Abstract
Background
Calcitonin gene-related peptide has shown to play a central role in cluster headache (CH) pathophysiology. A clinical trial with galcanezumab was carried out in chronic cluster headache (CCH) but did not meet its primay endpoint. However, its off-label use in patients with CCH refractory to other therapies could be considered. We aimed to asses the efficacy and safety of galcanezumab as CCH preventive treatment in a real-life setting.
Methods
An observational study was conducted. Patients with CCH who received at least one dose of 240 mg of galcanezumab.
Results
Twenty-one patients who tried a mean of 6.3 ± 1.9 preventive therapies, including onabotulinumtoxinA in 90.5%. At baseline, the median of frequency was 60 (37.5–105) monthly attacks with 10 (8.3–10) points in pain intensity (Numerical Rating Scale). After one month, the frequency decreased to 31 (10.5–45) (p = 0.003) with 8.5 (8–9.5) intensity (p = 0.007); 10 (47.6%) patients were 50% responders of whom four (19%) were 75% responders. Of the 15 patients with 3 months of follow-up, seven (46.6%) reduced their frequency by 50% and four (26.6%) by 75%, with 40 (10–60) monthly attacks (p = 0.07) and pain intensity of 8 (5–10) (p = 0.026). Some 52% patients experienced adverse events, mostly mild.
Conclusions
In our cohort of refractory CCH, galcanezumab was effective in almost 50% of patients. This finding supports individual off-label treatment attempts.
Introduction
Cluster headache
Cluster headache (CH) belongs to the trigeminal autonomic cephalalgias (TAC) group. It is characterised by episodes of intense, strictly unilateral pain lasting 15–180 minutes, accompanied by ipsilateral trigeminoautonomic symptoms (conjunctival injection, lacrimation, nasal congestion, rhinorrhoea, eyelid oedema, forehead and facial sweating, miosis and/or ptosis) and/or restlessness. They may have several attacks on the same day with a circadian pattern, predominantly in the evening. The International Classification of Headache Disorders, 3rd edition (ICHD-3) (1) distinguishes two forms. In episodic cluster headache (ECH), attacks occur separated by periods of pain-free remission lasting ≥3 months. The chronic form is defined by the presence of cluster headache attacks lasting one year or more without remission or with periods of remission lasting less than three months.
The prevalence of CH is about one per 1000 in the general population, with a male:female ratio of about 2.5:1 (2). The episodic form is six times more common than the chronic form (3). Chronic cluster headache (CCH) may be chronic from the onset or develop from an episodic form. After 10 years, about one-third of patients with ECH will progress to CCH, and the same proportion will improve from CCH to ECH (4).
It is crucial to have adequate treatments that allow us to prevent patients with ECH from chronification and to ensure that those with CCH can minimise the number of attacks they suffer.
We should bear in mind that CH, especially in its chronic form, is one of the most disabling pain syndromes, with significant impact on daily, work and social activities, in addition to the resulting major emotional impact (5).
Preventive treatment for chronic cluster headache
According to the recommendations of the European Federation of Neurology, verapamil is the drug with the highest level of evidence for CH. Other effective preventive treatments include lithium, topiramate, melatonin, valproate and baclofen (6). They all require slow titration to control side effects such as bradycardia (verapamil) or cognitive effects and weight loss (topiramate). Lithium requires frequent monitoring as a result of the risk of toxicity. All of this is a barrier to treatment adherence. OnabotulinumtoxinA is approved for the prevention of chronic migraine, although it may also be considered in patients with chronic cluster headache before invasive therapies are used (7).
For patients who are refractory to medical treatment, we can consider surgical options such as sphenopalatine ganglion (SPG) radiofrequency. A promising option for these patients is invasive neuromodulation, both peripheral such as occipital nerve stimulation (ONS) (8) or central with hypothalamic deep brain stimulation (9). However, these techniques are not without their own set of complications.
Despite all these therapeutic options, there is still a proportion of patients (not quantified) who are non-responders, which urges us to use all the tools at our disposal. In 2018, the European Headache Federation attempted to differentiate these patients with a specific section in the ICHD-3 for chronic refractory cluster headache (10). They defined it as at least three severe headache attacks per week that affect the patient's quality of life, despite adequate symptomatic and preventive treatment, and failure of at least three preventive treatment options.
The role of calcitonin gene-related peptide (CGRP) in cluster headache
CGRP is a 37-amino acid neuropeptide. It has been detected throughout the trigeminovascular system, in nerve fibres innervating the cranial vasculature, the trigeminal root and the trigeminal nucleus (11–15). Similar to migraine, CGRP plays an important role in the pathophysiology of CH (16). CGRP levels are elevated in jugular blood and tear fluid during active periods, both interictally and during the attack itself, and are reduced by effective symptomatic treatment such as oxygen therapy or triptans (which act in part by inhibiting CGRP release from trigeminal nerve fibres). In addition, CGRP infusion induces headache attacks in CH patients (17–19).
Evidence to support the use of galcanezumab for the prevention of cluster headache
Galcanezumab is a humanised immunoglobulin G4 monoclonal antibody that targets the CGRP molecule. Two randomised, double-blind, placebo-controlled phase III clinical trials with 300 mg subcutaneous (s.c.) monthly galcanezumab have been conducted in CH: one of them in ECH (20) and another one in CCH (21). Only the clinical trial of galcanezumab in ECH met its primary endpoints. As a result, the US Food and Drugs Authority approved galcanezumab for ECH in the USA in June 2019, whereas the European Medicines Agency rejected the indication for the prevention of cluster headache in February 2020. However, case series using galcanezumab for CCH have been published, suggesting that some of these patients, particularly those who are refractory to other therapies, may benefit from its use (22,23). Another real-life study analysed the response in cluster headache attack frequency in a patient with co-morbid migraine treated with galcanezumab, with similar positive results (24). Finally, results have recently been published from an extension phase clinical trial of galcanezumab in chronic cluster headache (25). This consists of a 52-week open-label treatment period and a 16-week washout period. The results confirm that, as previously shown in migraine, the safety profile of the drug is favourable and adverse events are mostly mild.
Objective
The present study aimed to analyse the response, in terms of efficacy and safety, of the off-label use of galcanezumab as a preventive treatment in patients with CCH who have failed other therapies. Although case series have limitations in their interpretation, given the disabling nature of this pathology, the difficulty of designing clinical trials in this population (26) and the discrepancy between the results of clinical trials vs real-life data, we consider that the information we can obtain from our clinical practice could be significant.
Methods
Population
Patients originated from the Headache Unit of Virgen del Rocío University Hospital (Seville, Spain), diagnosed with CCH according to ICHD-3 diagnostic criteria, with secondary causes excluded and fulfilling refractory CCH criteria (10). A monthly dose of 240 mg was administered in all cases.
Inclusion criteria were: (i) age of at least 18 years; (ii) diagnosis of CCH according to IHCD-III criteria; (iii) treatment of cluster headache with galcanezumab for at least one month; and (iv) ability to sign the informed consent.
It should be noted that the dose used in the clinical trials was 300 mg; however, we adjusted the dose to 240 mg (two prefilled pens of 120 mg) monthly because this is the only formulation available in Spain). Galcanezumab was offered on a compassionate basis within a public health system.
Data collection
A prospective descriptive study was conducted. Sociodemographic data and baseline characteristics were collected retrospectively from medical records. Efficacy (frequency, pain intensity, symptomatic treatment) and safety parameters were collected prospectively through clinical interviews and headache diaries.
Statistical analysis
Nominal (categorical) variables were presented as absolute values and percentages. Quantitative variables with a normal distribution were summarised using the mean ± SD, whereas non-normally distributed quantitative variables were described using the median and interquartile range. Statistical significance between baseline and the three-month follow-up was assessed using various methods: for quantitative variables, the Kolmogorov–Smirnov analysis was employed; for paired qualitative variables, the Cochran's Q-test was used. To identify trends over time, the Durbin–Conover test was applied. Adjustments for multiple comparisons were made using Bonferroni correction. Safety analyses, focusing on changes, were performed using McNemar's paired sample t-test. In cases where patients did not complete the three-month follow-up by the time of the present study, their data were considered as missing for the three-month analysis. p < 0.05 was considered statistically significant. Data analysis was performed using SPSS, version 23.00 (IBM Corp., Armonk, NY, USA) and R Project software (R Foundation, Vienna, Austria).
Results
Baseline characteristics of the population
We included 21 patients whose baseline characteristics are shown in Table 1. Most were male, with a mean age of 47.5 years. The mean time of evolution of their CH was approximately 12 years, with 6.5 years since chronification (42.9% had evolved from an episodic form). Most cases were right-sided and one patient had associated trigeminal neuralgia (cluster-tic). The average number of previous preventive treatments was 6.3, including onabotulinumtoxinA in 90.5% of cases. Thirteen (61.9%) had undergone repeated anaesthetic great occipital nerve (GON) blocks. In addition, eight patients had an occipital neurostimulator, two had occipital radiofrequency, two had SPG radiofrequency and one had bilateral GON section. Despite all these therapies, at baseline (Table 2), they still had an average frequency of 76.6 monthly attacks with a pain intensity of 8.9 points on the Numerical Rating Scale (NRS), which led to 61.9% of patients taking at least 10 days of triptans per month.
Baseline characteristics of the population.
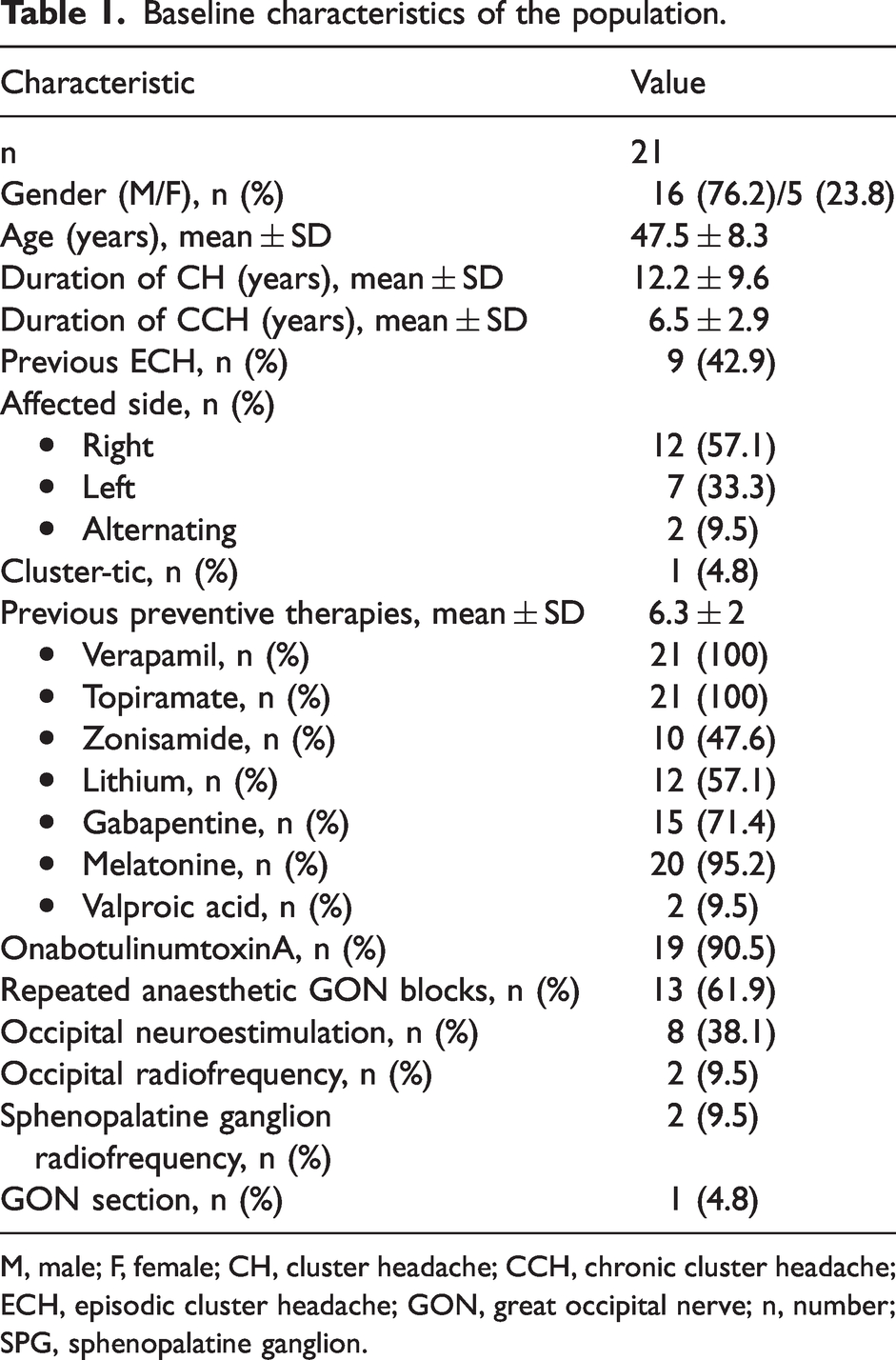
M, male; F, female; CH, cluster headache; CCH, chronic cluster headache; ECH, episodic cluster headache; GON, great occipital nerve; n, number; SPG, sphenopalatine ganglion.
Clinical status of patients at baseline.

n, number; IQR, interquartile range; NRS, Numerical Rating Scale.
Efficacy after one month of treatment
The results are summarised in Table 3.
Efficacy parameters after one month of treatment.

n, number; IQR: interquartile range; NRS, Numerical Rating Scale.
Of the initial 21 patients, one month after treatment (240 mg of galcanezumab), monthly attacks were reduced from a median of 60 to 31 (p = 0.003) and pain intensity from 10 to 8 points on the NRS (p = 0.007). Patients taking triptans more than 10 days per month decreased from 61.9% to 33.3%. Considering patients who reduced their monthly cluster headache attacks by 50% as responders, 47.6% of the population were responders. In addition, 19% of patients reduced their frequency by at least 75%.
After that visit, six patients discontinued: two at their own request because of clinical improvement, two because of ineffectiveness (clinical worsening), one because of intolerance (severe constipation) and another one as a result of loss to follow-up. Therefore, we have three-month follow-up data on 15/21 initial patients.
Efficacy after 3 months of treatment
The results of 15 patients who completed three months of treatment are summarised in Table 4. We can see the improvement in frequency and pain intensity in Figures 1 and 2. To avoid confusion in the interpretation of the results, only the data from the 15 patients who reached three months of treatment were used.
Efficacy parameters after three months of treatment.

n, number; IQR, interquartile range; NRS, Numerical Rating Scale.
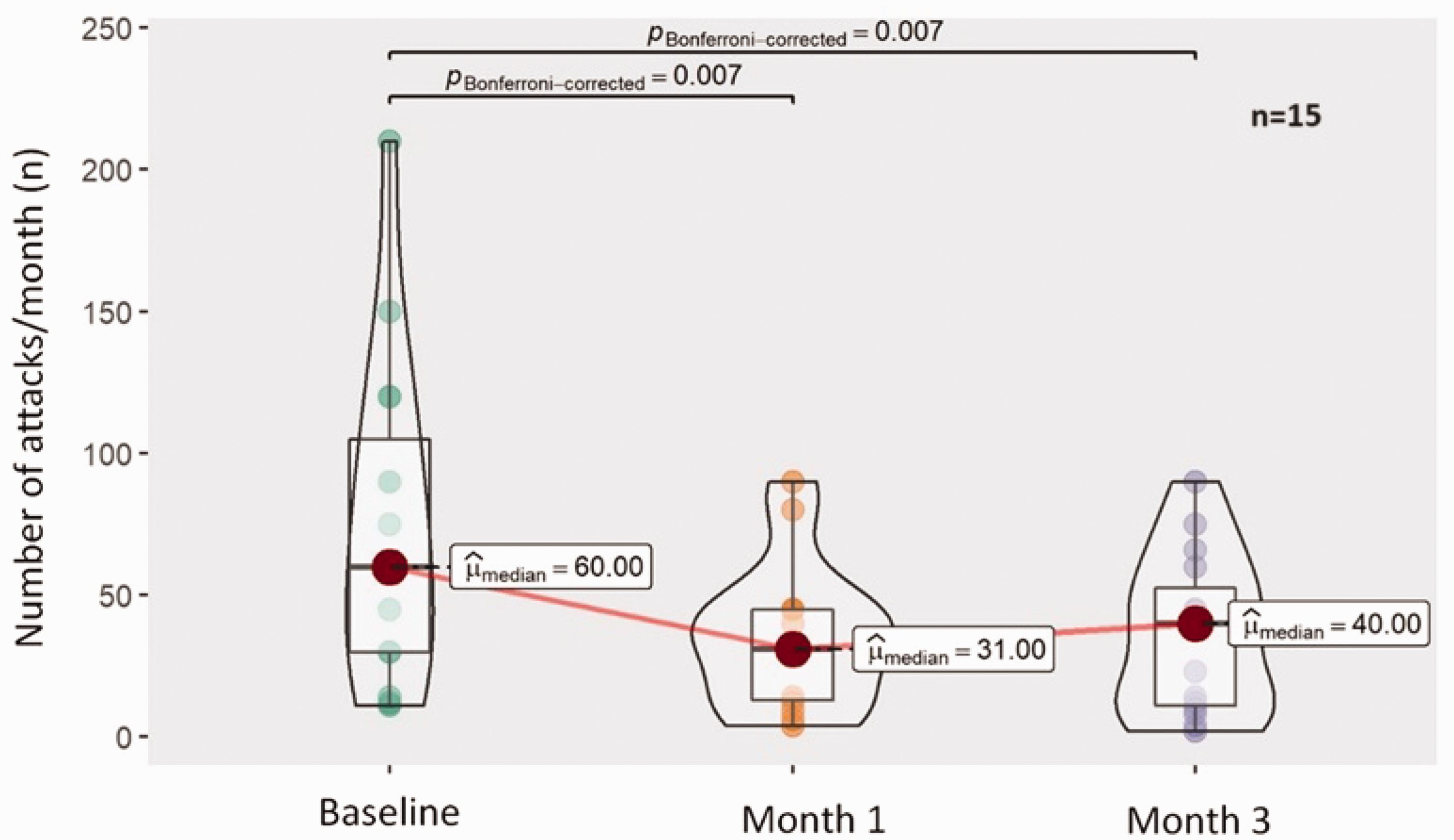
Frequency (attacks/month) of 15 patients who completed three months of treatment. Box-violin plot showing pairwise analysis with the Durvin–Conover test.

Pain intensity (Numerical Rating Scale (NRS)) of 15 patients who completed three months of treatment. Box-violin plot showing pairwise analysis with the Durvin–Conover test.
After three months of treatment, median frequency was 40 monthly attacks (p = 0.007), pain intensity was 8 points in NRS scale (p = 0.026) and only 27.7% of patients continued taking more than 10 days of triptans per month (p = 0.015). The responder rate, defined as a 50% reduction in monthly attacks, was 46.7%, with 20% of patients achieving at least a 75% frequency reduction. After that visit, another two patients dropped out of treatment: one because of poor adherence and one because the patient moved to another hospital.
The reasons for discontinuation are summarised in Table 5.
Reasons for treatment discontinuation.

Association of response with baseline population characteristics
No statistically significant relationship (p ≥ 0.05) was found between response parameters (frequency, intensity, triptan abuse, responder rate) and any of the baseline sociodemographic or clinical baseline characteristics (gender, age, duration of CH, duration of CCH, previous preventive treatments).
Safety of treatment
In total, seven different side effects were reported by 11 (52%) of the initial 21 patients. As shown in Figure 3, the most common was constipation (8.4%), which led to discontinuation in one patient. The following were flu-like symptoms, asthenia and diarrhea. No serious adverse events were reported.
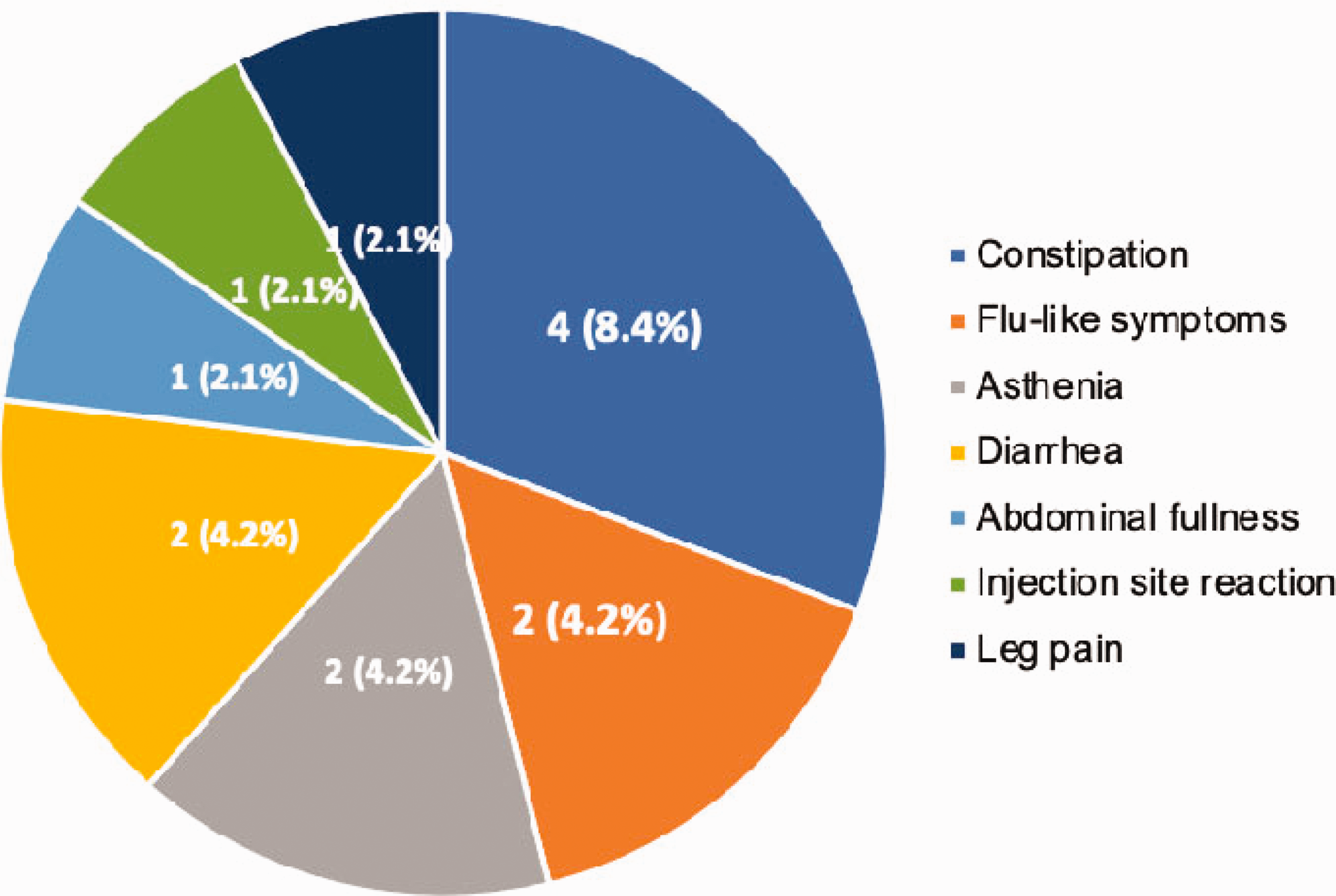
Adverse effects reported by patients. N (%) represents the number of patients who reported each adverse effect.
Discussion
The highest level of scientific evidence for the efficacy of a treatment comes from randomised, placebo-controlled clinical trials. In this case, the clinical trial of galcanezumab in patients with CCH was negative. However, we have to take into account that: (i) this is an isolated clinical trial; (ii) the design of clinical trials in this population is very difficult because of the specific characteristics of the pathology and must be carried out with special considerations; and (iii) the treatments recommended in clinical guidelines for the prevention of CH often do not have a high level of scientific evidence.
Descriptive studies not only have important limitations, but also great strengths. They are closer to the reality of our clinical practice, allowing the analysis of a population that is generally more refractory, with more comorbidity and concomitant treatments, and with fluctuations inherent in their pathology. Furthermore, descriptive studies allow for indefinite follow-up. On the other hand, the amount of missing data is greater, increasing the likelihood of bias. However, the sum of several descriptive studies in the same area can provide very valuable information and serve as a guide for specifically designed clinical trials.
In our case, although our population had a longer duration of disease and had failed a greater number of preventive treatments (including onabotulinumtoxinA and surgical treatments) regarding a randomised controlled trial (21), almost 50% of the sample responded to galcanezumab in terms of frequency (attacks/month) with statistically significant reductions in pain intensity and triptan abuse. These results are consistent with the study by Membrilla et al. (23) that included six patients with refractory CCH who received galcanezumab 240 mg s.c. monthly for three months, in which five (83.3%) reduced their attacks frequency by at least 50% and one patient reported a significant improvement in pain intensity. Similarly, in a study by Ruscheweyh et al. (22), which included 22 patients with CCH treated with galcanezumab 240 mg, erenumab 70 mg or erenumab 140 mg s.c. monthly, 55% of patients were responders with a significant reduction in both pain frequency and intensity.
In our analysis, we found a slight increase in the frequency of attacks between month 1 and month 3 of treatment, although we do not know whether this is because of a partial loss of effect of the drug or fluctuations inherent to the disease. Accordingly, studies with longer follow-up are needed to clarify this question.
It is also important to bear in mind that we used a dose of 240 mg monthly galcanezumab, which is lower than the clinical trial dose of 300 mg. We do not know whether using the randomised controlled trial dose would result in a similar response, or whether 240 mg would be insufficient in general population or in a population above a certain body mass index (we did not analyse the body mass index of our cohort).
As a limitation, we were ubnable to analyse the change in quality of life scales with treatment because many patients did not complete these scales at subsequent visits, making the analysis of no value.
Conclusions
In our cohort of 21 patients with CCH who had been refractory to an average of 6.3 previous preventive therapies, including in most cases onabotulinumtoxinA and various types of invasive treatments, almost 50% responded to 240 mg s.c. monthly galcanezumab in terms of frequency reduction. Pain intensity was also significantly reduced, with a median reduction of 2 points after three months of treatment. All of this led to a 50% reduction in triptan abuse. In most cases, the response started during the first month of treatment and was maintained until the third month.
Given that chronic cluster headache is one of the most disabling pain syndromes and that treatment options are limited, this finding could support individual off-label treatment attemps without major safety risks.
Clinical implications
Although clinical trial with galcanezumab in CCH was negative, real-world data suggest that it could be a therapeutic option for patients with poor response to other medications. One month after 240 mg of galcanezumab, almost 50% of patients became “responders”, with a significant reduction in pain intensity and triptan abuse. Adverse events were mostly mild, with the most common being constipation.
Footnotes
Declaration of conflicting interest
The authors declare that there are no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
The authors declare that ethics approval has been requested.
