response rate” and “change in MO status to no MO” (Figures 5 –7). Our judgments of the certainty of evidence using GRADE approach were “moderate” for the outcomes “change in monthly migraine days” and “response rate” due to concerns over MOH definition for short duration (Table 3). For the outcome “change in medication overuse status”, we downgraded the certainty of evidence by two levels due to indirectness in MOH patient’s definition, MOH was defined based on medication overuse duration for only four weeks in the majority of the studies.

Forest plot of comparison between botulinum toxin type A (BoNTA) versus placebo. Adverse events among patients with chronic migraine and medication overuse headache.

Forest plot of comparison between “human monoclonal antibodies (mABs) targeting calcitonin gene-related peptide or its receptor” versus placebo. Change in monthly migraine days from baseline to post-treatment among patients with chronic migraine and medication overuse headache.
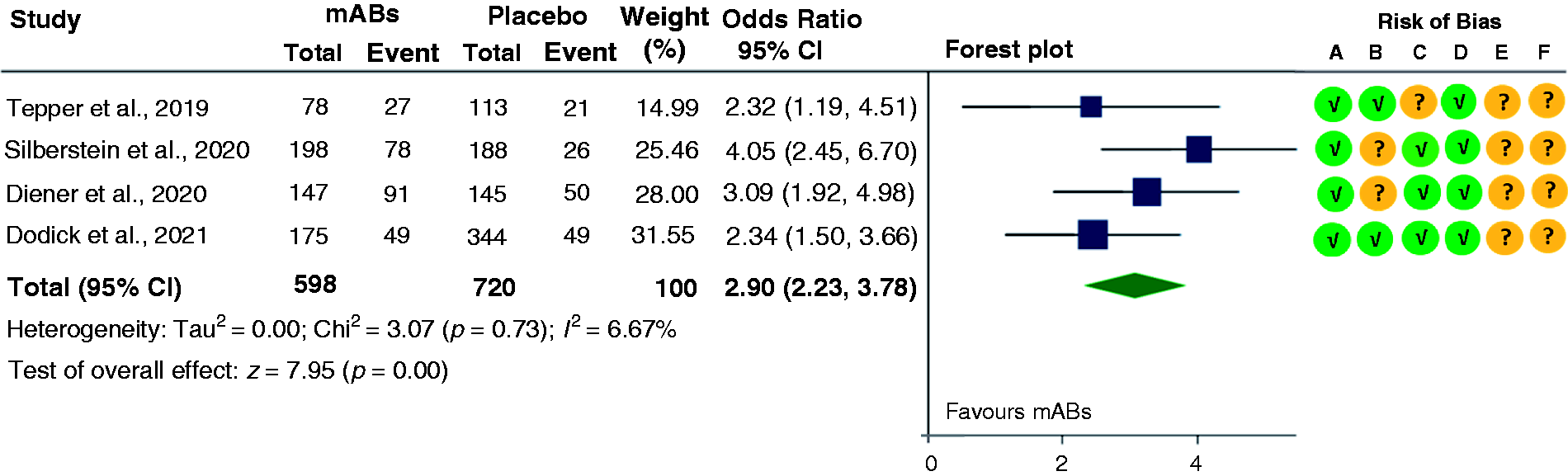
Forest plot of comparison between “human monoclonal antibodies (mABs) targeting calcitonin gene-related peptide or its receptor” versus placebo. ≥50% reduction from baseline in migraine days among patients with chronic migraine and medication overuse headache.

Forest plot of comparison between “human monoclonal antibodies (mABs) targeting calcitonin gene-related peptide or its receptor” versus placebo. Changing medication overuse status to no medication overuse among patients with chronic migraine and medication overuse headache.
The overall adverse event rate ranged from 43.3% to 68.3% and drop-out rate varied from 1.5% and 2.5%. Compared to placebo, no significant difference in the frequency of AEs was reported in the lower dose (70 mg to 120 mg) of mABs, whereas significant differences were observed in higher doses (140 mg to 300 mg) of mABs (Figures 8–9). The most common AEs includes injection site reaction such as pain, erythema, induration, hemorrhage, infections including nasopharyngitis, upper respiratory tract infection, sinusitis, urinary tract infection, muscle spasm, back pain, and fatigue.
Forest plot of comparison between “human monoclonal antibodies (mABs) targeting calcitonin gene-related peptide or its receptor (low dose)” versus placebo. Adverse events among patients with chronic migraine and medication overuse headache.
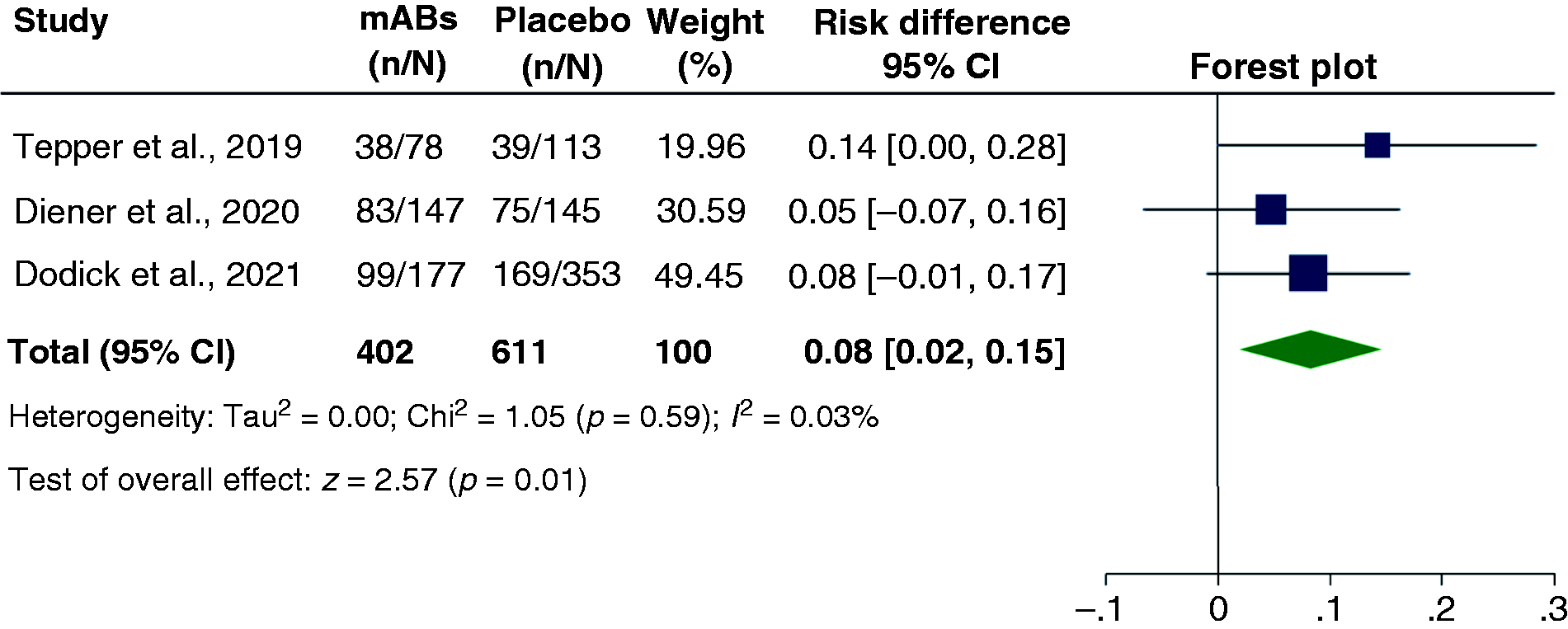
Forest plot of comparison between “human monoclonal antibodies (mABs) targeting calcitonin gene-related peptide or its receptor (high dose)” versus placebo. Adverse events among patients with chronic migraine and medication overuse headache.
Topiramate versus placebo
The publications by Diener et al. (35) and Mei et al. (36) reported a positive effect of topiramate in patients with CM and MOH. Both studies allowed a maximum dose of 100 mg per day. Diener et al. (35) reported a significant reduction in mean monthly migraine days by 3.5 ± 7.1 days in topiramate group compared to an increase of 0.8 ± 4.8 days in placebo. In this study, patients were allowed to take acute medication for rescue, and prophylactic medications were permitted with at least three months use and a stable dose (35). In Mei et al. (36) selected patients went through withdrawal therapy of other preventive treatments before the baseline phase, and reported a reduction in the number of headache days, response rate, and acute medication use (p < 0.0001) (36). Treatment emergent adverse events was observed in both studies varying from 30% to 75%. The most common AEs were paresthesia, nausea, dizziness, dyspepsia, fatigue, anorexia, taste alteration and disturbance in attention.
Risk of bias in included studies
Risk of bias assessments for each outcome, including five domain judgments of RoB2 tool is demonstrated at the right side the forest plots in Figures 2,3,5 –7 and published in the online Supplementary Material 3–4. Risk of bias reporting monthly headache days and response rate in the studies assessing efficiency of BoNTA varied from “some concerns” (Pijpers et al. [28]) to “high risk of bias” (Sandrini et al. [29] and Silberstein et al. (30)). Pipers et al. (28) and Silberstein et al. (30) had defined MOH based on four weeks evaluation during the baseline period. Sandrini et al. (29) had included MOH based on three months evaluation that was confirmed through personal communication with the author, but only patients with migraine without aura were included. The publication by Sandrini et al. (29) and Silberstein et al. (30) were suspected of a lack of blinding due to the fact that BoNTA injections can lead to visible differences in the frontal area of the treatment groups. In addition, 18% of the randomized patients in Sandrini et al. (29) have missing outcome data and were not included in the ITT analysis.
All studies evaluating efficiency of mABs in CM patients with MOH were post hoc analysis not described sufficiently in the original study protocol (31–34). Except for the outcome “changing medication overuse status”, risk of bias for outcomes across all studies assessing the efficacy of mABs was similar and predominately of “some concerns”. Risk of bias reporting the outcome “changing medication overuse status” varied from “some concerns” to “high risk of bias”. Three studies evaluating mABs targeting CGRPr (32–34) had defined MOH based on a four week baseline period only. In the publication by Tepper et al. (32) the definition of medication overuse changed over time and was not included in the analysis. As per personal communication with the author, the criteria for medication overuse for endline only requires monthly threshold of overuse, whereas stricter criteria were used during the randomization/enrollment. Thus, the total number of participants analyzed for this outcome was higher than randomized during the baseline period. A funnel plot to assess publication bias was not created for the outcomes for both BoNTA and mABs studies, given that there were less than 10 studies in the meta-analysis.
Discussion
This is the first systematic review to evaluate the relative effectiveness of topiramate, BoNTA and mABs targeting CGRP on CM patients with MOH. The meta-analysis of the included studies in this review illustrates that both BoNTA and mABs targeting CGRP are effective for the prevention of CM with MOH. Compared to placebo, treatment with BoNTA gave a reduction of almost two monthly headache days, whereas a reduction of 2.7 migraine days per month was found in patients treated with mABs. Nevertheless, the effectiveness of BoNTA should be interpreted with caution, since the results of this intervention are based on two reports with high risk of bias (28,29). Patients were almost three times as likely to have a ≥50% reduction in migraine days with mABs treatment compared to placebo.
BoNTA
The combined result for BoNTA in the current meta-analysis is consistent with the previous meta-analysis that evaluated the effect of BoNTA in patients with CM, showing a favorable outcome for 50% response rate with an OR of 1.51 (95% CI, 1.10 to 2.09) (40). All included studies reported reduction in headache frequency from baseline, but the results were insignificant in two studies with broad confidence intervals (Sandrini et al. [29] and Pijpers et al. [28]). The main source of uncertainty could be a consequence of small sample size. Furthermore, the estimates for achieving ≥50% response rates were inconsistent across the included studies. Pijpers et al. (28) has shown the estimates favoring placebo, most likely caused by the injection of 17.5 units of BoNTA in the frontal area of the placebo group. In addition, the inclusion of depression in this study may contribute to reduce the ≥50% response rate in the BoNTA and placebo group. The results in the current study must be interpreted with caution due to considerable differences in the MOH definition, intervention procedures, and inclusion and exclusion criteria identified. Dosing and injection procedure varied between the studies, subjects in two studies (Sandrini et al. [29] and Pijpers et al. [28]) received only one BoNTA treatment cycle while the other (Silberstein et al. [30]) received two treatment cycles with 12 weeks apart. Participants in Silberstein et al. (30) may possibly be benefited from a second dose of BoNTA. Nevertheless, no clinically significant serious adverse events were reported in any subjects, and clear trends were observed towards better performance of BoNTA treated patients for all considered secondary parameters including health related quality of life (HRQoL), HIT-6 scores, MIDAS scores, changes in medication overuse status (29,30).
mABs
For mABs targeting CGRP, all included trials demonstrated a clear statistically significant superiority for all measured primary outcomes. The overall estimates for ≥50% response rate was higher than the previous meta-analysis that reported only for CM patients (OR 2.36, 95% CI, 2.11 to 2.63) (40). Eptinezumab (Diener et al. (31)) benefited over placebo even in achieving 75% response rate (30% vs 15%). Fremanezumab (Silberstein et al. (33)) has the highest response rate, possibly an additional benefit of concomitant treatment with topiramate or propranolol and/or an effect of higher dosage. Although estimates for higher doses of mABs were calculated in the current meta-analysis, results for low doses also favored mABs compared to placebo (data not shown). The evidence for an outcome “changing MO status to no MO” needs to be interpreted with caution because the diagnosis of MOH was not defined according to the ICHD-3, but based on painkiller use during last four weeks only. In addition, patients with ICHD classification for opioid-overuse headache were excluded, thus inferences cannot be drawn about patients overusing these drugs. However, benefits of mABs were also evident from other outcomes reported in the included studies. Silberstein et al. (33) and Tepper et al. (32) had reported clinically relevant mean changes from baseline in MSQoL scores, HIT-6 scores and MIDAS total scores that were in favor of fremanezumab and erenumab. Dodick et al. (34) have shown that galcanezumab-treated patients transitioned from triptan overuse status to non-overuse at approximately twice the rate of placebo and switched from NSAID/aspirin overuse and multiple drug overuse to non-overuse status respectively at the rate of 30% and 50% greater than placebo. Treatment emergent adverse events in mABs were comparable to placebo, suggesting a good tolerability and safety. A limitation of the current data is that all studies evaluating mABs were post hoc analyses in a subset of MOH population. In post hoc analyses it may be more likely to use less rigorous method, non-representative subgroups, and to report positive result rather than negative results. Thus, the results need to be confirmed in well-designed randomized controlled studies preplanned for evaluating MOH patients.
Evidence from previous reports applying traditional withdrawal therapy in MOH patients demonstrated a high non-compliance rate with early discontinuation, and that patients could develop more severe symptoms including headaches, vomiting, nausea, anxiety, restlessness and sleep disturbances (41,42). None of the studies evaluating mABs applied withdrawal therapy before or during the treatment period, and patients were allowed to take acute headache medication as needed throughout the trial. The use of opioid and barbiturates, however, was limited to no more than three to four days per month. Thus, the results from the mABs studies indicate that patients can experience the benefit of mABs without discontinuation of overuse drugs and achieve reversion from MO to no MO. For studies evaluating BoNTA, intervention with withdrawal therapy varied. Treatment with withdrawal therapy was likely to be an effective approach to treat MOH as shown in Sandrini et al. (29) and Pijpers et al. (28) that showed improvements in primary outcomes in placebo treated patients. Silberstein et al. argues that patients with CM and MOH without a withdrawal therapy can be effectively treated with BoNTA. However, the level of evidence to support withdrawal therapy is low due to inadequate power, high loss in follow up and high risk of bias (Sandrini et al. [29] and Pijpers et al. [28]).
Overall completeness and applicability of evidence
This is the first review to include only randomized controlled trials to evaluate the effectiveness of BoNTA, mABs and topiramate in CM patients with MOH. All studies were published in the last 11 years. The findings of this review are externally valid and clinically relevant. Only one study was a single center study, and one had a small sample size. Studies reporting data on BoNTA were of generally poor quality, thus we are uncertain about the estimate for the outcomes reported. The quality of evidence was low to moderate for studies evaluating mABs. Thus, further research is very likely to have an important impact on our confidence in the estimate of effect. Studies assessing topiramate for the prevention of CM with MOH was scarce and of insufficient quality, thus they were not included in the meta-analysis. Topiramate should be further investigated among MOH patients with well-designed trials.
Quality of evidence and potential biases
Overall, the general limitation in the studies was the definition of MOH for the short duration of four weeks, and majority of the studies were a post hoc analysis. We have identified that only few studies had included MOH population based on three months of evaluation. Thus, we have included MOH populations based on both short- and long-term definition, and this alterations to our inclusion criteria from the published protocol is described in the method section as well.
Using the GRADE criteria, we assessed the quality of evidence as ranging from low to very low for all the outcomes measured for BoNTA. For studies evaluating mABs, the quality ranged from low to moderate for all measured outcomes. For BoNTA studies, we downgraded the certainty of evidence for monthly headache days to low due to imprecision (low number of participants in two studies), high risk of bias (lack of blinding, high dropouts), and indirect population (MOH defined for short duration). Certainty of evidence for response rate was downgraded by three levels to very low due to an inconsistent direction of effect, considerable heterogeneity (I2 = 91.63%), and imprecision. For studies evaluating mABs, we downgraded the certainty of evidence for monthly migraine days and response rate to moderate. This was due to an indirect population (definition of MOH for short duration), we therefore downgraded the certainty of evidence by one level. However, for the same reason we downgraded the level of evidence by two levels for the outcome “change in medication overuse status”. No relevant publication bias was detected for both medications.
Conclusion
Both BoNTA and mABs showed greater response rate over placebo in reducing number of headache or migraine days. Patient treated with mABs showed the greatest effect size and were almost three times as likely to have a ≥50% reduction in migraine days from baseline. The dropout rates were somewhat lower in the mABs groups compared to BoNTA, and no significant treatment emergent adverse events were identified in either of BoNTA or mABs groups compared to placebo. Only seven studies with sufficient data were included in this meta-analysis, and the quality ranged from very low to moderate. Thus, based on this meta-analysis, firm conclusions regarding treatment strategies for patient with CM and MOH could not be done. However, the present meta-analysis indicated that such patients may benefit of treatment with mABs and probably also BoNTA without needing withdrawal before or during treatment. There is insufficient data to evaluate the impact of topiramate in CM patients with MOH, thus this requires high-quality randomized trials in the future.
Clinical implications
Treatment with botulinum toxin type A and monoclonal antibodies against calcitonin gene-related peptide reduced monthly migraine days in chronic migraine patients with medication overuse headache, whereas ≥50% response rate was significant only for monoclonal antibodies. The frequency of adverse effect was similar compared to placebo for both botulinum toxin type A and low dose of monoclonal antibodies. The dropout rate was low and varied from 1.5% and 2.5% in monoclonal antibodies. Evidence to recommend topiramate for chronic migraine patients with medication overuse headache is inadequate.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231156922 - Supplemental material for Randomized controlled studies evaluating Topiramate, Botulinum toxin type A, and mABs targeting CGRP in patients with chronic migraine and medication overuse headache: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-cep-10.1177_03331024231156922 for Randomized controlled studies evaluating Topiramate, Botulinum toxin type A, and mABs targeting CGRP in patients with chronic migraine and medication overuse headache: A systematic review and meta-analysis by Samita Giri, Erling Tronvik, Mattias Linde, Sindre Andre Pedersen and Knut Hagen in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024231156922 - Supplemental material for Randomized controlled studies evaluating Topiramate, Botulinum toxin type A, and mABs targeting CGRP in patients with chronic migraine and medication overuse headache: A systematic review and meta-analysis
Supplemental material, sj-pdf-2-cep-10.1177_03331024231156922 for Randomized controlled studies evaluating Topiramate, Botulinum toxin type A, and mABs targeting CGRP in patients with chronic migraine and medication overuse headache: A systematic review and meta-analysis by Samita Giri, Erling Tronvik, Mattias Linde, Sindre Andre Pedersen and Knut Hagen in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
