Abstract
Aim
To systematically evaluate the safety and tolerability of calcitonin-gene-related peptide binding monoclonal antibodies from the results of randomized controlled trials.
Methods
Online databases were searched on calcitonin-gene-related peptide binding monoclonal antibodies for the prevention of episodic migraine. Overall withdrawal, withdrawal due to adverse events, adverse events, serious adverse events and specific adverse events were extracted from the included studies. A meta-analysis was performed with Revman 5.3.0 software.
Results
Ten studies that investigated four drugs (galcanezumab, erenumab, fremanezumab and eptinezumab) with 5817 participants were included in this study. Serious adverse events, overall withdrawals, withdrawal due to adverse events and any adverse events were not significantly associated with monoclonal antibody treatment. Injection site pain and erythema were significantly higher in the calcitonin-gene-related peptide binding monoclonal antibodies treatment group than in the placebo group. The rates of serious adverse events were significantly higher in the galcanezumab 120 mg group. Injection site erythema was associated with galcanezumab 120 mg and 240 mg. Injection site pain and nasopharyngitis were associated with galcanezumab 150 mg and 5 mg, respectively. Overall adverse events were significantly higher with erenumab 70 mg and 140 mg. Treatment-related adverse events were significantly higher with fremanezumab 225 mg/month and 675 mg/quarter.
Conclusions
This study provides data on the safety and tolerability profiles of calcitonin-gene-related peptide binding monoclonal antibodies and confirms their potential use as preventive treatments for episodic migraine. In addition to the acceptable withdrawal rates, serious adverse events were rare, and the severity of most adverse events was mild to moderate. Injection site reaction may be the major adverse event associated with galcanezumab.
Introduction
Approximately 15% of the population worldwide suffers from migraine (1). According to the 3rd edition of the International Classification of Headache Disorders (ICHD-III), migraine can be classified as episodic migraine (EM), defined as migraine less than 15 migraine days or headache days per month with or without aura, and chronic migraine (CM) with more than 14 headache days per month (of which ≥8 are migraine days with or without aura) for more than 3 months (2). Previous therapies have not met the challenge of alleviating the headache pain and economic burden induced by migraine (3,4). In addition, the tolerability and patient adherence to the current preventive treatments are limited (5). Therefore, there is an urgent need for novel, well-tolerated and effective treatments for migraine.
Calcitonin-gene-related peptide (CGRP) has been shown to be a promising target for treating migraine in previous studies (6,7). Initially, monoclonal antibodies (mAbs) against CGRP receptors were the first confirmed to be effective for the acute treatment of migraine (8). Recently, several phase 2 or 2b and phase 3 clinical trials focusing on preventive therapy for both EM and CM have demonstrated that mAbs against both the CGRP ligand [galcanezumab (LY29517542), eptinezumab (ALD403), fremanezumab (TEV-49125)] and the receptor [erenumab (AMG 334)] were effective in reducing mean monthly migraine days and migraine frequency (9–18). While previous reviews reported the efficacy and safety of a specific CGRP mAb, only several phase 2 or 2b studies were included (19–21). Comprehensive safety profiles of CGRP mAbs were absent.
In this study, we performed a meta-analysis to evaluate the safety and tolerability of CGRP binding mAbs for the preventive therapy of episodic migraine.
Methods
All meta-analyses were performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (22).
Data sources and search
Online databases including PubMed, Embase (via Ovid interface), Cochrane Online Library and Clinicaltrials.gov were searched with the language restricted to English. The last search date was 1 June 2018. The search terms included the following: Episodic migraine, galcanezumab, LY29517542, eptinezumab, ALD403, fremanezumab, TEV-49125, erenumab and AMG 334.
Selection of studies
Two reviewers independently reviewed the titles and abstracts from the search results, and eligible studies were selected according to predefined inclusion and exclusion criteria. Any disagreement was resolved by consensus between reviewers.
Inclusion criteria
Study design: Randomized, double-blind, placebo-controlled trials. Participants: Adults aged 16–65 years with a diagnosis of episodic migraine according to ICHD-III for at least one year prior to enrollment (2). Interventions: Galcanezumab, eptinezumab, fremanezumab and erenumab at different doses and placebo as a control group. Outcomes: All data came from full journal publications or results from clinicaltrials.gov; at least one of the following was provided: Serious adverse events (SAEs), withdrawal, withdrawal due to adverse events (AEs) and treatment-emergent adverse events (TEAEs).
Exclusion criteria
Study design: Open label studies; post hoc analysis and interim analyses were excluded. Participants: Participants were excluded if they received more than one kind of CGRP binding mAb. Trials of healthy participants were also excluded. The use of acute migraine medications was allowed. Outcomes: The study was excluded if over 15% of the included participants withdrew.
Data extraction and evaluation of evidence
Two reviewers independently extracted relevant information from eligible studies. The extracted information included the Clinicaltrials.gov registration number, study design, inclusion criteria, type of mAbs, doses, route of injection, number of participants (intention to treat, ITT), number of males and females, age, the duration of the double-blind period, type of migraine, SAEs, TEAEs, withdrawal rates, withdrawals due to AEs, number of participants who developed antidrug antibodies (ADA), treatment-emergent antidrug antibodies (TEADAs) and any AEs. The evaluation of the evidence was performed according to the guideline for assessing the risk of bias in the Cochrane Handbook 5.1.0 (23). Any divergence was resolved by consensus between investigators.
Outcome measures
SAEs, TEAEs, and withdrawals due to AEs were investigated to measure the safety and tolerability of CGRP binding mAbs. SAEs were defined as life-threatening AEs that could result in death, a persistent or significant disability, or a congenital birth defect or hospitalization.
Data analysis
All meta-analyses were performed with Review Manager 5.3 software. For dichotomous variables, risk ratios (RRs) and 95% confidence intervals (95% CIs) were selected for analysis using the Mantel-Haenszel method (24). Statistical heterogeneity was assessed with I2 (25). For I2 > 50%, the heterogeneity was regarded as unacceptable and was analyzed with a random effects model or subgroup analysis. A fixed effects model was applied for I2 < 50%. A sensitivity test was performed by switching statistical values. For example, RR was converted to an odds ratio (OR) or a fixed effects size model was converted to a random effects size model. p < 0.05 was regarded as the level of statistical significance.
Results
Study selection
The search of online databases yielded 422 studies including 46 in PubMed, 171 in Embase, 167 in Cochrane Library and 38 in clinicaltrials.gov. Six trials, in which the focus was either on CM or healthy volunteers, were excluded (26–31). A total of 10 studies were finally included in our meta-analysis. The details of the screening procedure are shown in Figure 1.
Flowchart of study selection.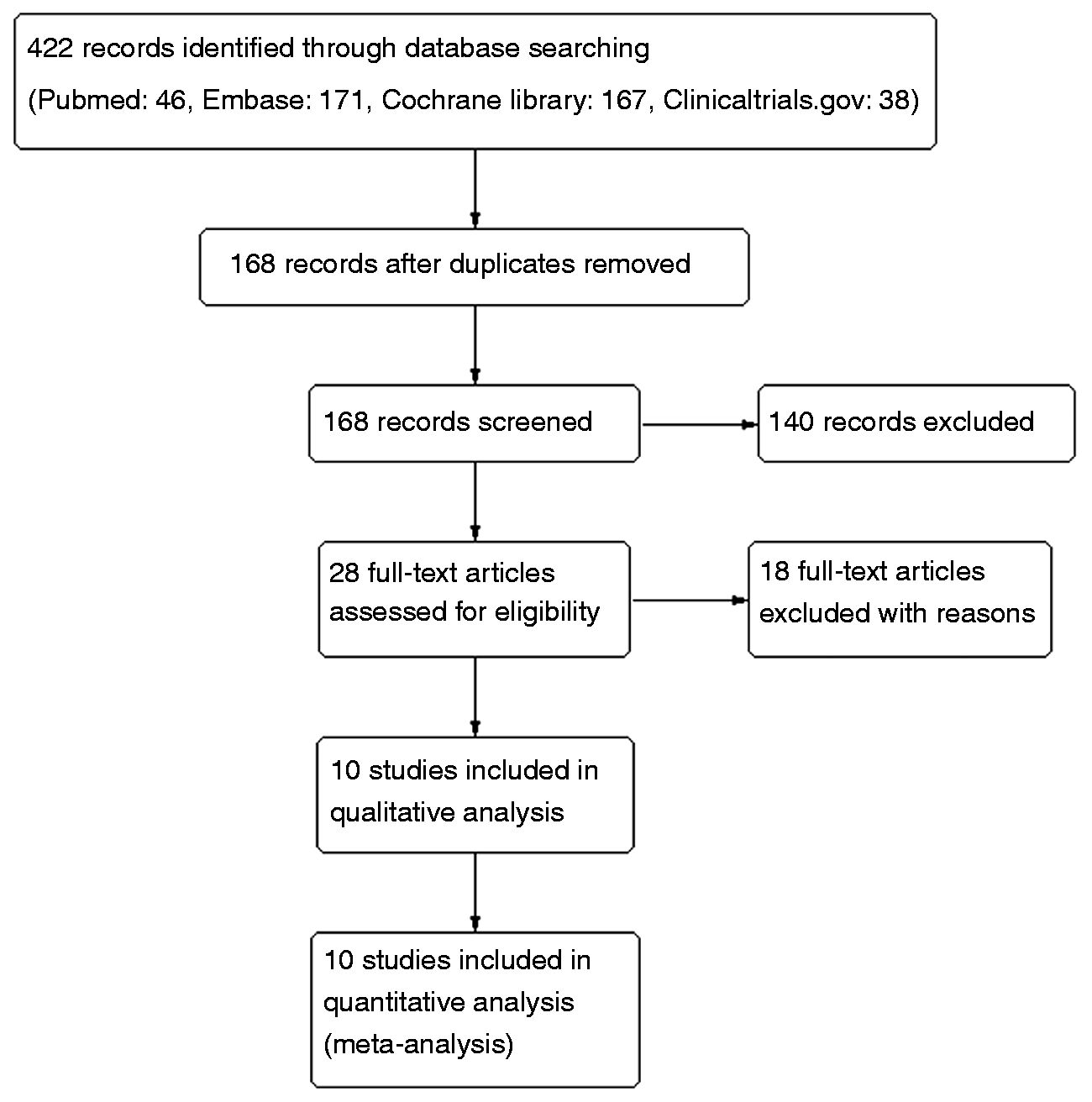
Characteristics of included studies
Baseline characteristics of the included studies.
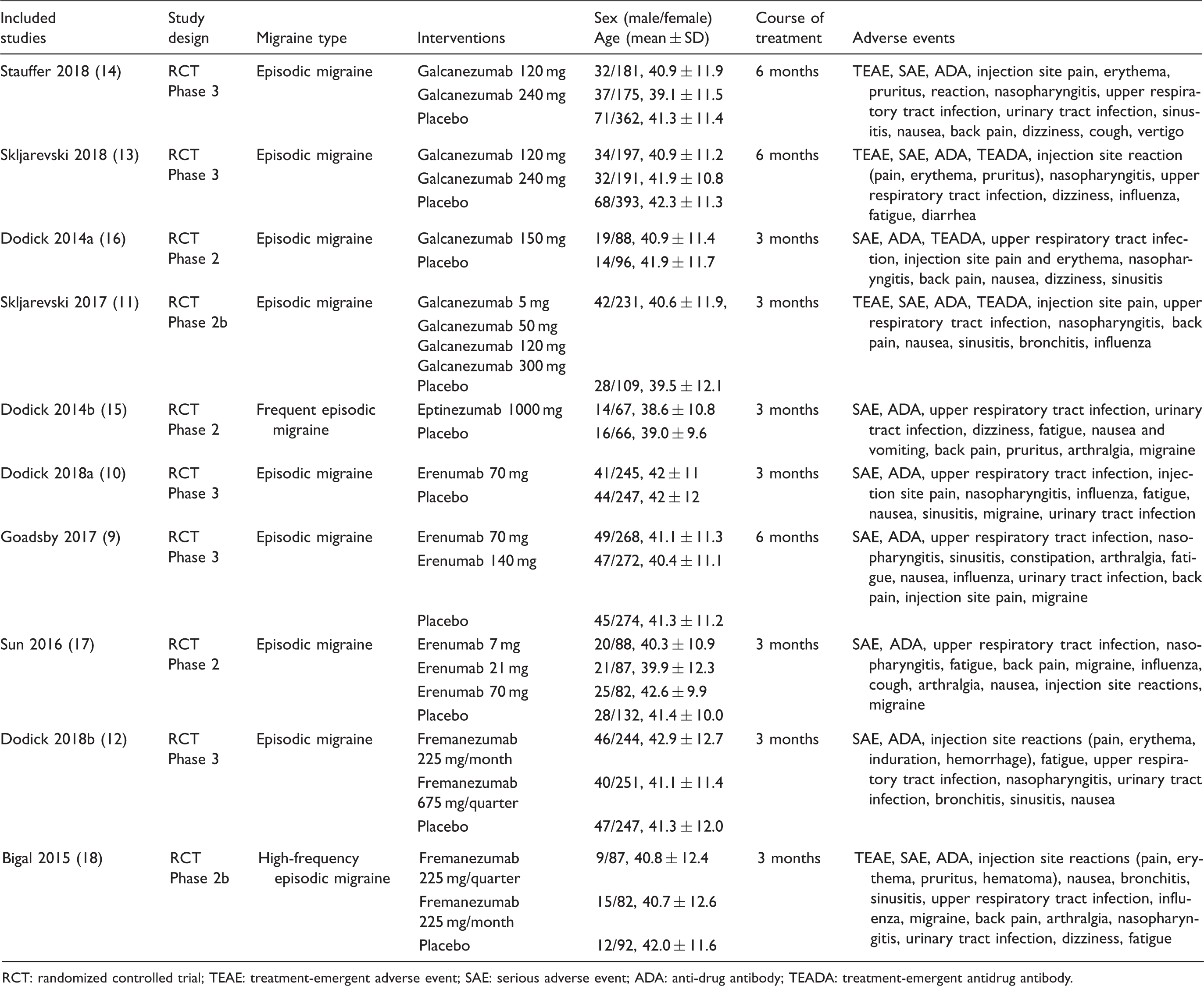
RCT: randomized controlled trial; TEAE: treatment-emergent adverse event; SAE: serious adverse event; ADA: anti-drug antibody; TEADA: treatment-emergent antidrug antibody.
Risk of bias of the included studies
In accordance with the Cochrane Handbook of Systematic Review, random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data and selective reporting were fully considered when assessing bias (23,24). All included studies were considered low risk and high quality. The detailed information is shown in Figure 2.
Risk of bias for the included studies.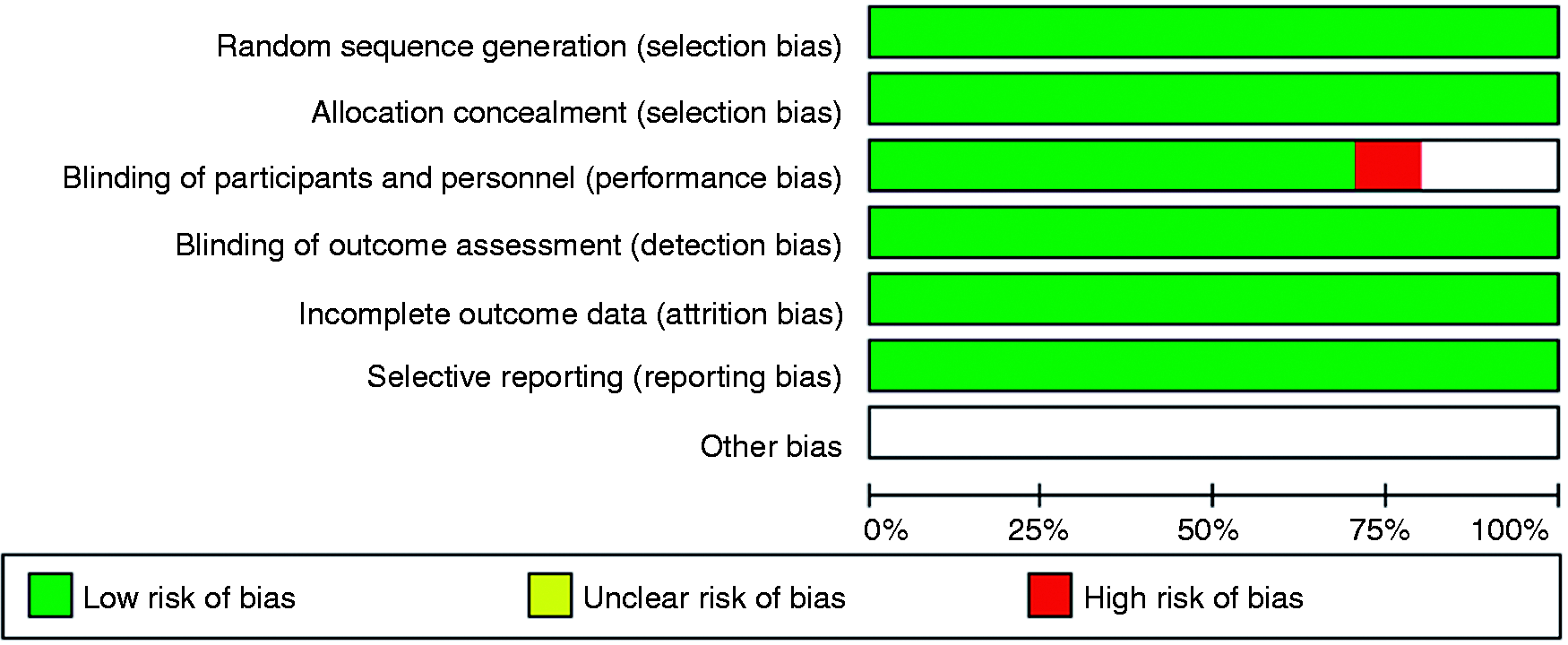
Overall withdrawal
All included studies reported overall withdrawals. There was no significant difference in overall withdrawals between the CGRP binding mAbs and placebo groups (RR 0.90, 95% CI: 0.77–1.05, p = 0.17, I2 = 0%, Figure 3).
Comparison results of overall withdrawal between CGRP binding mAbs and placebo.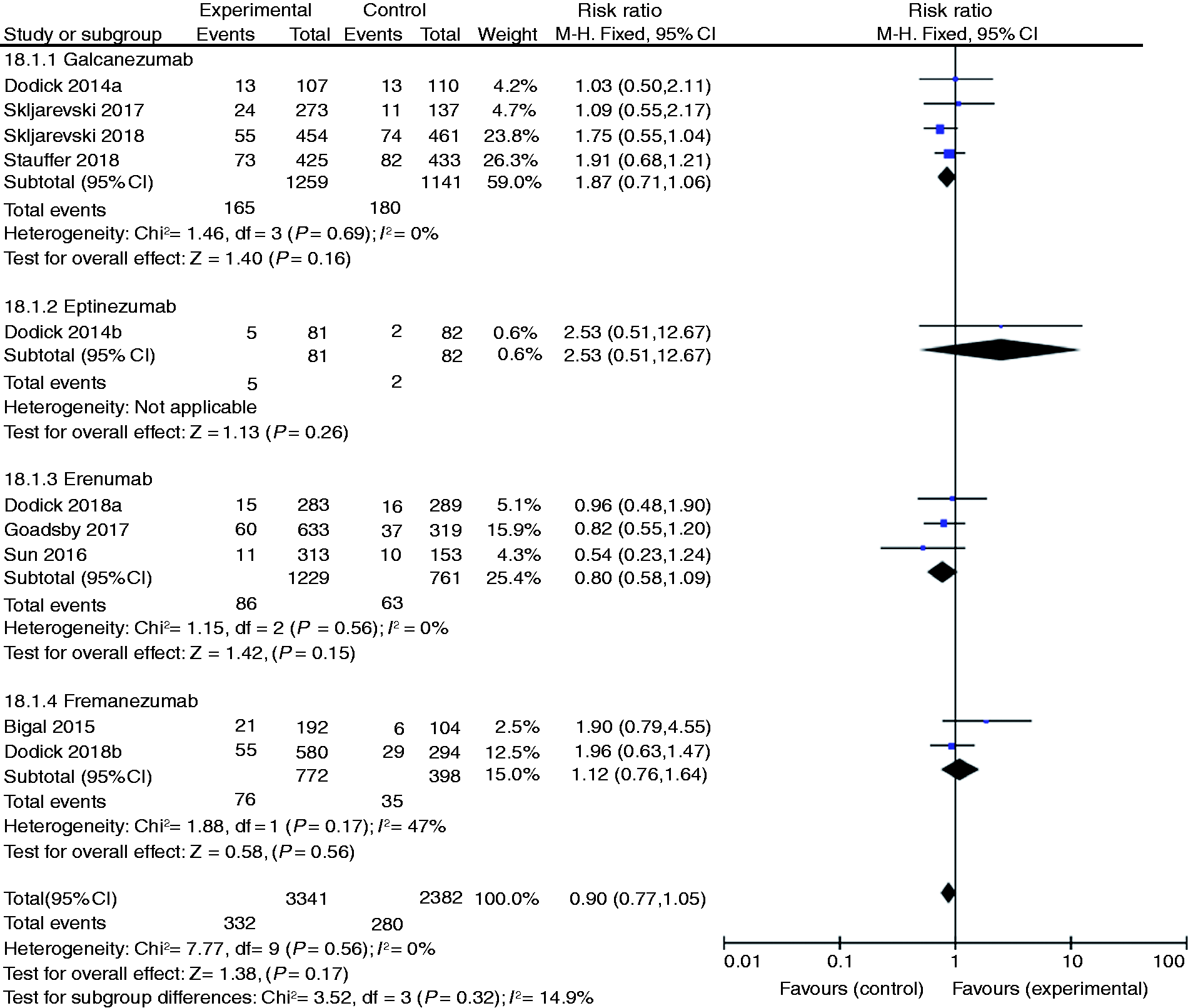
Withdrawal due to AEs
All included studies reported the withdrawal rates due to AEs. No differences were observed between CGRP binding mAbs and placebo groups (RR 1.39, 95% CI: 0.95–2.04, p = 0.09, I2 = 0%, Figure 4].
Comparison results of withdrawal due to AEs between CGRP binding mAbs and placebo.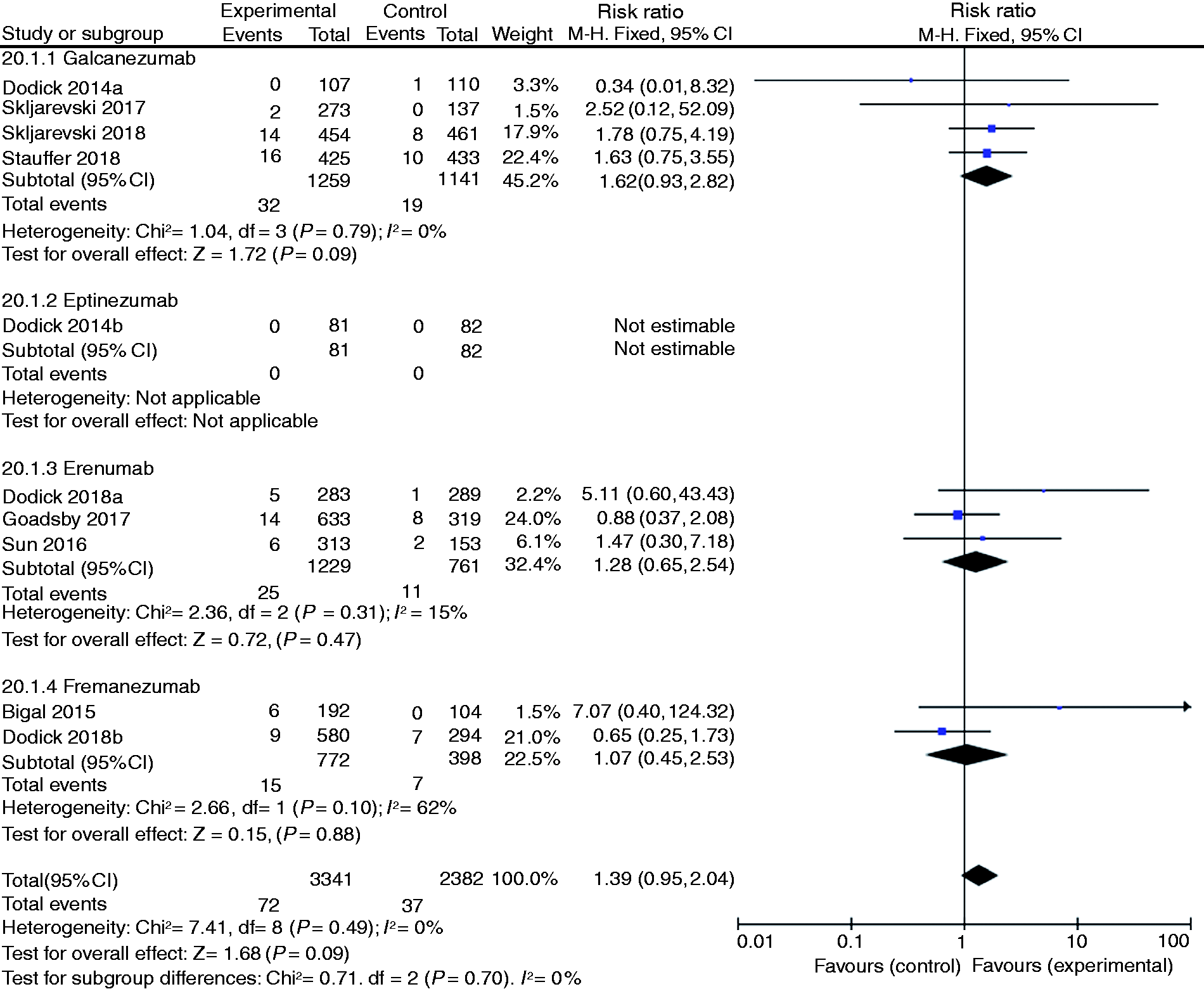
SAEs
SAEs were reported in all included studies. The meta-analysis found no significant difference between the two interventions (RR 1.13, 95% CI: 0.74–1.72, p = 0.56, I2 = 0%, Figure 5].
Comparison results of SAEs between CGRP binding mAbs and placebo.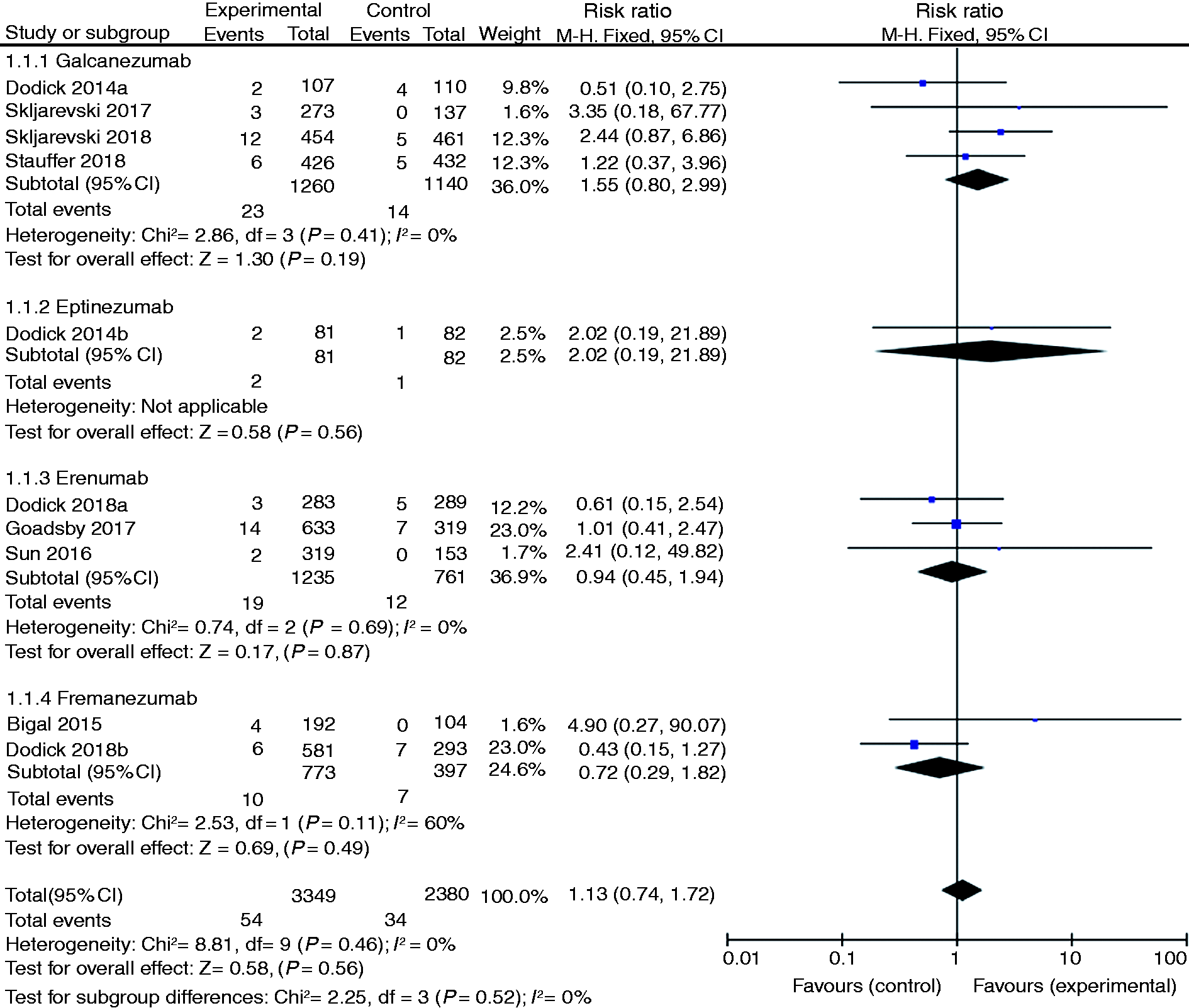
Any AE
Comparison results between CGRP binding mAbs placebo in relation to specific AEs.
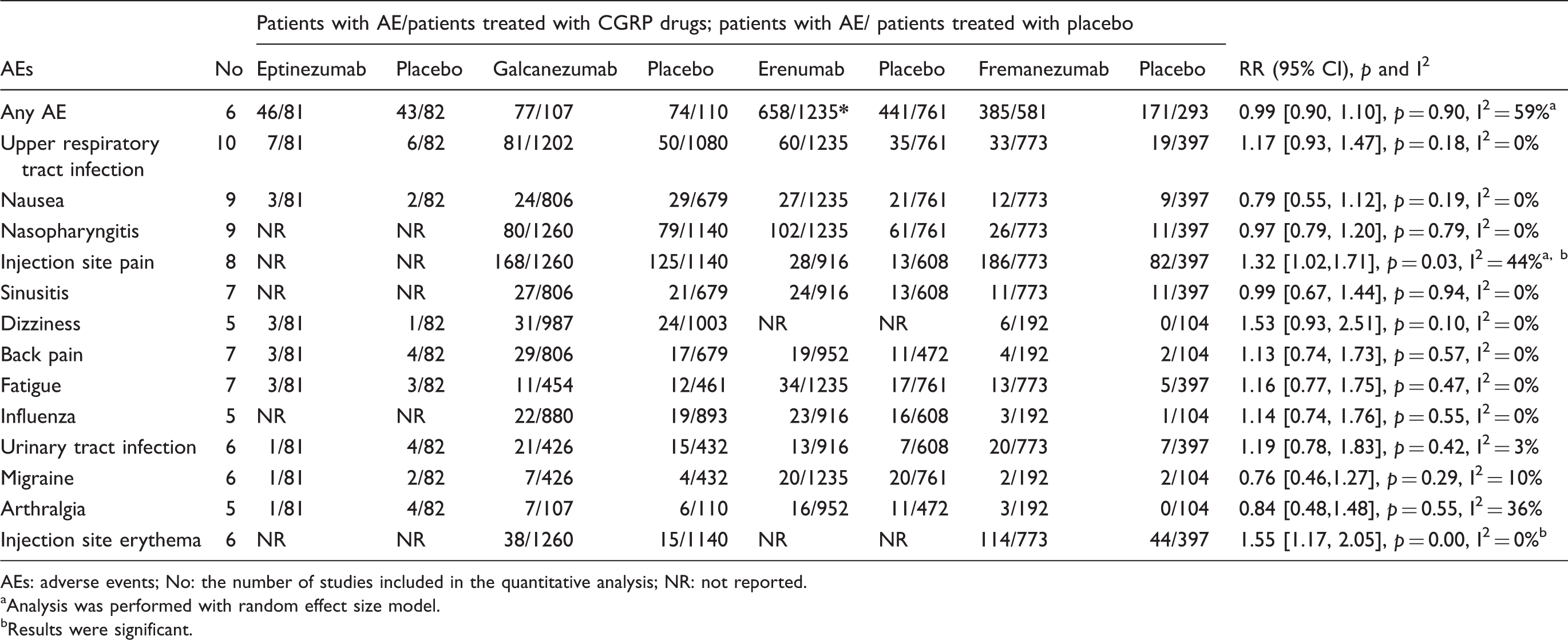
AEs: adverse events; No: the number of studies included in the quantitative analysis; NR: not reported.
Analysis was performed with random effect size model.
Results were significant.
Specific AEs reported in included studies
A total of 13 AEs, including upper respiratory tract infection, nausea, nasopharyngitis, injection site pain and erythema, sinusitis, dizziness, back pain, fatigue, influenza, urinary tract infection, migraine, and arthralgia, were reported in ≥ 2% of the participants. The results showed that the rates of injection site pain and injection site erythema were significantly higher in the CGRP binding mAbs group as compared to the placebo group (RR 1.32, 95% CI: 1.02–1.71, p = 0.03, I2 = 44%, random effect model; RR 1.55, 95% CI: 1.17–2.05, p = 0.00, I2 = 0%, respectively]. No significant difference was detected between the two groups in terms of other AEs. The detailed results are shown in Table 2.
Results of different doses of galcanezumab compared to placebo
Comparison results of different doses of galcanezumab.
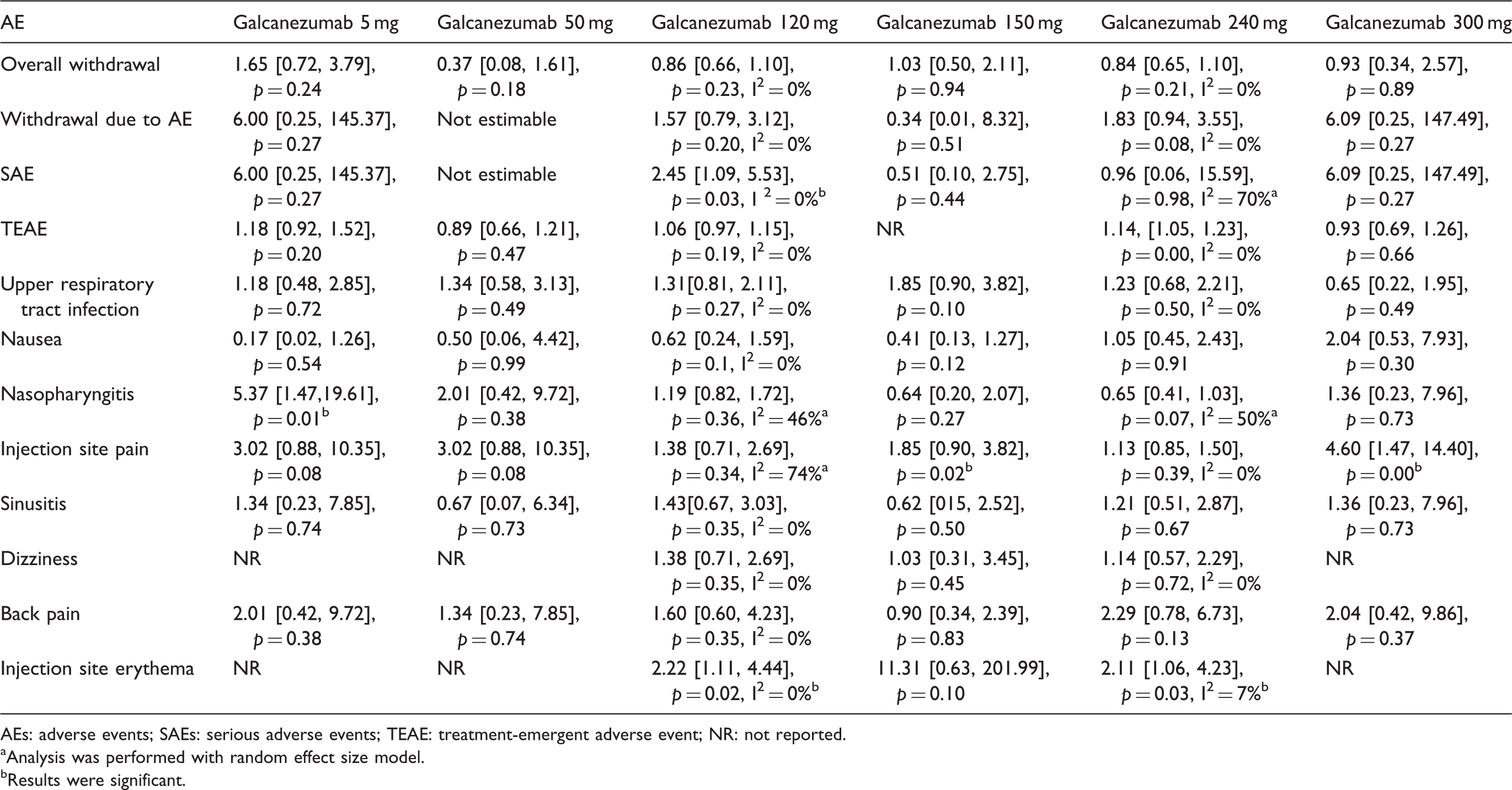
AEs: adverse events; SAEs: serious adverse events; TEAE: treatment-emergent adverse event; NR: not reported.
Analysis was performed with random effect size model.
Results were significant.
Results of different doses of erenumab compared to placebo
Comparison results of different doses of erenumab.
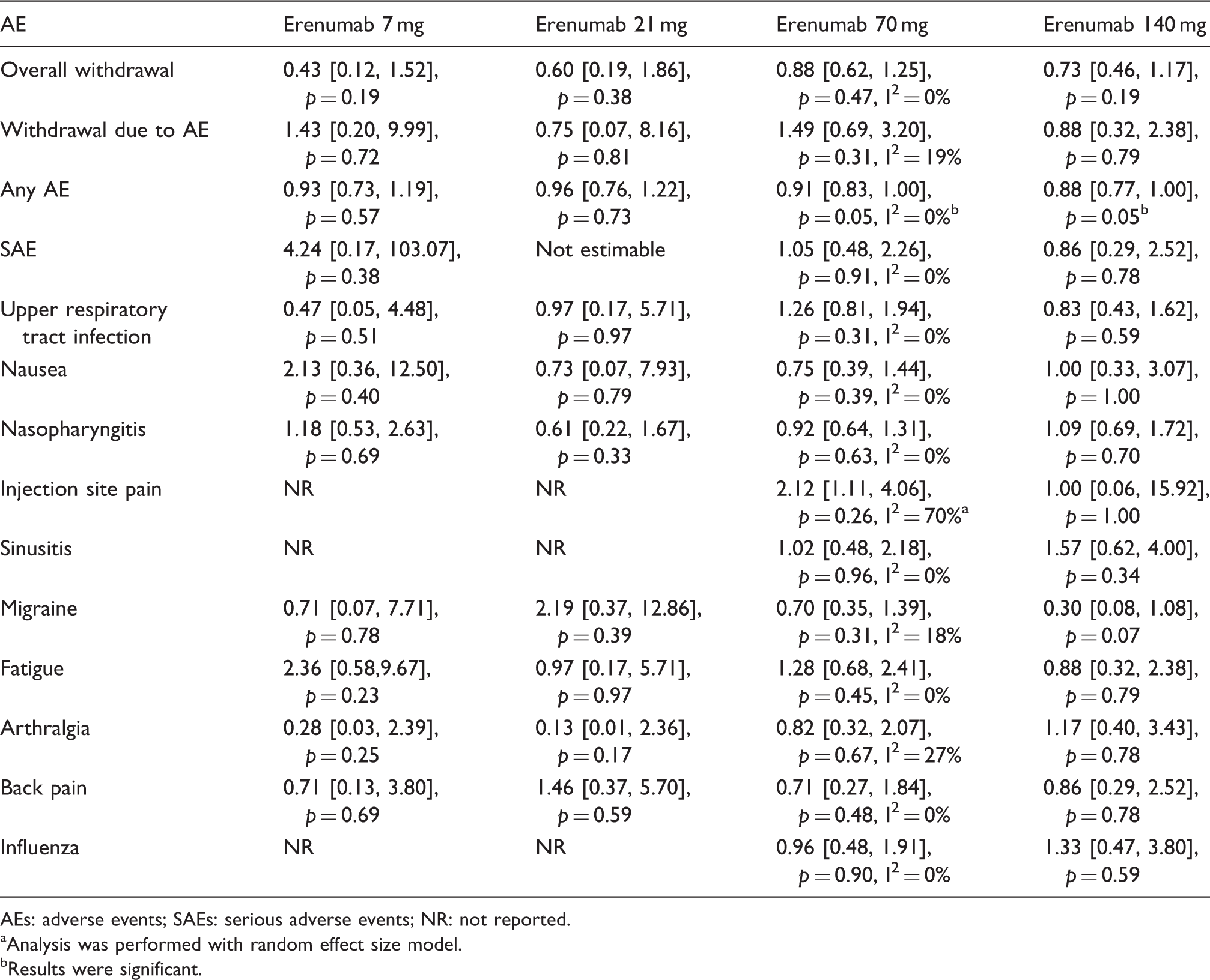
AEs: adverse events; SAEs: serious adverse events; NR: not reported.
Analysis was performed with random effect size model.
Results were significant.
Results of different doses of fremanezumab compared to placebo
Comparison results of different doses of fremanezumab.
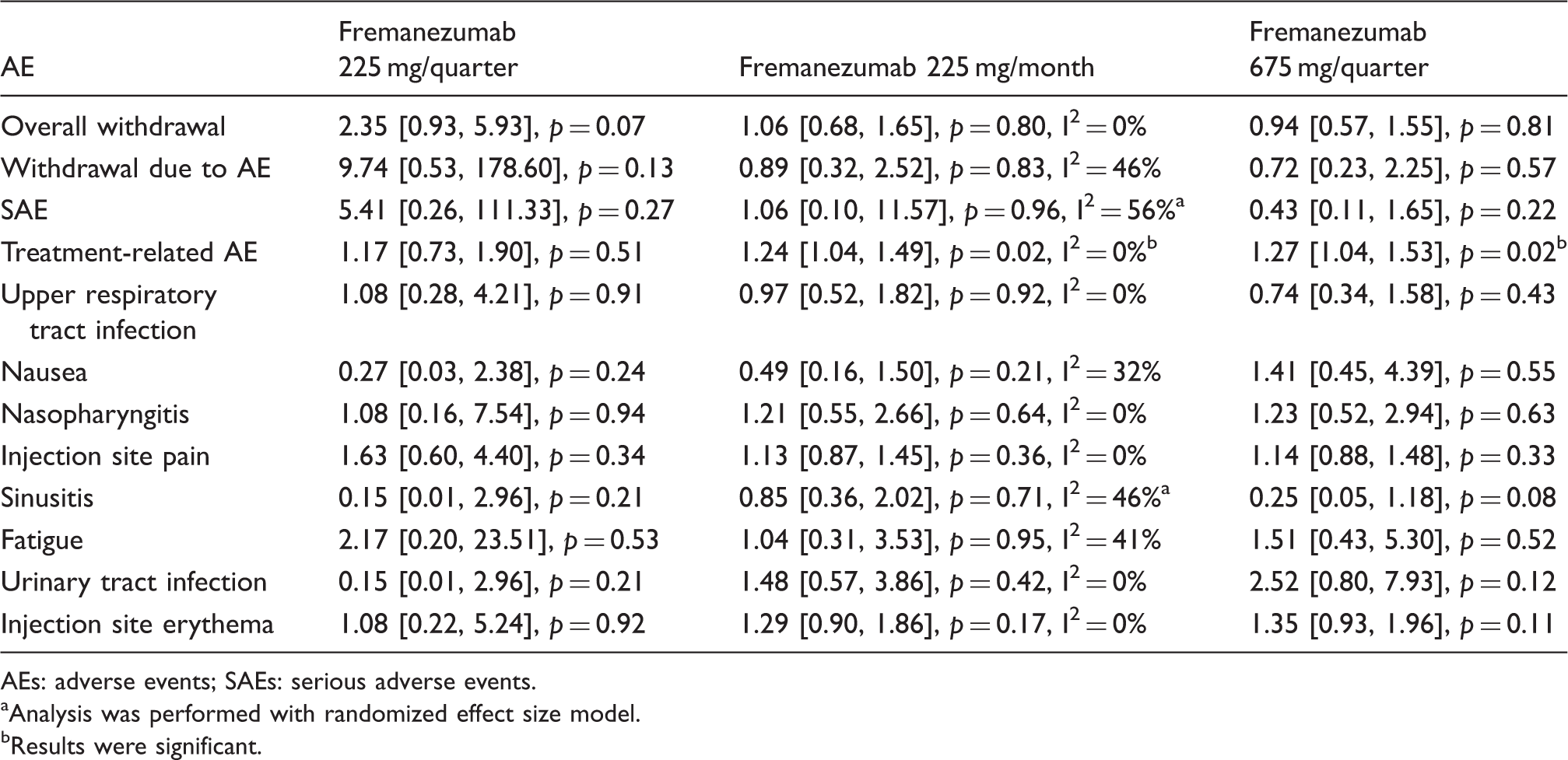
AEs: adverse events; SAEs: serious adverse events.
Analysis was performed with randomized effect size model.
Results were significant.
Results of eptinezumab compared to placebo
Comparison results between eptinezumab and placebo.
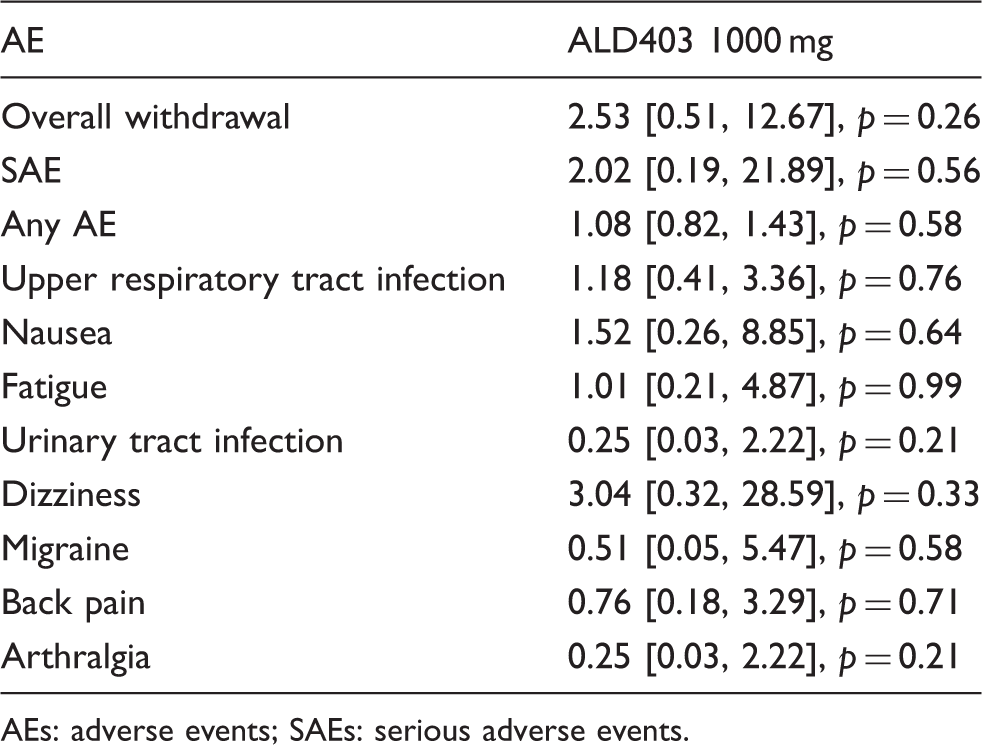
AEs: adverse events; SAEs: serious adverse events.
Results of ADA + reported in included studies
Postbaseline ADA + events and TEADA + events reported with galcanezumab.
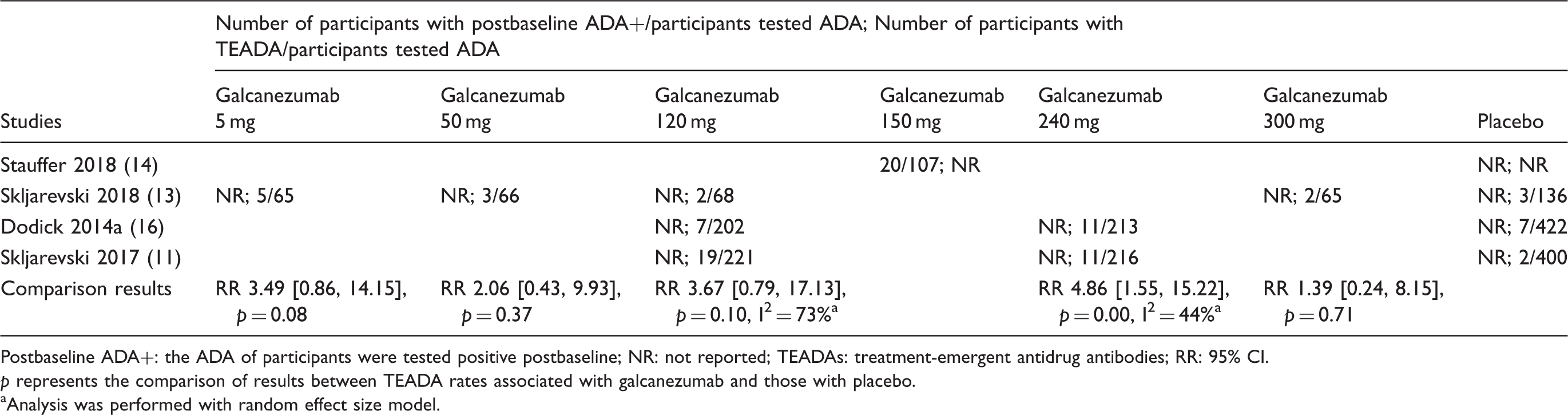
Postbaseline ADA+: the ADA of participants were tested positive postbaseline; NR: not reported; TEADAs: treatment-emergent antidrug antibodies; RR: 95% CI.
p represents the comparison of results between TEADA rates associated with galcanezumab and those with placebo.
Analysis was performed with random effect size model.
Postbaseline ADA + events in erenumab.

NR: not reported.
Postbaseline ADA + events in fremanezumab.

NR: not reported.
Postbaseline ADA + events in eptinezumab.
NR: not reported.
Sensitivity test
The sensitivity test was performed by converting RR to an odds ratio, and no difference was found. We also changed the fixed effects size model to a random effects size model, again with no significant differences.
Discussion
This study compared the safety and tolerability between four different CGRP binding mAbs and placebo when used as preventive treatment for episodic migraine. The safety profiles of all the doses of each CGRP binding mAb, galcanezumab, eptinezumab, fremanezumab and erenumab, were assessed individually. A previous meta-analysis related to this topic only focused on one specific CGRP binding mAb, namely erenumab, which targets the CGRP receptor (21). However, only phase 2 or 2b studies were included in the previous review (20). To the best of our knowledge, this is the first comprehensive study that includes five phase 3 trials to evaluate the safety and tolerability of CGRP binding mAbs.
In our review, a total of 10 high-quality studies reported 13 types of AEs. In general, CGRP binding mAbs were well tolerated, since the overall withdrawal rates and withdrawals due to AEs between treatment and placebo groups were similar. All included studies reported the occurrence of the rate of SAEs, which was low and similar between the CGRP binding mAbs and placebo groups. Moreover, SAEs were deemed not to be related to mAb treatment in most of the included studies. ‘Any AE’ was reported in six studies and was also similar between the two interventions. In essence, the safety of mAbs appears to be a consistent finding.
Regarding specific types of AEs, upper respiratory tract infection was the most frequently reported in all included studies. The occurrence rate was approximately 5.5% in the CGRP binding mAbs group and did not exceed the rate in the placebo group. Injection site reactions such as pain and erythema were more frequently associated with the CGRP binding mAbs group. However, the severity of these two reactions was mild to moderate and is well tolerated according to the literature (13). In our subgroup analysis, injection site pain appeared to be associated with different doses of galcanezumab. Moreover, in two phase 3 studies of galcanezumab 120 mg and 240 mg, the occurrence rate of injection site erythema was significantly higher than that of placebo (13,14). Injection site reactions appear to be the major adverse event associated with galcanezumab. There was no difference in the rate of occurrence of the remaining AEs such as nausea and nasopharyngitis between the mAb treated and placebo groups.
In the specific analysis of each dose of galcanezumab, 120 mg was associated with a higher rate of SAEs, though the investigators claimed that none of the SAEs were related to galcanezumab treatment (13). The occurrence rate of nasopharyngitis was approximately 6.5% among all galcanezumab-treated participants and was significantly higher in those taking galcanezumab 5 mg. The mechanism underlying the high frequency of nasopharyngitis is unclear, especially because the higher dose of galcanezumab did not lead to nasopharyngitis. This finding needs to be verified in long-term follow-up studies. Injection site reactions including pain and erythema were the main AEs associated with galcanezumab. However, since only four participants discontinued the study due to this AE, the tolerability of galcanezumab was accepted despite the high incidence of injection site reactions (12). The phase 3 studies that evaluated the 120 mg and 240 mg doses of galcanezumab reported a similar efficacy between these two doses, with both reaching the primary and secondary end points. These similar AE profiles persisted over a 6-month period. However, from a safety and economic perspective, subcutaneous administration of 120 mg may be the optimal monthly dosing of galcanezumab.
The Food and Drug Administration (FDA) has recently approved the CGRP binding mAb erenumab (AIMOVIG) for the prevention of migraine (32). Erenumab is the only mAb that targets the CGRP receptor instead of the CGRP ligand. The occurrence rate of any AE was marginally but significantly higher with the 70 mg and 140 mg doses of erenumab as compared to placebo. Nonetheless, there was no difference in specific AEs such as upper respiratory tract infection between erenumab treatment and placebo. However, migraine AE did appear to be more frequent with erenumab 140 mg when compared with placebo.
CGRP is the most potent vasodilator peptide known to date, and vascular safety is the main concern (33). However, none of the included studies reported any cardiovascular events during the 3 or 6-month treatment periods. Overall, all doses of erenumab were safe and well-tolerated. This outcome confirms erenumab as a promising treatment for migraine, as demonstrated by previous studies (21).
Regarding fremanezumab, three different doses were reported in two studies. In one study that enrolled participants with high frequency EM of at least 8 days of headache per month, both 225 mg/month and 225 mg/quarter were shown to be effective in preventing EM. A dose of 675 mg/quarter was first used in one of our included studies as a single higher dose among the four mAbs (12). This single higher subcutaneous dose was much more convenient than the monthly dose and demonstrated similar efficacy. In addition to the similar reduction in mean monthly migraine days, AEs associated with 225 mg/month and 675 mg/month were also similar in terms of the significantly higher rate of treatment-related adverse events (TRAEs). Interestingly, other than the marginally increased frequency of sinusitis with fremanezumab 675 mg/quarter, no difference was found in specific AEs between the two treatments. According to the literature, injection site pain was reported to be the most common fremanezumab-associated AE, and the incidence was higher than that associated with placebo (12). In our meta-analysis, the incidence of this AE with each dose of fremanezumab was not significantly higher than that with placebo, despite a slight increase. The use of this single higher dose should perhaps be the focus of future studies because of its strengths in terms of convenience and equivalent efficacy.
Only one phase 2 trial reported the safety of eptinezumab 1000 mg. There was no difference in the occurrence rate of all AEs between the two interventions. The withdrawal rate was also acceptable. However, this phase 2 trial only enrolled 163 participants, which was fewer than in other trials. Therefore, the safety of eptinezumab should be considered with prudence and confirmed in future large randomized controlled trials. There are three ongoing trials (NCT 02985398, NCT02974153 and NCT02559895) of eptinezumab in the treatment of EM and CM, and the results are scheduled to appear in the near future.
Additionally, we calculated the postbaseline ADA + rates and TEADA rates. Three studies of galcanezumab reported the rates of TEADA in both the intervention and control groups, and no difference was found. All included studies claimed that none of the ADA + -related AEs were noted, and ADA + did not influence the efficacy of CGRP binding mAbs. It seems that the effect of ADA + was small, but this was only examined over a short period of time. The long-term effect of ADA + on the efficacy and safety of CGRP binding mAbs should be investigated in future studies.
For the sensitivity test, the statistical results of our study were found to be reliable. No significant changes were found after the RR was converted to OR and a fixed effects size model to a random effects size model.
Previous oral preventive therapies for migraine were associated with a high withdrawal rate (34). In addition, perhaps because of tolerability and safety issues, both adherence and persistence with current medications tend to be low. The withdrawal rate of approximately 2.15% for CGRP binding mAbs is likely due to the low incidence of AEs. This finding is notably lower than withdrawals associated with the use of oral medications such as topiramate, which has a withdrawal rate of 17% and 27% for the 50 mg and 100 mg doses, respectively (34). Monthly dosing is more convenient than the daily dosing regimen of oral medications. Besides, a single higher dose of fremanezumab, which showed a similar therapeutic effect as monthly dosing, is simpler and probably easier to be accepted than the monthly dosing used in galcanezumab, eptinezumab and erenumab. It would be very worthwhile to investigate this treatment approach in future studies.
The mechanism underlying the limited AEs associated with CGRP binding mAbs remains unclear. Numerous studies have reported that monoclonal antibodies do not readily penetrate the blood-brain barrier, which may in part explain this result (33). The therapeutic effect is thought to be primarily achieved peripherally by targeting the CGRP pathway, which is sufficient for migraine prophylaxis. The main AE of CGRP binding mAbs, injection site reaction, is focal and regional, probably owing to its vasodilator nature and peripheral action.
There are some limitations in our analysis. First, due to the limited data, we only focused on the short-term side effects of mAbs during the double-blind period and neglected the long-term effects. Second, the inclusion criteria were not the same for all of our included studies. Some studies only enrolled patients who were not receiving migraine preventive medications, while other studies allowed the enrollment of patients with concomitant use of medications. In our meta-analysis, these studies were all included and analyzed. Third, the double-blind period was not the same in our included studies, ranging from 3 months to 6 months, which might contribute to heterogeneity. Finally, some adverse events, such as constipation, were only mentioned in a few studies and did not undergo a meta-analysis.
The long-term safety of CGRP binding mAbs remains unknown and needs to be investigated in large, randomized trials.
Conclusions
The results of this meta-analysis demonstrate that the use of CGRP binding mAbs was safe and well-tolerated in the short-term. The withdrawal rate was low and acceptable. SAEs were rare, and the severity of most AEs was mild to moderate. Injection site reaction may be the major AE associated with galcanezumab. At present, no obvious ADA+-associated AEs were identified. Future studies should focus on the long-term safety of CGRP binding mAbs.
Footnotes
Clinical implications
This meta-analysis demonstrates that the use of CGRP binding mAbs is safe and well-tolerated in the short term.
Injection site pain may be the major AE associated with galcanezumab despite the low withdrawal rate.
No obvious ADA+-associated AEs were identified.
Future studies should focus on the long-term safety of CGRP binding mAbs.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
