Abstract
Background
Cluster headache (CH) is one of the most severe types of pain, yet pharmacological treatment options are limited. In the present study, we aimed to systematically evaluate the effect of treatment with monoclonal antibodies (mAbs) blocking calcitonin gene-related peptide (CGRP) in the prevention of episodic cluster headache (eCH) and chronic (cCH) cluster headache.
Methods
We searched Embase, Medline, Cochrane CENTRAL and Web of Science. Included were all clinical trials, case series and single case reports that reported the effects of anti-CGRP treatment on CH. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) were used for abstracting data. Risk of bias was evaluated using the Risk of Bias 2 (RoB 2) tool for randomized trials and Risk Of Bias In Non-randomized Studies – of Interventions (ROBINS-I) for non-randomized studies. The collected data from all studies were summarized using descriptive statistics. Data from randomized trials were additionally reported as odds ratio (OR) with 95% confidence interval (CI) using a random-effects meta-analysis. Given the variability in study design and the diverse formats of reported outcome data, we primarily focused on the ≥ 50% responder rate reported closest to the 4-week point following drug administration, which was consistently reported in almost all studies.
Results
From 734 identified records, 25 articles were included, comprising a total of 1587 patients. Meta-analysis of randomized, placebo-controlled trials suggests that anti-CGRP treatment had a statistically significant positive effect in eCH (OR = 1.65, 95% CI = 1.07–2.55, p = 0.02), with galcanezumab 300 mg and eptinezumab 400 mg being more effective than placebo in achieving a ≥ 50% responder rate in eCH attacks at the 4-week mark following administration. Simultaneously, a significant mean reduction in weekly attack frequency at week 4 was observed only for galcanezumab 300 mg (p = 0.04). Despite findings from some non-randomized trials, the meta-analysis showed no statistically significant effect in cCH (OR = 1.07, 95% CI = 0.78–1.48, p = 0.68).
Conclusions
Our analysis revealed a beneficial effect of anti-CGRP mAbs in eCH, while no effect was observed in cCH. Despite these positive findings, the favourable results with galcanezumab and eptinezumab in eCH should be interpreted with caution due to discrepancies in the outcome data and the challenges associated with selecting endpoints in CH trials. The discrepancy between real-world data and findings from controlled trials further highlights the need for continued discussions among experts to develop more adequate methods for conducting CH trials.
Trial Registration
PROSPERO ID: CRD420250609351.
This is a visual representation of the abstract.
Introduction
The potential of monoclonal antibodies (mAbs) targeting calcitonin gene-related peptide (CGRP) or its receptors for preventing cluster headache (CH) garnered significant optimism, driven by their established efficacy in migraine prevention. Past experiences nourished this hope: some 25 years prior, triptans, then a migraine treatment, had first been successfully used to treat cluster headache attacks. 1 Additionally, elevated CGRP levels during cluster headache attacks, potentially surpassing those in migraine attacks, further supported this therapeutic rationale.2–4
Despite this promise, the first clinical trials evaluating galcanezumab for CH yielded mixed results. While the drug showed no efficacy in preventing chronic cluster headache (cCH), 5 it demonstrated a statistically significant benefit over placebo in episodic cluster headache (eCH). 6 As a result, the Food and Drug Administration approved galcanezumab for the treatment of eCH in the USA. In contrast, European regulatory authorities (European Medicines Agency) considered the evidence insufficient for approval, judging the observed efficacy of galcanezumab to be too low compared to verapamil, which remains the standard first-line preventive therapy.
These outcome discrepancies led to speculation that the trial endpoints may not have adequately captured the drug's therapeutic impact, prompting questions about the true efficacy of galcanezumab. Subsequent studies of similar CGRP-targeted mAbs were discontinued, likely due to insufficient efficacy, despite adjustments to trial endpoints aimed to achieve a reduction of at least 50% from baseline in weekly attack frequency. Consequently, pharmaceutical companies ceased efforts to pursue licensure for these agents in CH prevention. In contrast, case reports and small case series have still suggested that some patients experience meaningful benefits from these treatments.
Efficacy (i.e. how well a treatment works under ideal conditions) and effectiveness (i.e. how well it works in real-world settings) very often differ. This phenomenon, referred to as the efficacy-effectiveness gap, 7 has led regulatory authorities to insist on conducting post-licensing observational studies with increasing frequency. 8 However, drugs generally perform better in clinical studies than in daily clinical practice, not worse. 8 The reasons for this comprise the more restrictive selection of the participants, the closer surveillance and the more insisting encouragement to adhere to the treatment in clinical trials.7,8 In the case of CGRP antagonists, real-world experience suggests the opposite. 9 These differences may stem from the absence of standardization, control groups, placebo arms or randomization in real-world data, which can introduce biases. 10 Alternatively, the discrepancy could reflect differing conceptualizations of therapeutic success in clinical versus real-world settings.
The present study systematically reviews the available data on the efficacy and effectiveness of monoclonal antibodies directed against CGRP and its receptors in preventing cluster headache attacks.
Methods
The protocol was registered in the Prospero database (CRD420250609351).
Eligibility
Peer-reviewed articles reporting a drug trial meeting the following criteria were eligible.
Population: The study population consisted of patients ≥ 18 years with eCH or cCH, diagnosed according to the diagnostic criteria published in the International Headache Classification, 3beta or 3rd editions (ICHD-3beta, ICHD-3).11,12 Intervention: The study population received a mAb directed against CGRP or its receptor (i.e. erenumab, galcanezumab, fremanezumab, or eptinezumab) to prevent cluster headache attacks. Comparison: We included studies with and without comparators. Outcome: We did not pre-specify the included outcome measures. Time frame: We included studies published up to 29 June 2025. Study design: We included clinical trials, case series and single case reports.
Search strategy
On 29 June 2025, we searched Embase, Medline (through Pubmed), Cochrane CENTRAL, and WebofScience for the terms: “erenumab,” “galcanezumab,” “fremanezumab” and “eptinezumab” combined with the term “cluster headache.”
Study selection
Two reviewers (LK and MTK) independently screened all articles identified with the above search strategy for eligibility based on their titles and abstracts. Next, they evaluated all full-text articles that were not eliminated during screening for suitability. Conflicts were resolved by a third reviewer (HP).
A Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) chart visualizes the number of articles at each step (identification, screening, evaluation of eligibility and inclusion of studies). We selected the articles using the Covidence software (Veritas Health Innovation Ltd, Melbourne, VIC, Australia).
Data extraction
Data extraction from the included studies was performed using Excel (Microsoft Corp., Redmond, WA, USA). We extracted demographic data, the precise headache diagnosis, headache severity at baseline (including intensity and frequency), the intervention (including dose and injection frequency), any deviation from the intended intervention (including adherence issues), the sampling strategy, the duration of the observation period, and all primary and secondary outcome measures. We also collected information on the frequency and types of adverse events (AEs).
Risk of bias assessment
Risk of bias was evaluated by two independent authors (LK and MTK) using the Risk of Bias 2 (RoB 2) tool for randomized trials and Risk Of Bias In Non-randomized Studies – of Interventions (ROBINS-I) for non-randomized Studies.13,14 Conflicts were resolved by consensus after discussion with a third reviewer (HP). To assess the risk of bias, we checked the information included in the publication and its supplements, as well as the National Clinical Trial (NCT) protocol (www.clinicaltrials.gov), if available.
Data analysis
Due to the variability in study design and the significantly different formats of reported outcome data, we decided to assess treatment response by determining the ≥50% responder rate, defined as a reduction of at least 50% from baseline in the weekly frequency of cluster headache attacks, as our main outcome factor. We selected this outcome parameter because corresponding data were available for all studies, except for one. In the study by Riesenberg et al. (2022), the ≥ 50% responder rate was not reported. 15 In line with a previous systematic review on this topic, treatment response was instead assessed using the Patient Global Impression of Improvement (PGI-I) scale, with scores of 1 (‘very much better’) or 2 (‘much better’) considered indicative of improvement. 16
Due to heterogeneity in follow-up durations and endpoint timings across the included studies, which ranged from 1 week to more than 1 year, we used the outcomes reported closest to the 4-week mark after administration as the reference time point in our analysis. This time point was chosen because most studies either defined it as their endpoint or provided data corresponding to it, which ensures greater consistency and accuracy in the analysis.
For double-blind, placebo-controlled trials, we also reported the mean reduction in weekly attack frequency at week 4, which was available for all studies except one. Due to the absence of published categorical data, we were unable to calculate an exact p-value; our assessments were therefore conducted indirectly, based on the information presented in the publications and on the corresponding NCT websites. For non-randomized trials, this parameter could not be assessed because reporting was inconsistent.
Other interesting outcome parameters, such as changes in the use of acute abortive treatments, PGI-I scale scores, or the duration and intensity of attacks, were not reported systematically, limiting a proper comparison.
Collected data were summarized using descriptive statistics. Continuous demographic and clinical variables are presented as the means with standard deviations (SD) or medians with interquartile ranges (IQR), depending on their distribution. Categorical variables are reported as frequencies and percentages. For double-blind, placebo-controlled trials, analyses were conducted using random-effects meta-analyses. Subgroup analyses were performed by categorizing studies based on participants with eCH or cCH. Non-randomized prospective trials and retrospective reports are presented separately due to their different designs, methodologies and potentially different risk of bias.
Statistical analyses were performed using SPSS, version 29.0.0.0 (IBM Corp., Armonk, NY, USA) and Excel 2016 (Microsoft Corp.). p < 0.05 was considered statistically significant.
Results
Included studies
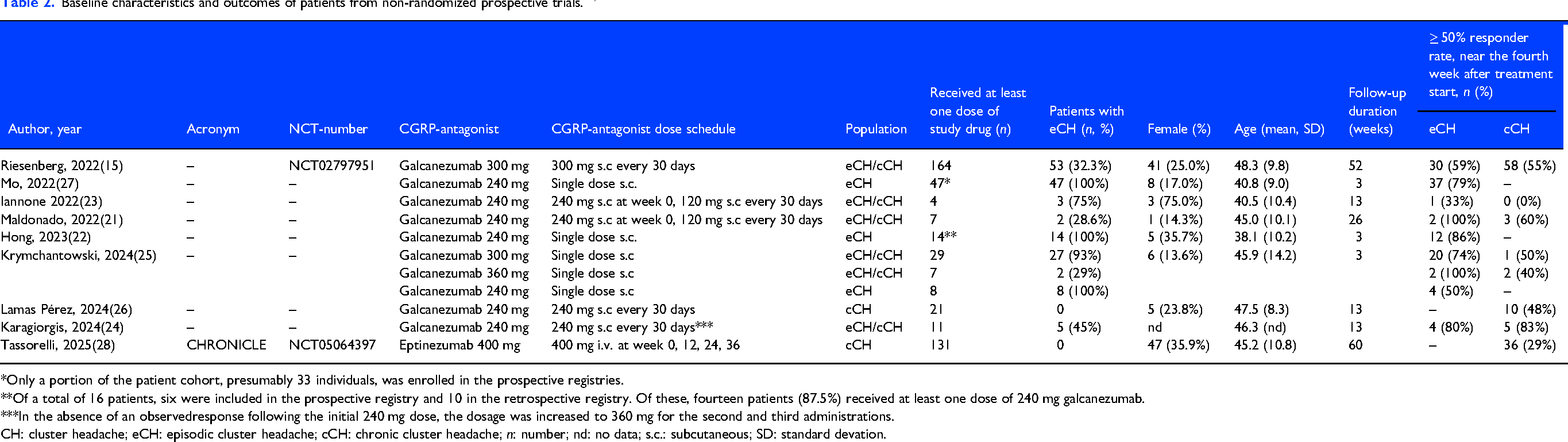
The initial search identified 734 records. After removing duplicates and excluding ineligible studies, 25 articles remained and were assessed in full against the inclusion criteria. The detailed study selection process is illustrated in Figure 1. Among the included studies, there were six double-blind, placebo-controlled trials,5,6,17–20 nine non-randomized prospective studies15,21–28 and 10 retrospective case series and case reports.29–38
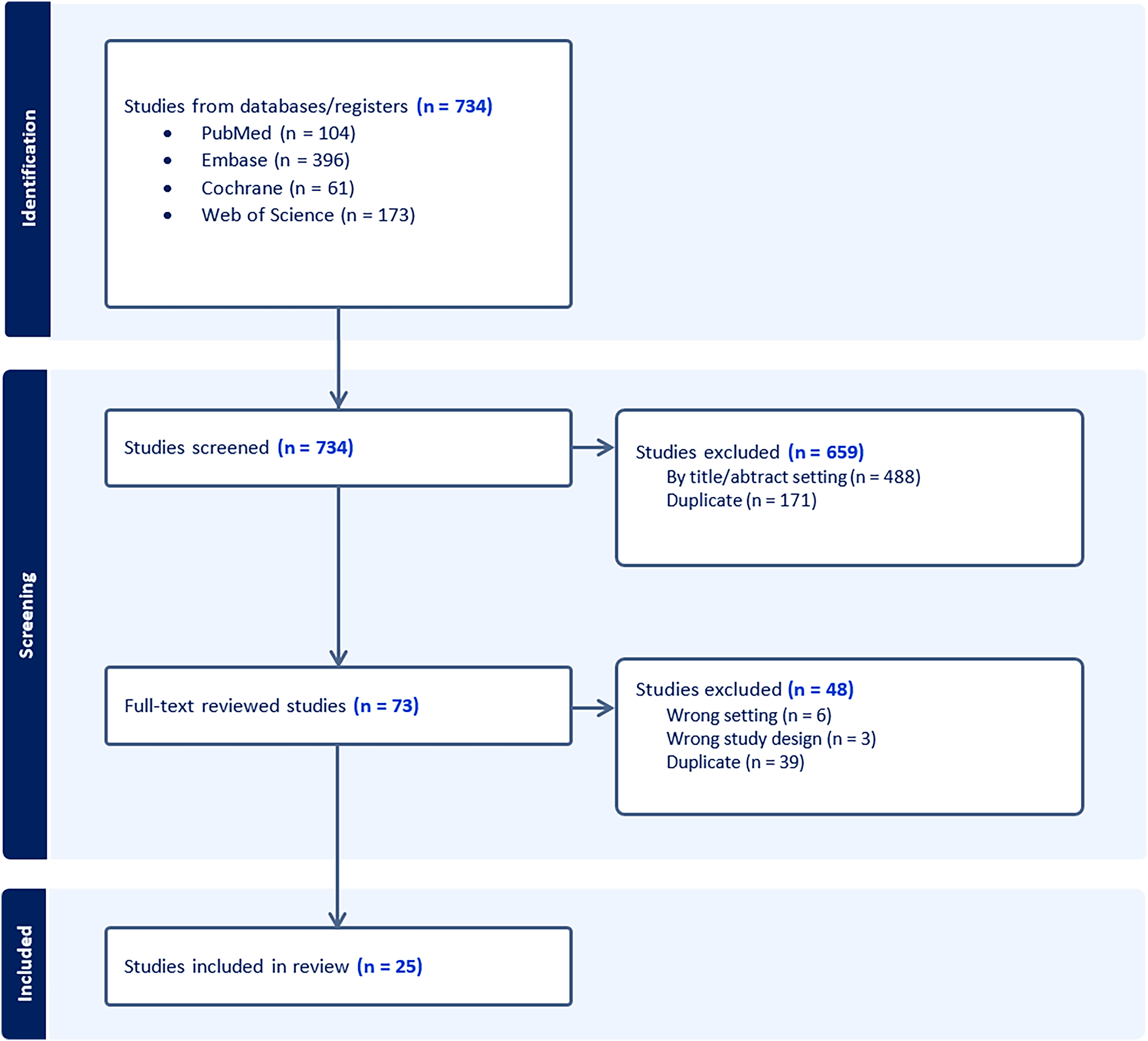
Flowchart outlining the study inclusion.
Out of a total of 1587 patients, the prospective randomized trials included 1081 (68.12%) individuals, of whom 504 (46.62%) had eCH and 577 (53.38%) cCH. Among these participants, 769 were male (71.14%), with a mean age of 45.55 years. In total, 491 patients (45.42%) received a CGRP antagonist: 166 (15.36%) were treated with galcanezumab, 281 (25.99%) with fremanezumab, 102 (9.44%) with eptinezumab and 41 (3.79%) with erenumab. The remaining 590 patients (54.58%) received placebo. All patients treated with galcanezumab received a monthly dose of 300 mg. Those treated with eptinezumab received 400 mg, while patients receiving erenumab were administered 240 mg. Fremanezumab was administered at a dose of 900 mg in 142 patients (50.53%) and 675 mg in 139 patients (49.47%).
In the non-randomized studies, 506 patients (31.88%) were included, of whom 173 (34.19%) had eCH and 333 (65.81%) had cCH. Among them, 370 (73.12%) were male, with a mean age of 42.22 years. In total, 344 patients (67.98%) received galcanezumab, 131 (25.89%) received eptinezumab, 16 (3.16%) received erenumab and 15 (2.96%) were treated with fremanezumab. Galcanezumab was administered at doses of 300 mg (n = 194; 56.40%), 240 mg (n = 143; 41.57%) and 360 mg (n = 7; 1.94%). Fremanezumab was given at doses of 675 mg in two patients (13.33%) and 225 mg in 13 patients (86.67%). Erenumab was administered at 140 mg in 11 patients (68.75%) and 70 mg in five patients (31.25%). Eptinezumab was used exclusively at a dose of 400 mg. A summary of the demographic and baseline characteristics of all the included studies is presented in Tables 1–3.
*Data are presented closest to the fourth week after the start of treatment.
**We were unable to calculate the precise p-value due to the lack of published categorical data. Our assessments were conducted indirectly, based on the data presented in the publication and on the NCT page.
***The study was terminated early due to interim futility analyses suggesting that achieving the primary endpoint was unlikely.
****12-week double-blind, placebo-controlled treatment period and a 52-week open-label period.
*****The study was terminated early due to interim futility analyses suggesting that achieving the primary endpoint was unlikely.
******Change from baseline in the overall monthly average number of CH attacks; mean reduction in weekly cluster headache attack frequency was not reported.
CH: cluster headache; eCH: episodic cluster headache; cCH: chronic cluster headache; n: number; ns: not significant; s.c.: subcutaneous; SD: standard devation.
*Only a portion of the patient cohort, presumably 33 individuals, was enrolled in the prospective registries.
**Of a total of 16 patients, six were included in the prospective registry and 10 in the retrospective registry. Of these, fourteen patients (87.5%) received at least one dose of 240 mg galcanezumab.
***In the absence of an observedresponse following the initial 240 mg dose, the dosage was increased to 360 mg for the second and third administrations.
CH: cluster headache; eCH: episodic cluster headache; cCH: chronic cluster headache; n: number; nd: no data; s.c.: subcutaneous; SD: standard devation.
*Was reduced to 120 mg in subsequent months in two patients.
**Was increased to 140 mg in subsequent months in all patients and changed to galcanezumab 240 mg in the third month in one patient.
eCH: episodic cluster headache; cCH: chronic cluster headache; n: number; nd: no data; s.c.: subcutaneous; SD: standard devation
Outcome
The meta-analysis of randomized, placebo-controlled trials showed that anti-CGRP treatment had a statistically significant effect in eCH, reducing the weekly frequency of cluster headache attacks by at least 50% from baseline at week 4 (odds ratio (OR) = 1.65, 95% confidence interval (CI) = 1.07–2.55, p = 0.02) with galcanezumab 300 mg (95% CI = 1.00–5.05, p = 0.05) and eptinezumab 400 mg (95% CI = 1.12–3.43, p = 0.02) showing greater efficacy than placebo. No significant effect was observed in cCH (OR = 1.07, 95% CI = 0.78–1.48, p = 0.68).
Only galcanezumab 300 mg showed a significant mean reduction in weekly attack frequency in eCH at week 4 (p = 0.04); no CGRP antagonist was superior to placebo in cCH. Meta-analysis for this parameter could not be performed due to the lack of published categorical data; p-values were therefore assessed indirectly based on the data reported in publications and the corresponding NCT websites.
Heterogeneity was low to moderate (i-squared 24%, tau-squared 0.04). However, given the low number of included studies, the estimates have to be interpreted with caution, as heterogeneity measure may be imprecise when few studies are included.
A detailed analysis is provided in Tables 1–3 and Figure 2.

As shown in Figures 3(a),(b), the proportion of patients achieving a ≥ 50% responder rate varies widely by study design, with the highest response rates observed in retrospective case series and case reports (100% for eCH and 65% for cCH), and the lowest in double-blind, placebo-controlled trials (67% for eCH and 37% for cCH). In non-randomized prospective trials, the mean proportion of ≥ 50% responders was 73% for eCH and 46% for cCH.
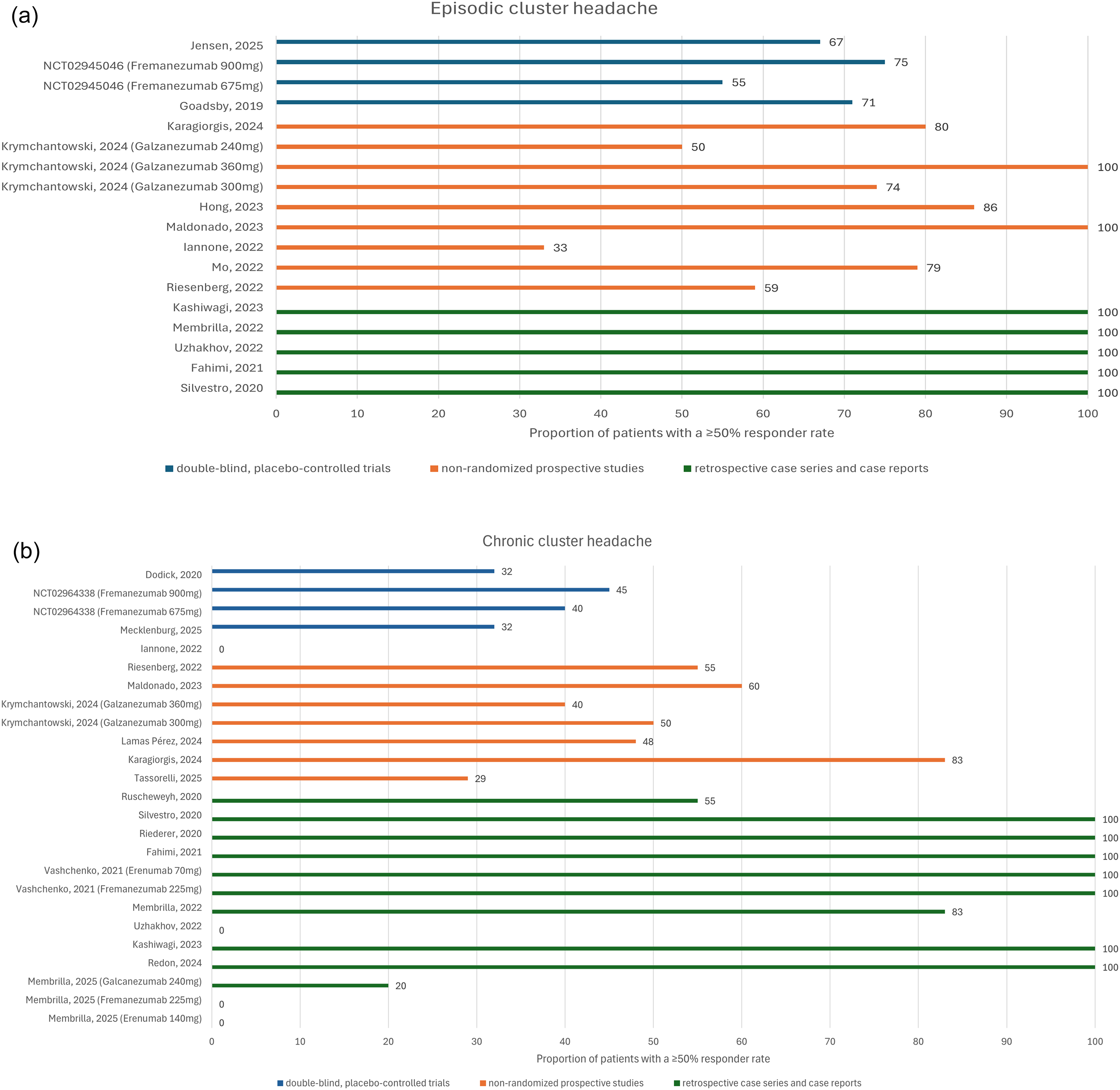
(a) Proportion of episodic cluster headache (eCH) patients with a ≥ 50% responder rate near the fourth week after treatment initiation, based on data from double-blind, placebo-controlled trials; non-randomized prospective studies; and retrospective case series and case reports.5,6,15–38 (b) Proportion of cCH patients with a ≥ 50% responder rate near the fourth week after treatment initiation, based on data from double-blind, placebo-controlled trials; non-randomized prospective studies; and retrospective case series and case reports.5,6,15–38
Adverse effects
During the treatment of patients from double-blind, placebo-controlled trials, 11 serious adverse events (SAEs) and 224 treatment-emergent adverse events (TEAEs) were reported. The most common adverse events included injection site reactions (n = 127) and upper respiratory tract infections (n = 55). No deaths related to the treatment were reported.
The highest percentage of adverse effects was observed with galcanezumab 300 mg (1.2% of patients with SAE and 64% with TEAE) and erenumab 400 mg (4.9% of patients with SAE and 60.98% with TEAE). A higher dose of medication correlated with an increased incidence of adverse effects, as seen with fremanezumab 900 mg compared to 675 mg (SAE: 2.11% vs. 1.4%; TEAE: 24.65% vs. 21.68%). The detailed data are presented in Table 4.
n: number; nd: no data; SAE: serious adverse event; TEAE: treatment emergent adverse events.
Risk of bias assessment
All randomized studies had a low risk of bias, while the majority of the observational studies had at least moderate risk. Due to the high risk of bias inherent in the methodology and design of retrospective case series and reports, this assessment was restricted to prospective studies only. The results of the risk of bias assessment for prospective trials are shown in Figures 4(a),(b).

(a) Risk of bias assessment for randomized studies. (b) Risk of bias assessment for prospective non-randomized studies.
Since the publications by Láinez et al. 39 and Dodick et al. 5 referred to the same NCT number, we included the study by Dodick et al. 5 in our analysis because it was the first to be published. One study was considered at serious risk of bias, due to the lack of information about deviations from the intended intervention. 15
Discussion
In this systematic review and meta-analysis, we assessed the efficacy and safety of all four mAbs directed against CGRP or its receptor (i.e. galcanezumab, fremanezumab, eptinezumab and erenumab) in both eCH and cCH. It is important to note that there are no head-to-head trials in the literature evaluating anti-CGRP treatment in this context and all comparisons between treatments are indirect.
The results of our meta-analysis of six placebo-controlled trials suggest that anti-CGRP treatment has a statistically significant positive effect in achieving a ≥ 50% responder rate at week 4 in eCH (OR = 1.65, 95% CI = 1.07–2.55, p = 0.02), with galcanezumab 300 mg and eptinezumab 400 mg being more effective than placebo. A significant reduction in mean weekly attack frequency at week 4 (p = 0.04) was observed only in the trial with galcanezumab. No statistically significant effect was observed in cCH (OR = 1.07, 95% CI = 0.78–1.48, p = 0.68). This pattern was further supported by findings from non-randomized prospective studies, in which the mean proportion of patients achieving a ≥ 50% response was 73% for eCH and 46% for cCH.
However, these positive results for eCH must be interpreted with caution. Although galcanezumab demonstrated efficacy in eCH during the initial 3-week observation period, this was not statistically significant at any subsequent follow-up until the end of the observation period at week 8.6,20 Similarly, although a significantly higher proportion of participants receiving eptinezumab achieved a ≥ 50% response compared to those receiving a placebo at weeks 2, 3 and 4, the primary endpoint of the trial, comprising a change in the number of weekly attacks from baseline during weeks 1 to 2, was not met.(20) The mean reduction at week 4 was also not significant. 20 In contrast, at week 4, EQ-5D-5L VAS scores assessing patients’ subjective well-being showed a significant mean improvement of 7.8 points with eptinezumab compared to placebo (95% CI = 1.49–14.03, p = 0.02), further complicating the interpretation of the outcome data. 20 These discrepancies raised concerns among the authors, leading to post hoc sensitivity analyses using PRESS (i.e. predicted residual error sum of squares) residuals. These analyses identified a small number of participants with a high influence on the results. After removing these cases (five eptinezumab and one placebo), the change-from-baseline analysis favored eptinezumab across all weeks, while 50% responder rates remained largely unchanged, supporting the robustness of the treatment effect. 20 This suggests that patient pre-selection might be a viable strategy, although the optimal method for its implementation remains undefined.
No monoclonal antibody has demonstrated superiority over placebo in cCH. An open-label safety trial of eptinezumab (CHRONICLE) suggests a clinical efficacy over 12 months, with 52% of participants achieving a ≥ 50% reduction in cCH frequency from baseline. 28 However, these findings should be interpreted with caution because the study lacked a placebo control group and a formal statistical analysis. In addition, the absolute number of patients reaching this threshold after 12 months did not increase substantially compared to week 4 but the total number of participants decreased (36/125 (29%) vs. 44/84 (52%)). This result suggests a selective dropout of non-responders, and thus an attrition bias. Lastly, the responder rate at three months was similar to the placebo responses reported in the controlled cCH studies.5,18,19
As shown in Figures 3(a),(b), our study revealed a striking discrepancy between uncontrolled, retrospective real-world evidence and controlled trials. The results reported in case reports and series overwhelmingly describe positive outcomes, even for cCH. This represents a type of “reverse efficacy-effectiveness gap,” which describes a rare scenario where a drug shows greater effectiveness in real-world practice than in clinical trials, contrary to the typical pattern where trial efficacy exceeds real-world effectiveness.7,8
As indicated by several real-world studies, anti-CGRP mAbs generally appear to have an effectiveness that may surpass their clinical efficacy. 9 However, the most likely explanation for this discrepancy in our analysis is publication bias, as studies with positive treatment outcomes are more likely to be published, while negative or inconclusive results often remain unpublished. An additional layer of concern can arise from the endpoint selection inherent to retrospective real-world analyses. Unlike randomized controlled trials, which strictly predefine primary and secondary endpoints alongside a detailed statistical analysis plan to limit bias, retrospective studies often exercise great latitude in choosing their outcome measures after the data collection. Moreover, many non-randomized studies lack statistical modeling and rely primarily on descriptive reporting. Consequently, these results must be interpreted with caution and, at best, serve as hypothesis-generating data that need to be confirmed in rigorously designed prospective trials.
All randomized studies set their primary endpoints as the mean change in weekly or monthly attack frequency; however, the time point for assessment varied between 2 and 12 weeks following the initiation of treatment. In contrast, non-randomized trials more frequently focused on the 50% responder rate, which was also assessed across a broad range of time points. The selection of endpoints and the length of the observation period represent a significant challenge in interpreting the efficacy of anti-CGRP therapies in CH. If the evaluation window is too short, clinically relevant treatment effects may be underestimated because data from migraine trials suggest that CGRP-targeted therapies may require time to exert their full biological impact. 40 Conversely, longer observation periods introduce the risk that spontaneous changes in attack frequency, as driven by the natural history of CH, including fluctuation and remission, may obscure true treatment effects.41–43 This is particularly relevant in eCH, where spontaneous improvement may occur in both active and control groups, thereby reducing the ability to discriminate drug-specific effects. To our knowledge, no study has analysed how frequently patients with cCH experience a spontaneous 50% decrease in their attack frequency despite unchanged prophylactic medication. However, there is evidence indicating that the attack frequency can change drastically over time. 44 Consequently, currently used endpoints may reflect a composite of treatment-related and disease-intrinsic changes. To date, an optimal methodological approach to fully account for spontaneous disease dynamics while capturing delayed biological effects has not yet been established, underscoring the need for further refinement of endpoint selection in future CH trials.
Another noteworthy observation is the substantial placebo effect observed in randomized trials. However, this is not an isolated phenomenon in CH trials, as placebo effects of a similar magnitude are also observed in migraine trials. 45 Beyond the possibility of a genuine biological mechanism underlying the placebo response, 45 an important consideration in CH patients is that they are often enrolled during periods of peak disease activity, with spontaneous remissions and fluctuating attack frequencies influencing trial outcomes.
The differential response to anti-CGRP mAbs observed between eCH and cCH may reflect more than differences in endpoint selection or study design. Increasing evidence suggests that the relative contribution of peripheral nociceptive mechanisms differs between these subforms. In eCH, peripheral trigeminal sensitization appears to play a significant role, supported by findings such as reduced nociceptive flexion reflex thresholds that normalize outside active bouts. 46 In contrast, these changes are absent in cCH, indicating a reduced role of peripheral nociceptor sensitization. Consistent with this view, surgical interruption of trigeminal afferent input has failed to reliably reduce attack frequency in cCH, suggesting that peripheral input may no longer be the primary driver of attacks in this disease stage.47,48 Since anti-CGRP mAbs primarily act outside the central nervous system, treatments targeting peripheral mechanisms may therefore be more effective in eCH than in the chronic form. These considerations support the notion that eCH and cCH may differ in relevant pathophysiological mechanisms, with important implications for both treatment responsiveness and endpoint selection in clinical trials.
Sensitivity to CGRP also varies between CH subtypes.40,49 CGRP infusions are less likely to trigger cluster headache attacks in patients with cCH than in those with eCH. In addition, plasma CGRP in CH patients rises during active periods, whereas interictal plasma CGRP levels are reported to be normal or even lower than in healthy controls.49,50 This contrasts with migraine, in which CGRP levels remain persistently elevated and play a central role in pathophysiology, a pattern that is also reflected in the stronger therapeutic response to anti-CGRP mAbs observed in migraine compared with CH. 51
Another factor complicating the interpretation of results in CH studies is that, for ethical reasons, other preventive therapies in parallel with CGRP treatment are generally accepted in trials of cCH but not in trials of eCH, which may also influence the outcome by either masking the effect of the CGRP-targeting treatment, or complicating the interpretation of efficacy.
Importantly, our findings do not exclude that CGRP antagonists at different doses may have an effect on CH. In the galcanezumab trial, the researchers used the highest previously tested dose, although higher or alternative dosing strategies may offer additional benefits. 52
Lastly, we must consider that the drugs may not yet have reached their maximum effect at the end of the study period. 53 As a result, any differences in efficacy between treatment groups may not have been fully captured within the timeframe of our analysis. Thus, our current methodology may underestimate the true efficacy of the intervention, but, as seen in the current trials, the efficacy of anti-CGRP treatment in CH declined rather over the longer follow-up period.5,6
Limitations
Several key limitations must be acknowledged. Most notably, the number of included trials is small and the study designs are highly heterogeneous in terms of sample size, follow-up duration and inclusion criteria. There are no head-to-head trials in the literature evaluating anti-CGRP treatment in this context and all comparisons between treatments are indirect. The early termination of two clinical trials (NCT02945046 and NCT02964338) further limits the proper interpretation of the available data.
Second, due to the heterogeneity of the available data, we chose the proportion of participants achieving a ≥ 50% reduction in attack frequency from baseline at week 4 as a single objective measure of treatment efficacy. While this approach provides consistency across studies, it may oversimplify the complex clinical picture by excluding other important outcomes, such as the use of acute treatments and patients’ subjective well-being. Additional time points would also be of interest, but we selected this endpoint as it was the most commonly reported across trials.
Third, many agents have only been evaluated in a single trial that was prematurely terminated due to a lack of preliminary efficacy signal, others, such as galcanezumab, are disproportionately represented in the literature.
Conclusions
Galcanezumab 300 mg and eptinezumab 400 mg were superior to placebo in achieving a ≥ 50% reduction in attack frequency in eCH at the 4-week mark following administration. However, the mean reduction in weekly attack frequency in eCH at week 4 was statistically significant only for galcanezumab 300 mg (p = 0.04). Despite some positive findings from non-randomized prospective studies, no placebo-controlled trial has demonstrated significant efficacy of any CGRP antagonist in the treatment of cCH.
The positive findings in eCH should be interpreted with caution because the discrepancies in the outcome data underscore significant methodological challenges in CH trials. A major limitation of the primary outcome in CH trials is its susceptibility to fluctuations caused by baseline and post-baseline variability in attack frequency, which is an inherent characteristic of CH. Furthermore, the substantial discrepancy between real-world data and controlled studies suggests that only the latter may provide consistently reliable results. This underscores the need for ongoing discussions among experts to reach a consensus and develop more appropriate methodologies for conducting CH trials.
Our findings do not exclude the potential efficacy of the tested drugs at alternative dosages. Adverse events were generally mild, with the most common being injection site reactions and gastrointestinal symptoms.
Article highlights
This systematic review and meta-analysis suggests that anti-CGRP monoclonal antibody treatment may have a beneficial effect in eCH, but not in cCH.
Positive findings should be interpreted with caution due to inconsistent outcomes, challenges in endpoint selection and discrepancies between real-world data and controlled trials, underscoring the need for expert consensus and improved trial methodologies in CH.
Footnotes
Author contributions
Project conceptualization and design: LK, MTK, SW and HP. Data collection: LK, MTK and HP. Systematic review and meta-analysis: Record screening and data extraction were performed by LK and MTK, with conflicts resolved by HP. Risk of bias assessment: LK and MTK, with conflicts resolved by HP. Data analysis and statistical testing: LK. Tables and figures: LK. Manuscript writing: LK, with final contributions from MTK, SW and HP. All authors reviewed and approved the final version of the manuscript submitted for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LK und MTK received travel grant and conference participation support from TEVA and AbbVie; SW received research funds from the Swiss National Science Foundation, the UZH Clinical research priority program (CRPP) stroke, the Zurich Neuroscience Center (ZNZ), the Baugarten foundation, the Hartmann Müller Foundation, the Koetser Foundation, the Philas Foundation and the Swiss Heart Foundation, as well as speaker honoraria from Amgen, Springer, Advisis AG, Teva Pharma, Boehringer Ingelheim, Lundbeck, Astra Zeneca and FoMF, and a consultancy fee from Bayer and Novartis via institution for research; HP received honoraria and/or speaking fees from AbbVie, Eli Lilly, Novartis and Teva Pharmaceuticals, and funding from the Werner Dessauer Stiftung. There are no conflicts of interest in regard to this manuscript.
Data availability statement
All data generated or analysed during this study are included within the published article. These data are unavailable elsewhere.