Abstract
It is a general belief that patients with medication overuse headache (MOH) need withdrawal of acute headache medication before they respond to prophylactic medication. In this 1-year open-labelled, multicentre study intention-to-treat analyses were performed on 56 patients with MOH. These were randomly assigned to receive prophylactic treatment from the start without detoxification, undergo a standard out-patient detoxification programme without prophylactic treatment from the start, or no specific treatment (5-month follow-up). The primary outcome measure, change in headache days per month, did not differ significantly between groups. However, the prophylaxis group had the greatest decrease in headache days compared with baseline, and also a significantly more pronounced reduction in total headache index (headache days/month x headache intensity x headache hours) at months 3 (P = 0.003) and 12 (P = 0.017) compared with the withdrawal group. At month 12, 53% of patients in the prophylaxis group had ≥ 50% reduction in monthly headache days compared with 25% in the withdrawal group (P = 0.081). Early introduction of preventive treatment without a previous detoxification programme reduced total headache suffering more effectively compared with abrupt withdrawal. (ClinicalTrials.gov number, NCT00159588).
Introduction
Medication overuse headache (MOH) is believed to affect 1% of the adult general population worldwide (1–6), and approximately one out of four patients referred to a neurologist for headache suffers from this condition (7). To fulfil the diagnostic criteria of MOH the patients should have ≥ 15 headache days/month, regular overuse of acute headache medication for at least 3 months, and the headache should have developed or markedly worsened during medication overuse (8).
It is generally accepted that the treatment of choice in patients with MOH is discontinuation of the overused drug(s). Previous studies have not evaluated the long-term course of patients with MOH without specific treatment, i.e. these studies have not included an adequate control group. It has been a general belief that patients with MOH rarely respond to preventative medications while overusing acute medication, which is also stated in the International Classification of Headache Disorders, 2nd edn (ICHD-II) and a revised version of these specific criteria (9, 10). However, many headache experts introduce prophylactic treatment early in the management of MOH (11), but randomized studies evaluating the effect of early start of prophylactic medication are lacking.
The aim of this randomized, prospective, multicentre study was to evaluate the effect of early introduction of prophylactic treatment compared with abrupt withdrawal and with a control group in patients with MOH.
Methods
An open-label, randomized, multicentre trial was conducted over a 4-year period, and the study was approved by the Regional Committee for Ethics in Medical Research, the Norwegian Data Inspectorate, the Norwegian Medicines Agency, and was registered at ClinicalTrials.gov before 13 September 2005 (ClinicalTrials.gov number, NCT00159588). Each patient signed a written informed consent. The study was conducted in accordance with Good Clinical Practice (12). An audit by the Norwegian Data Inspectorate evaluated the way data were recorded and stored. Data from the clinical report forms and headache diaries were entered into a raw data file by a study nurse (G.G.) who did not participate in the data analyses, and this raw data file was saved unchanged in case of an external audit of the study result.
Patients
From 1 January 2004 to 31 December 2006 patients from the out-patient clinics of the neurological departments at five University Hospitals in Norway were included. Patients with suspected MOH according to the physician referral letter were mailed a headache diary to be completed for at least 3 months before attending the first visit. This period served as a baseline.
The inclusion criteria were: (i) age between 18 and 70 years, (ii) MOH defined as headache ≥ 15 days/month for at least 3 months combined with intake of ergots, triptans, opioids and/or combination medication (simple analgesics combined with caffeine) for ≥ 10 days/month, or of simple analgesics ≥ 15 days for a minimum of 3 months (7). Patients were excluded if (i) there were contraindications for all types of prophylactic drugs, (ii) there had been no improvement of the headache at previous trials to stop overused medication for at least 3 weeks, (iii) the patient had a history of hemicrania continua, chronic paroxysmal hemicrania or cluster headache, (iv) the patient used analgesics frequently for other complaints than headache, or (v) the patient was pregnant, breastfeeding or not using effective contraception.
Randomization and treatment
Computer-generated randomization with randomly permuted blocks stratified by centre and gender was used, administrated by the Unit for Applied Clinical Research at the Norwegian University of Science and Technology, Trondheim, Norway. Patients recruited from St Olav's Hospital were stratified by primary diagnosis because we expected less favourable prognoses for individuals with tension-type headache (TTH) than for those with migraine.
All groups used the same headache diary throughout the study, and all received at the start the same written general information about MOH, informing them among other things that withdrawal of acute attack medication was the standard treatment. The patients were randomly assigned to three different groups.
The withdrawal group underwent a standard out-patient detoxification programme consisting of: (i) advice to withdraw abruptly the overused medication, (ii) written information about potential withdrawal symptoms and (iii) telephone call from the doctor after 2 weeks. If necessary, they were: (i) allowed to use rescue medication up to 2 days/week [10–25 mg amitriptyline (if lack of sleep), 50 mg diclofenac or 500 mg naproxen orally, and/or 20 mg metoclopramide] (16); (ii) offered a sick leave for up to 2 weeks; (iii) offered in-patient detoxification if they failed to complete the out-patient detoxification programme; and (iv) offered to start with preventive treatment after 3 months.
The prophylaxis group was not explicitly advised to withdraw the overused medication. Preventive treatment was started on day 1, based on a priority list of medicines. For each patient the preventive agent was chosen on the basis of a list of specifications, taking into consideration the primary headache diagnosis, the drug's side-effect profile, coexisting conditions and, in addition, the patient's preferences and what they had tried before (Table 1).
Priority list of preventive medication∗ for different primary headache diagnoses

∗Topiramate was not approved by the Norwegian Medicines Agency for migraine prophylaxis when this study was planned and approved.
The control group did not get a new preventive medication or direct advice to stop using analgesics or medication prescribed by their physician. The controls finished the study period after 5 months’ observation, but were then offered the treatment considered to be optimal for them (withdrawal or prophylactic treatment) and further follow-up visits.
After inclusion, patients in all groups underwent blood screening tests and brain imaging (computed tomography or magnetic resonance imaging) for those who had not undergone imaging during the past 2 years.
Study procedure
Follow-up visits were scheduled at months 1, 3, 5 and 12 after inclusion, and a telephone call was scheduled at month 2. A telephone call from the doctor was also scheduled after 2 weeks for patients in the withdrawal group.
At inclusion, at month 5 and at month 12 (only the withdrawal group and the prophylaxis group) the patients were asked to fill in the Hospital Anxiety and Depression Scale (HADS) and a health-related quality of life-questionnaire (HRQoL) using the Short Form (SF)-12 (13, 14). The SF-12 measures HRQoL in two main domains, a mental health component score (MCS-12) and a physical health component score (PCS-12).
Headache diary
In the headache diary, which was applied both during the baseline period and after randomization, patients recorded daily whether they had headache or not. They were asked to record the duration of headache when awake, if they had nausea, the brand name and number of doses taken, and absence from work. The headache severity was scored on a three-point scale from 1 to 3, explained in the diary as: 1, mild (does not inhibit work or other activities); 2, moderate (inhibits but does not exclude work or other activities); 3, severe (excludes work or other activities).
Some patients did not indicate headache duration in hours, but used descriptions. ‘The whole day’ was stipulated to be 16 h, whereas ‘morning’, ‘lunchtime’, ‘afternoon’ and ‘evening’ were stipulated as 4 h each. A headache day was defined as a day with headache lasting > 30 min.
Outcome measures
The primary outcome measures were the change in number of headache days, compared with the baseline period, during month 3 and month 5 (all groups), and month 12 (the withdrawal group and prophylaxis group only). Change at month 3 was the major primary outcome measure because up to this point the treatment differed markedly between the groups. After 3 months the withdrawal group could receive preventive treatment if necessary.
The secondary outcome measures included: change from baseline in days with use of acute headache medication per month, change from baseline in headache hours, change from baseline in ‘headache index’ (HI) per month calculated by the sum of the products of ‘headache days/month’ combined with ‘mean daily hours with headache’ and ‘mean daily headache severity’ on days with headache, change from baseline in sick leave days per month, change from baseline in anxiety and depression measured by HADS, and change from baseline in SF-12 measured by the two main domains MCS-12 and PCS-12. In addition, responder rates in the three groups were compared. A responder was defined as being without medication overuse and with ≥ 50% reduction in monthly headache days compared with baseline.
Power calculation
The main efficacy variable was the number of headache days per month. In a previous Swedish study the mean number of headache days/month at inclusion was 26.9 with a standard deviation (
Statistics
Non-parametric tests were used for comparisons between groups (Kruskal–Wallis test, Mann–Whitney U-test and χ2 test) and within groups (Wilcoxon test) because of skewed distribution of data. If statistical significance at the 0.05 level using Kruskal–Wallis test was achieved, Bonferroni correction was used for post hoc comparisons of pairs of groups (P < 0.017). Because Bonferroni-type corrections can be associated with type II errors, and because the Bonferroni adjustment, even when applicable, is too conservative when variables co-vary, no further adjustments were made for multiple comparisons (16, 17).
The efficacy analysis was carried out for those who met the inclusion criteria and had at least one post-baseline observation (at least 1 month's follow-up) (intension to treat population). For drop-outs after this stage, missing data were filled in using the method ‘last post-randomized measurement carried forward’. Analyses were carried out using
Results
Three out of 64 randomized patients were excluded because they had had < 15 headache days/month at baseline (Fig. 1). Of the 61 who met the inclusion criteria, five did not deliver a post-baseline headache diary and were excluded from the efficacy analyses. A total of 13 patients (21%) did not complete the study according to the protocol (Fig. 1). The drop-out rate tended to be somewhat higher for patients in the withdrawal group. At month 5 it was 18% in the withdrawal group (compared with 10% in the two other groups, P = 0.38), and after 12 months it was 36% (compared with 16% in the prophylaxis group, P = 0.14). During the study period the drop-out rate was lower among patients included at St Olav's Hospital (10%) than for those included at the four other centres (47%) (P = 0.001). The reasons given for dropping out were: did not tolerate abrupt withdrawal (two in the withdrawal group), worsening of gonarthritis when not using non-steroidal anti-inflammatory drugs (one in the withdrawal group), suicide (one in the withdrawal group, but not related to headache according to the patient's general practitioner), hospitalized for other reasons (one in the prophylaxis group), successful withdrawal that gave few headache attacks (one control who himself decided to withdraw), and no specific reason given (four in the withdrawal group, two in the prophylaxis group, and one control).

Study flow chart.
Table 2 presents the demographic and clinical characteristics of the remaining 56 subjects who were randomized to the three different groups. No significant differences emerged between the three groups with regard to sociodemographic variables, headache type, number of headache days per month, number of analgesic days per month or type of overused medication.
Study population characteristics at baseline

∗Chi-squared test.
†Krusal–Wallis test.
HDs, headache days; IH, intensity of headache; HHs, headache hours; HADS, Hospital Anxiety and Depression Scale; MCS, mental health component score; PCS, physical health component score.
None of the patients in the withdrawal group was detoxified as an in-patient. At the 3-month follow-up, individuals in the withdrawal group were offered prophylactic treatment, and 13 out of 18 individuals (72%) who attended the 3-month visit accepted. Of the five subjects who did not start with preventive medication, three improved considerably during the withdrawal period and did not need preventive medication, and two did not want it.
Among the 30 individuals (13 in the withdrawal group and 17 in the prophylaxis group) who received prophylactic treatment, 18 tried one type during the follow-up, five tried two types, whereas the remaining seven subjects tried three to five types due to either intolerance or lack of effect of the first attempts. During the follow-up a total of 53 attempts with preventive medication were performed; angiotensin II blocker (candesartan) was tried 17 times, amitriptyline 15 times, gabapentin 10 times, valproate seven times and β-blockers four times. At the end of follow-up, 24 participants were on medical preventive treatment, the most common drug was angiotensin II blocker (n = 9), followed by amitriptyline (n = 8), gabapentin (n = 3), valproate (n = 3) and a β-blocker (n = 1).
Headache days
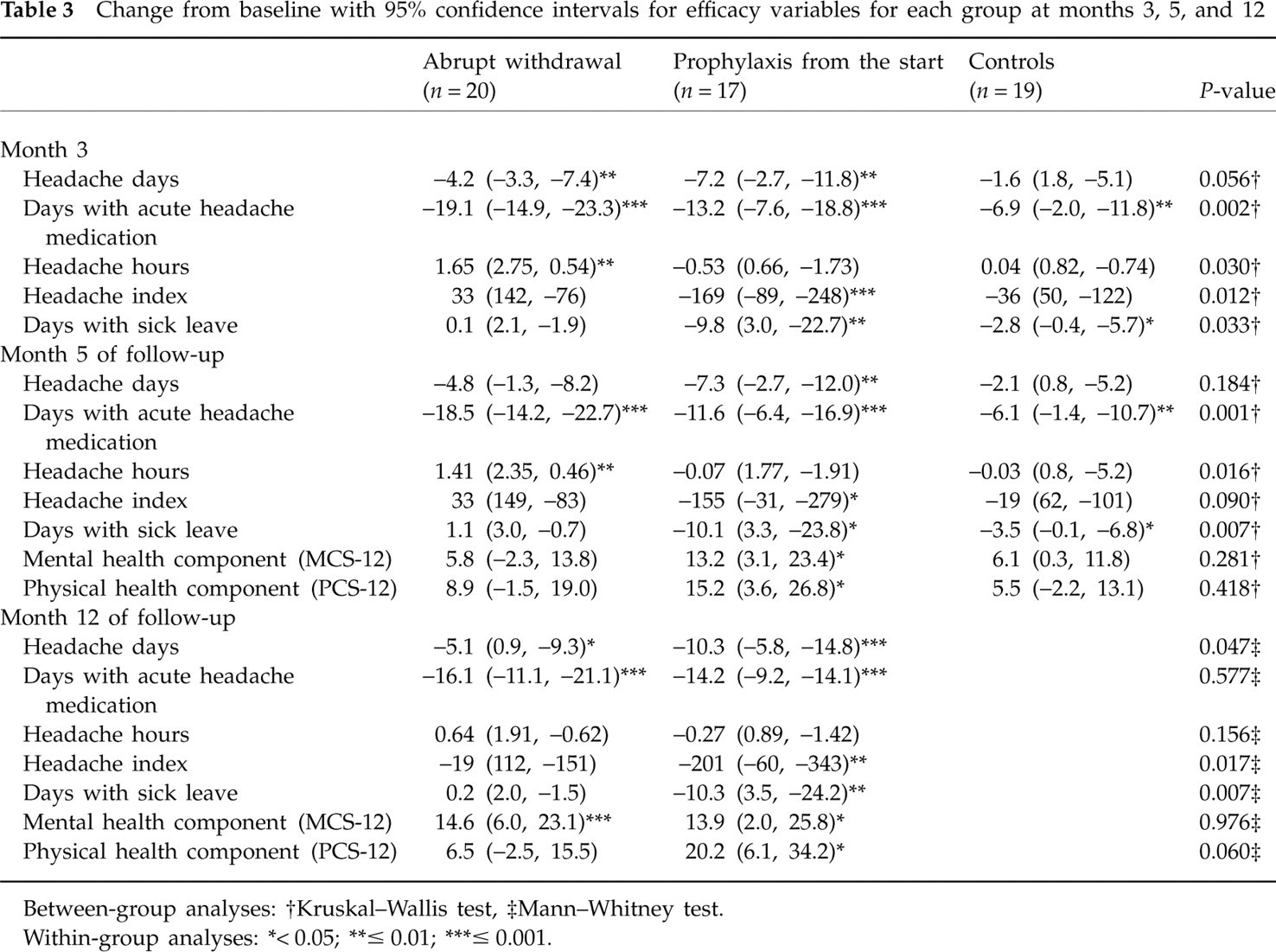
The prophylaxis group had the most consistent reduction in headache days/month compared with baseline (Fig. 2), being 7.2 days at month 3 (P = 0.002), 7.3 at month 5 (P = 0.009) and 10.3 at month 12 (P = 0.001). The corresponding figures for the withdrawal group were 4.1 (P = 0.005), 4.7 (P = 0.064) and 5.1 (P = 0.035), whereas for controls the reduction in mean headache days/month was 1.6 at month 3 (P = 0.46) and 2.1 at month 5 (P = 0.41). Change in headache days per month did not differ significantly between groups during the follow-up (Table 3).
Change from baseline with 95% confidence intervals for efficacy variables for each group at months 3, 5, and 12
Between-group analyses:
†Kruskal–Wallis test,
‡Mann–Whitney test.
Within-group analyses:
∗< 0.05;
∗∗≤ 0.01;
∗∗∗≤ 0.001.

Headache days per month.
Overall, the reduction in monthly headache days was most pronounced for the 39 individuals who had tried none or only one prophylactic drug before inclusion in the study compared with 17 patients who had tried two or more, e.g. at month 5, respectively 6.1 days and 1.3 days (P = 0.022). This was most pronounced for the prophylaxis group, with a reduction in monthly headache days at the end of follow-up of 12.6 days for individuals who had tried fewer than two preventive drugs at baseline compared with 2.7 days for those who had tried two or more (P = 0.032).
The patients who were considered to have had ‘pure’ migraine before they developed MOH had fewer monthly headache days at baseline than those with TTH or mixed headache (20.7 days vs. 27.6 days, P < 0.001). However, the headache type did not influence prognosis (change in monthly headache days from baseline) in the three different groups.
Medication intake
All three groups had a significant (P < 0.009) reduction in days with use of acute headache medication per month compared with baseline during the follow-up (Fig. 3). Change in acute headache medication per month was significantly more pronounced in the withdrawal group compared with controls at months 3 and 5 (P < 0.001), whereas pairwise comparisons between controls and the prophylaxis group, and between the prophylaxis and withdrawal groups did not achieve Bonferroni-adjusted significance level at months 3 and 5 (P ≥ 0.04). Among the controls, five out of 19 (26%) actively performed withdrawal of acute attack medication on their own.

Days with use of acute headache medication per month.
Headache hours
The withdrawal group had a significant (P < 0.009) increase in headache hours compared with baseline at months 3 and 5 (Table 3). At month 5 the withdrawal group had an increase of 1.4 headache hours, whereas the prophylaxis group had a decrease of 0.1 h (P = 0.017).
Headache index
Regarding change in HI/month from baseline, the prophylaxis group had a significantly (P = 0.003) more pronounced reduction at month 3 compared with the withdrawal group (Table 3). As demonstrated by Fig. 4, the withdrawal group had an increase in HI/month during the first 5 months of follow-up compared with baseline. At month 12 the prophylaxis group had a reduction of HI with an average of 201 per month (41% reduction from baseline), whereas the withdrawal group had a decrease of only 20 (5% reduction from baseline) (P = 0.017).

Headache index (HI) per month.
Disability
The prophylaxis group had the most prominent reduction in sick leave days/month compared with baseline, being 9.8 days at month 3 (P = 0.008), 10.0 days at month 5 (P = 0.028) and 10.3 days at month 12 (P = 0.008). During the whole follow-up, the number of days with sick leave/month was higher in the withdrawal group than in the prophylaxis group at months 3 (P = 0.016), 5 (P = 0.007) and 12 (P = 0.007) (Table 3).
The prophylaxis group had also the most prominent increase in physical health component score (PCS-12) compared with baseline: 15.2 at month 5 (P = 0.018) and 20.2 at month 12 (P = 0.012). The corresponding figures for the MCS-12 were 13.2 at month 5 (P = 0.024) and 13.9 at month 12 (P = 0.039) (Table 3). The withdrawal group had a significant increase in MCS-12 at month 12, being 14.6 (P = 0.001) (Table 3). No significant difference was found between the groups regarding PCS-12 and MCS-12.
At month 12 the prophylaxis group had a decrease in total anxiety and depression scores measured by HADS of 2.9 compared with baseline (P = 0.049), whereas the withdrawal group had an increase of 0.6 (group comparison: P = 0.058).
Responders
The proportion of responders without medication overuse combined with ≥ 50% reduction in monthly headache days tended to be highest for the prophylaxis group during the follow-up, being significantly higher at month 5 for prophylaxis group than for the controls (41% vs. 5%, P = 0.010, Fig. 5). At month 12, 53% of the patients in the prophylaxis group had ≥ 50% reduction in monthly headache days compared with 25% in the withdrawal group (P = 0.081, Fig. 5).

Responder rates (responder defined as being without medication overuse combined with 50% reduction in headache days per month). ∗P = 0.010.
Change in headache diagnosis
The results of blood screening tests and brain imaging had no influence on the suggested headache diagnosis. Individuals with unclassified headache at the first screening visit were all classified as having an original migraine, TTH, or migraine coexisting with TTH during the follow-up visits. After the end of the follow-up period, the headache diagnosis was revised in one case because the headache did not improve despite withdrawal of analgesics (from MOH to cervicogenic headache).
Discussion
The main finding in this randomized, open-label, 1-year follow-up study of patients with MOH was that early introduction of prophylactic medication without a detoxification programme was an effective way to reduce headache days and total headache burden during the first 3 months, and the improvement was sustained during the whole follow-up period. The effect was most pronounced in individuals who had previously tried no more than one type of preventive medication.
The optimal way to evaluate new treatment strategies is to perform a randomized, double-blind, placebo-controlled trial, but so far no blinded placebo-controlled trials involving withdrawal therapy among patients with MOH have been performed. To the best of our knowledge, this is the first randomized study evaluating the efficacy of early introduction of preventive medication compared with the traditional withdrawal therapy. Although we could not include as many patients as intended, power calculations indicate that the study should be able to detect a difference in treatment effect between the groups of clinical interest.
The reduction in days with use of acute headache medication per month was most pronounced in the withdrawal group, but the group who received prophylactic treatment at the start also had > 50% reduction of days with acute headache medication during the first 3 months. The prophylaxis group was not explicitly advised to withdraw the overused medication. However, the general written information stated that abrupt withdrawal was the standard treatment of MOH. This information may have influenced both patients in the prophylaxis group and controls to be more restrictive in their use of acute attack medication. This may explain why 26% of the controls markedly reduced intake of acute medication on their own. However, we consider it likely that the reduction of days with use of acute medication among the prophylaxis group was due, at least in part, to an overall reduction of headache days/month caused by the prophylactic medicines and not only to an active effort to reduce the acute medication. Thus, our results do not support the tenet that patients with MOH rarely respond to prophylactic medications while overusing acute attack medication. This tenet has also been challenged in a recent double-blinded, placebo-controlled trial evaluating the efficacy of topiramate among patients with chronic migraine, of whom the majority had medication overuse (18). In this 16-week trial, topiramate significantly reduced the mean number of monthly migraine days compared with placebo, despite the fact that use of acute medication did not differ between the groups (18).
The spontaneous long-term course of MOH is mostly unknown, and no previous prospective studies have included a control group receiving no active treatment or strong advice of withdrawal of the overused medication. One may argue that our control group was not totally unaffected by the intervention, since investigation by a neurologist and participation in a MOH trial (including general information about the condition) is also an intervention. Because 26% of the controls tapered off the overuse of medication on their own, information alone about the negative effects of medication overuse seems to have an impact on the overuse in some individuals. Previously it has been shown that advice alone gives a similar success rate to a standardized detoxification programme in a 2-month follow-up of patients with the combination of migraine and MOH (19). Conceivably, the spontaneous long-time course of MOH could be demonstrated in double-blinded placebo-controlled trials including patients with chronic headache with medication overuse, but to date such patients have been excluded from most trials evaluating the efficacy of prophylactic treatment. So far, the placebo response in a few studies evaluating the efficacy of topiramate among migraine patients with mediation-overuse has been inconsistent. No change in migraine days or days with intake of acute medication compared with the 4 weeks’ baseline period was reported in the placebo group during a 16-week follow-up (18). In another study the placebo group of 14 subjects had a mean decrease of headache days per month from 24.5 to 15.4, with a corresponding reduction in amount of acute medication taken monthly from 30.8 to 15.4 during a 12-week follow-up (20). To make valid conclusions about the spontaneous long-term course of MOH more double-blinded, placebo-controlled studies are needed on patients with medication overuse combined with chronic headache (TTH included) evaluating prophylactic drugs, or prospective studies without intervention.
During the first 3 months only 15% of the individuals in the withdrawal group had a > 50% reduction in headache frequency. In contrast, the corresponding success rate of withdrawal therapy has been reported to be > 70% in several previous studies (1). However, almost all previous studies have been based on migraine patients, and lack of information about drop-outs and lack of patients with TTH in these studies make direct comparisons with the present study difficult. The relatively low response rate is similar to that in a recently published Danish study reporting that only 45% of those with MOH had at least 1% reduction in headache frequency after a 2-month drug-free period (11). The low response rate in the Danish study was in part caused by the fact that individuals with TTH had a median relative reduction in headache frequency of 0%. In the present study, > 30% of the participants had TTH, and this may in part explain the rather low response rate. Based on the results of the Danish study and the present study, it is reasonable to assume that definite MOH is less prevalent than previously thought. In the withdrawal group of the present study 50% had not improved at the 3-month visit and hence did not fulfil the original criteria for definite MOH (9). However, the prevalence of definite MOH is influenced by the way a responder is defined. There is no universally accepted definition of a ‘responder’ for studies evaluating treatment effect of patients with MOH. In the D criterion of MOH it is stated that ‘headache reverts to its previous pattern within 2 months after discontinuation of the analgesics’ (9). This criterion is difficult to incorporate in a study definition of a ‘responder’, because reliable data regarding the previous headache pattern are usually lacking. According to the ICHD-II criteria the effect of withdrawal therapy shall be evaluated after 2 months, and this is also recommended by some authors (11). However, there is little evidence to support a 2-month withdrawal as being necessary or sufficient. In the present study the withdrawal group had to wait 3 months before preventive medication was considered to be absolutely certain that withdrawal had been tried sufficiently long. As indicated by Fig. 2, the headache frequency tended to decrease from month 2 to month 3, probably illustrating that the 2-month limit mentioned in the original ICHD-II criteria was somewhat arbitrary. During the first 3 months none of the patients in the withdrawal group started to overuse attack medication.
Until now, abrupt withdrawal of acute attack medication has been the treatment of choice in patients with MOH. However, the most common problems with such a strategy are the withdrawal headache and that many patients are not able to complete the recommended drug-free period. In the present study these problems were reflected in the somewhat higher overall drop-out rate in this group (36% vs. 16% in the prophylaxis group) and in the increase in overall suffering (high HI in the withdrawal group). Similarly, 36% of patients with MOH were not able to be drug-free for a 2-month period in the Danish study (11). Problems with withdrawal headache and high drop-out rates probably explain why many headache experts are reluctant to recommend a long drug-free period before they introduce prophylactic treatment in the management of MOH (11).
During the whole study period it was evident that those who received preventive medication from day 1 had the lowest number of headache days, days with sick leave, HI/month and the highest response rate compared with the two other groups. The results from the present study do not support the tenet that patients with MOH rarely respond to prophylactic medications while overusing acute medication. On the contrary, it indicates that early introduction of preventive medication is an attractive alternative to abrupt withdrawal of acute attack medication. In the Danish study, 55% of those who managed to stop use of acute medication had no change in headache frequency or got worse (21). In the follow-up of these, a 26% reduction in headache frequency was found, at least in part explained by use of preventive medication. We suggest that some of these individuals would have improved earlier if preventive medication had been started before. In our study the group who received preventive medication from the start seemed to have a ‘flying start’, which remained during the whole study period and which reduced the total burden of headache and lowered the drop-out risk. In contrast, the withdrawal group continued to have a high HI during the whole 12-month follow-up. Because headache days/month decrease with 5.1 days, the lack of decrease in HI was caused by more headache hours combined with higher headache intensity, probably because of the more restrictive use of pain killers than in the group receiving preventive medication from the start.
The study has also demonstrated a significant reduction of headache days per month among patients in the withdrawal group during the first 3-month withdrawal period. The optimal way to treat these patients may therefore be to recommend withdrawal or reduction of analgesics, together with early introduction of prophylactic medication.
In conclusion, this randomized, multicentre, 1-year follow-up trial has shown that prophylactic treatment from the start without abrupt detoxification was superior to abrupt withdrawal in reducing total headache burden. Regarding the decrease in headache days compared with baseline, our study has also demonstrated a significant effect of withdrawal during the first 3-month withdrawal period, but it has also indicated that abrupt withdrawal may not be necessary before starting with prophylactic medications. The notion that patients with MOH need withdrawal of acute medication in order to respond to prophylactic medication seems to be incorrect. In future studies on treatment of MOH it may be advisable to have a group with both prophylactic treatment and withdrawal of acute medication from the start.
