Abstract
Background
To develop and validate an easy-to-use scoring system to predict the response to the first epidural blood patching in patients with spontaneous intracranial hypotension.
Methods
This study recruited consecutive patients with spontaneous intracranial hypotension receiving epidural blood patching in a tertiary medical center, which were chronologically divided into a derivation cohort and a validation cohort. In the derivation cohort, factors associated with the first epidural blood patching response were identified by using multivariable logistic regression modeling. A scoring system was developed, and the cutoff score was determined by using the receiver operating characteristic curve. The findings were verified in an independent validation cohort.
Results
The study involved 280 patients in the derivation cohort and 78 patients in the validation cohort. The spontaneous intracranial hypotension-epidural blood patching score (range 0–5) included two clinical variables (sex and age) and two radiological variables (midbrain-pons angle and anterior epidural cerebrospinal fluid collections). A score of ≥3 was predictive of the first epidural blood patching response, which was consistent in the validation cohort. Overall, patients who scored ≥3 were more likely to respond to the first epidural blood patching (odds ratio = 10.3).
Conclusion
For patients with spontaneous intracranial hypotension-epidural blood patching score ≥3, it is prudent to attempt at least one targeted epidural blood patching before considering more invasive interventions.
Keywords
Background
Spontaneous intracranial hypotension (SIH) is caused by spinal cerebrospinal fluid (CSF) leakage and the main symptom is acute disabling orthostatic headache, which may last for months or even years without adequate treatment (1,2). Delayed diagnosis and treatment could lead to prolonged disability and even potentially life-threatening complications, such as subdural hematoma, uncal herniation, brainstem ischemia, Duret hemorrhage or cerebral venous thrombosis (1,3–6). Based on relevant brain imaging findings, a scoring system (range 0–9), including the presence of engorgement of venous sinus, diffuse pachymeningeal enhancement, subdural fluid collection, suprasellar cistern ≤4 mm, prepontine cistern ≤5 mm, and mamillopontine distance ≤6.5 mm, was developed for patients with SIH to predict the likelihood of finding a CSF leak on spinal imaging with a score of ≥5 points (7). However, as the neuroimaging findings tend to be less severe in the earlier stages, a diagnosis of SIH should be highly suspected for patients who scored ≥3 within 17 days of disease onset (8). Such a scoring system is useful in facilitating the diagnosis and therefore preventing delays in the initiation of proper treatment.
The mainstay of treatment for SIH is epidural blood patching (EBP), especially when conservative treatment has failed (1,2,9–11). EBPs, particularly targeted EBPs, usually lead to rapid improvement due to changes in craniospinal CSF mechanics, as well as a more sustained effect resulting from tamponade and sealing of the dural defect (12,13). Overall, approximately 40–50% of patients with SIH require more than one EBP (11,14,15). As a result, factors that can be used to predict the response to the first EBP are of clinical importance. Several factors have been proposed in previous studies, including early bladder activity on radioisotope cisternography, diencephalic-mesencephalic deformity and descent of iter below the incisural line on brain magnetic resonance imaging (MRI), or a high neutrophil-to-lymphocyte ratio (10,16,17). However, the findings in different studies were inconsistent. In addition, some of the above factors involve a lumbar puncture, which could be technically challenging in patients with SIH and may potentially aggravate CSF leakage. In our previous study involving 163 patients with SIH, it was demonstrated that anterior epidural CSF collection involving <8 segments and a midbrain-pons angle ≥40° were associated with a good response to a single EBP (15). However, these findings have not yet been validated. In addition, whether patients with more than one predictor could have an even higher probability of having a good outcome warrants further study.
We aimed to conduct a large-scale study to verify the role of these pre-EBP factors and to develop and validate an easy-to-use scoring system to predict the treatment response to the first EBP in patients with SIH.
Methods
Patients and clinical setting
This was a retrospective analysis of a prospectively enrolled cohort consisting of consecutive patients with SIH from Taipei Veterans General Hospital (TVGH), a tertiary medical center in the capital of Taiwan. Patients were included if they i) were hospitalized for the diagnosis and treatment of SIH, ii) fulfilled the International Classification of Headache Disorders, third edition (ICHD-3) criteria for 7.2.3 Headache attributed to spontaneous intracranial hypotension (18) (applied retrospectively for patient enrolled before 2018), and iii) had treatment outcome at three months available. Patients were excluded from the analysis if i) they refused EBP, ii) EBP was not indicated (due to spontaneous recovery, recovery after conservative treatment, recovery after treatment after treatment at another hospital, etc.), or iii) brain MRI and/or heavily T2-weighted (HT2W) magnetic resonance myelography (MRM) was not available for analysis.
The patients received a thorough clinical assessment with a standardized protocol, including a detailed history, neurological examination, pre- and post-gadolinium brain MRI, and HT2W MRM (and/or computed tomography (CT) myelogram before 2003) as described previously (15,19–21). Only patients who received at least one EBP were included in the analysis. Patients were chronologically divided into a derivation cohort (80%) and an independent validation cohort (20%) by the 80–20 rule (Figure 1). The study was approved by the Institutional Review Board of TVGH (VGH IRB No. 2021-07-012AC).

Study flow chart of patient inclusion in the derivation and validation cohorts. (a) Study flow chart of enrolled patients in the derivation cohort and (b) Study flow chart of enrolled patients in the validation cohort.
Epidural blood patch
As targeted EBPs are more effective than nontargeted EBPs (22), targeted EBPs were attempted as the first-line treatment in addition to bed rest and hydration for all the patients with periradicular leaks and/or epidural fluid collections. HT2W MRM (and/or CT myelography before 2003) was routinely carried out to localize periradicular leaks, which were the main target for EBPs. The procedure was performed by an experienced anesthesiologist (S-SH). Briefly, an 18-gauge epidural Tuohy needle was inserted via the midline approach with the patient lying in a lateral decubitus position, and autologous blood was slowly injected at the levels of periradicular leaks identified. When periradicular leaks could not be localized on HT2W MRM (and/or CT myelogram before 2003), EBPs were delivered at the level with the most prominent epidural CSF collections or meningeal diverticulum (20). When patients had neither periradicular leaks nor spinal epidural CSF collections, EBPs were delivered at the cervico-thoracic and/or thoraco-lumbar junctions, which rendered them non-targeted.
Derivation cohort
The derivation cohort involved consecutive patients with SIH admitted between December 1997 and December 2018 (Figure 1A). The outcome measure was the response to the first EBP (15). Patients were categorized as responders if they had complete remission of symptoms lasting for three months follow up at our neurology outpatient clinic after the first EBP. In contrast, those with persistent or recurrent symptoms within three months or only partial relief after the first EBP in our or other hospitals, which necessitated further EBPs, were labeled nonresponders (15). The response to subsequent EBPs was evaluated with the same standard. The number of EBPs needed for each patient to have a complete and sustained resolution of symptoms lasting for at least three months was recorded. Pre-EBP factors associated with a treatment response were determined by using multivariable logistic regression analyses. An outcome stratification scale (i.e., the SIH-EBP score) was then developed by using the pre-EBP factors identified. The cutoff value of the SIH-EBP scores predictive of a treatment response was determined by using the receiver operating characteristic (ROC) curve.
Validation cohort
This SIH-EBP score was subsequently validated in an independent cohort of 78 consecutive patients enrolled between January 2019 and January 2021. The patients were diagnosed and treated through a standard protocol as the derivation cohort (Figure 1B). The accuracy, sensitivity, specificity, and positive and negative predictive values (PPV and NPV) of the scoring system were determined.
Some of the patients (54%) in the derivation cohort were included in one of our previous studies (15) investigating treatment response to the first EBP, but there was no overlapping between the derivation and the validation cohorts. Therefore, the validation process was carried out in an independent cohort of newly recruited patients.
Statistical analysis
Statistical analysis was performed with IBM SPSS version 26.0 (IBM Corp., Armonk, NY, USA). Descriptive analysis was performed using frequencies and percentages for categorical variables and the mean ± standard deviation (SD) or median [interquartile range (IQR)] for continuous variables, and comparisons between groups were made by using the χ2 and Student’s t tests for categorical and continuous variables, respectively. Statistical significance was defined as P < 0.05. Although it was found that EBP volumes were positively correlated with treatment outcome in our previous study (15), only variables available to the clinicians before the first EBP were used in the construction of the scoring system. As the scoring system was designed to predict the treatment response, EBP volume was not included as a variable. In the development of the SIH-EBP scoring system, cutoff values of certain variables were based on the results of our previous study, including the length of the anterior epidural CSF collection (Figure 2A) and midbrain-pons angle (Figure 2B) (15), as well as determined by using classification and regression tree (CRT) analysis, which a nonparametric, binary partitioning of the predictor variable to reduce mixing at each split (23). Although epidural CSF collections could be anterior or posterior in location (24), only the length of “anterior" epidural collections was included in the scoring system, as one of the aims of the present study was to verify the findings of our previous work (15). Multivariable logistic regression analyses were performed, initially including all potential predictor variables in the model, with stepwise elimination of variables not contributing to the model, and the variable retention criterion was set to P < 0.10 to minimize the possibility of losing important variables (25). The clinical utility of the scoring system was determined in the derivation and validation cohorts, as well as the entire study cohort. Mantel-Haenszel χ2 test for trend was carried out to determine whether a higher SIH-EBP score was associated with a higher response rate. Finally, logistic regression modeling was performed to estimate the odds ratio (OR), as well as the 95% confidence interval (CI), for a favorable outcome between patients who scored above and below the cutoff of the SIH-SBP score.

Measurement of anterior epidural CSF collection and midbrain-pons angle in SIH. (a) Sagittal MRI of the thoracic spine in one patient showing anterior epidural CSF collection in 8 segments (T5–T12) (arrow). Heavily T2-weighted MR myelography of cross-sectional views of the 5th and 11th thoracic spines showing anterior epidural CSF collection (arrow), comparable with conventional T2-weighted images of the same levels and (b) Midline sagittal brain MRI in another patient showing the angle between the line tangential to the anterior margin of the midbrain and the line tangential to the superior margin of the pons, i.e., midbrain-pons angle (43.7°).
Results
Study participants
In total, 350 patients with SIH who presented to our neurology outpatient clinic or emergency department between December 1997 and December 2018 were screened. Two patients who refused EBP treatment, and 61 patients who did not receive EBP treatment because of spontaneous recovery, recovery after conservative treatment, or recovery after treatment at another hospital were excluded. Seven patients were excluded due to unavailability of brain MRI (n = 4) or HT2W MRM (n = 3). The remaining 280 patients (178 F/102 M, mean age 39.9 ± 9.8 years) with SIH who received at least one EBP constituted the derivation cohort.
In the derivation cohort, 203 (72.5%) patients with diffuse pachymeningeal enhancement, 78 (27.9%) with subdural effusion or hematoma were demonstrated on brain MRI and 233 (83.2%) with periradicular leaks and 263 (93.9%) with epidural CSF collections were demonstrated on HT2W MRM. The mean duration between disease onset and diagnosis was 25.8 ± 47.0 days (range 1–597 days). A total of 149 patients (53.2%) recovered after the first EBP, and 94 (33.6%), 31 (11.1%), and 5 (1.8%) patients received two, three, and four EBPs, respectively. Surgical repair was necessary for one patient (0.4%) who failed four unsuccessful EBPs.
The validation cohort consisted of 78 patients (48 F/30 M, mean age 41.0 ± 9.6 years) with SIH recruited between January 2019 and January 2021. Of these 78 patients, 56 (71.8%) with diffuse pachymeningeal enhancement, 20 (25.6%) with subdural effusion or hematoma were demonstrated on brain MRI and 71 (91.0%) with periradicular leaks and 73 (93.6%) with epidural CSF collection were demonstrated on HT2W MRM. A total of 42 patients (53.8%) recovered after the first EBP, and 27 (34.6%), six (7.7%), two (2.6%), and one (1.3%) patients received two, three, four, and five EBPs, respectively. The injected volumes of EBPs were larger in the validation cohort than in the derivation cohort (36.7 ± 13.3 ml vs. 29.6 ± 11.0 ml, P < 0.001), as larger amounts of blood were injected into these patients due to the finding in our prior study that the volumes of EBPs were related to the treatment outcome (15). Otherwise, there were no statistically significant differences in demographics, clinical manifestations, pre-existing migraine, preceding trivial trauma, time from onset to diagnosis and the first EBP injection or the treatment response between the derivation and validation cohorts (Table 1).
Demographics, clinical characteristics and treatments of the derivation and validation cohorts.

CSF, cerebrospinal fluid; EBP, epidural blood patch; SD, standard deviation.
Factors associated with treatment response
The clinical and radiological features are shown in Table 2. The responders were more likely to be women (73.2% vs. 52.7%, P < 0.001) and had larger volumes of EBPs (30.8 ± 11.5 ml vs. 28.2 ± 10.2 ml, P = 0.048), greater midbrain-pons angles (43.5 ± 17.0° vs. 34.4 ± 18.3°, P < 0.001), and shorter anterior epidural CSF collections (8.0 ± 5.3 segments vs. 14.3 ± 6.0 segments, P < 0.001) when compared with the nonresponders. For patients without anterior epidural CSF collection, the response rate was 100% (n = 10/10). The corresponding figures were 86.8% (n = 66/76), 44.5% (n = 65/146), and 16.7% (n = 8/48) in patients with 1–7, 8–18, and ≥19 segments of anterior epidural collections, respectively. Otherwise, clinical manifestations, proportions of pre-existing migraine and preceding trivial trauma, and time from onset to diagnosis or the first EBP injection were not significantly different between the responders and nonresponders.
Clinical and radiological features of the responder and nonresponder groups.

CSF, cerebrospinal fluid; EBP, epidural blood patch; SD, standard deviation.
Development of the SIH-EBP Score
CRT analysis was performed to derive the cutoff values for pre-EBP continuous variables, i.e., age, time from onset to diagnosis, time from onset to first EBP injection, midbrain-pons angle and length of anterior epidural CSF collection (23). In order to make the scoring system simple and easy to use, cutoff values close to those derived from CRT analysis were used. Age was divided using a cutoff value of 50 years (cutoff value derived from CRT analysis = 48.5 years). A cutoff value of 40° for midbrain-pons angle (cutoff value derived from CRT analysis = 36.6°, which was similar to that of a prior study [42.6°]) (15) and the length of anterior epidural CSF collection was partitioned at 8 and 18 segments (cutoff value derived from CRT analysis = 7.5 segments at the first level and 17.5 segments at the second level). It was not feasible to partition the time from onset to diagnosis and time from onset to first EBP by using CRT analysis, and these two factors were not included in the scoring system.
Furthermore, multivariable logistic regression modeling was developed with stepwise elimination of variables not contributing to the model (25), and four variables remained and constituted the SIH-EBP score (online Supplemental Table S1). The scoring system included two clinical variables, woman (1 point) and age ≥50 years (1 point), as well as two radiological variables, midbrain-pons angle ≥40° (1 point) and anterior epidural CSF collection <8 segments (including 0 segments) (2 points) or 8–18 segments (1 point) (Table 3). The total score ranged from 0 to 5. By using ROC analysis, a score ≥3 was predictive of a favorable outcome following the first EBP, with an area under the ROC curve of 0.80 (95% CI = 0.75–0.85) (Figure 3A). The accuracy was 76%, and the sensitivity, specificity, PPV and NPV were 77%, 75%, 78% and 74%, respectively (online Supplemental Table S2).
The SIH-EBP score.

The SIH-EBP score included two clinical variables, woman (1 point) and age ≥50 years (1 point), as well as two radiological variables, midbrain-pons angle ≥40° (1 point) and anterior epidural CSF collection <8 segments (including 0 segments) (2 points) or 8–18 segments (1 point). The total score was the sum of the points of these variables, ranging from 0 to 5.
CSF, cerebrospinal fluid; EBP, epidural blood patch; SIH, spontaneous intracranial hypotension.

ROC curves for the SIH-EBP score.
When the original cutoff values derived from CRT analysis, i.e., age ≥48.5 years (1 point), were used instead, the findings were consistent (Figure 3B).
Validation of the SIH-EBP Score
The clinical utility of the SIH-EBP score was then tested in the validation cohort. In the validation cohort (n = 78), a cutoff value of SIH-EBP score predictive of a treatment response to the first EBP was determined at ≥3, with an area under the ROC curve of 0.83 (95% CI = 0.73–0.92) (Figure 3A). The accuracy was 77%. The sensitivity, specificity, PPV and NPV were 74%, 81%, 82% and 73%, respectively (online Supplemental Table S2).
Prognostic performance of the SIH-EBP Score
In the entire study population (n = 358), which consisted of both the derivation and validation cohorts, the overall response rate to the first EBP was 53.4% (n = 191). A higher SIH-EBP score was associated with a higher response rate to the first EBP (P < 0.001, Mantel-Haenszel χ2 test for trend) (Figure 4A). Patients who scored ≥3 were more likely to respond to the first EBP (OR = 10.3, 95% CI = 6.3–16.8, P < 0.001) when compared with those with a score <3 (n = 146/186, 78.5% vs. n = 45/172, 26.2%; P < 0.001) (Figure 4B).
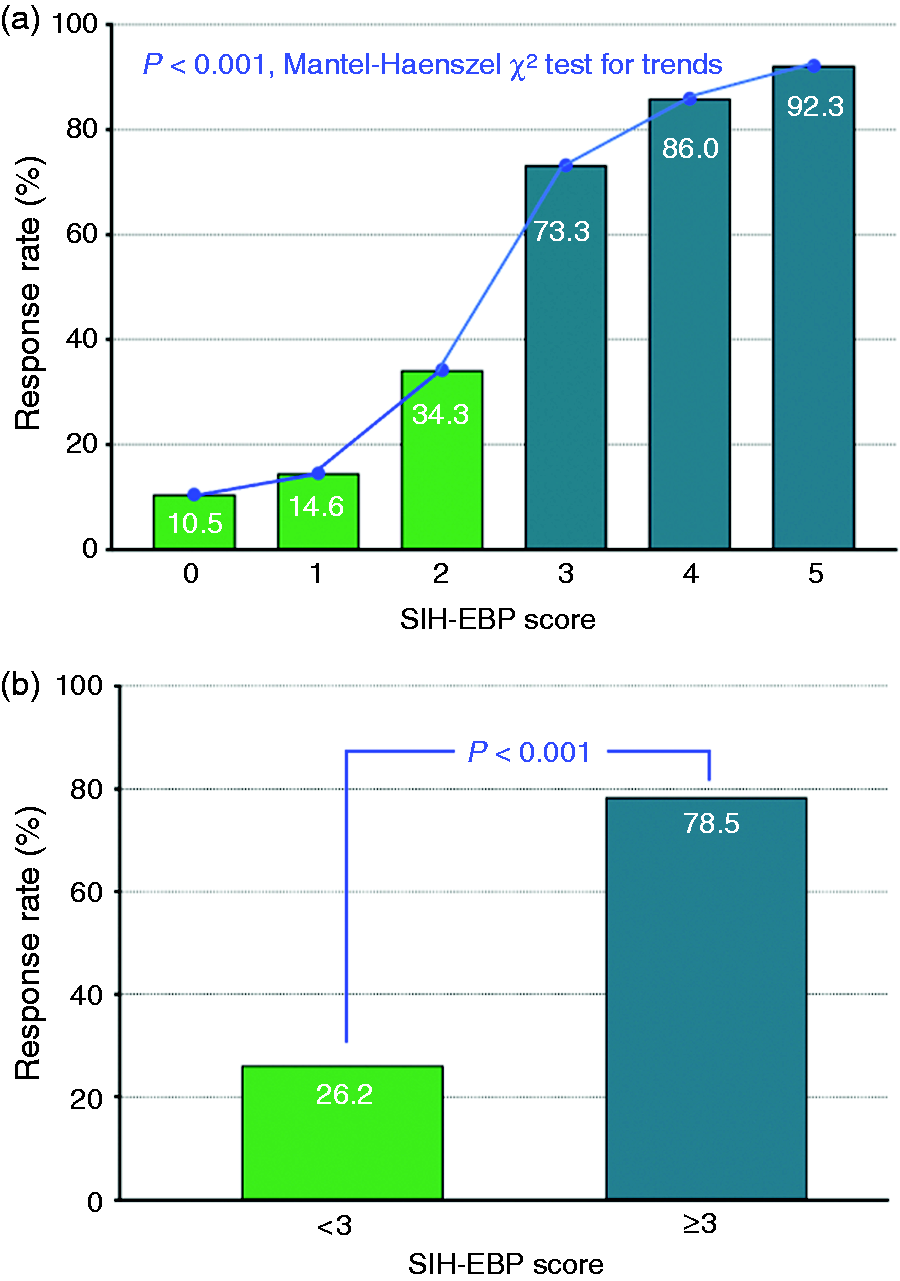
The SIH-EBP score and EBP response rate.
Discussion
In the present study, a prognostic scoring system incorporating clinical and radiological features, namely, the SIH-EBP score, was developed in a large cohort of patients with SIH (n = 280). Besides, the validity of the SIH-EBP score was verified in an independent cohort (n = 78). A higher score was associated with a greater likelihood of having a favorable outcome (Figure 4A) and a cutoff score ≥3 increased the odds of having a favorable treatment response to the first EBP by 10-fold (OR = 10.3, 95% CI = 6.3–16.8, P < 0.001) (Figure 4B). As a result, a high score should prompt at least one attempt of targeted EBP before considering surgical intervention. On the other hand, for patients who score <3 and fail the first EBP, more invasive imaging techniques, such as digital subtraction myelography or CT myelography, may be considered to localize the spinal CSF leaks more accurately or to identify CSF-venous fistulas, based on which further interventions could be implemented. The scoring system is simple and easy to use and can play an important role in the clinical management of patients with SIH.
One of the clinical factors in the SIH-EBP scoring system, i.e., older age, was reported in some prior studies. It was shown that older patients were more likely to have a complete rather than partial response to EBPs (9). However, female sex was a newly identified outcome predictor in patients with SIH. Since women tend to have smaller spinal canals (26,27), especially in cervical and lumbar regions, it is likely that EBPs of the same volumes might be more effective in packing or sealing the CSF leaks (13) in women than in men. However, further studies are still needed to confirm our findings and to determine factors accounting for gender differences in the treatment response. On the other hand, the response rate to the first EBP (53.2%) in the current series was lower than that reported in a meta-analysis (64%) (28). This was probably attributed to the intractable nature of the present cohort. Some of our patients were referred to our medical center after treatment failure in other hospitals.
Classical radiological features of SIH, such as subdural fluid collections, enhancement of the pachymeninges, engorgement of venous structures, pituitary hyperemia, and sagging of the brain (1,11) were not predictive of the treatment response. Instead, our previous study showed that the midbrain-pons angle and length of the anterior epidural CSF collections were more useful (15), which was confirmed in the present study. It was shown that the mean midbrain-pons angle of patients with SIH was significantly less than that of normal controls (48.9 ± 10.5° vs. 60.4 ± 7.3°, P < 0.001) (7), and narrowing of the midbrain-pons angle may result from brain sagging (29,30). On the other hand, longer anterior epidural CSF collections were predictive of a poorer treatment response (15). Since MRI of the spine was acquired in a supine position, the length of the anterior epidural CSF collection may better reflect the severity of CSF leakage. Both above factors could be indicators for disease severity (30), and were therefore, associated with the treatment response.
The majority of patients were treated with EBPs targeted at the CSF leaks identified, which was believed to be more effective than nontargeted EBPs. In a recently published meta-analysis, it was concluded that targeted and nontargeted EBPs were equally effective (28). However, interpretations could be difficult given the heterogeneities in patient characteristics, imaging techniques used to localize the CSF leaks, definitions of targeted and nontargeted EBPs, and other factors. To date, there were only a limited number of retrospective studies providing the response rates of both targeted and nontargeted (or blind) EBPs in SIH patients. In one study, targeted EBPs were more effective than nontargeted EBPs delivered at lumbar or upper thoracic levels (87.1% vs. 52%, P < 0.05) in a non-randomized study (n = 56) (22), which is supportive of our treatment strategy. On the other hand, the response rates were comparable in two other reports (31,32). However, the findings should be interpreted with caution. In one of the studies (n = 95), the blind EBP group referred to patients in whom the CSF leakage sites could not be identified, and it was not clear where blind EBPs were delivered (31). In the other (n = 116), patents in the blind EBP group actually had EBPs delivered at both the cervicothoracic and thoracolumbar junctions at the same time (32), rather than at the lumbar region. Theoretically, EBPs placed at the level of CSF leaks should be more effective than those delivered remotely. Further studies are needed to provide more supportive evidence for the implementation of targeted EBPs in SIH patients.
Study strengths
One of the most important strengths of the present study was the large sample size (n = 358), which could reduce the selection bias, decrease the influences of data variability, and yield more accurate estimates. In addition, the study utilized a two-stage design. The validity of the scoring system was consistent in two independent cohorts, which increased the credibility of the findings. Third, the patients were evaluated and treated with a standardized protocol. Therefore, the data were of high quality and reliability.
Study limitations
This study has some limitations. First, only a limited number of neuroimaging factors were included in the analysis as the current study aimed to validate our previous findings (15), based on which a simple scoring system was developed. Second, CSF-venous fistula, which is gaining attention recently (33,34), may be overlooked in our cohort as HT2W MRM, rather than digital subtraction myelography or dynamic computed tomographic myelography, was used in the current study. This could potentially limit the generalizability of our findings. However, the aim of the present study was to identify predictors for treatment outcome based on information available from medical records or images acquired previously. Further studies are needed to determine whether the scoring system could be applied to patients with CSF-venous fistulas. Third, although EBP volume was identified as an outcome predictor in our previous study (15), it is not included in the scoring system. Besides, although the majority of patients received targeted EBPs, the EBPs were delivered at cervico-thoracic and/or thoraco-lumbar junctions, i.e., nontargeted, in a small proportion (0.8%), when neither periradicular leaks nor epidural collections could be identified. This could also have had an impact on the treatment response. As the study was to identify pre-EBP factors predictive of treatment outcome, only factors available before treatment were taken into consideration. Fourth, the characteristics of the present cohort were different from those in some other studies, which could lead to concerns about generalizability of our findings. For instance, the proportion of patients with epidural CSF collection (93.9% in the derivation cohort and 93.6% in the validation cohort) was higher than that in other series of SIH (e.g. 50.5% in a US study) (33). However, the percentages were close to that (91%) in a recent meta-analysis involving studies evaluating the diagnostic yield of MR myelography (35). Besides, in the present study, only a small proportion of patients received surgical repair for CSF leaks (0.4%). In some studies, 20.3–50.2% of the cases underwent surgical intervention for the CSF leaks (33,36). However, the findings of the present study are consistent with some other series, in which patients who failed multiple EBPs constituted only a minority (0–6.9%) (10,17,22,37). The discrepancies could reflect the heterogeneity of SIH patients, which could be accounted for by differences in ethnicities, patient characteristics, clinical settings, imaging techniques, treatment strategies, or other factors.
Conclusion
The SIH-EBP score, which includes two clinical and two radiological variables, is a simple and validated scoring system that could be used to predict the response to the first EBP in patients with SIH with high accuracy. Patients with a score ≥3 could have a favorable treatment outcome; therefore, it encourages patients going forward with an EBP.
Clinical implications
The SIH-EBP score (range 0–5) includes four clinical and radiological variables (sex, age, midbrain-pons angle, and anterior epidural cerebrospinal fluid collections). The SIH-EBP score is a simple and validated system for outcome prediction in spontaneous intracranial hypotension. Patients whose SIH-EBP score ≥3 increase the odds of having the first epidural blood patch response by 10 folds.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221147488 - Supplemental material for The SIH-EBP Score: A grading scale to predict the response to the first epidural blood patch in spontaneous intracranial hypotension
Supplemental material, sj-pdf-1-cep-10.1177_03331024221147488 for The SIH-EBP Score: A grading scale to predict the response to the first epidural blood patch in spontaneous intracranial hypotension by Po-Tso Lin, Yen-Feng Wang, Shu-Shya Hseu, Jong-Ling Fuh, Jiing-Feng Lirng, Jr-Wei Wu, Shu-Ting Chen, Shih-Pin Chen, Wei-Ta Chen, Shuu-Jiun Wang in Cephalalgia
Footnotes
Acknowledgement
The authors would like to thank the patients for their participation.
Author Contributions
P-T Lin had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: P-T Lin, Y-F Wang and S-J Wang. Acquisition, analysis, or interpretation of data: P-T Lin, Y-F Wang, S-S Hseu, J-L Fuh, J-F Lirng, J-W Wu, S-T Chen, S-P Chen, W-T Chen and S-J Wang. Drafting of the manuscript: P-T Lin, Y-F Wang and S-J Wang. Critical revision of the manuscript for important intellectual content: Y-F Wang and S-J Wang. Statistical analysis: P-T Lin, Y-F Wang and S-J Wang. Obtained funding: Y-F Wang and S-J Wang. Administrative, technical, or material support: Y-F Wang and S-J Wang. Supervision: Y-F Wang and S-J Wang.
Availability of data and materials
Anonymized data that support the findings of this study are available from the corresponding author upon reasonable requests from any qualified investigator.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Y-F Wang has received honoraria as a speaker from Taiwan branches of Allergan/AbbVie, Eli Lilly, Novartis, Pfizer, Sanofi, UCB, and Viatris, and Orient EuroPharma. He has received research grants from the Taiwan Ministry of Science and Technology, and Taipei Veterans General Hospital. S-J Wang has served on the advisory boards of Daiichi-Sankyo, Eli Lilly and Novartis; has received honoraria as a moderator from Allergan/AbbVie, Pfizer, Eli Lilly, Biogen and Eisai and has been the PI in trials sponsored by Eli Lilly, Novartis, and Allergan/AbbVie. He has received research grants from the Taiwan Ministry of Science and Technology, Brain Research Center, National Yang Ming Chiao Tung University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan, Taipei Veterans General Hospital, Taiwan Headache Society and Taiwan branches of Eli Lilly, Novartis, and Pfizer. P-T Lin, S-S Hseu, J-L Fuh, J-F Lirng, J-W Wu, S-T Chen, S-P Chen, and W-T Chen reported no disclosures relevant to the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored in part by Taiwan Ministry of Science and Technology [MOST 106-2314-B-075-021, MOST 107-2314-B-075-018-MY2, MOST 109–2314-B-075-054, and MOST 110-2314-B-075 -041-MY3 (to Y-F Wang), and MOST 104-2314-B-010-015-MY2, MOST106-2321-B-010-009, MOST 107-2321-B-010-001, MOST 108-2321-B-010-014 -MY2, 108-2321-B-010 001, MOST 108-2314-B-010-023-MY3, and MOST-110-2321-B-010-005 (to S-J Wang)]; and Taipei Veterans General Hospital [V108-C-092, V109-C-096, and V110-C-111 (to Y-F Wang), and V105C-127, V106C-106, V107C-135, V108C-105, and V109C-090 (to S-J Wang)]; this work was also supported by the Brain Research Center, National Yang-Ming University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
