Abstract
Background
Monoclonal antibodies against calcitonin gene-related peptide (CGRP) or its receptor (anti-CGRP(-R) mAbs) and small-molecule CGRP receptor antagonists (gepants) are new mechanism-based prophylactic drugs developed to address the unmet needs of pre-existing migraine prophylactic medications. However, several uncertainties remain in their real-world applications.
Methods
This is a narrative review of the literature on the use of CGRP-targeting novel therapeutics in specific situations, including non-responders to prior therapy, combination therapy, switching, and treatment termination. In the case of lack of available literature, we made suggestions based on clinical reasoning.
Results
High-quality evidence supports the use of all available anti-CGRP(-R) mAbs (erenumab, galcanezumab, fremanezumab, and eptinezumab) in non-responders to prior therapy. There is insufficient evidence to support or reject the efficacy of combining CGRP(-R) mAbs or gepants with oral migraine prophylactic agents or botulinum toxin A. Switching from one CGRP(-R) mAb to another might benefit a fraction of patients. Currently, treatment termination depends on reimbursement policies, and the optimal mode of termination is discussed.
Conclusions
New prophylactic drugs that target the CGRP pathway are promising treatment options for patients with difficult-to-treat migraine. Individualized approaches using a combination of new substances with oral prophylactic drugs or botulinum toxin A, switching between new drugs, and adjusting treatment duration could enhance excellence in practice.
Introduction
Migraine prophylactic treatment is essential to reduce the number of days with migraine and associated disabilities in patients with high-frequency episodic migraine (EM) or chronic migraine (CM), those with highly disabling attacks despite acute treatment, or those who are intolerable or contraindicated to acute migraine medication (1). However, prophylactic treatments for migraine have been unsatisfactory due to a lack of disease-specific drugs, slow mode of action, and poor tolerability. These characteristics lead to low adherence rates (2,3).
Novel migraine prophylactic drugs, such as anti-calcitonin gene-related peptide (CGRP) or CGRP receptor monoclonal antibodies (anti-CGRP(-R) mAbs: erenumab, fremanezumab, galcanezumab, and eptinezumab) and small-molecule CGRP receptor antagonists (gepants: atogepant and rimegepant), have been developed and approved by the United States Food and Drug Administration (1,4,5). While these new drugs are believed to address some unmet needs of preexisting migraine prophylactic medications, several uncertainties remain in their real-world applications. Here, we review the existing evidence and focus on the common challenges in the daily use of new prophylactic drugs for migraine. We outline the effects of novel therapeutic use in patients without treatment success who underwent multiple previous prophylactic treatments, discuss strategies for non-responders to the initial anti-CGRP(-R) mAb therapy (switching between novel therapeutics, combination therapy and conventional drugs), and highlight data regarding medication pauses after successful anti-CGRP(-R) mAb therapy to reach an optimal mode of treatment termination.
Efficacy and safety of new therapies in patients who failed conventional migraine preventive treatments
Randomized clinical trial (RCT)
Several double-blind, placebo-controlled RCTs have assessed the efficacy and safety of anti-CGRP(-R) mAbs over 12 weeks in patients with difficult-to-treat migraine who underwent prior unsuccessful prophylactic migraine treatment (Table 1). Patients who previously had an inadequate response to two to four migraine prophylactic medications (or categories) were recruited in the erenumab (LIBERTY), fremanezumab (FOCUS), galcanezumab (CONQUER), and eptinezumab (DELIVER) studies (Table 1) (6–9). Although patient characteristics differed among studies (100% EM, 61% CM, 42% CM, and 46% CM in LIBERTY, FOCUS, CONQUER, and DELIVER, respectively), all these three-month trials showed the superiority of anti-CGRP(-R) mAbs in such patient populations.
Randomized clinical trials (RCT) with anti-calcitonin gene-related peptide (CGRP) or CGRP receptor monoclonal antibodies in patients with prior unsuccessful prophylactic treatments.
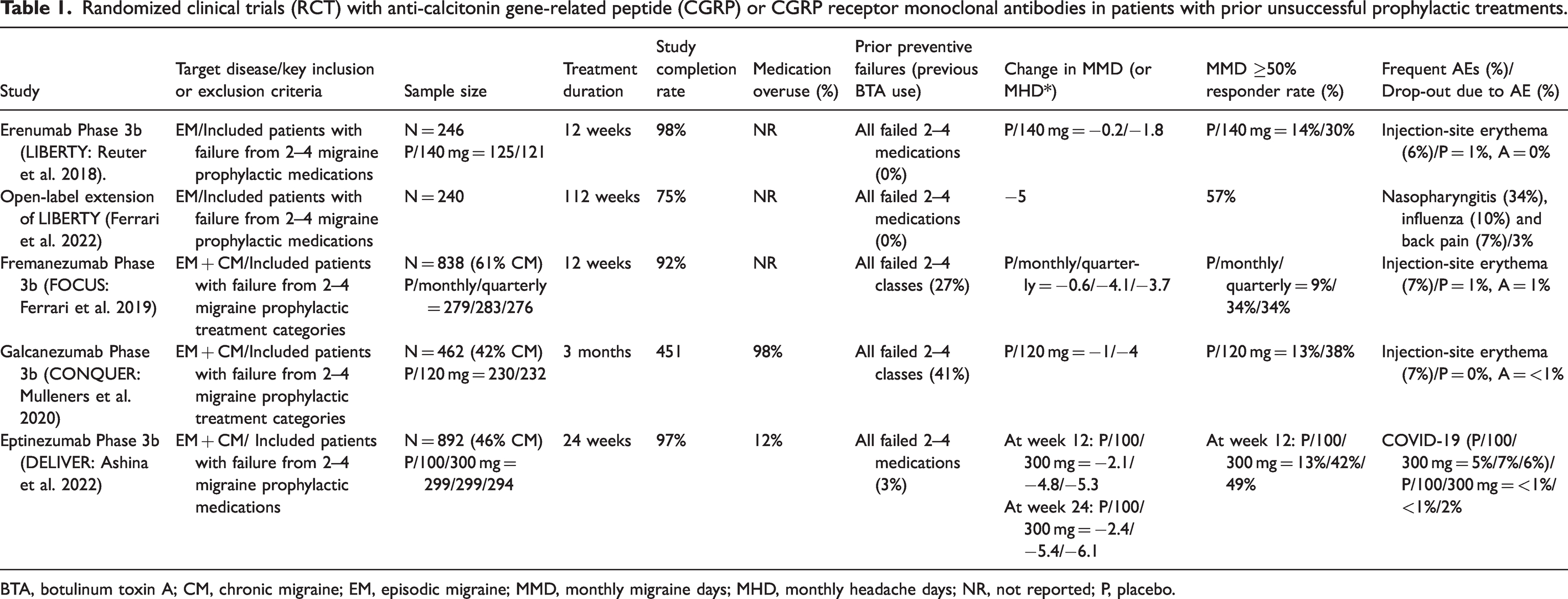
BTA, botulinum toxin A; CM, chronic migraine; EM, episodic migraine; MMD, monthly migraine days; MHD, monthly headache days; NR, not reported; P, placebo.
LIBERTY included EM patients (N = 246) and reported that a monthly erenumab dose of 140 mg led to a significantly higher proportion of patients (30%) reaching ≥50% reduction in monthly migraine days (MMD) and a significant reduction in MMD (−1.8) compared to placebo (Table 1). Patients from LIBERTY (n = 240) entered the open-label extension phase (OLEP) (24 months), with 122 patients switching from placebo to erenumab, and 118 patients continuing with erenumab. The efficacy outcome of OLEP included the proportion of patients with a ≥50% reduction in MMD from the baseline in the double-blind phase. After reaching the initial ≥50% response threshold, a <40% response versus baseline was considered a non-response in the following assessments. An MMD reduction between 40% and 50% was acceptable, and the participant was considered a ≥50% responder if the response at the next visit was ≥50%. A participant with <50% response at two consecutive visits was considered a non-responder for both periods. The main finding of the OLEP study was that the ≥50% responder rate was 57.2%, the reduction in MMD was five days, and approximately one-third of non-responders (failed to reach ≥50% reduction in MMD) converted to being responders after continuous treatment (10). FOCUS included EM and CM patients (n = 838) and reported that fremanezumab (monthly 225 mg or quarterly 675 mg) led to a significantly higher proportion of patients (34%), reaching ≥50% reduction in MMD and a significant reduction in MMD (−4) compared to placebo. CONQUER included EM and CM patients (n = 462) and reported that monthly galcanezumab 120 mg led to a significantly higher proportion of patients (38%), reaching ≥50% reduction in MMD and a significant reduction in MMD (−4) compared to placebo. DELIVER included EM and CM patients (n = 892) and reported that monthly eptinezumab (100 or 300 mg) led to a significantly higher proportion of patients (42%/49%), reaching ≥50% reduction in MMD and a significant reduction in MMD (−5.4/−6.1) compared to placebo.
Collectively, RCTs revealed anti-CGRP(-R) mAbs (erenumab, fremanezumab, galcanezumab, and eptinezumab) to have a superior efficacy as compared to placebo treatment in patients with EM and CM, without success in previous prophylactic treatments. Across studies, the safety profiles and adverse events leading to discontinuation of anti-CGRP(-R) mAbs were similar to those of the placebo (Table 1).
RCTs with oral gepants (atogepant 10, 30, or 60 mg and rimegepant 75 mg) for the prophylactic treatment of EM included patients with multiple prior unsuccessful prophylaxis regimens (11,12). Conclusions regarding the efficacy of gepants for the treatment of difficult-to-treat migraines cannot be drawn because subgroup data have not been reported to date.
Real-world studies
Real-world studies are heterogeneous in terms of the study setting, design, and outcome measurement. Nonetheless, valuable information can be obtained from real-world evidence that cannot be captured in the RCT setting. For example, most RCTs on difficult-to-treat migraine included patients who had unsuccessful responses to drugs from two to four oral medication categories and did not allow the recent or concomitant use of botulinum toxin A (BTA). Patients included in real-world studies had worse clinical profiles than of those participating in RCTs. We summarize the details of real-world studies regarding erenumab in Table 2 and fremanezumab and galcanezumab in Table 3. To date, no real-world studies of eptinezumab and gepants have been published.
Real-world studies with erenumab.

AMD, monthly acute medication days; BTA, botulinum toxin A; DE, dose escalation; MHD, monthly headache days; MMD, monthly migraine days; NR, not reported. Percentage of patients with adverse events leading to discontinuation ranged 0–12% (not shown). Percentage of patients with no efficacy leading to discontinuation ranged 0-22% (not shown). Most reported adverse event was constipation (ranged 10-65%). *The assessment period was the last month of treatment compared to the baseline. **The number of patients included in the efficacy analysis.
Real-world studies with galcanezumab and fremanezumab.
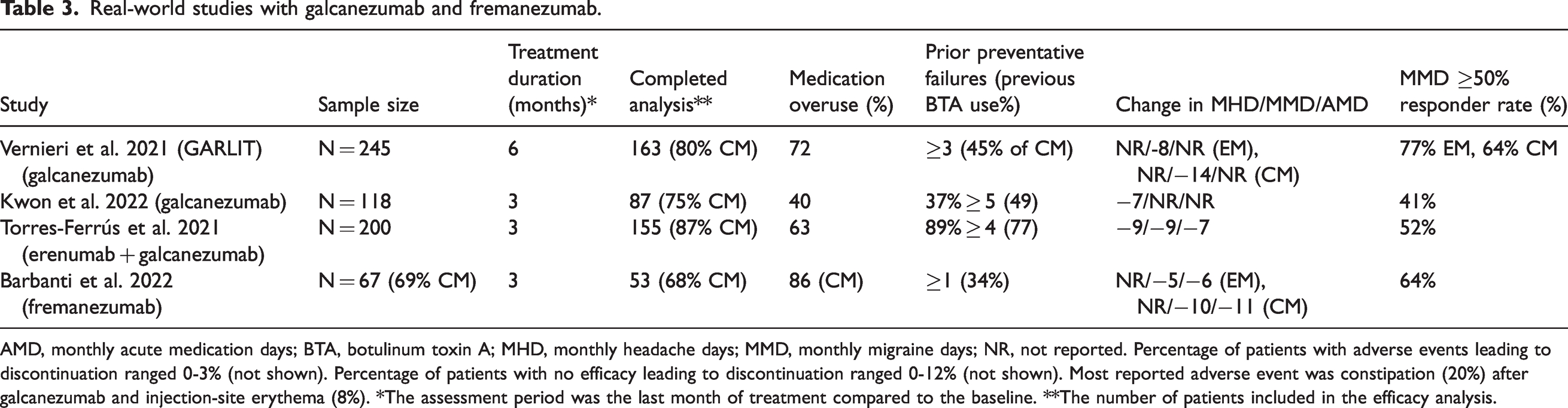
AMD, monthly acute medication days; BTA, botulinum toxin A; MHD, monthly headache days; MMD, monthly migraine days; NR, not reported. Percentage of patients with adverse events leading to discontinuation ranged 0-3% (not shown). Percentage of patients with no efficacy leading to discontinuation ranged 0-12% (not shown). Most reported adverse event was constipation (20%) after galcanezumab and injection-site erythema (8%). *The assessment period was the last month of treatment compared to the baseline. **The number of patients included in the efficacy analysis.
Real-world studies (13–30) assessed the efficacy of erenumab using the following parameters: mean reduction in monthly headache days (MHDs), monthly migraine days (MMDs), monthly acute medication days (AMDs) and the proportion of patients reporting a ≥50% reduction in MMD in patients with EM and CM (Table 2). The treatment duration ranged from two to 12 months, with a commercially available dose (i.e., erenumab 70 or 140 mg every four weeks). Patient profiles were usually worse than those in RCTs in terms of prior unsuccessful prophylactic treatments (1–9), medication overuse (19%–100%), and non-responsiveness to BTA (up to 100%). Real-world data showed that 27%–88% of patients reported a ≥50% reduction in MMDs (Table 2). A large percentage of patients (30%–94%) required dose escalation, suggesting a higher effectiveness of erenumab 140 mg (16–18,23,27–29). Treatment interruption due to lack of effectiveness and adverse events was 0%–22% and 0%–12%, respectively. Real-world studies of erenumab reported that 22%–83% of patients converted from CM to EM (16,17,19,27,30), and one subgroup analysis showed a higher rate of conversion among patients with difficult-to-treat migraine with a long history of CM and multiple prior unsuccessful prophylactic treatments, including BTA (31).
A limited number of real-world studies are available for fremanezumab and galcanezumab (Table 3). In a 12-week multicenter open-label study of fremanezumab, all patients had at least one unsuccessful prophylactic medication (average therapy, 4.5) (32). In this study, the 50% responder rates were 77% and 58% in patients with EM and CM, respectively (Table 3). In a six-month multicenter study of galcanezumab (33), patients with EM and CM with three or more prior unsuccessful prophylactic treatments (maximum 12 therapies) received galcanezumab (loading dose 240 mg and 120 mg monthly thereafter), and the proportion of patients reporting a ≥50% reduction in MMDs was 77% and 64% for patients with EM and CM, respectively. Another real-world study (34) supported the efficacy of galcanezumab even in patients with five unsuccessful prophylactic medication categories (37% of patients). Torres‑Ferrús et al. (35) administered erenumab (n = 109) and galcanezumab (n = 46) to 155 patients with EM and CM with four or more prior unsuccessful prophylactic treatments and reported that 52% of the patients were 50% responders in MMDs.
Taken together, real-world experience supports the efficacy of anti-CGRP(-R) mAbs, even in patients who were excluded from the RCTs owing to multiple prior unsuccessful migraine prophylactic treatments.
Use of new therapies in patients with difficult-to-treat migraine
By interpreting the evidence from clinical trials and real-world data discussed above, it is evident that novel CGRP-targeting therapies provide a new horizon for migraine treatment. However, the high cost of these new drugs has prevented their widespread use. Currently, patients who previously had an insufficient response to old migraine prophylactic drugs are considered a priority target for new therapies, and anti-CGRP(-R) mAbs and gepants are currently defined as third-line treatments under national or private insurance programs in most countries worldwide. We agree with the compelling necessity of new therapies in patients with an insufficient response to multiple migraine prophylactic medications. Nevertheless, although the study designs were not directly comparable, the 50% responder rate from most trials targeting difficult-to-treat migraine seems to be smaller than that reported in trials where patients who had an insufficient response to multiple medications were excluded (Table 4) (36–39). This may suggest that earlier intervention with anti-CGRP(-R) mAb or gepants may help prevent disease progression to an indeed “difficult-to-treat” migraine. Similar to our point of view, the expert panel suggested including anti-CGRP(-R) mAbs as a first-line treatment option in the 2022 update on the European Headache Federation (EHF) guidelines (40). Currently, reimbursement in Europe is not in line with the use of anti-CGRP(-R) mAb derived from most clinical trials in which treatment-naïve patients were recruited. Although not reimbursed by insurance services, anti-CGRP(-R) mAbs are increasingly preferred as a first-line treatment for patients who want to avoid oral prophylactics owing to potential adverse effects, drug-drug interactions, or slower onset of action.
50% responder rates in clinical trials with anti-calcitonin gene-related peptide (CGRP) or CGRP receptor monoclonal antibodies in patients without vs. with prior unsuccessful prophylactic treatments.
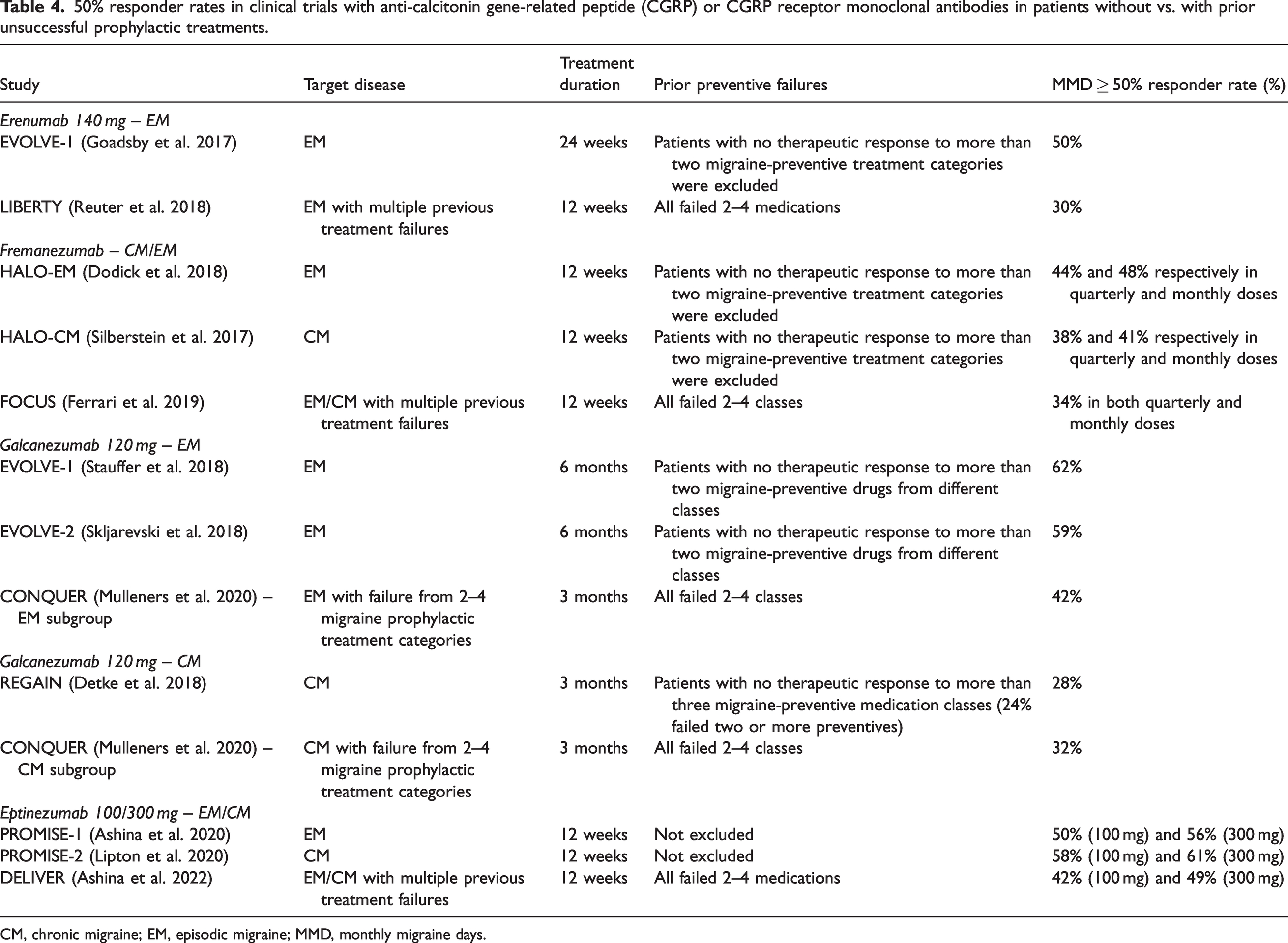
CM, chronic migraine; EM, episodic migraine; MMD, monthly migraine days.
Combination therapy
Under the current reimbursement policies of most countries, new migraine preventive therapies, including anti-CGRP(-R) mAbs and gepants, can be reimbursed only in patients who do not show an adequate response to or have contraindications or poor tolerability to multiple migraine prophylactic treatments. There is no global consensus on how to handle previously used migraine prophylactic medications when initiating anti-CGRP(-R) mAbs. Therefore, whether new therapies will be added to previous treatments or will be used alone is dependent on reimbursement policies and national guidelines and is debatable in several instances. The 2018 EHF guidelines suggested discontinuing oral prophylactics before starting anti-CGRP(-R) mAbs in patients with EM unless the patient had a previous history of CM, whereas it was recommended to add anti-CGRP(-R) mAbs to the preexisting oral prophylactics in patients with CM and consider later withdrawal of the oral drugs (41). In the 2022 update of the EHF guidelines, the combined use of other prophylactics was considered optional and left to individual consideration (40). The American Headache Society guidelines state that new therapies can be added to one or more existing regimens based on clinical judgment (42). Regarding BTA, the recommendation in the 2018 EHF guidelines is to discontinue the BTA injection when starting the anti-CGRP(-R) mAbs if BTA previously provided an inadequate treatment response (41), whereas individualized decisions are more emphasized and combination treatments seem more accepted in the American Headache Society (AHS) guideline (43). Herein, we review the rationale and clinical evidence for combination therapy in terms of efficacy and safety.
Rationale of combination therapy with oral preventive medications or BTA
Although controversy exists, a combination of two or more prophylactic treatments with different mechanisms is widely used in practice to enhance the efficacy or avoid adverse effects of high-dose monotherapy (44–46). This can be achieved by combining low-dose oral preventive drugs or non-pharmacologic options with oral preventive drug(s) for patients with a poor response or tolerability to each drug. However, there is no high-quality study comparing monotherapy and combination therapy. Nevertheless, the absence of high-quality studies does not mean futility, but it can be partly attributed to funding issues, and several case series or small-sized randomized trials have shown the potential efficacy of combination therapy, although they can provide only limited evidence (47–51). Given that patients receiving anti-CGRP(-R) mAbs or gepants are more likely to have had insufficient responses to multiple evidence-based migraine prophylactic agents, and the response rates of these new drugs are not excellent in such populations (Table 1) (6–8), combination therapy may eventually be required.
The rationale for combining novel CGRP-targeting therapeutics with oral prophylactic drugs is based on their different mechanisms of action. Anti-CGRP(-R) mAbs and gepants affect trigeminal nociceptive pathways by selectively acting on CGRP or its receptors. In contrast, oral prophylactics can also act centrally on multiple mechanisms of migraine, including central sensitization, neuronal excitability, and cortical spreading depression (52). Thus, the combination of anti-CGRP(-R) mAbs or gepants with oral migraine prophylactics can be theoretically beneficial. From a safety perspective, the pharmacokinetics and pharmacodynamic profile of anti-CGRP(-R) mAbs do not change with oral medications or vice versa. This is because the absorption and metabolism of mAbs are distinct from those of oral medications (53). Anti-CGRP(-R) mAbs were rated to have a low likelihood of interacting with anti-epileptic drugs (54). In contrast, gepants can interact with oral drugs that inhibit or induce the hepatic enzyme CYP3A4 (53). Among first-line migraine preventive drugs, topiramate is a weak inducer of CYP3A4 (55). In addition, verapamil and amlodipine can increase the plasma concentration of gepants (53,55). Despite these theoretical concerns, the concomitant use of verapamil and topiramate was not prohibited during the rimegepant trial (12).
The combination of CGRP-targeting treatment with BTA has also been investigated. Anti-CGRP(-R) mAbs preferentially block the release of CGRP from Aδ fibers, whereas BTA inhibits the stimulated release of CGRP from C fibers (56). This difference could be the biological rationale for the combination of anti-CGRP(-R) mAbs and BTA. Gepants seem to act outside the blood–brain barrier (57,58), and atogepant partially inhibits both Aδ and C fibers (59), the pattern of which is different from that of BTA (56). A combination of atogepant and BTA resulted in robust inhibition of neurons in the spinal trigeminal nucleus, probably by blocking both Aδ and C fibers in a preclinical study (60).
Clinical evidence of efficacy and safety of combination therapy
To date, no study has directly compared the efficacy of anti-CGRP(-R) mAb monotherapy and combination therapy with oral migraine prophylactic drugs. Several clinical trials of anti-CGRP(-R) mAbs have included a subpopulation (3–99% of the total population) who were on the concomitant use of one (61–63) or more (64) migraine prophylactic medications. A subgroup analysis comparing participants with and without concomitant oral prophylactic drugs is currently unavailable, and patient characteristics may not have been balanced between the two groups. Clinical trial data have shown that the combination of oral prophylactic medications with anti-CGRP(-R) mAbs is generally accepted and considered safe, and thus even allowed in such trials. In real-world studies of anti-CGRP(-R) mAbs, oral migraine prophylactics were used more frequently (42–79%). This was probably because they included patients who were more refractory to prior treatment than those who were eligible for clinical trials. However, the rate of serious adverse events remained low. Taken together, there is insufficient evidence to support the efficacy of combination therapies. Nonetheless, the combined use can be safe, and individualized decisions should be made in patients with an insufficient response to anti-CGRP(-R) mAbs.
Unlike oral prophylactics, the concomitant use of BTA was not allowed in clinical trials of anti-CGRP(−R) mAbs. However, the combination of BTA and anti-CGRP(-R) mAb has been frequently reported in real-world studies, with 34% in pooled data (65–67). The combined efficacy seems to be an additive effect of the therapeutic gains from each therapy (67). When classified into previous responses to BTA, partial responders may benefit from combination therapy with anti-CGRP(-R) mAbs, whereas non-responders do not show a superior treatment response to combined therapy compared with anti-CGRP(-R) mAb monotherapy (66). Based on the current data, no new safety concerns were raised owing to the combined use of BTA and anti-CGRP(-R) mAbs.
For gepants, combination therapy with other oral prophylactics was allowed in a phase 2b/3 rimegepant trial (12), and a recent atogepant trial for CM also included a subset of patients using one concomitant oral preventive medication (ClinicalTrials.gov identifier: NCT03855137). In the rimegepant trial, BTA was allowed if used at a stable dose and frequency (12). Subgroup analysis is currently not available, and there are no real-world studies regarding the preventive use of gepants.
Switching between new therapies
The main difference between the available anti-CGRP(-R) mAbs is that the site of action is the CGRP receptor (erenumab) and the CGRP ligand (fremanezumab, eptinezumab, and galcanezumab). Although anti-CGRP(-R) mAbs generally have a good safety profile, adverse events may occur, resulting in treatment discontinuation. If the reported adverse events are related to a specific class (e.g., constipation related to erenumab), switching to a different class may be appropriate based on clinical experience.
However, switching based on efficacy is much more complex due to data scarcity. A real-world retrospective study reported that 32% of erenumab non-responders showed a clinically meaningful reduction in MHD (≥30%) after switching to galcanezumab or fremanezumab (68). A ≥50% response was achieved in 12% of the participants (68). Interestingly, patients with persistent daily headaches did not benefit from switching treatment. Some case reports (69,70) also demonstrated a clinically meaningful reduction in headache days after switching to galcanezumab, despite erenumab treatment failure. These data indicate that switching is a promising treatment option.
There are no available data regarding the comparative efficacies of different mAb classes. Switching from anti-CGRP-R mAb to anti-CGRP mAb was most frequently encountered, owing to the earliest approval of erenumab among anti-CGRP(-R) mAbs in most European countries and the United States. Whether non-responders to anti-CGRP mAb treatment may benefit from treatment with a different anti-CGRP mAb class or an anti-CGRP-R mAb remains to be elucidated. A possible explanation for the variation in therapeutic efficacy may be the existence of different subtypes of migraine, in which other neuropeptides, such as amylin, adrenomedullin, and pituitary adenylate cyclase-activating polypeptide, may play predominant role (71–73).
The switching algorithm is presented in Figure 1. In addition to differences in the site of action, considerations to support a switch between anti-CGRP(-R) mAbs include the opportunity for dose escalation (from erenumab 70 to 140 mg), differences in the administration schedule (monthly versus quarterly), and differences in formulations (subcutaneous versus intravenous) (Figure 1). Eptinezumab is the only intravenous anti-CGRP(-R) mAb. Eptinezumab reaches its maximum serum concentration within minutes to hours, whereas other antibodies require up to one week to reach their maximum levels (74–76). Notably, in an acute migraine treatment setting, switching between drugs within the same class (e.g., triptans) is an accepted strategy. Despite insufficient evidence, switching to another anti-CGRP(-R) mAb or gepants might be an option for patients with an inadequate response to one anti-CGRP(-R) mAb.

Switching algorithm. Selection of mAb class is dependent on availability and patient’s comorbidities and preferences but not on the efficacy. Assessment of the benefits of mAbs after six to 12 months.
No study has directly compared the efficacy profiles of anti-CGRP(-R) mAbs and gepants. However, anti-CGRP(-R) mAbs and gepants share a similar target (the CGRP pathway), and some adverse reactions are unique to a specific class (injection site reaction in all mAbs, constipation in erenumab, anaphylactic reactions reported in erenumab and eptinezumab users, but potentially in all other mAbs). Thus, considerations to support a switch from mAb classes to gepants and vice versa are currently not based on comparative efficacy but on a difference in the administration schedule (monthly and quarterly [mAb classes] versus once daily [gepants]) and differences in formulations (subcutaneous and intravenous [mAb classes] versus oral [gepants]). Additionally, a switch between gepants (atogepant and rimegepant) is yet to be elucidated.
Treatment termination
How long the new preventive therapies should be continued is a common question for patients. Currently, there is no clear evidence-based answer. In practice, treatment duration is affected by the cost and reimbursement program. In this section, we review the current recommendations for treatment duration and the clinical data regarding treatment termination.
Current recommendation from local guidelines
The EHF guidelines recommend stopping mAbs after six to 12 months of treatment (41). Most local guidelines do not specify the treatment duration or termination of new CGRP-targeting therapies, leaving it to the physician’s discretion (42,77). However, insurance programs usually reimburse anti-CGRP(-R) mAb treatments for only six to 12 months and require a certain period of discontinuation to determine the necessity of treatment re-initiation. In Germany, patients who show a significant response in the initial three months can continue for another six to nine months (78). Thereafter, treatment interruption is highly recommended to assess the necessity of continuing anti-CGRP(-R) mAb treatment (78). In Italy, patients who show a sustained treatment response at three and six months of treatment can be reimbursed for a one-year prescription of anti-CGRP(-R) mAbs, and three months (now one month) of drug holidays are required to evaluate the necessity of restarting treatment. Other local policies have similar structures but differ in detail across the globe, whereas not all anti-CGRP(-R) mAbs are reimbursed by the national health insurance program in some countries (e.g., South Korea); thus, no external regulations regarding prescription exist.
Clinical perspective
Ideally, physicians should make a clinical decision regarding treatment termination not based on reimbursement policies but on the treatment goals developed for each patient. Generally, a 50% (or 30%) reduction in MMDs (or MHDs) is considered the cutoff for treatment response. However, this may not be sufficient for the patient’s well-being. In contrast, some patients benefit from new treatments, even though they are classified as non-responders (79). After the discontinuation of anti-CGRP(-R) mAb treatment, symptoms of most patients tended to deteriorate (27,80–83). Furthermore, 70%–90% of patients require treatment re-initiation after three months of discontinuation (82,83). Baseline disability and outcome measures during discontinuation are associated with a sustained response after discontinuation (83).
From a clinical perspective, it seems beneficial to continue anti-CGRP(-R) mAbs until an optimal treatment outcome is achieved. For oral prophylactics, the goal is ≤4 headache days per month when treatment termination is determined. This treatment goal can be applied to maintain and terminate CGRP-targeting therapies. If necessary, oral prophylactic drugs or BTA should be administered to achieve an optimal response. If the optimal outcome is achieved and stably maintained for three to six months, the treatment can be discontinued. The mode of discontinuation can be either abrupt or tapered (i.e., increasing the injection interval), of which the abrupt method is more common in most countries under the current guidelines, and the tapered method is adopted by some experts where available. Therefore, patients should be closely monitored during treatment or tapering. Re-initiation can be performed with the same anti-CGRP(-R) mAb if the initial treatment is successful (80). Figure 2 displays an algorithm for treatment termination that we suggest. For gepants, no data are available on the duration and termination of therapy.

Suggested algorithm for treatment termination.
Suggestions for future research
The introduction of novel CGRP-targeting therapeutics has led to a new and promising era in the treatment of migraine. Reliable data support the safety and efficacy of anti-CGRP(-R) mAbs and gepants in patients with difficult-to-treat migraine with up to four prior unsuccessful prophylactic treatments; however, several challenges remain. No published trial has compared the efficacy of an anti-CGRP mAb, an anti-CGRP-R mAb, or gepants, and the choice is up to the physician. Several factors influence this choice, including availability, pharmacokinetic properties, comorbidities, and patient preferences. Objective biomarkers to predict the efficacy of treatment initiation with anti-CGRP(-R) mAbs, gepant, or switches remain missing. Total freedom from migraine is rarely achieved with new prophylactics, which demonstrates the complexity of the disease.
To date, it is not understood why the majority of patients respond to migraine prophylactics and some do not. Adjustment of some parameters, including treatment duration and dose escalation, may increase effectiveness and present an option before switching, particularly in light of data showing a higher response rate upon prolonged treatment duration or after dose escalation (16–18). A randomized, placebo-controlled, single-ascending-dose study assessing the safety and efficacy of higher-dose anti-CGRP(-R) mAbs in non-responders is necessary. Meeting individualized needs may also involve combining older and newer treatments or the administration of a complex of receptor and ligand mAbs. A trial designed to determine the nature and extent of any clinically meaningful synergistic effects of these treatments is warranted. Migraine attack frequency fluctuates over time, and migraine may improve or remit; therefore, it is important to re-evaluate the therapeutic response and determine whether to continue or, if possible, taper or discontinue treatment if patients no longer meet the criteria for preventive treatment. A randomized withdrawal trial might provide insight into the natural history of migraine after discontinuation of anti-CGRP(-R) mAbs and identify the risk factors for migraine relapse and progression. Eventually, non-responders to anti-CGRP(-R) mAbs and gepants pose a challenge for clinicians, and treatment options for these subgroups, if any, are very limited.
Conclusion
Novel therapeutics that target the CGRP pathway are promising treatment options for patients with difficult-to-treat migraine. With an excellent safety and tolerability profile, the efficacy of anti-CGRP(-R) mAbs has been documented in well-designed trials, specifically targeting patients who have an insufficient response in terms of efficacy or tolerability to multiple migraine prophylactic drugs. Nevertheless, treatment outcomes and response rates remain suboptimal in this patient population. Thus, combination therapy and switching between new drugs may be the next step despite the paucity of high-quality evidence. The early application of new therapeutics and maintenance of optimal treatment outcomes can be helpful in preventing disease progression and worsening after treatment.
Clinical implications
Novel migraine prophylactic drugs (i.e., anti-CGRP(-R) mAbs and gepants) are promising treatment options for patients with difficult-to-treat migraine. There is high-quality evidence of the efficacy of anti-CGRP(-R) mAbs in patients with an insufficient response to previous migraine prophylactic drugs. In non-responders to anti-CGRP(-R) mAbs or gepants, combination therapy with oral prophylactics or BTA or switching between CGRP-targeting therapeutics can be considered and supported by pharmacological properties, preclinical evidence, small case series, and safety profiles. Treatment termination is currently regulated by the reimbursement. Nonetheless, individualized treatment may benefit patients and help achieve an optimal treatment outcome.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MJL has received personal or institutional honoraria for participation in clinical trials, speaker fees, or as consultants from Abbvie, Biohaven, Eli Lilly, Lundbeck, Novartis, Sanofi-Aventis, SK Pharm, Teva (Otsuka), Yuhan Company, and YuYu Pharma, and research support from the National Research Foundation of Korea and Seoul National University. MAA reports no conflicts of interest. UR received personal or institutional honoraria for participation in clinical trials, speaker fees, or as consultants from Abbvie, Allergan, Alder, Amgen, Eli Lilly, Lundbeck, Medscape, Novartis, Pfizer, StreaMedUp, Teva, and research support from the German Ministry for Education and Research and Novartis (CHERUB01).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the New Faculty Startup Fund from Seoul National University, a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIP; No. 2020R1A2B5B01001826 to MJL), the Research Fund of Rigshospitalet Denmark (E-23327-04 to MAA), and the Lundbeck Foundation (R310-2018-3711 to MAA).
