Abstract
Objectives
In the application of the Monro-Kellie doctrine in spontaneous intracranial hypotension, the brain tissue volume is generally considered as a fixed constant. Traditionally, cerebral venous dilation is thought to compensate for decreased cerebrospinal fluid. However, whether brain tissue volume is invariable has not yet been explored. The objective of this study is to evaluate whether brain tissue volume is fixed or variable in spontaneous intracranial hypotension patients using automatic quantitative methods.
Methods
This retrospective and longitudinal study analyzed spontaneous intracranial hypotension patients between 1 January 2007 and 31 July 2015. Voxel-based morphometry was used to examine brain volume changes during and after the resolution of spontaneous intracranial hypotension. Brain structure volume was analyzed using Statistical Parametric Mapping version 12 and FMRIB Software Library v6.0. Post-treatment neuroimages were used as surrogate baseline measures.
Results
Forty-four patients with spontaneous intracranial hypotension were analyzed (mean [standard deviation] age, 37.8 [8.5] years; 32 female and 12 male). The whole brain tissue volume was decreased during spontaneous intracranial hypotension compared to follow-up (1180.3 [103.5] mL vs. 1190.4 [93.1] mL, difference: −10.1 mL [95% confidence interval: −18.4 to −1.8 mL], p = 0.019). In addition, ventricular cerebrospinal fluid volume was decreased during spontaneous intracranial hypotension compared to follow-up (15.8 [6.1] mL vs. 18.9 [6.9] mL, difference: −3.2 mL [95% confidence interval: −4.5 to −1.8 mL], p < 0.001). Longer anterior epidural cerebrospinal fluid collections, as measured by number of vertebral segments, were associated with greater reduction of ventricular cerebrospinal fluid volume (Pearson’s r = −0.32, p = 0.036).
Conclusion
The current study found the brain tissue volume and ventricular cerebrospinal fluid are decreased in spontaneous intracranial hypotension patients. The change in ventricular cerebrospinal fluid volume, but not brain tissue volume change, was associated with the severity of spinal cerebrospinal fluid leakage. These results challenge the assumption that brain tissue volume is a fixed constant.
Keywords
Introduction
Spontaneous intracranial hypotension (SIH) results from cerebrospinal fluid (CSF) leakage (1–6). The most common symptom of SIH is orthostatic headache (1,6–10). According to the International Classification of Headache Disorders, 3rd edition (ICHD-3), diagnosis of SIH requires neuroimaging evidence of CSF leakage and a temporal relationship between headache onset and occurrence of low CSF pressure or CSF leakage (22). Patients with SIH may have a wide variety of brain magnetic resonance imaging (MRI) abnormalities, including diffuse pachymeningeal enhancement (11), pituitary hyperemia (11,12), cerebral venous dilation (13–15), brain sagging (11,15), and subdural effusion or hematoma (16–18).
The Monro-Kellie doctrine postulates a constant total volume of blood, brain, and CSF, such that changes in the volume of one component results in reciprocal alterations in one or both of the other two components (19). Currently, the Monro-Kellie doctrine is widely accepted as an explanation of neuroimaging abnormalities associated with SIH (1,3). Therefore, reduction of CSF volume in patients with SIH may result in a compensatory increase of brain and/or intracranial blood volume. Brain tissue volume, however, is generally considered invariable, and therefore, compensation would occur through an increase of blood volume, specifically venous blood, since veins are more adjustable than arteries (1). Prior studies have reported deformities of certain brain structures in SIH patients (15,20,21), and therefore, whether total brain tissue volume is invariable remains unknown. The primary aim of the current study was to evaluate whether brain tissue volume is fixed or variable in SIH patients using automatic quantitative methods. We hypothesized that brain tissue volume is altered in patients with SIH. In addition, the associations between brain and spinal MRI findings in SIH patients were explored.
Methods
Demographics and clinical profiles
We retrospectively analyzed the medical records and neuroimaging findings of patients with SIH who were admitted to the Neurology ward of Taipei Veterans General Hospital, a medical center in Taiwan, between 1 January 2007 and 31 July 2015. Diagnosis of SIH was made according to the proposed criteria for headache secondary to SIH (7.2.3) in the ICHD-3 (22). The onset-neuroimaging interval was defined as the time interval between the onset of the first symptom of SIH and the date of brain and spinal MRI studies. The interval between pre-treatment brain MRI and follow-up post-recovery brain MRI was also recorded.
Inclusion and exclusion criteria
The inclusion criteria were as follows: a) Confirmation of ICHD-3 criteria of SIH (7.2.3); b) spinal CSF leakage successfully treated via epidural blood patching (EBP), with complete remission of symptoms after the epidural blood patching and evidences of complete resolution of CSF leak in the follow-up neuroimaging studies; c) pre-treatment brain MRI completed in Taipei Veterans General Hospital – however, hydration or analgesic usage prior to the pre-treatment brain MRI were acceptable; d) follow-up MRI >7 days and ≤3 years after complete recovery. Patients with a follow-up brain MRI exceeding 3 years were excluded from the study to avoid age-related brain volume change (23); e) pre-treatment and follow-up brain MRI completed within the protocol parameters described in the ‘Brain neuroimaging’ section below. The exclusion criteria were as follows: i) Incomplete recovery after EBP, including partial improvement, recurrence, or intractable spinal CSF leak during follow-ups and ii) for the accuracy of voxel-based morphometry analysis, data from patients with subdural hematoma or fluid collection in brain MRIs were excluded.
Brain neuroimaging
Brain MRIs were performed on a 1.5-T scanner. The MRI protocol has been described in previous publications (5,15,24). All patients received post-contrast (gadopentetate dimeglumine (Gd) 0.1 mmol/kg b.w.) MRIs. Brain images were obtained in the transverse plane with T1- and T2-weighted spin-echo sequences. The section thickness was 1 mm, with an acquisition matrix of 256 × 256. MRI signs of SIH were recorded as continuous variables, including midbrain-pons angle (15,20,24,25), descent of the cerebral aqueduct (depicted as iter descent distance (in mm) below the incisural line connecting the tuberculum sellae with the confluence of the vein of Galen into the straight sinus) (11,21), and the severity of transverse sinus venous distension (15). The measurements of these continuous variables were described in our previous publications (15,24). The circadian variability of brain volume at different time points within a day may be a confounding factor for the brain volume comparisons (26). In this study, the timing of scanning of pre-treatment and follow-up MRI were both left to chance between 8 am and 11 pm; therefore, we analyzed the difference of the timing of scanning (within-day) between them.
Voxel-based morphometry was used to examine brain volume changes during and after SIH resolution by using T1-weighted MR imaging (27). We applied the widely used FMRIB Software Library v6.0 (FSL6) Brain Extraction Tool (BET) for skull stripping and Statistical Parametric Mapping version12 (SPM12) for volumetric measurement (28,29). The reason for choosing SPM12 is that its accuracy is not inferior to or even better than other segmentation software (30,31). After skull removal and bias field correction, brain regions were segmented into the following categories: Total intracranial fluid, CSF, and brain tissue, and the lowest level of segmentation was the McRae line. The measurement of ventricular CSF volume has been used to compare the CSF volume changes before and after spaceflight (32). Prior studies have shown that direct measurement of cerebral blood volume depends on hemodynamic MRI techniques (33,34). Therefore, in the current study, the “estimated intracranial blood volume” was measured by subtracting CSF volume from total intracranial fluid volume.
Spine neuroimaging
Heavily T2-weighted non-contrast magnetic resonance myelography (MRM) was performed with a phased-array spine coil on a 1.5T superconducting system (Signa HD or Signa Excite twin, GE Medical Systems) without using intrathecal gadolinium as described previously (5). The number of vertebral segment(s) were calculated to measure the length of anterior epidural fluid collection. According to our previous studies, the length of anterior epidural fluid collection reflects the severity of SIH (15,24).
Statistics
Since pre-SIH diagnosis brain MRIs were not available, MRI data collected after recovery during follow-up were used for comparisons. Mean values with standard deviations and median values with interquartile ranges (IQR) are reported. Comparisons among categorical and continuous variables were analyzed using chi-square or t-tests as appropriate. A Mann-Whitney U test was used to compare non-normally distributed continuous variables. Differences between pre-treatment and follow-up measurements were assessed with independent-sample t-tests with a significance threshold of p < 0.05. Pearson correlations were calculated to examine potential associations between continuous variables. Bonferroni’s corrections for multiple comparisons were applied in correlational analyses, including correlations between volume changes in brain structures (corrected for 15 pairwise comparisons: p = 0.05/15 tests = 0.003). The associations between brain structural volume changes and brain or spinal MRI signs were exploratory, with a significance threshold of p < 0.05.
Ethics
The study protocol was approved by the Institutional Review Board of Taipei Veterans General Hospital (2017-07-001AC#1).
Results
Patients
A total of 161 patients with SIH were recruited. After exclusions, data on 44 patients (12 males and 32 females) were analyzed (Figure 1). The mean (standard deviation [SD]) patient age was 37.8 [8.5] years, and the median (interquartile range [IQR]) interval between pre-treatment and follow-up MRIs was 94.5 (40.5–447.3) days. The demographics, clinical profiles, and neuroimaging findings of both included and excluded patients are summarized in Table 1. The median (IQR) onset-neuroimaging interval was 12 (4.3–23.3) days in the included patient group and 22 (8–37) days in the excluded patient group (Mann-Whitney U test, p = 0.014). The mean difference in the timing of scanning (within-day) was 1.8 (95% CI: −0.01 to +3.65, p = 0.051) hours (mean timing of pre-treatment MRI vs. follow-up MRI: 15.9 [4.3] vs. 14.1 [3.4] of 24 h).

Study schematic flow chart.
Demographics, clinical profiles, and neuroimaging findings in SIH patients.

*Results were considered significant by p < 0.05. All patients with subdural fluid collection or hematomas were excluded from the study.
**p-value calculated with the Mann-Whitney U test.
Brain volume change during SIH
Compared to the follow-up brain MRIs, the whole brain tissue volume significantly decreased during SIH (mean [SD] volume for pre-treatment vs. follow-up: 1180.3 [103.5] mL vs. 1190.4 [93.1] mL, mean difference (or Δ): −10.1 mL [95% CI: −18.4 to −1.8 mL], p = 0.019). In addition, the mean [SD] ventricular CSF volume was significantly lower at pre-treatment compared to follow-up (pre-treatment vs. follow-up: 15.8 [6.1] mL vs. 18.9 [6.9] mL, Δ: −3.2 mL [95% CI: −4.5 to −1.8 mL], p < 0.001). Also, the mean [SD] extraventricular CSF volume was significantly lower at pre-treatment compared to follow-up (mean [SD] volume for pre-treatment vs. follow-up: 123.5 [52.5] mL vs. 147.8 [57.5] mL, mean difference (or Δ): −24.2 mL [95% CI: −43.6 to −4.8 mL], p = 0.016). The mean [SD] estimated intracranial blood volume was significantly greater at pre-treatment compared to follow-up (pre-treatment vs. follow-up: 182.8 [54.6] mL vs. 152.4 [54.3] mL, Δ: 30.4 mL [95% CI: 11.7 to 49.0 mL], p = 0.002).
Brain tissue was separated into supratentorial and infratentorial regions. While the mean [SD] volume of supratentorial brain tissue was significantly lower at pre-treatment compared to follow-up (pre-treatment vs. follow-up: 981.5 [103.3] mL: vs. 991.1 [92.8] mL, Δ: −9.6 mL [95% CI: −18.2 to −1.0 mL], p = 0.029), there was no significant difference in mean [SD] infratentorial brain tissue (pre-treatment vs. follow-up: 198.9 [19.5] mL vs. 199.3 [19.9] mL, p = 0.487). Brain structure volume changes are depicted in Table 2.
Changes in brain structure volume and brain MRI signs before treatment and at follow-up.

*Results were considered significant at p < 0.05.
Associations between volume changes of different brain structures
Associations between changes in brain tissue volume and intracranial blood volume from pre-treatment to follow-up were investigated. Brain tissue volume changes were significantly associated with supratentorial tissue volume changes (Pearson’s r = 0.99, p < 0.001) and estimated intracranial blood volume changes (Pearson’s r = −0.48, p = 0.001), but not with infratentorial tissue volume changes (Pearson’s r = −0.13, p = 0.419). Also, there was no significant correlation between the volume changes of ventricular CSF and supratentorial brain tissue (Pearson’s r = 0.12, p = 0.436). The correlations between brain structural volume changes are depicted in Table 3.
Associations between changes in volume of brain structures, brain MRI signs (continuous variables), and spinal MRI signs (Pearson’s coefficient r value, p-values).

*Bonferroni’s correction for multiple comparisons was applied to the associations between volume changes of different brain structures, p < 0.003 was considered significant after corrections for 15 pairwise comparisons: p = 0.05/15 tests = 0.003).
#p < .05 was considered significant in exploratory results, i.e. brain tissue volume changes and brain and spinal MRI signs. Bold values are statistically significant.
Exploratory analyses: Changes in MRI signs in SIH and associations with brain structure volume changes
Among brain descent-related MRI signs (15), the mean (SD) midbrain pons angle (pre-treatment vs. follow-up: 42.9° [15.2]° vs. 56.7° [11.9]°, Δ: −13.9° [95% CI: −19.9° to −7.8°], p < 0.001) and brain sagging (mean distance between iter and incisural line, mm) (pre-treatment vs. follow-up: 5.7 [4.4] mm vs. 1.5 [3.4] mm, Δ: 4.2 mm [95% CI: 3.3 to 5.1 mm], p < 0.001) significantly differed from pre-treatment to follow-up. Among cerebral vein-related MRI signs (15), the transverse sinus angle was significantly larger at pre-treatment compared to follow-up (mean [SD] for pre-treatment vs. follow-up: 70.0° ± 19.7° vs. 53.6°±13.9°, Δ: 16.4° [95% CI: 10.7° to 22.2°], p < 0.001) (see Table 2).
The associations between brain structural volume changes from pre-treatment to follow-up and neuroimaging signs are depicted in Table 3. Changes in ventricular CSF volume were significantly correlated with the length of anterior epidural CSF collections (Pearson’s r = −0.32, p = 0.036). However, changes in brain structure volume did not significantly correlate with changes in brain MRI signs, and changes in brain tissue volume and estimated intracranial blood volume did not significantly correlate with the length of anterior epidural CSF collections (Pearson’s r = −0.20, p = 0.195 and Pearson’s r = 0.07, p = 0.633, respectively).
Discussion
The current study found a decrease in total brain tissue volume and ventricular CSF, along with an increase in “estimated” intracranial blood volume in SIH patients prior to treatment. In addition, the change in ventricular CSF volume, but not brain tissue volume change, was associated with the severity of spinal CSF leakage.
This is the first study demonstrating a decrease in brain volume in SIH, challenging the widely accepted Monro-Kellie doctrine that brain tissue volume is invariable (1,35). According to Liu et al., normal brain volume is 1217.0 ± 27.81 mL, which is comparable to the SIH patients after treatment in the current study (36–38). Also, the values of the ventricular and extraventricular CSF volumes in this study are comparable with the results in previous studies (32,39). The CSF volume changes in present study (−16.9% in ventricular CSF and −16.3% in extraventricular CSF) are much larger than daily variations of ventricular volume (±1.63%) reported by Dickson et al. (40). While no prior studies have used voxel-based morphometry to analyze brain volume changes during SIH, the volumetric analysis has been applied in several neurological diseases and clinical conditions, such as dehydration (40), post-spaceflight (32,41), and normal pressure hydrocephalus (42). Interestingly, studies on dehydration and post-spaceflight have also demonstrated decreased brain tissue volume (32,40,41). Furthermore, studies on patients with normal pressure hydrocephalus have revealed either increased or unchanged MR volume after removal of CSF (42,43). These findings suggest that brain tissue volume is not fixed in response to changes in CSF dynamics or microgravity.
There are some differences between the present study and previous publications in SIH. In this study, the onset-diagnosis interval was shorter in our study (44), which was due to the easy access of the healthcare system in Taiwan (24). Another difference is that almost all patients with SIH were hospitalized for investigation and treatment; that is, epidural blood patching, which is the standard protocol of our hospital (5,24). Therefore, our “hospitalized” patients do not reflect a selection bias of more severe cases. It is noteworthy that previous studies found vasogenic edema and brain swelling during SIH, especially in the deep brain structures and diencephalon (21,26). Cauley et al. analyzed 13 patients with probable intracranial hypotension and found higher apparent diffusion coefficient (ADC) values in tegmental midbrain, basal ganglia, thalamus, and frontal white matter (26). Savoiardo et al. compared the ADC value between SIH with brain swelling, SIH without brain swelling, and control group (21). Compared with control group, SIH patients with brain swelling had higher ADC values in midbrain, basal ganglia, thalamus, and parietal white matter (21). On the other hand, there were no differences in ADC values between “SIH without brain swelling” and “control” groups (21). According to their findings, some but not all patients with SIH developed brain swelling with variable severity (21). The possible mechanisms underlying brain swelling in a subgroup of SIH patients may be venous stagnation or functional stenosis of venous outflow caused by brain sagging, leading to impairment of deep venous drainage and vasogenic edema (21). In contrast, the present study showed the “total” brain tissue volume decreased during SIH. Still, our finding is not sufficient to exclude the possibility of brain swelling or vasogenic edema in diencephalic-mesencephalic structures; further studies are needed to analyze the regional brain volume changes.
The Monro-Kellie doctrine postulates that the sum of the total volume of brain tissue, CSF, and blood vessels is a constant, and that brain tissue volume is not changeable (1). Consistent with previous studies using venous sinus angle or diameter measurements (11,13–15), the current study found increased intracranial blood volume and an increased transverse sinus angle in SIH patients. We used an indirect method to estimate the intracranial blood volume (subtracting CSF volume from total intracranial fluid volume) since it is not possible to measure the true volume of intracranial vessels through voxel-based morphometry directly (33). Therefore, caution must be used when interpreting the associations between changes of “estimated intracranial blood volume” and other brain structures. In this study, the sum of brain tissue, CSF, and “estimated intracranial blood volume” is not a constant, and the possible reasons are a) methodological limitation of estimating intracranial blood volume, and b) other unaccounted variables, such as craniospinal elastance (45). In addition, we found that brain tissue volume changes did not correlate with the severity of brain sagging, which excluded brain sagging as an explanation of brain volume changes.
We propose reduced free water diffusion in the brain parenchyma (Figure 2) as an explanation to account for the brain tissue volume decrease (0.85%) in SIH patients. This diffusion theory is supported by previous studies on MR volume during dehydration (40,46,47). Duning et al. showed a brain tissue volume decrease of 0.55% after 16 hours of fluid restriction followed by a 0.72% increase in brain tissue volume after rehydration with 1.5 L of water (48). Moreover, more recent studies have found a significant brain volume decrease after spaceflight (0.35%), similar to our neuroimaging findings (32,41,49). Redistribution of free water in the cerebral cortex and decreased CSF production during microgravity have been postulated to explain post-spaceflight brain volume decreases (49–51). Similar to post-spaceflight and hydration studies, the current study found brain volume decreases mostly localized to the supratentorial region (41,48). Lee et al. found free water redistribution mainly located in supratentorial regions, but not in infratentorial brain structures (50). In addition, intrathecal gadobutrol MRI has shown contrast widely distributed throughout the entire brain, with a higher tendency to accumulate in supratentorial periventricular regions (52). These studies suggest that supratentorial brain tissue is more likely to be affected by changes in hydration status. Further studies are needed to corroborate the hypotheses behind the brain volume changes in SIH.
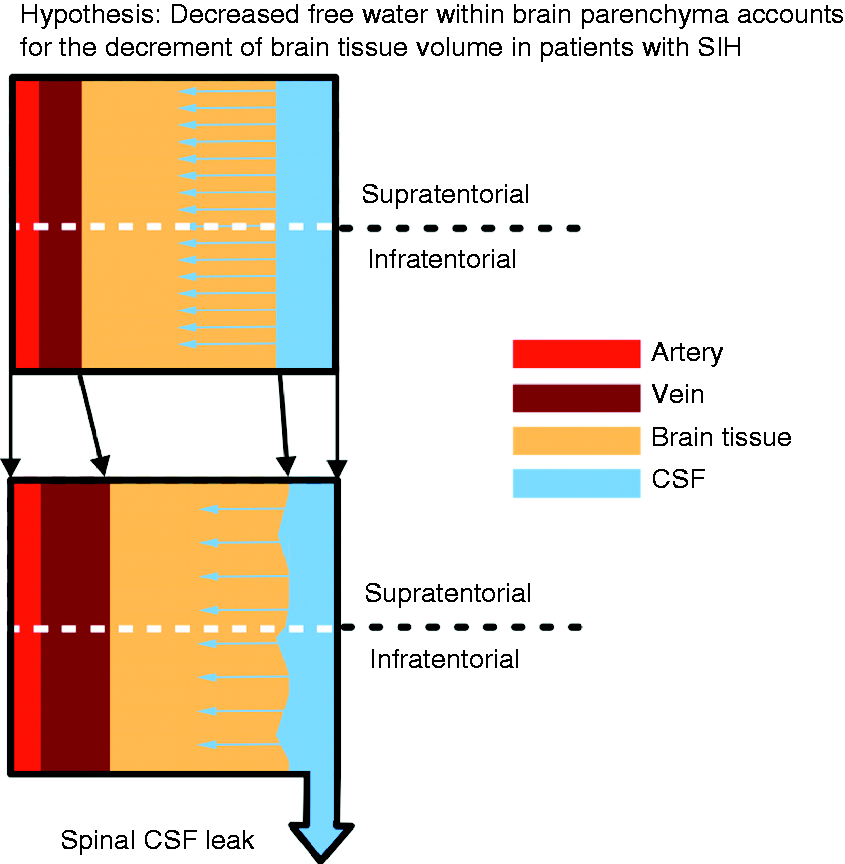
Conceptual diagram demonstrating a hypothesis for the decrease in brain tissue volume in patients with SIH.
Currently, quantitation of brain volume is not routinely performed in SIH patients, and this is the first study to demonstrate that brain volume is changeable in SIH. Brain volume changes were not associated with brain MRI signs. However, the severity of ventricular CSF volume change was associated with spinal MRI findings (more extensive anterior epidural CSF collections). Therefore, in agreement with our previous studies, the length of anterior epidural CSF collections is a reliable clinical parameter for disease severity (15,24).
The current study has limitations that must be considered when interpreting the results. First, there are standard errors in studies using automatic volumetric software. Therefore, we applied the widely used FSL6 Brain Extraction Tool for skull stripping and SPM12 for volumetric measurement because the accuracy of the software is not inferior to or is even better than the other segmentation software (30,31). Second, due to logistic issues of MRI availability in our hospital, our study did not control the timing of the brain MRI studies to avoid circadian variability of structural brain MRI. However, the timing of scanning was left to chance through our hospital’s electronic appointment system, which could minimize the circadian variability of structural brain MRI. Although there was a trend of significance of the mean difference (1.8 hours) in the timing of scanning (within-day), the “Δ timing of scanning” did not correlate with the changes of whole brain tissue volume (Pearson’s r = 0.03, p = 0.852). Third, follow-up neuroimaging during recovery was used as a surrogate for a baseline measurement for comparison of brain volume changes during SIH. However, the possibility of persistent structural changes due to SIH cannot be excluded. Moreover, due to the retrospective design, our study did not have a control group of healthy subjects for comparisons who finished two images within on average 3 months. Fourth, patients with complicated subdural fluid collections or hematoma were excluded from study participation for the following reasons: a) subdural fluid collections or hematoma may interfere with the automatic segmentation algorithms and b) neurosurgical procedures may be confounding factors for measurement of tissue volume in follow-up MRIs. Therefore, the current results can only reflect volume changes in patients without subdural fluid collections or hematoma. On a related note, the median onset-diagnosis interval was longer in the excluded patients group because median onset-diagnosis interval is longer in patients with subdural fluid collection or hematoma (median, 33 days [IQR, 23–58 days] vs. 15 days [IQR, 6–28 days]; Mann-Whitney U test; p < 0.001). After excluding patients with subdural fluid collection or hematoma, the median onset-neuroimaging interval did not differ between the included and excluded patient groups (Mann-Whitney U test; p = 0.407).
Conclusions
The current study found a decrease in brain tissue volume in patients with SIH, challenging the traditional concept that brain volume is constant during SIH. Further studies are warranted to investigate the underlying mechanisms of brain volume changes.
Article highlights
The study findings showed brain volume is decreased during spinal CSF leak, challenging the assumption that the brain tissue volume is invariable in SIH. The brain tissue volume changes did not correlate with the severity of brain sagging, which excluded brain sagging as an explanation of brain volume changes. We found that the severity of ventricular CSF volume change was associated with spinal MRI findings (more extensive anterior epidural CSF collections).
Footnotes
Acknowledgement
We wish to thank the participants of the International Headache Academy (iHEAD) for inspiring discussions during the meeting.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from Taipei Veterans General Hospital [V108C-105, V107C-135], Ministry of Science and Technology of Taiwan [MOST 108-2321-B-010-014-MY2, MOST 108-2321-B-010-001, MOST 108-2314-B-010-023-MY3], Ministry of Health and Welfare, Taiwan [MOHW 107-TDU-B-211-123001, MOHW 108-TDU-B-211-133001], and Brain Research Center, National Yang-Ming University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan.
