Abstract
Background
Migraine is a complex neurological disorder involving generalized abnormalities in processing sensory information. Adopting evidence that central sensitization imposes major hurdles in the treatment of migraine, we hypothesized that it is the non-ictal (rather than ictal) allodynia that may determine the outcome of migraine prevention with peripherally-acting drugs.
Methods
To test this hypothesis, we used Quantitative Sensory Testing to determine whether it is possible to identify a patient’s response to prophylactic treatment with galcanezumab based on presence/absence of cephalic and/or extracephalic allodynia during the pre-treatment non-ictal phase of migraine.
Results
Using strict criteria for allodynia (heat 32–40°C, cold 32–20°C, mechanical <60 g), we report that (a) the incidence of pre-treatment non-ictal cephalic allodynia was 21% in the 24 responders (>50% decrease in monthly migraine days) and 85% in the 19 non-responders; (b) the incidence of non-ictal extracephalic allodynia distinguishes responders from non-responders less accurately; and that (c) the incidence of non-ictal cephalic allodynia was similar in the chronic migraine and high-frequency episodic migraine groups.
Conclusions
Clinically, the findings suggest that presence/absence of non-ictal allodynia can be used to identify galcanezumab responders with nearly 80% accuracy and galcanezumab non-responders with nearly 85% accuracy. Mechanistically, the presence of non-ictal allodynia (reflecting a state of activity-independent central sensitization) in both chronic migraine and high-frequency episodic migraine patients raises the possibility that the state of non-ictal allodynia may be attributed to physiological properties of central trigeminovascular neurons that are due to the genetic load of the individual patient rather than their migraine frequency.
Introduction
Migraine is a complex neurological disorder involving multiple cortical, subcortical, and brainstem areas that regulate autonomic, affective, cognitive, and sensory functions (1,2). Of relevance to the current study is the mounting evidence for generalized abnormalities in the processing of sensory signals that originate in visual, auditory, olfactory, and somatosensory organs (3). While the abnormally heightened sensitivities to light, sound and smell are commonly attributed to enhanced excitability and responsivity of cortical neurons involved in the generation of visual, auditory and olfactory perceptions (4–6), it is thought that the abnormal perception of pain or its induction by innocuous stimuli depends on enhanced excitability and responsivity of peripheral and central trigeminovascular neurons that feed into the somatosensory cortices. According to the revised International Association for the Study of Pain (IASP) – Basic Pain Terminology (7), allodynia, defined as pain in response to a normally non-nociceptive stimulus (i.e., reflecting the fact that the nature of the stimulus and the response modes differ), is mediated by peripheral activation of tactile, warm and cold receptors that gain access to central nociceptive processing (8).
Adopting this view of allodynia and the continuously growing knowledge about central sensitization (defined as increased responsiveness of nociceptive neurons in the central nervous system to their normal or subthreshold afferent input) and the hurdles it poses to the treatment of multiple pain conditions including migraine, we showed nearly 20 years ago that ictal allodynia is detrimental to the success of rendering migraine patients pain-free with triptans (9,10). Incorporating the concept that the baseline excitability and responsivity state of central trigeminovascular neurons (as reflected by enhanced sensitivity to skin stimulation) that mediate the headache phase of migraine could determine outcome of prophylactic treatment of migraine, Wang and his colleagues (11) reported recently that the prophylactic treatment of chronic migraine (CM) with the calcium channel blocker flunarizine was significantly more effective in those participants whose skin sensitivity to mechanical and thermal stimulation of the periorbital region was relatively normal, in comparison to those whose periorbital skin sensitivity was abnormally heightened. Somewhat similarly, May and colleagues (12) reported that the pre-treatment forearm heat pain threshold of migraine patients whose number of migraine days per month (MMD) decreased by >30% following a three-month period of galcanezumab treatment was significantly higher (45.2°C) than in those who failed to achieve the 30% reduction in MMD (44.3°C).
Fundamental differences between allodynia that occurs during the ictal phase (i.e., sensitization occurring during a time when central trigeminovascular neurons receive massive nociceptive input from active meningeal nociceptors) vs non-ictal (i.e., sensitization occurring during a time when central trigeminovascular neurons receive no or subliminal nociceptive input from meningeal nociceptors) phases of migraine have led us to hypothesize that while ictal allodynia can determine response outcome to acute treatment of migraine, it is the non-ictal allodynia that can determine the outcome of migraine prevention with migraine prophylactic drugs that interact with peripheral but not central trigeminovascular neurons. Accordingly, the goal of the current study was to determine whether it is possible to prospectively identify patients’ response to galcanezumab, a monoclonal antibody against calcitonin gene-related peptide (CGRP-mAb approved in the US for the preventive treatment of migraine in adults), based on the presence or absence of cephalic and/or extracephalic allodynia during the non-ictal phase.
Methods
This prospective, observational, open-label, cohort study was part of a multifaceted project whose main goal was to investigate the many mechanisms by which peripherally acting drugs restore brain functions and structure in areas involved in the generation of the complex migraine attack. Here, only the results of the QST studies are reported. The study was approved by our local institutional review board and was conducted according to Good Clinical Practice and the Declaration of Helsinki. Patients provided written informed consent before participating in the study.
Study population
Study participants were recruited at the Beth Israel Deaconess Medical Center (BIDMC) Comprehensive Headache Center, Boston, MA.
Patients received diagnosis of migraine according to the criteria of the third edition of the International Headache Classification of the International Headache Society (ICHD-3) (13) by a neurologist and a (SA) headache specialist after filling out a four-week e-diary. Inclusion criteria for the study were: between 18 and 65 years of age, previous diagnosis of migraine (with or without aura) in accordance with the ICHD-3 criteria, experience of headache ≥8 days per month (based on participants recalling their experiences during the last three months, and on e-diary data during the four-week baseline period), and onset of migraine at age 50 years or younger. Participating patients agreed to refrain from initiating or changing the type, dosage, or frequency of any prophylactic medications for indications other than migraine that may interfere with the study objectives (e.g., antidepressants, anticonvulsants, beta-adrenergic blockers, etc.). Exclusion criteria were current regimen of one or more migraine preventative therapies, pregnancy, breastfeeding, significant psychiatric or cognitive disorder and/or behavioral problems that could interfere with the study, other significant pain problem (e.g., cancer pain, fibromyalgia, other head or facial pain disorder), known or suspected severe cardiac disease (e.g., symptomatic coronary artery disease, prior myocardial infarction, congestive heart failure), known or suspected cerebrovascular disease (e.g., prior stroke or transient ischemic attack, symptomatic carotid artery disease, prior carotid endarterectomy or other vascular neck surgery), report of abnormal electrocardiogram within the last year (e.g., second or third-degree heart block, prolonged QT interval, atrial fibrillation, atrial flutter, history of ventricular tachycardia or ventricular fibrillation, clinically significant premature ventricular contraction), uncontrolled high blood pressure (systolic >160 mm Hg, diastolic >100 mm Hg), known history or suspicion of secondary headache, known history or suspicion of substance abuse or addiction (within the last five years), current use of marijuana (including medical marijuana) or has used marijuana (including medical marijuana) or cannabidiol oil within the last one year. Other exclusion criteria were current use of simple analgesics or non-steroidal anti-inflammatory drugs (NSAIDs) >15 days per month or triptans, ergots, or combined analgesics >10 days per month for headaches or other body pain. The patient could not participate if they currently used opioids for headaches or other body pain, had undergone nerve block (occipital or other) in the head or neck within the last three months, had received botulinum toxin or anti-CGRP monoclonal antibody treatment within the last six months.
Study design
Sixty-four patients were initially recruited to the study. Participation included three visits to the BIDMC Comprehensive Headache Center (Figure 1). In the first visit, participants received a detailed explanation about the study, signed an informed consent, provided detailed medical and headache history, and received training on how to complete an electronic daily headache diary and instructions about the Quantitative Sensory Testing (QST). Those who were headache-free for >12 hours, underwent the QST as described below. Those who were at the ictal state or headache-free for <12 hours, were offered to take the QST again when at the non-ictal state (see definition below) and before initiation of treatment. Those who completed the daily e-diary for 30 days were scheduled for a second visit. In the second visit, the daily headache diary was reviewed, a urine pregnancy test was obtained from all female participants, a QST was offered to those who did not take the test in the first appointment, and the initiation dose (240 mg) of galcanezumab was administered. Patients continued to fill in the daily e-diary for three more months. During this period, they either self-administered or returned to the Headache Clinic to receive the second and third galcanezumab injections (120 mg). The 43 participants who completed the four-month e-diary were scheduled for a final visit. In this visit, they were given the opportunity to ask any question, provide their impressions of the treatment, and review a summary of the data they entered electronically.

Study flow chart. QST, Quantitative Sensory Testing.
E-diary
All participants filled a daily e-diary for at least four months (one month before and three months after treatment initiation (Figure 1). The daily e-diary was administered in the form of a REDCap survey using an email link that participants accessed from their personal computer/electronic device. It consisted of a questionnaire that helped us determine the daily/monthly incidence of headache and migraine, laterality (unilateral, bilateral), pain intensity, occurrence of associated symptoms (nausea, vomiting, throbbing, photophobia, phonophobia, osmophobia), and use of abortive medications.
Classification and definition of responders
Those experiencing eight to 14 migraine days per month (MMD) were classified as high-frequency episodic migraine (HFEM) patients. Those experiencing ≥15 MMD were classified as chronic migraine (CM) patients. Galcanezumab responders were patients whose percentage of migraine days decreased by >50% during the three-month treatment period (compared to the percentage of migraine days in the one-month pre-treatment period) (Figure 2). Conversely, galcanezumab non-responders were patients whose percentage of migraine days/month decreased by <50% during the three-month treatment period (compared to the percentage of migraine days in the one-month pre-treatment period) (Figure 2). The 50% change in migraine days per month was adopted as it is the standard in defining responders in phase 3 placebo-controlled studies (14–17).

Identification of galcanezumab responders and non-responders. Patient’s daily e-diary entry data were used to compute percentage of monthly migraine days during the one-month pre-treatment period and the three-month treatment period. Red dots represent days in which the participant indicated experiencing headache fulfilling migraine criteria. Blue dots represent days in which the participant indicated experiencing no headache. Each dot represents the participant’s entry in each of the 120 days. Responders were those whose percentage of migraine days decreased by >50% during the three-month treatment period. Non-responders were those whose migraine days decreased by <50% during the three-month treatment period.
Definition of the non-ictal phase
To be assessed during a non-ictal phase, participants had to be pain-free for at least 24 hrs, 12 hrs prior and 12 hrs post the QST session. These criteria were adopted to accommodate those CM patients whose pain-free intervals were rarely longer than one to two days (see explanation/justification in Discussion). In cases in which the number of MMD was above 20, treatment initiation was delayed (up to two months) until the participant was headache-free for the required number of hours.
Quantitative sensory testing (QST)
To address the main goal of the current study in the most objective and unbiased way, participants’ pain thresholds to heat, cold and mechanical stimulation were determined only once, during a non-ictal phase, and before the administration of the first dose of galcanezumab (i.e., before knowing whether the galcanezumab treatment reduced the MMD and by how much). Heat, cold, and mechanical pain thresholds were determined using QST as described in detail in our previous study (18). Briefly, all QST sessions were conducted in a quiet room (room temperature 24°C) away from noise and distraction. To increase consistency and reliability, and to decrease anxiety or fear that the sensory stimuli would be too painful to handle or cause tissue damage, we first took the time to explain to participants the difference between pain threshold (the earliest time point at which a stimulus is felt painful) and pain tolerance (the latest time point at which they can tolerate the stimulus). We then asked them to hold the thermode in their hand (so that they could let go if they could not stand the pain) while the temperature increased (heat) or decreased (cold) gradually and press the stop button as soon as they felt pain. For the mechanical pain threshold testing, we gave them the opportunity to try the different monofilaments and see that they do not penetrate the skin or cause any damage. At the end of this ‘learning’ phase, pain thresholds to heat, cold and mechanical stimulation were determined first at the thenar eminence skin, and then at the periorbital area, both on the most common side of the headache (i.e., if the headache was mostly unilateral) or at the side where the headache was usually most painful. Thermal skin stimuli were delivered through a 30 × 30 mm2 thermode (Q-Sense 2016, Medoc, Ramat-Yishai, Israel) attached to the skin at a constant pressure. Pain thresholds were determined using the Method of Limit (19) as described in detail in Burstein et al., 2000 (18) and 2004 (20). To determine heat pain thresholds, the skin was allowed to adapt to a temperature of 32°C for one minute and then warmed up at a slow rate (1°C/sec) to a maximum of 50°C or until the earliest (lowest) temperature that pain sensation was perceived, at which point the subject stopped the stimulus by pressing a button on a patient response unit. Heat stimuli were repeated three times each at 30-second intervals, and the mean of the recorded temperatures was considered threshold. To determine cold pain thresholds, the skin was allowed to adapt to a temperature of 32°C for one minute and then cooled down at a slow rate (1°C/sec) to a minimum of 16°C or until the earliest (lowest) temperature that pain sensation was perceived, at which point the subject stopped the stimulus by pressing a button on a patient response unit. Cold stimuli were repeated three times each at 30-second intervals, and the mean of the recorded temperatures was considered threshold. Pain threshold to mechanical stimuli were determined by using calibrated von Frey hairs (VFH, Stoelting). Each monofilament was applied to the skin three times (for two seconds) in an ascending order (2g, 4g, 6g, 8g, 10g, 15g, 26g, 60g, 100g, 180g, 300g) and the smallest VFH force inducing pain at two out of three trials was considered threshold.
Definition of allodynia
Cutaneous allodynia is defined as pain resulting from a normally non-noxious stimulus of the skin. In this study the presence of allodynia was determined using the value of the pain thresholds for heat, cold and mechanical stimuli measured at the periorbital skin area while the patient was pain-free for a minimum of 24 hours, 12h before and 12 h after the testing. Patients were defined as allodynic if their heat pain thresholds were between 32 and 40°C, if their cold pain thresholds were between 32 and 20°C, or if their mechanical pain thresholds were below 60 g (Figure 3) (21–23). These values are based on the following: (a) heat threshold of c-mechano-heat nociceptors in the human skin is around 41°C (24–27), and heat pain threshold in the periorbital area of pain-free migraine patients is well above 40°C; (b) cold threshold of c-polymodal nociceptors innervating the human skin is 19°C (28), and cold pain threshold in the periorbital area of pain-free migraine patients is well below 20°C; and (c) mechanical pain threshold in the periorbital area of pain-free migraine patients is above 60 g (11,18,29).

Definition of allodynia. Adopting the traditional definition of allodynia (pain in response to a non-nociceptive stimulus that does not normally provoke pain) and the principle that the test stimulus does not activate nociceptors, we defined thermal allodynia (a) below 40°C for heat and above 20°C for cold based on existing data on activation threshold of nociceptors innervating the human skin (see refs. in Methods), and on multiple psychophysical studies in which pain thresholds to different sensory modalities have been defined in healthy subjects. In the absence of such data for mechanical threshold, mechanical allodynia (b) was defined as induction of pain by skin indentation with a force that is smaller than 60 g based on psychophysical studies in healthy subjects and migraine patients, falling within the more general term of hyperalgesia.
Statistics
The database consisted of pain threshold measurements in 43 patients with migraine, taken at the non-ictal phase (QST was not conducted in participants who were at an ictal phase during the pre-treatment visit). The resulting distributions were tested for normality (Shapiro-Wilk test), and their descriptive statistics computed. The differences between those whose MMD decreased by >50% (i.e., the responders) and those whose MMD decreased by <50% (i.e., non-responders) were computed, and the criteria for allodynia were applied, yielding two groups of patients – those with and those without cutaneous allodynia. The proportions of patients with allodynia in each group were tested for statistical significance using Fisher Exact Test. Differences in mean pain thresholds between the respective responder and non-responder groups were performed using non-parametric and non-paired Mann Whitney U test. The person computing and comparing incidences of allodynia, and mean pain thresholds in the different groups (responders, non-responders) and classes (CM and HFEM) of patients was blinded to the group and class of each individual.
Results
Participants
Sixty-four CGRP-mAb naïve episodic and chronic migraine patients were recruited to this study. Forty-three of them fulfilled the four criteria for being included in the data analysis: i) complete the four-month electronic diary (one month before and three months during treatment), ii) not miss a single treatment cycle, iii) undergo QST before treatment initiation, and iv) during a non-ictal phase (as defined above). Based on the percent reduction in migraine days per month, 24 patients were classified as responders (>50% reduction in migraine days per month during the first three months of treatment, example in Figure 2A) and 19 were classified as non-responders (<50% reduction in migraine days per month, example in Figure 2B). Their demographics are presented in Tables 1 and 2, respectively. The 24 responders (79% female, 21% male) were 33.8 ± 8.6 (mean ± SD) years old, experienced migraine for 15.2 ± 9.7 years, recorded 13.7 ± 4.2 migraine days per month in the e-diary, and had a BMI of 25.2 ± 4.4. The 19 non-responders (95% female, 5% male) were 34.3 ± 12.5 (mean ± SD) years old, experienced migraine for 17.0 ± 12.2 years, recorded 16.2 ± 6.8 migraine days per month, and had a BMI of 24.4 ± 6.0. Comparisons between corresponding values in responders vs non-responders yielded no significant differences (age: p = 0.77, years with migraine: p = 0.75, migraine days/month: p = 0.09, BMI: p = 0.12 Mann-Whitney).
Demographic data, migraine classification, treatment response and values of heat, cold and mechanical pain thresholds at cephalic and extra-cephalic sites among the 24 responders (>50% decrease in MMD).

Pain threshold values considered within the allodynic range are marked in red.
BMI: body mass index; CM: chronic migraine; HFEM: high frequency episodic migraine; MIDAS: migraine disability assessment; MMD: mean migraine days/month; QST: quantitative sensory testing.
Demographic data, migraine classification, treatment response and values of heat, cold and mechanical pain thresholds at cephalic and extra-cephalic sites among the 19 non-responders (<50% decrease in MMD).

Pain threshold values considered within the allodynic range are marked in red.
BMI: body mass index; CM: chronic migraine; HFEM: high frequency episodic migraine; MIDAS: migraine disability assessment; MMD: mean migraine days/month; QST: quantitative sensory testing.
Relative frequency of non-ictal cephalic allodynia
Of the 24 responders, 79% did not have non-ictal cephalic allodynia and 21% did (Figure 4A, Table 1). In contrast, among the 19 non-responders, 15.7% did not have allodynia whereas 84.3% had allodynia (Figure 4A, Table 2). These relative differences in frequencies of allodynia were statistically significant (χ2 = p < 0.0001, DF = 1).
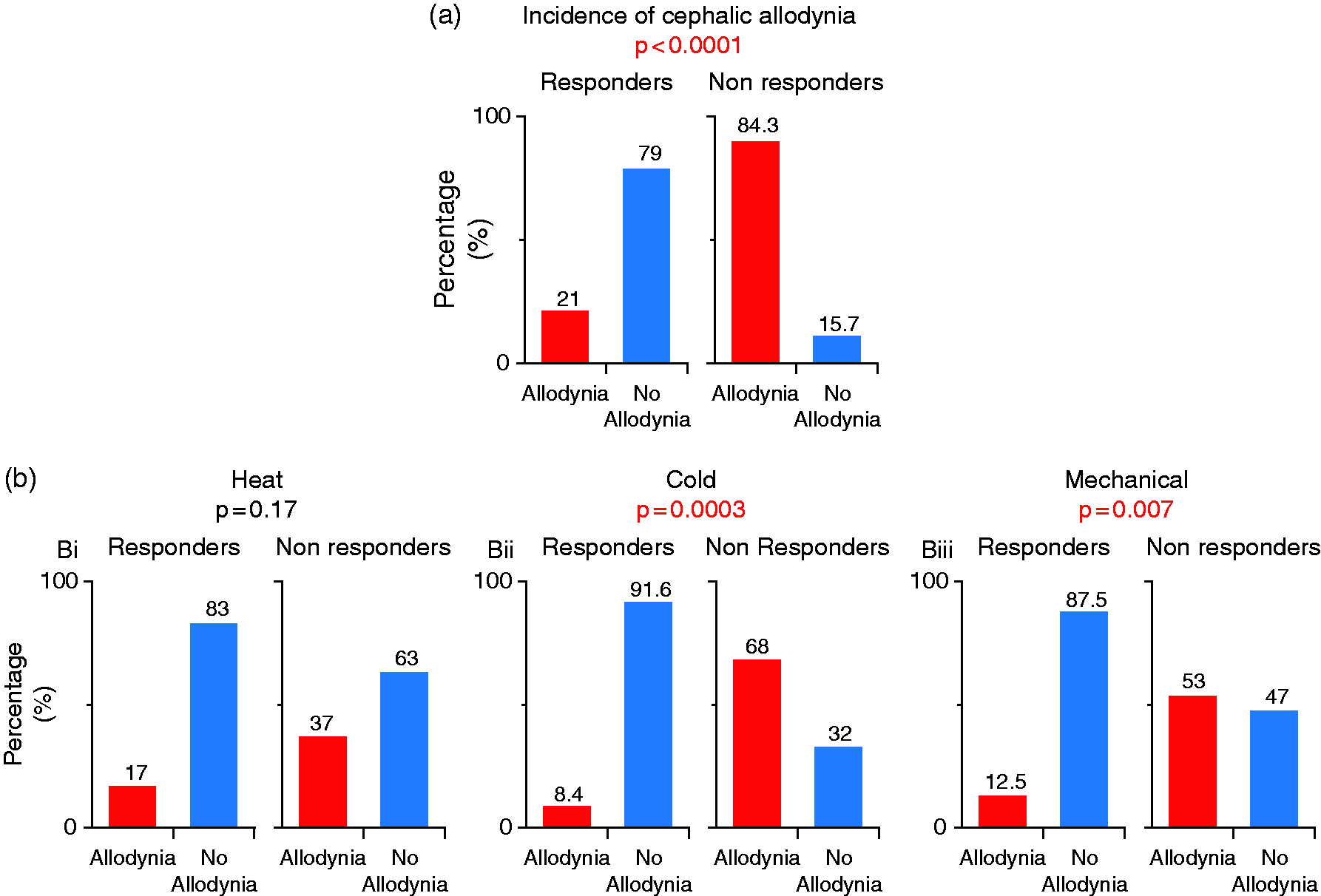
(a) Incidence of cephalic allodynia in responders and non-responders (in this analysis, patients were considered allodynic if one of their pain thresholds (i.e., either heat or cold or mechanical) was within the allodynic zone as defined in Figure 1). Note the significant difference in the incidence of cephalic allodynia between the two groups (Fisher Exact Test) and (b) Incidences of heat (Bi), cold (Bii) and mechanical (Biii) cephalic allodynia in responders and non-responders. Note that the predictive value of cephalic allodynia was significant in response to cold and mechanical, but not heat stimulation (Fisher Exact test).
Relative frequency of heat, cold and mechanical cephalic allodynia
In the 24 responders, heat allodynia was detected in four (17%), cold allodynia in two (8.4%), and mechanical allodynia in three (12.5%) subjects (Figure 4B, Table 1). In the 19 non-responders, heat allodynia was detected in seven (37%), cold allodynia in 13 (68%), and mechanical allodynia in 10 (53%) subjects (Figure 2, Table 2). These relative frequency differences were statistically significant for cold (χ2 = p = 0.0001, DF = 1) and mechanical (χ2 = p = 0.004, DF = 1), but not for heat (χ2 = p = 0.13, DF = 1) pain threshold.
Of the five patients with allodynia among the responders, two exhibited heat, cold and mechanical allodynia, one exhibited cold allodynia only, one exhibited heat allodynia only, and one exhibited mechanical allodynia only. Of the 17 patients with allodynia among the non-responders, five exhibited heat, cold and mechanical allodynia, three exhibited cold and mechanical allodynia, one exhibited heat and mechanical allodynia, four exhibited cold allodynia only, three exhibited mechanical allodynia only and one exhibited heat allodynia only ( Tables 1, 2).
Cephalic heat, cold, and mechanical pain thresholds
Among the 24 responders, median [IQR] heat 45.3° [42.7–46.5], cold 16.0° [16.0–16.5], and mechanical 100 g [60–180] pain thresholds were all outside the allodynic range (Figure 5A–C). In contrast, among the 19 non-responders, median [IQR] mechanical pain threshold (26 g [15–60]) was within the allodynic range, cold pain threshold (20.5°C [16.0–24.3]) was nearly at the allodynic range, while heat pain threshold 42.0°C [39.5–45.6] was clearly outside the allodynic range (Figure 5A–C). Comparisons of these corresponding values yielded statistically significant differences between responders and non-responders (heat: p = 0.03, cold: p = 0.001, mechanical: p = 0.004, Mann Whitney U test).

Heat (a, d), cold (b, e) and mechanical (c, f) pain thresholds recoded in cephalic and extra-cephalic sites of responders and non-responders. Differences between responders and non-responders are shown in box-and-whisker plots combined with scatterplots of individual values. Note distinction between pain thresholds inside and outside the allodynic zones (marked by orange area). Also note that responders differed (*<0.05) from non-responders in their cephalic pain thresholds of all modalities (heat: p = 0.03, cold: p = 0.001, mechanical: p = 0.004, Mann Whitney U test).
Relative frequency of extra-cephalic allodynia
Of the 18 responders who provided extracephalic QST values, 88% did not have allodynia and 12% had allodynia (Figure 6A, Table 1). In contrast, among the 19 non-responders, 58% did not have allodynia, whereas 42% had allodynia (Figure 6A, Table 2). These relative frequencies of allodynia were statistically different (χ2 = p = 0.03, DF = 1).

(a) Incidence of extra-cephalic allodynia in responders and non-responders. Note that the observed tendencies are statistically insignificant (Fisher Exact Test), suggesting that measurements of extra-cephalic pain thresholds cannot be used to determine treatment response and (b) Incidence of heat (Bi), cold (Bii) and mechanical (Biii) extra-cephalic allodynia in responders and non-responders. Note that no modality can distinguish between the two groups (Fisher Exact test).
Relative frequency of heat, cold and mechanical extra-cephalic allodynia
In the responders, heat allodynia was detected in one (5%), cold allodynia in one (5%), and mechanical allodynia in two (10%) subjects (Figure 6B, Table 1). In the 19 non-responders, heat allodynia was detected in one (5%), cold allodynia in four (21%), and mechanical allodynia in three (15%) subjects (Table 2, Figure 6B). These relative frequencies were statistically non-significant for heat, cold, and mechanical (χ2 = p > 0.05, DF = 1).
Extra-cephalic heat, cold, and mechanical pain thresholds
Among the 18 responders, median [IQR] heat 45.6°C [44.3–46.8], cold 16.0°C [16.0–16.5], and mechanical 140 g [100–180] pain thresholds were all outside the allodynic range (Figure 5D–F). Similarly, among the 19 non-responders, median [IQR] heat 43.7°C [41.1–45.3], cold 17.0°C [16.0–18.8], and mechanical 100 g [60–180] pain thresholds were also outside the allodynic range (Figure 5D–F). Comparisons of these corresponding values yielded statistically significant differences for heat (p = 0.01, Mann Whitney U test) but not for cold or mechanical (p > 0.05, Mann Whitney U test).
Relative frequency of non-ictal cephalic allodynia in episodic vs. chronic migraine
Of the 43 participants, 24 were classified as episodic (66% responders, 34% non-responders) and 19 as chronic (42% responders, 58% non-responders) migraineurs (χ2 = p = 0.10, DF = 1). The relative frequency of non-ictal cephalic allodynia was similar among the episodic (45%) and chronic (58%) patients (Figure 7A, χ2 = p = 0.43, DF = 1).

(a) Incidence of cephalic allodynia in HFEM and CM. Note that the incidence is similar (Fisher Exact test) in the two groups, suggesting that the development of allodynia and central sensitization is independent of attack frequency. Incidences of heat (B),Continued.cold (c) and mechanical (d) cephalic allodynia in HFEM and CM. Note that no modality can distinguish between the two groups (Fisher Exact test). Pain thresholds for heat (e), cold (f) and mechanical (g) recorded in the cephalic site of HFEM and CM. Differences between HFEM and CM are shown in box-and-whisker plots combined with scatterplots of individual values. Note distinction between pain thresholds inside and outside the allodynic zones (marked by orange area). Also note that the two groups did not differ in any of their pain thresholds (p > 0.05 Mann Whitney U test).
Relative frequency of non-ictal heat, cold and mechanical cephalic allodynia in episodic vs. chronic migraine
In the 24 episodic migraine patients, heat allodynia was detected in five (20%), cold allodynia in eight (33%), and mechanical allodynia in seven (29%) subjects (Figure 7B–D, Tables 1, 2 ). In the 19 chronic migraine patients, heat allodynia was detected in five (26%), cold allodynia in seven (36%), and mechanical allodynia in eight (42%) subjects (Figure 7B–D, Tables 1, 2). These relative frequencies were statistically non-significant (χ2 = p > 0.05, DF = 1).
Non-ictal cephalic heat, cold, and mechanical pain thresholds in episodic vs. chronic migraine
Among the 24 episodic migraine patients, median [IQR] heat 45.3° [39.8–46.2], cold 16.0° [16.0–23.6], and mechanical 100 g [34.5–180] pain thresholds were similar (p > 0.05 for all modalities, Mann Whitney U test) to those recorded in the 19 chronic migraine patients (heat 43.4° [40.4–46.0], cold 16.0° [16.0–20.5], mechanical 60 g [15.0–180]), (Figure 7E–G, Tables 1–2).
Combined pain thresholds of all patients
Combining QST results of all 43 patients (regardless of their classification into responders or non-responders, or whether they are diagnosed with HFEM or CM) showed that in the non-ictal state, (mean ± SD; Mann Whitney) cephalic and extra-cephalic heat (43.2 ± 3.3 vs. 44.3 ± 2.7°C, p = 0.24), cold (19.1 ± 4.2 vs. 17.5 ± 2.3°C, p = 0.33), and mechanical (89.7 ± 67.3 vs. 119.4 ± 59.5 g, p = 0.03) pain thresholds were far above the allodynia threshold (>40°C for heat, <20°C for cold, and below 60 g for mechanical).
Sensitivity, specificity and accuracy
Based on the number of non-ictal allodynic and non-allodynic among the responders and non-responders, we calculated the positive (PPV) and negative (NPV) predictive values as follow: PPV = 86% (True Positive [TP] = 19, False Positive [FP] = 3) (PPV = TP/[TP + FP]). NPV = 76% (False Negative [FN] = 5, True Negative [TN] = 16) (NPV = TN/[FN + TN]). Accordingly, the overall sensitivity is 79%, specificity 84% and accuracy 81%.
Discussion
Summary of Findings
The goal of this study was to determine whether presence of non-ictal cephalic allodynia can be used to identify responders (and non-responders) to preventive treatment of migraine with galcanezumab, a CGRP-mAb whose principal site of action in migraine prevention is outside the brain. Using practical QST paradigm to assess patients’ skin sensitivity while pain-free and before initiation of treatment with galcanezumab, we show that by using strict criteria for non-ictal periorbital allodynia, it is possible to identify responders with nearly 80% accuracy and non-responders with nearly 85% accuracy. In contrast, the assessment of non-ictal extra-cephalic allodynia with QST was of little value as it missed nearly 50% of the allodynic patients among the non-responders (accuracy rate of 42%) and added little to the assessment of allodynia among the responders. The finding that the relative frequency of non-ictal periorbital allodynia in the CM group was similar to the relative frequency of allodynia in the HFEM group suggests that the two groups share a similar pathophysiological state (in this study, it is the establishment of activity-independent central sensitization – the neural correlates of non-ictal allodynia). Along this line, it was surprising to find that presence or absence of allodynia was unrelated to the number or hours or days that the patients were headache-free prior to the sensory testing. This is yet another novel finding that points to the possibility that the state of non-ictal allodynia may be attributed to molecular, cellular and/or physiological properties of central trigeminovascular neurons and brain areas that regulate their excitability (30) that are due to the genetic load of the individual patient rather than the pathophysiological state of disease.
QST vs ASC-12 and pain threshold vs allodynia
Two methodological aspects distinguish the current study from most previous studies on cutaneous allodynia in migraine patients. The first is attributed to the evaluation method and the second to the fundamental difference between pain threshold and allodynia. Regarding the first, it is worthwhile noting that nearly all studies conducted between 2004 and 2021 used the 12-item Allodynia Symptom Checklist (ASC-12), a questionnaire that assesses symptoms of cutaneous allodynia during headache by subjective post-hoc reporting by memory, rather than QST that assesses the physiological presence or absence of allodynia at the time of testing (whether conducted in the ictal or inter-ictal phase) (31,32). Due to the nature of these studies, it has not been possible to determine with reasonable level of reliability whether answers to the ASC-12 questions relate to patients’ life-time experience, to their experience during acute migraine attacks, or to their experience during a headache-free period. Regarding the second methodological aspect, large inconsistencies have been reported in studies in which QST was used to evaluate thermal and mechanical pain thresholds and their contribution to migraine pathophysiology (31,32). Possible sources of these inconsistencies could be attributed to different sets of instructions given to patients regarding the time they need to stop the stimulus, cultural differences between different populations, time of testing (ictal vs. non-ictal vs. peri-ictal vs. post-ictal), age, frequency of attacks, and use of different criteria for determination of allodynia. While different pain threshold values have been reported for the four different phases of migraine (33–35), it is unclear whether these statistical differences carry with them any clinically meaningful significance as all reported heat and cold pain thresholds fall above 40°C (heat) and below 20°C (cold) – a range of temperatures that are well within the activation threshold of nociceptors innervating the human skin. While all reported pain thresholds in these studies were judged as reliable, they cannot serve as evidence of allodynia as they do not fall outside the range of activation of nociceptors and within the activation range of warm and cold receptors (7,36,37). By adopting the criteria defined by Loeser and Treede (7), whereby allodynia should be considered only when a pain threshold falls outside the activation threshold of the nociceptor, and by analyzing patients individually, we show that absence or presence of non-ictal allodynia influences the outcome of migraine prevention with galcanezumab. Of note, our class (HFEM vs CM) and group (responders vs non-responders) analyses of heat, cold, and mechanical pain thresholds in the periorbital and extracranial regions show similar values to those reported in the other studies mentioned above (34,35,38–40). Accordingly, we conclude that the value of studying the role of cutaneous allodynia and central sensitization in migraine pathophysiology will increase exponentially if the herein proposed criteria are used to define allodynia with QST, and if analyses take into consideration the individual patient rather than the group performance.
By far the most important aspect of the current study is the finding that pre-treatment measurement of non-ictal allodynia can be used to identify with high degree of accuracy which individual patient will experience a reduction in MMD that is larger than 50% following galcanezumab therapy. It is also highly accurate in identifying which patient will not. The rationale for our assumption – that it is the non-ictal rather than ictal allodynia that can help us to determine response outcome to treatment with galcanezumab – stems from the fact that the efficacy of this prophylactic drug derives from its ability to reduce CGRP signaling in the dura and consequently attenuates sensitization and eventual activation of meningeal nociceptors (Figure 8). As shown in the figure, at the pre-treatment non-ictal phase (Figure 8a), both the peripheral and central trigeminovascular neurons are inactive and the patients are pain-free (thus during a non-ictal phase). As the non-responder is allodynic when all neurons are inactive, it is reasonable to propose that the central neurons in this patient are sensitized and hyperexcitable. In contrast, the absence of allodynia in the responder suggests that the central neurons of this patient are not sensitized when pain-free. Given the overwhelming amount of data on the consequences of neuronal sensitization, hyperexcitability and hyper-responsivity (41,42), in Figure 8b we propose that at the post-treatment ictal phase activation of the peripheral neurons in responders is attenuated enough by the treatment to prevent the activation of non-sensitized central trigeminovascular neurons in the responders, but not the activation of sensitized central trigeminovascular neurons in the non-responders. Consequently, the non-responder patient experiences a headache despite treatment, whereas the responder does not. Scientifically, one way to explain the activation of the sensitized (but not non-sensitized) central neuron by the reduced input it gets from the nociceptor is that at the sensitized state, far fewer excitatory post-synaptic potentials are required to generate an action potential (43). Although not presented in Figure 8 (for simplicity), we must take into consideration the likely possibility that the hyper-responsiveness of the central neurons is also attributed to dysfunction of the endogenous pain control system (30,44,45), which could be due to the individual genetic load or acquired by the prolonged exposure to pain. Additionally, the findings that the non-ictal extra-cephalic heat pain threshold was significantly lower in the non-responders than in the responders raise the possibility that consequential sensitization of thalamic trigeminovascular neurons may also influence responsivity to galcanezumab, a concept presented in several of our previous studies (18,46,47).

Proposed explanation for why and how non-ictal allodynia interferes with galcanezumab’s ability to prevent the initiation of the next headache. (a) At the pre-treatment non-ictal phase, both the peripheral and central trigeminovascular neurons are inactive (subthreshold baseline activity is insufficient to generate action potentials) and the patient is pain-free. Because the non-responder is allodynic when all neurons are inactive, it is assumed that the central neuron in this patient is sensitized and hyperresponsive. In contrast, the absence of allodynia in the responder suggests that the central neuron in this patient is not sensitized or hyperresponsive when the patient is pain-free and (b) At the post-treatment ictal phase, activation of the peripheral neurons during the ictal phase is attenuated enough (by the mAb ability to neutralize the CGRP peptide and stop it from binding to the CLR-RAMP1) to prevent the activation of the non-sensitized central trigeminovascular neurons in the responders, but not the activation of the sensitized central trigeminovascular neurons in the non-responder. Consequently, the non-responder patient experiences a headache, whereas the responder does not. For simplicity, the contribution of descending pain modulatory inputs to the sensitization state of the neuron is not shown.
One of the more surprising findings of this study is that the presence of non-ictal allodynia is unrelated to the number of years patients experienced migraine, their baseline attack frequency, or the number of hours or days that they were headache-free prior to the sensory testing. It is surprising because it does not ‘fit’ current notions that the sensitized state of the central neuron depends either on the nociceptive input it receives from the periphery (43,48) or on descending input it receives from the brainstem (49–51) at the peri-ictal phase of an attack. Rather, it raises the possibility that the tendency to become sensitized depends on genetic factors that are due to the individual (much like the cortical hyperexcitability theory of migraine aura). If this theory is correct, one would expect to find different patterns of expression and/or different functional states of ionic channels and receptors on membranes of central trigeminovascular neurons of responders vs non-responders, a proposal supported indirectly by mounting evidence for abnormal expression of genes that promote hyperexcitability in central neurons of certain migraine patients (52).
Limitations
As with all observational, open-label, cohort studies, interpretation of results must take into consideration participants’ expectations. However, given that all patients received the same galcanezumab treatment, the same information about the goals of the study, the same training and instructions on when to stop the sensory stimuli (i.e., as soon as they perceived them as painful, not before, and not when they could no longer stand the pain), and given that the efficacy of the treatment (56% achieved >50% decrease in MMD and 44% did not) was similar to the efficacy data reported in the phase 3 studies (14), the potential impact on the results of the absence of a placebo-controlled group is somewhat lessened. Although not included in this paper, all 24 responders (after three months of treatment) continued to be responders after 12 months of treatment.
While documenting accurately the number of days or hours that participants were pain-free prior to the time of sensory testing, we were not able to collect with sufficient accuracy the time to the next migraine attack. As previous studies suggested that the time to the next migraine attack may influence the QST results, the findings of the current study may have been somewhat influenced by the relatively novel distinction between the different non-ictal phases (peri-ictal, interictal, post-ictal). However, as shown in the study, our analyses of pain thresholds in the two groups (responders, non-responders) and two classes (CM, HFEM) of patients are similar to those reported in previous and more recent studies (31–35).
As participants in the current study had to fulfil inclusion criteria for treatment with galcanezumab, we could not include those with fewer than eight migraine days per month. Consequently, our discussion and conclusions apply only to the HFEM and CM patients but not to those with LFEM or even those with less than one migraine day per month.
Conclusions
Detection of non-ictal cutaneous allodynia with a simplified paradigm of QST may provide a quick, affordable, non-invasive, and patient-friendly way to prospectively distinguish between responders and non-responders to the prophylactic treatment of migraine with drugs that reduce CGRP signaling. The implementation of this technique must take into consideration the individual patient rather than only her/his migraine classification status.
Article highlights
Non-ictal cephalic allodynia can be used to identify galcanezumab responders with nearly 80% accuracy and galcanezumab non-responders with nearly 85% accuracy. Mechanistically, the findings that Non-ictal allodynia is present in patients with 10–24 migraine days per month raise the possibility that the state of non-ictal allodynia may be attributed to molecular, cellular and physiological properties of central trigeminovascular neurons that are inherent to the genetic load of the individual patient rather than their attack frequency (i.e., the pathophysiological state of disease).
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SA received honoraria for consulting from Allergan/AbbVie, Amgen, Biohaven, Eli Lilly, Impel NeuroPharma, Novartis, Satsuma, Supernus, Theranica, Percept.
RB is the John Hedley-Whyte Professor of Anesthesia and Neuroscience at the Beth Israel Deaconess Medical Center and Harvard Medical School. He has received research support from the NIH: R01 NS094198-01A1, R37 NS079678, R01NS095655, R01 NS104296, R21 NS106345, Allergan, Teva, Dr. Reddy, Eli Lilly, Trigeminal and the Migraine Research Foundation. He is a reviewer for NINDS, holds stock options in AllayLamp, Theranica and Percept; serves as consultant, advisory board member, or has received honoraria from: Alder, Allergan, Amgen, Autonomic Technologies, Avanir, Biohaven, CGRP Diagnostic, Dr. Reddy’s Laboratory, ElectroCore, Eli Lilly, GlaxoSmithKline, Merck, Pernix, Theranica, Teva, and Trigemina. CME fees from Healthlogix, Medlogix, WebMD/Medscape, and Patents 9061025, 11732265.1, 10806890, US2021-0015908, WO21007165, US2021-0128724, WO21005497. BIDMC owns patent US 10,766,952 B2. Other authors declare that they have no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Parts of this study were funded by Eli Lilly, the National Institutes of Health (R37 NS079678, RO1 NS094198, RO1 NS106345), and the Anesthesia Department at Beth Israel Deaconess Medical Center. Galcanezumab was provided by Eli Lilly.
