Abstract
Background
The present prospective, real-world study aims to assess anti-calcitonin gene-related peptide (CGRP) monoclonal antibodies (mAbs) effectiveness across three consecutive one-year treatment cycles by means of a conservative intention-to-treat (ITT) analysis.
Methods
We enrolled 179 subjects (75.4% females, 51.3 years 95% confidence interval [49.2–53.4] years), 87.2% with chronic migraine and medication overuse) who started mAbs between 2018 and 2020. We recorded clinical data supported by a prospectively filled headache diary up to three one-year treatment cycles. The ITT analysis was performed with a multivariate linear mixed model considering the entire population.
Results
We observed a marked and consistent reduction in monthly migraine days (MMDs) across the three one-year cycles of treatment: −12.7 )[−11.4 – −14.1] at end of the first year of treatment (C1), −12.4 [−11.0 – −13.8] at the end of the second year (C2) and −12.9 [−11.4 – −14.3] at the end of the third year (C3). Baseline and residual MMDs progressively decreased across the three cycles (p = 0.008): from 21.1 [19.8–22.4] to 9.6 [8.3–11.0] in C1, from 19.0 [17.4–20.5] to 9.6 [8.1–11.1] in C2, and from 15.9 [14.3–17.5] to 8.5 [6.9–10.1] in C3. At the end of C3, the 50% response rate was 38.5% (69/179).
Conclusions
In our cohort, mAbs induced a meaningful and sustained reduction in MMDs across three consecutive one-year cycles of treatment. The ITT analysis revealed a remaining high burden of disease. While confirming mAbs effectiveness in migraine prevention, these findings underscore the need for more treatment approaches and for exploring other non-CGRP dependent pathways.
This is a visual representation of the abstract.
Introduction
Monoclonal antibodies (mAbs) targeting the calcitonin-gene related peptide (CGRP) pathway have rapidly changed the treatment landscape of migraine prevention. They block the activity of CGRP, a vasoactive neuropeptide responsible for peripheral and central mechanisms underpinning migraine pathophysiology (1).
Galcanezumab, fremanezumab and eptinezumab are humanized mAbs directed against the ligand, while erenumab is the only fully human mAb directed against the CGRP receptor (2).
Randomized clinical trials (RCTs), and associated open-label studies, provided strong evidence of good efficacy and safety (3,4). Real-world (RW) data showed an even larger effectiveness and good tolerability in the follow-up periods and in difficult-to-treat individuals (5).
While high quality studies corroborate mAbs use as a potential first-line preventive treatment (6,7), healthcare drug costs led to selection of strict criteria for treatment subsidization on a national basis (8,9). Reimbursement criteria of Italian Medicines Agency (AIFA) prioritized migraine frequency and related disability, required multiple previous treatment failures and limited the duration of treatment to one-year repeatable cycles, mandatorily separated by a suspension period of at least one month (10).
While a wealth of RCTs and RW studies corroborate the positive effect of mAbs over the short and middle term, evidence from treatment periods extending beyond one year is relatively scarce. Two open-label extensions (OLE) of a RCT on episodic migraine have reported the performance of erenumab over a three-to-five years period without suspensions, with more than half of participants achieving at least a 50% reduction in monthly migraine days (MMDs) during the whole follow-up period (11,12). Tolerability was good and the rate of treatment persistence high.
When considering RW studies, most were focused on the effect of mAbs suspension and restart with follow-up periods lasting from one to three years. These studies consistently reported a clinical worsening during the discontinuation period after six to twelve months of treatment, with a high variability of the worsening entity (13–25). Some of the studies also assessed the occurrence and the percentage of response following suspension (14,16,18,21–26).
Most of the above-mentioned studies adopted a per-protocol analysis, which intrinsically carries the risk of overestimating treatment effectiveness, especially in RW studies that are usually characterized by an important reduction of subjects remaining on treatment over time (27,28). OLE studies may suffer from this limitation as well, and they are usually designed to assess tolerability and safety, rather than effectiveness. This is a result of the roll-over process of participants from the parent double-blind treatment period, during which participants who poorly tolerated the study drug are early discontinued.
To the best of our knowledge, no real-life studies have evaluated migraine outcomes during mAbs treatment according to a conservative intention-to-treat (ITT) analysis over a follow-up period longer than two years. Our primary aim was to assess mAbs effectiveness across three consecutive one-year cycles of treatment with a conservative ITT approach. This approach considers data from all participants, including those who discontinue treatment for any reason. It therefore provides an unbiased estimate of the efficacy of the intervention on the primary study outcome at the level of adherence observed. This method of analysis results in a more accurate, unbiased estimate than that yielded from a per-protocol approach, which will also be presented in the present study.
Methods
Subjects
We enrolled all consecutive subjects with migraine who started treatment with one of the three mAbs available on the market at the time of the study (erenumab, galcanezumab and fremanezumab) for whom we had clinical data supported by a prospectively filled headache diary for the entire follow-up. We included 179 subjects with migraine who started treatment between December 2018 and January 2020. All participants were enrolled among those attending the outpatient clinics of the Headache Science & Neurorehabilitation Unit of the IRCCS Mondino Foundation (Pavia), the Headache Center of Careggi University Hospital (Florence) or the Neuroalgology Unit and Headache Center, Fondazione IRCCS Istituto Neurologico Carlo Besta (Milan).
Enrolled subjects satisfied the following inclusion criteria: (i) diagnosis of migraine with aura, without aura or chronic migraine (CM) according to the criteria of the International Classification of Headache Disorders, 3rd edition (ICHD-3) (29); (ii) at least eight MMDs during a three-month screening period; (iii) received at least one dose of mAbs; and (iv) attended at least the first three-month follow-up visit.
Exclusion criteria were: (i) contraindication to mAbs treatment according to AIFA (namely uncontrolled hypertension, unstable angina or other severe cardiologic comorbidities, previous ischemic stroke or transient ischemic attack, thromboembolic events) and (ii) pregnancy and breastfeeding.
The study was part of the Registro Italiano Cefalee (RICe) study (RICe project, Approval Number: ID 14591_oss Em. 2023–204 and subsequent amendments), and locally approved by the Ethics Committee of Pavia (P-20190105434). All participants enrolled signed the RICe informed consent.
Study procedures
This is a multicentric, open-label and observational, real-world study. All clinical parameters were prospectively recorded using an ad hoc headache diary.
The protocol foresaw three consecutive one-year treatment cycles with mAbs (C1: C1start to C1end; C2: C2start to C2end; and C3 C3start to C3end). Each cycle was separated by a suspension period (S1 between C1 and C2, and S2 between C2 and C3) lasting at least one month as per Italian regulations. At C1start, all subjects underwent a complete clinical evaluation, including a detailed anamnestic collection, a full neurological and general examination, and revision of headache diaries of the three preceding months. A psychological interview using the Structured Clinical Interview for DSM-5 Disorders (SCID-5) was performed by a psychologist with a background in the headache field (30). The first mAb dose was administered in the hospital setting, individuals were instructed to self-administer the subsequent doses at home. Participants returned to the Center every 12 weeks for the follow-up visits (M3 – M6 – M9 – M12) in each cycle (Figure 1) . After an at least one-month suspension period, subjects could restart mAbs treatment according to the following AIFA criteria: (i) presence of at least eight MMDs and (ii) Migraine Disability Assessment (MIDAS) score ≥11. At each visit, and during the suspension periods, we collected: monthly headache days (MHDs), MMDs, days and doses of acute drug intake, and occurrence of adverse events.
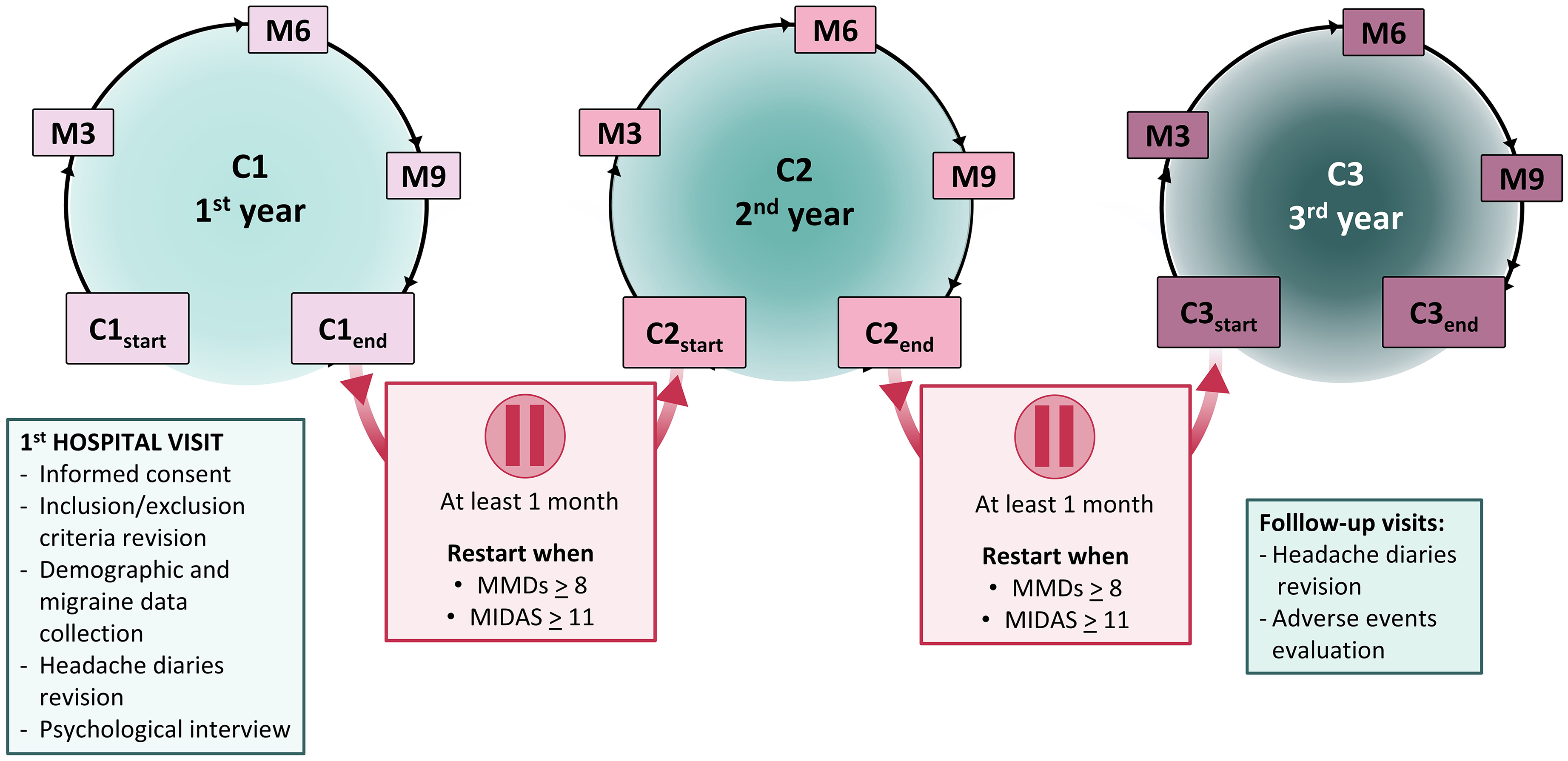
Study design. Legend: C1 = first one-year cycle of treatment; C2 = second one-year cycle of treatment; C3 = third one-year cycle of treatment; MMDs = monthly migraine days; MIDAS = Migraine Disability Assessment scale; M0 = baseline; M3 = three months from treatment initiation or re-initiation; M6 = six months from treatment initiation or re-initiation; M9 = nine months from treatment initiation or re-initiation.
Outcomes
ITT analysis
The primary outcomes were: (i) the reduction of MMDs from baseline to the last month of treatment and (ii) the overall residual MMDs at the end of each treatment cycle. All subjects enrolled were included in the ITT analysis and estimated marginal means were calculated according to a linear mixed model (LMM).
As secondary outcomes, we assessed the 30% and 50% response rate across each cycle defined as subjects achieving at least a 30% or a 50% reduction in MMDs in the last month of C1 (C1end), of C2 (C2end) and of C3 (C3end) compared to C1start. Among Drop-outs (for definition, see below) subjects who did not restart treatment due to sustained effectiveness were considered Responders, whereas drop-outs for any other reason were considered Non-responders.
Per-protocol analyses
In the per-protocol sub-analyses, we considered only those subjects who received all mAbs injections across the three one-year cycles (Completers).
All participants who discontinued mAbs for any reason were defined as Drop-outs. The Drop-out group included participants who early discontinued treatment for: (i) ineffectiveness; (ii) lost to follow-up; (iii) sustained effectiveness; or (iv) non-serious adverse events, or de novo appearance of a contraindication.
We planned two separate per-protocol analyses to detail:
the reasons and timing of treatment discontinuation in Drop-outs; MMDs reduction, residual MMDs and responder rate in Completers across the three one-year cycles.
Statistical analysis
A formal sample size calculation was not performed, but we estimated a proper sample size according to data from literature regarding real-world studies with up to two years of follow-up (25,31). These studies enrolled 164 and 178 subjects, respectively. Our final population is comparable to previous studies. Finally, an a posteriori power analysis considering the observed reduction in MMDs showed a power above 95% (G*Power, version 3.1.9.6; Kiel University, Kiel, Germany). Statistical analysis was conducted using SPSS, version 21 (IBM Corp., Armonk, NY, USA). Normality was tested with the Kolgorov–Smirnov test; several variables showed a non-normal distribution; thus, we adopted non-parametric tests for the analyses.
Categorical data are reported as absolute numbers and percentages, with continuous variables reported as mean with 95% confidence intervals (CI) [5% CI - 95% CI].
Categorical variables were analyzed with the Chi-square test, Fisher's exact test or McNemar test, as appropriate. Continuous variables were analyzed with Mann–Whitney U-test or Kruskall–Wallis test.
The primary ITT analysis was performed using an LMM to detail the estimated marginal means of MMDs reduction from the timepoint observed and the baseline value of C1, as well as the residual MMDs across the three one-year cycles. The LMM included as fixed factors: (i) factor CYCLE (C1, C2 and C3); (ii) factor MONTHS (follow-up visits within each one-year cycle, namely M0 = Cstart – M3 – M6 – M9 – M12 = Cend); and (iii) the interaction CYCLE × MONTHS. The LMM also included age, sex, presence of any psychopathological disorder and overall months of follow-up (number of months under mAbs treatments + months of suspension until three-year period completion or mAbs discontinuation) as fixed factors. We included a random subjects intercept to account for inter-individual variability. We used a restricted maximum likelihood estimation method and, in accordance with repeated measures design, we used an autoregressive – AR(1) – covariance structure. Factor MONTHS detailed the change in MMDs independently from the cycle of treatment, whereas factor CYCLE detailed the average MMDs within each specific one-year treatment period. Separate analyses were considered only in case of significant CYCLE × MONTHS interaction. The p-values were corrected within each LMM and, in addition, according to Bonferroni's method to account for multiple comparisons. Two sensitivity analyses using a multiple imputation by means of a predictive mean matching and last observation carried forward methods, confirmed the validity of the results of the LMM analysis.
For the first per-protocol analysis, we adopted a Kaplan–Meier approach to detail the causes of premature treatment interruption. In the second, we applied the previously described LMM including only those subjects who completed the overall three one-year cycles. For all the performed tests, the level of significance was set at α = 0.050.
Results
Study population
The study population included 179 subjects (75.4% females, 51.3 [49.2–53.4] years), with disease onset at 15.6 [14.3–16.9] years). The majority of them (96.1%) had a diagnosis of CM, with a history of chronicity of 14.2 [12.6–15.8] years). Within the CM population, 90.7% of subjects (156/172) fulfilled the IHS criteria for acute medication overuse (MO) (29). The subjects enrolled had previously failed 5.1 [4.9–5.4] classes of preventive medications, and 36.9% of them were still taking a preventive treatment at baseline.
One hundred seventeen individuals were treated with erenumab 140 mg subcutaneously (s.c) per month, 53 with galcanezumab 120 mg s.c per month, with a loading dose of 240 mg at C1start. Nine individuals were treated with fremanezumab 225 mg s.c. monthly or 675 mg quarterly.
The most prevalent comorbidities collected through medical records were psychopathological disorders (29.6%), predominantly represented by anxiety disorders (15.1%) or associated anxiety and mood disorders (12.3%). Individuals also reported insomnia (19.6%) or controlled arterial hypertension (10.6%). Detailed clinical and demographic features among study groups are reported in Table 1.
Clinical and demographic features.

Legend: BoNT-A = onabotulinum toxin type A; CM = chronic migraine; CM + MO = chronic migraine associated with medication overuse; HIT-6 = Headache Impact Test-6; MIDAS = Migraine Disability Assessment; NSAIDs = non-steroidal anti-inflammatory drugs. Continuative data are presented as means [95% confidence interval], whereas categorical variables are presented as absolute values (%). Statistical analysis was conducted with the Mann–Whitney test for independent samples for continuous variables that demonstrated a non-normal distribution and one-way ANOVA for continuous variables that demonstrated a normal distribution. A Chi-square test was used for categorical variables. Significant values are indicated in bold.
Of the starting population, 43.6% (n = 78) completed the overall three one-year cycles (Completers group), whereas 56.4% (n = 101) did not complete the three consecutive one-year treatment periods due to different reasons further detailed in the text (Drop-out group). The study flowchart is graphically represented in Figure 2.
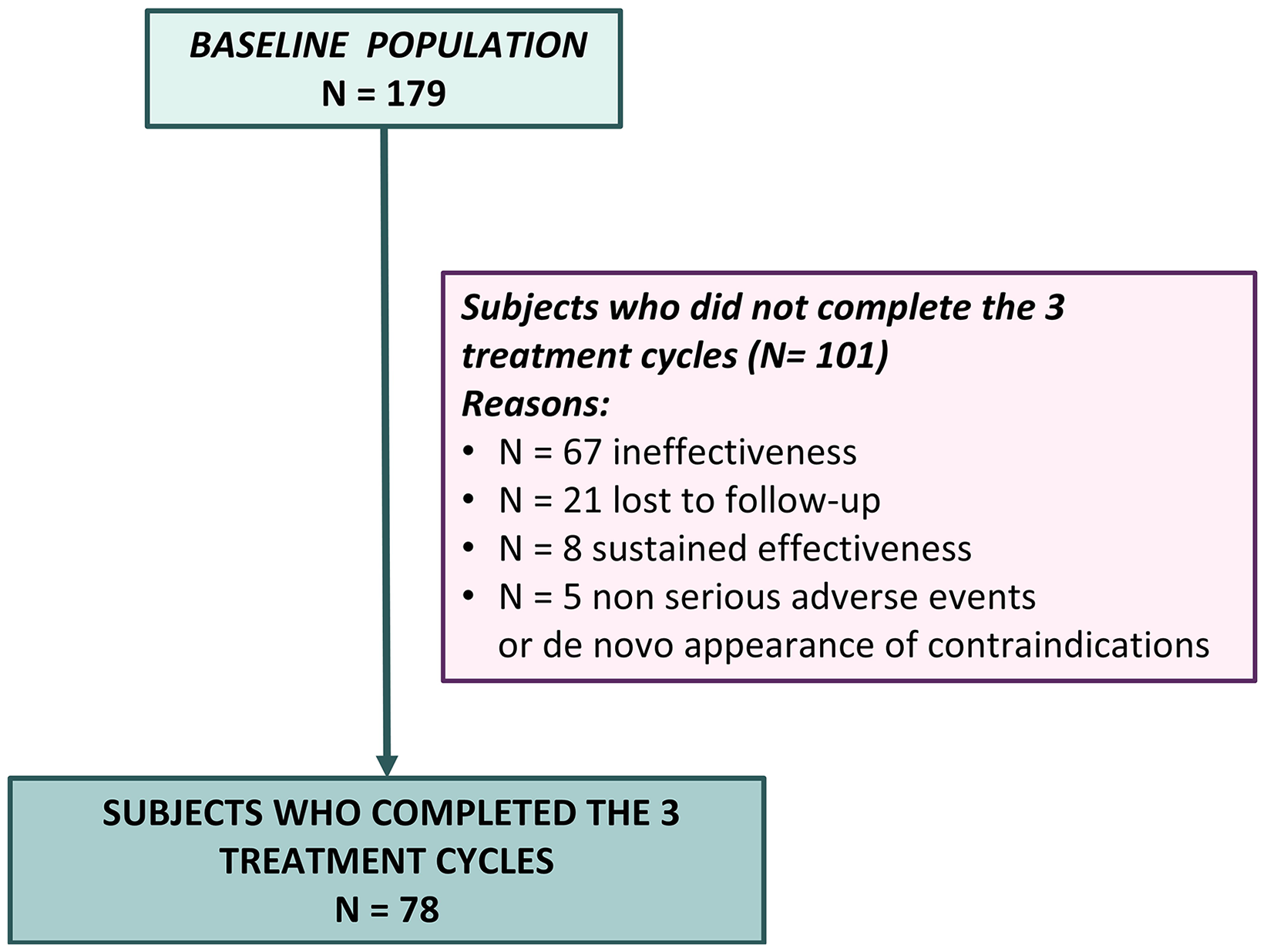
Flowchart of enrolled subjects.
ITT analysis
Analysis of monthly migraine days across the three one-year treatment cycles
In the ITT analysis, MMDs consistently decreased from baseline in each cycle, from 21.1 [19.8–22.4] to 9.6 [8.3–11.0] in C1, from 19.0 [17.4–20.5] to 9.6 [8.1–11.1] in C2, and from 15.9 [14.3–17.5] to 8.5 [6.9–10.1] in C3 (factor MONTHS: F = 212.457, p = 0.008; CYCLE × MONTHS interaction: F = 4.430, p = 0.008) (Figure 3).

Monthly migraine days (MMDs) across the three one-year treatment cycles according to the intention to treat analysis. Comparison between average number of MMDs at the beginning of each cycle are reported. Legend: C1 = first one-year cycle of treatment; C2 = second one-year cycle of treatment; C3 = third one-year cycle of treatment; M3 = three months from treatment initiation or re-initiation; M6 = six months from treatment initiation or re-initiation; M9 = nine months from treatment initiation or re-initiation.
The average MMDs were 12.2 [11.1–13.4] across C1, 11.5 [10.3–12.7] across C2 and 10.4 [9.2–11.7] across C3 (factor CYCLE: F = 8.288, p = 0.008) (see supplementary material, Table S1). Overall, across the three cycles of treatment, individuals reported 11.4 [10.3–12.5] MMDs.
MMDs decrease during each cycle (C1, C2 and C3) was already significant after the first three months of treatment and remained stable throughout the entire cycle (factor MONTHS: F = 3.868, p = 0.072).
No differences were detected when comparing MMDs at M3 (p = 0.677), M6 (p = 0.184), M9 (p = 0.344) and M12 (p = 0.581) of each cycle.
The average number of MMDs at the beginning of each cycle showed a gradual decrease across the three consecutive cycles. MMDs at C2start and at C3start were lower compared to C1start (C1start vs. C2start p = 0.008; C1start vs. C3start p = 0.008). MMDs at C3start were also lower compared to C2start (C2start vs. C3start p = 0.016) (Figure 3).
The entity of MMDs decrease from C1start was comparable among C1, C2 and C3 (CYCLE × MONTHS interaction: F = 0.702, p = 0.648). MMDs reduction was −12.7 [−11.4 – −14.1] during C1, −12.4 [−11.0 – −13.8] during C2 and −12.9 [ −11.4 – −14.3] during C3 (factor CYCLE: F = 0.753, p = 0.472), with an average reduction in MMDs across the three-year treatment period of −12.7 [ −11.4 – −14.0]. MMDs reduction across the three cycles is graphically represented in Figure 4 and detailed in the supplementary material (Table S1).
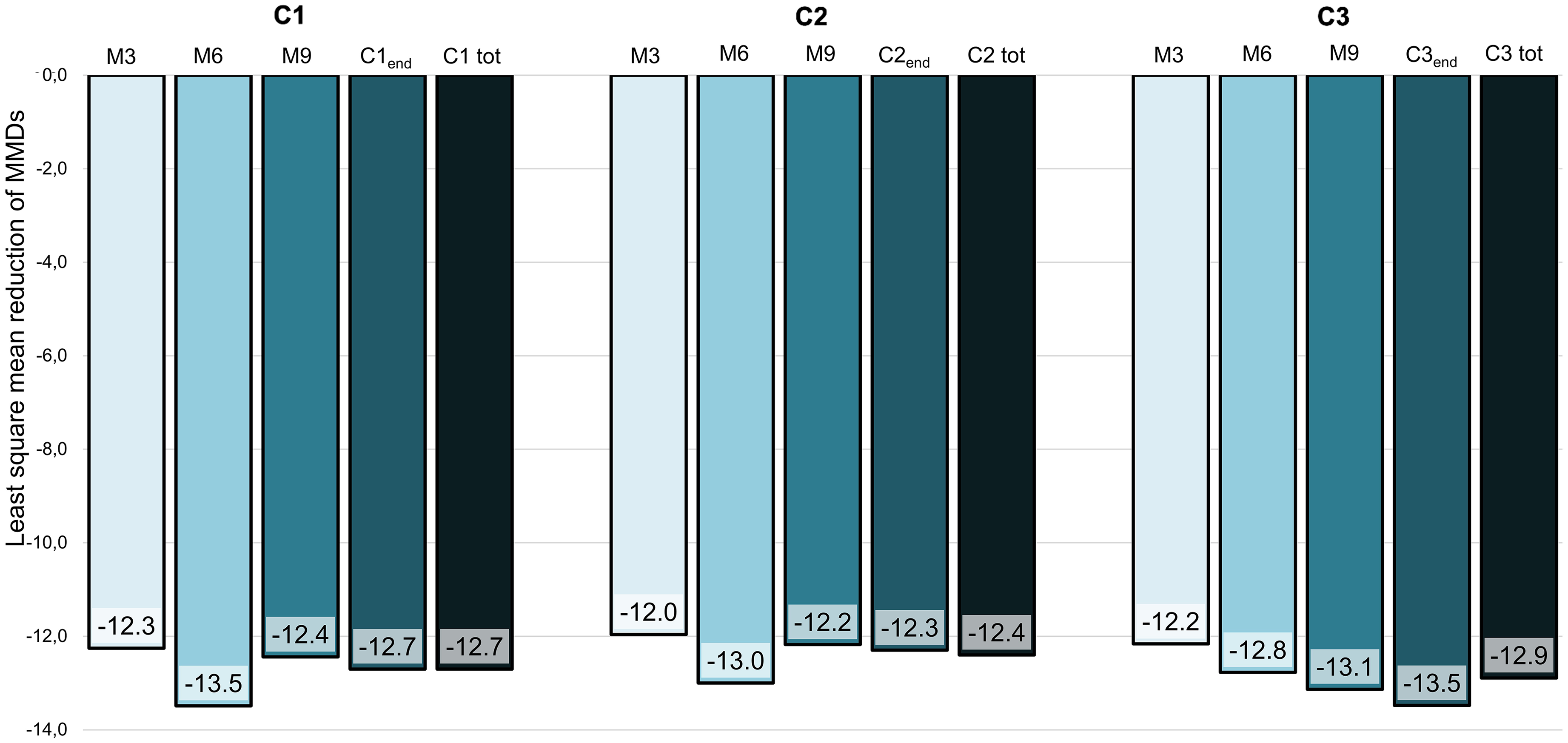
Monthly migraine days reduction during quarterly timepoints compared to C1start across the three one-year treatment cycles according to the intention to treat analysis. Legend: C1 = first one-year cycle of treatment; C2 = second one-year cycle of treatment; C3 = third one-year cycle of treatment; M3 = three months from treatment initiation or re-initiation; M6 = six months from treatment initiation or re-initiation; M9 = nine months from treatment initiation or re-initiation.
The suspension period between C1 and C2 (S1) lasted 4.9 [4.4–5.5] months), whereas that between C2 and C3 (S2) lasted 3.8 [3.3–4.3] months. At a post-hoc analysis, MMDs showed a worsening during both suspension periods (C1end vs. C2start p = 0.008; C2end vs. C3start p = 0.008).
Responder rate analysis
In the ITT analysis, the rate of 50% responders varied across cycles (p = 0.002). The 50% responders at C1end were 48.0% (86/179), at C2end were 38.0% (68/179) and at C3end were 38.5% (69/179).
When considering the 50% response over the three cycles, 55.9% (100/179) of subjects qualified as 50% responders in at least one cycle, of whom 15.6% (28/179) were Responder in only one, 11.2% (20/179) in two and 29.1% (52/179) in all the three cycles (Figure 5).

Fifty-percent response rate across the three one-year treatment cycles. Fifty-percent response rate was defined as subjects achieving at least a 50% reduction in MMDs at C1end, C2end and C3end compared to C1start. Individuals who did not restart treatment due to sustained effectiveness were considered Responders. Drop-outs for any other reason were considered as Non-responders.
Compared to Non-responders, the 50% Responders presented a lower prevalence of psychopathological comorbidities (namely anxious and mood disorders) (p = 0.020) and a lower chance of concomitant preventive treatment at baseline (p = 0.004).
The rate of 30% responders varied among cycles (p = 0.001). The 30% responders at C1end were 62.0% (111/179), at C2end were 46.9% (84/179) and at C3end were equal to 44.7% (80/179).
When considering the 30% response over the three cycles, 60.3% (108/179) of subjects qualified as 30% responders in at least one cycle, of whom 18.4% (33/179) were Responder in only one, 8.4% (15/179) in two and 39.7% (71/179) in all the three cycles.
Compared to Non-responders, the 30% Responders were characterized by a lower number of previously failed preventive treatments (p = 0.008), a lower prevalence of psychopathological comorbidities (p = 0.032), a lower chance of concomitant preventive treatment at baseline (p = 0.004) and lower doses of monthly acute drug intake at baseline (p = 0.024).
Per-protocol analyses
Survival analysis and in-depth analysis of reasons for drop-out
Out of the 179 enrolled subjects, 101 did not complete the three one-year cycles. The Kaplan–Meier analysis demonstrated that the number of injections differed across reasons for discontinuation (p = 0.008). Out of the 101 subjects who discontinued treatment, 66.3% (n = 67) suspended for ineffectiveness after a mean of 10 [7.1–12.9] injections; 20.8% (n = 21) were lost to follow-up after a mean of 19 [13.1–24.9] injections; 7.9% (n = 8) showed a sustained effectiveness after a mean of 15 [9.5–20.5] injections; finally 5.0% (n = 5) stopped for non-serious adverse events, or de novo appearance of a contraindications, after a mean of 12 [9.8–14.1] injections. Individuals who discontinued treatment for ineffectiveness were characterized by a higher number of previously ineffective preventive treatments (p = 0.002), a higher rate of ongoing preventive treatment at baseline (p < 0.001), and a higher number of baseline MMDs (p = 0.007) and monthly days of acute drug intake (p = 0.040) compared to subjects who discontinued for any other reason.
Analysis of monthly migraine days across the three one-year treatment cycles in completers group
Out of 179 participants, 78 completed the overall three one-year treatment cycles (Completers group). In this per-protocol analysis, we detailed the absolute number of MMDs, as well as their reduction, across the three cycles within the Completers group. We presented the estimated marginal means calculated with the LMM, as previously described.
Within the Completers group, MMDs consistently decreased during each cycle (from 22.6 [ 20.9–24.3] to 8.2 [ 6.5–9.8] in C1, from 17.8 [16.0–19.5] to 8.4 [6.8–10.1] in C2, from 14.9 [13.2–16.6] to 7.6 [ 5.9–9.2] in C3; factor MONTHS: F = 151.428, p < 0.001; CYCLE × MONTHS interaction: F = 7.160, p < 0.001).
The average MMDs were 11.4 [10.1–12.7] during C1, 10.4 [9.1–11.7] during C2 and 9.5 [8.2–10.8] during C3 (factor CYCLE: F = 10.913, p < 0.001). The overall number of MMDs across the three treatment cycles was 10.4 [9.2–11.6].
The average number of MMDs at the beginning of each cycle showed a gradual decrease across the three consecutive cycles. MMDs at C2start and at C3start were lower compared to C1start (C1start vs. C2start p < 0.001; C1start vs. C3start p < 0.001). MMDs at C3start were also lower compared to C2start (C2start vs. C3start p = 0.010).
The entity of MMDs decrease was comparable among C1, C2 and C3 (CYCLE × MONTHS interaction: F = 0.700, p = 0.649). MMDs showed a reduction of −12.9 [10.0 – −14.4] during C1, −12.3 [−10.1 – −14.5] during C2 and −12.7 [−10.5 – −14.9] during C3 (factor CYCLE: F = 0.717, p = 0.488). The average MMDs reduction across the 3-year treatment period was −12.4 [−10.3 – −14.5].
The reduction in MMDs was higher in the Completers group (−12.4 [−10.3 – −14.5]) and in the Drop-out group (−11.7 [−12.5 – −10.8]) compared to participants who discontinued due to ineffectiveness (−6.4[−6.8 – −6.0]) (p = 0.001 for both comparison).
MMDs at the beginning and at the end of each cycle and MMDs reduction are reported in the supplementary material (Table S2).
Response rate analysis
In the Completers group, the rate of 50% responders was stable across cycles (p = 0.779). The 50% responders were 75.6% at C1end (59/78), 74.4% at C2end (58/78) and 78.2% at C3end (61/78).
When considering the 50% response over the three cycles, 92.3% (72/78) of subjects qualified as 50% responders in at least one cycle, of whom 12.8% (10/78) were Responder in only one, 23.1% (18/78) in two and 56.4% (44/78) in all the three cycles.
The rate of 30% responders was stable across cycles (p = 0.807). The 30% responders were 91.0% at C1end(71/78), 93.6% at C2end (73/78) and 92.3% at C3end (72/78).
When considering the 30% response over the three cycles, 98.7% (77/78) of subjects qualified as 30% responders in at least one cycle, of whom 1.3% (1/78) were Responder in only one, 16.7% (13/78) in two and 80.8% (63/78) in all the three cycles.
In the Completers per-protocol analysis, no predictors of 30% or 50% response were identified.
Safety and tolerability analysis
Sixty-nine subjects (38.5%) reported at least one non-serious adverse event. The most prevalent adverse events were constipation (24.6% - 44/179), local cutaneous reactions (10.1% - 18/179) and fatigue (11.2% - 20/179). Only five subjects discontinued the treatment due to adverse events. No serious adverse events were reported.
Discussion
In this real-world, open-label study, 179 people with migraine were treated with CGRP targeting mAbs for three consecutive one-year cycles to evaluate MMDs changes. The number of consecutive treatment cycles evaluated, and the adoption of a conservative ITT analysis to assess the results, represent novelties and peculiarities of our study.
Our results can be summarized as follows: (i) the number of MMDs decreased during each treatment cycle with a meaningful reduction in the first three months, further reaching stable values (C1end: 9.6, C2end: 9.6, C3end: 8.5); (ii) the entity of MMDs reduction was comparable among the three treatment cycles (C1: −12.7, C2: −12.4, C3: −12.9); (iii) after correction for age, sex, follow-up duration and the presence of psychopathological disorders, we found a global reduction of −12.7 MMDs across the three cycles, with a residual number of 11.4 MMDs; and (iv) the 50% responder rate slightly decreased across the three cycles (C1end: 48.0%, C2end: 38.0%, C3end: 38.5%).
The ITT analysis also showed a worsening of MMDs during both suspension periods, although the number of MMDs at the beginning of C2 and C3 showed a gradual decrease compared to C1start.
These findings are consistent with real-world evidence of a rapid migraine worsening during suspension periods, still not reaching pre-treatment values (16,19–23,25). It should be noted, however, that the suspension period was quite short (4.9 months for S1 and 3.8 months for S2) with the possibility to restart treatment as soon as a meaningful worsening of migraine frequency was detected. Thus, it is reasonable that this observation is explained by an early mAbs re-initiation per clinical decision, rather than a disease modifying effect of these novel drugs. Despite migraine worsening during suspensions, mAbs demonstrated a comparable effectiveness across the three cycles. MMDs presented a reduction soon after the first three months of treatment, followed by stable values throughout the whole one-year cycle. This evidence is consistent with previous literature showing a similar fast mAbs benefit during the first and second treatment cycles despite the related “drug-holiday” (14,16,18,21–23,25).
We also observed a slight response-rate decrease across the three cycles. This evidence deserves a careful interpretation as, contrary to previously cited studies, we adopted a very conservative statistical method. Indeed, in the ITT analysis we classified the Drop-outs as Non-responders. The growing number of Drop-outs across the three cycles determined a consequent reduction in the response rate. Notably, despite the adoption of such a rigorous statistical analysis, we only observed a slight response-rate reduction with a maintenance of 50% response in approximately two out of five individuals across the three one-year treatment cycles. An even stronger data is the evidence of a high consistency of 50% response rate across the three cycles.
Interestingly, the reduction in MMDs was similar when applying the per-protocol analysis on the 78 subjects who completed the three treatment cycles, but the percentage of 50% responders was markedly higher (C1end: 75.6%, C2end: 74.4%, C3end: 78.2%) compared to the ITT analysis. Indeed, our ITT analysis showed a lower 50% response rate compared to previous long-term open-label studies (OLE) with similar follow-up periods that used per-protocol approaches (4,11,12). The response rate of our ITT analysis was also lower compared to most of the previously conducted real-life observational studies that adopted per-protocol designs (14,17,19–21,23–26,32).
For example, the recently published I-GRAINE study provided an overview of mAbs effectiveness across a three-year period by means of a per-protocol analysis (26). Out of the 2064 individuals with high frequency or chronic migraine enrolled at the beginning of the study, only 212 participants completed the three one-year treatment cycles. Noteworthy, the 50% responder rates were 92.9% at the end of the first year of treatment, and 96.2% at the end the third treatment cycle.
By contrast, Andreou et al. (31) assessed response rate across two years of mAbs treatment by means of a ITT analysis showing a 50% response rate of 24% (38/160).
We also acknowledge that in previous reports drop-outs may have occurred for several reasons, including adverse events, loss to follow-up and sustained effectiveness, and do not necessarily represent only individuals who failed mAbs treatment. Thus, the presence of drop-outs may not always lead to a clear overestimation of effectiveness even in studies without an ITT approach.
According to our data, subjects who stopped for ineffectiveness presented a higher number of previously failed and ongoing preventive treatments compared to those who completed the three cycles, as well as higher baseline migraine frequency. The same was true for 30% responders in the ITT analysis. Another relevant finding is the lower prevalence of psychopathological disorders (namely anxious and mood disorders) in 30% and 50% responders.
The first evidence reflects previous literature data, well summarized in a recent metanalysis (33) and narrative review (34), showing that mAbs response is less likely to occur in those individuals who previously failed a higher number of preventive treatments. The evidence that migraine frequency could influence response was also supported by two recent large real-world studies, suggesting that an early mAbs treatment could be more effective (35,36). Finally, the previous literature supports our findings on the role of psychopathological disorders as negative predictors of response (33,37,38).
Intriguingly, despite a high entity of MMDs reduction over a prolonged treatment schedule, at the end of each cycle, the residual MMDs were consistently above eight. This result is confirmed in both the ITT and per-protocol analyses. The lack of a cumulative MMDs reduction over a two-year treatment follow-up has been previously reported in real-world studies (23,31). A possible pathophysiological explanation could be the persistence of sensitization processes despite CGRP antagonism. Indeed, the CGRP pathway is just a part of the complex (and still far from clear) mechanisms involved in migraine pathogenesis. We can thus speculate that acting on the CGRP alone may not be enough to obtain further reduction of migraine frequency as other pathways are likely to be concomitantly involved (39). This evidence raises the need for the identification of different dysfunctional pathways in migraine individuals. Longer treatment periods, as well as the availability of migraine treatments directed to different pathways (peptides, endocannabinoid system, ion channels, hormones, to name a few) (40), are needed to confirm this speculation.
To our knowledge, this is the first real-life evaluation of monthly migraine days during three consecutive one-year cycles that applies an ITT analysis for assessing mAbs effectiveness.
We must acknowledge some limitations of this study. First of all, our population mainly included difficult-to-treat individuals with CM-MO, and this can be attributed to the fact that the study was conducted in tertiary headache centers, where subjects with more complex and treatment-resistant conditions are typically referred. Furthermore, our analyses were carried out on the first individuals treated, who were usually those individuals with greater needs and more severe conditions. Further evaluation of subjects with high frequency episodic migraine is needed to confirm our conclusions. We also need to consider the limitation imposed by the Italian regulation that allowed mAbs restart only in those individuals presenting with more than eight MMDs, determining a potential underestimation of responders. We also considered a population mostly treated with erenumab and galcanezumab because they were the first available in our country, so the data are not completely generalizable to all currently available mAbs treatments. Moreover, it should be considered that the Italian treatment schedule, which foresees a mandatory suspension period after one-year treatment, may intrinsically carry a higher risk of dropping-out.
On the other hand, the ITT analysis, the prolonged period of assessment and the diary-based data prospectively collected represent relevant strengths of the study.
Conclusions
This real-world study confirms sustained effectiveness across three one-year cycles of mAbs treatment and two related suspensions. The ITT analysis revealed a marked and comparable entity of MMDs reduction across three one-year cycles, with an overall decrease from baseline of −12.7 MMDs, but with a relevant burden of disease in terms of residual MMDs. Our results underscore the importance of the identification of potential alternative concomitant non-CGRP dependent dysfunctional pathways in migraine individuals.
Article highlights
Monoclonal antibodies directed against the CGRP pathway induced a meaningful and sustained reduction in monthly migraine days (MMDs) across three one-year treatment cycles in a cohort of 179 individuals.
According to the intention-to-treat analysis, the MMDs reduction was −12.7 with a 8.5 residual MMDs at the end of the third year.
At the end of each year of treatment, residual MMDs were persistently >8, revealing a remaining high burden of disease and the need for exploring other non-CGRP dependent treatments.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251353421 - Supplemental material for Real world effectiveness of anti-CGRP monoclonal antibodies over three consecutive one-year treatment cycles: An intention-to-treat analysis
Supplemental material, sj-docx-1-cep-10.1177_03331024251353421 for Real world effectiveness of anti-CGRP monoclonal antibodies over three consecutive one-year treatment cycles: An intention-to-treat analysis by Gloria Vaghi, Michele Corrado, Maria Magdalena Pocora, Federico Bighiani, Francescantonio Cammarota, Alessandro Antoniazzi, Luca Costantino, Daniele Martinelli, Marta Allena, Natascia Ghiotto, Elena Guaschino, Sara Bottiroli, Luigi Francesco Iannone, Francesco De Cesaris, Danilo Antonio Montisano, Licia Grazzi, Grazia Sances, Cristina Montomoli, Cristina Tassorelli and Roberto De Icco in Cephalalgia
Footnotes
Acknowledgements
We thank the Research Nurse Team and the clinical staff of the Headache Centers of IRCCS Mondino Foundation for their precious assistance in all of the activities.
Author contributions
RDI and GV: Study concept and design; Subjects enrollment; Data collection; Statistical analysis; Interpretation of data; Writing the first draft. MC, MMP, FB, FC, AA, LC, DM, MA, NG, EG and SB: Subjects enrollment; Revision of the manuscript for content. LFI, FDC, DAM and LG: Subjects enrollment; Data Collection; Data interpretation; Revision of the manuscript for content. CM: Critical reappraisal of the manuscript, statistical analysis. GS and CT: Study concept and design; Subjects enrollment; Interpretation of data; Drafting/revision of the manuscript for content. All authors read and approved the final version of the manuscript submitted for publication.
Data availability
The dataset analyzed during the current study is available in the Zenodo repository with the DOI: 10.5281/zenodo.14260579. The dataset is available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article (past 4 years). GV received personal fees for participating in speaking at scientific events from Lundbeck. MC, FB, FC, AA, LC, SB and CM: None. MMP reports travel grants from Teva. DM reports support from Abbvie for participating in advisory boards; consulting fees from LifeSciences Consultants, fees from Medscape for organizing educational programs; personal fees from Lundbeck for lecturing at symposia, travel grants from Pfizer. MA received personal fees as speaker from TEVA. NG received personal fees as speaker from Eli Lilly, Pfizer and Lundbeck. EG received personal fees as speaker from Lundbeck and Novartis. LFI received honoraria for scientific presentations and travel fee from TEVA, Eli Lilly, Pfizer, Organon, Abbvie and Lundbeck. Research founding from the International Headache Society. FDC received speaker's fees for TEVA, Lundbeck and Novartis. DAM received speaker's fees from Lundbeck. LG received consultancy and advisory fees from: Abbvie; Eli Lilly; Lundbeck; Teva; Pfizer; Moreover Collaborator for RCTs sponsored by: Eli Lilly; TEVA Pharm Ind. GS received personal fees as speaker or Advisory Board from: Eli Lilly, TEVA, Lundbeck, Pfizer and travel grants from TEVA. CT reports support from Abbvie and Novartis for investigator-initiated trials; consulting fees from Abbvie, Eli Lilly, Dompé, Ipsen, Lundbeck, Pfizer, TEVA and Medscape for participating in advisory boards; support from Abbvie, Eli Lilly, Dompé, Ipsen, Lundbeck, Pfizer and TEVA for attending meetings; and personal fees from Abbvie, Eli Lilly, Lundbeck, Pfizer and TEVA for lecturing at symposia. She is principal investigator of clinical trials sponsored by Abbvie, Biohaven, Eli Lilly, Ipsen, Lundbeck, Pfizer and TEVA. She has received research grants from the European Commission, the Italian Ministry of Health and the Migraine Research Foundation. RDI received personal fees for participating in speaking at scientific events from Eli-Lilly, TEVA and Lundbeck. He has participated in advisory boards for Pfizer and AbbVie.
Ethical statement
The study was approved by the local Ethics Committee of Pavia, Italy (P-20190105434). All participants signed a written informed consent before enrollment. The study procedures were carried out following the guidelines for proper human research conduct in accordance with the Helsinki Declaration of the World Medical Association and its revisions.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Research Grant from the Italian Ministry of Health to IRCCS Mondino Foundation (Ricerca Corrente 2025–2027).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
