Abstract
Objective
To systemically review clinical studies investigating the role of prolactin and its receptors in headache and migraine.
Background
Migraine prevalence is more common in women compared to men. As prolactin is a crucial regulator of the hypothalamus-pituitary-gonadal axis, prolactin and its receptors might contribute to signaling mechanisms underlying migraine.
Methods
In this systematic review, we searched PubMed and EMBASE with the terms: prolactin, hyperprolactinemia, macroprolactinemia, hypoprolactinemia, migraine, headache, head pain and trigeminal pain pathway for clinical studies investigating prolactin signaling in headache and migraine. Two reviewers independently screened 841 articles for population, intervention, comparison, outcome, and study design. Studies were restricted to the English language and were excluded if they had a nonexperimental methodology.
Results
Nineteen clinical studies met the inclusion criteria and were included in the qualitative and quantitative analysis. The main findings were that serum prolactin levels were found to be higher in individuals with migraine compared to healthy controls, and prolactinomas (prolactin-secreting pituitary adenomas) were correlated with higher incidence of headache in otherwise healthy individuals and migraine attacks in individuals with migraine.
Conclusion
Considerable evidence suggests a key role of prolactin and its receptors in migraine pathophysiology. Further randomized and placebo-controlled clinical studies targeting prolactin signaling are needed to further clarify influences of prolactin in migraine attack initiation.
Introduction
Prolactin (PRL) is a polypeptide hormone expressed at lactotrophic cells in the anterior pituitary gland. PRL release and serum PRL levels are affected by physiological changes including breastfeeding, menstruation, pregnancy, and sleep, pathological factors including hypothyroidism and pituitary adenoma, and numerous drugs like dopamine, morphine, and serotonin (1,2). Thus, PRL is involved in a wide range of cellular signaling pathways, and increasing evidence suggests PRL implication in mechanisms leading to pain.
Migraine is the sixth most common of all medical conditions and the second leading cause of years lived with disability (3–5). The cumulative lifetime risk of migraine in the general population is ∼33% in women and 18% in men. Although, the prevalence of migraine before puberty and after 50 years of age is not sex dependent (∼5%), the prevalence of migraine during puberty rises in girls, and between 30 and 50 years of age the prevalence of migraine in women (30%) is three to four times higher than in men (4). About 20–25% of female migraine patients report menstrual migraine (e.g., attacks occurring in the perimenstrual period: the five-day window from two days before to two days after the first day of menstruation) (6). Moreover, the clinical feature of migraine attacks including characteristics, duration, frequency, severity and associated symptoms are highly dependent on age and sex. The sexual dimorphic etiology of migraine suggests that perturbations in PRL, which has a crucial role in controlling the hypothalamus-pituitary-gonadal (HPG) axis, might be involved in signaling mechanisms underlying migraine. Here, we highlight the structural and functional properties of PRL and its receptor, outline their expression and regulation, and provide a systematic review of the possible involvement of PRL in headache and migraine pathophysiology.
Molecular structure and receptor signaling
PRL is a member of the prolactin/growth hormone/placental lactogen family (7) encoded by a single gene on chromosome 6 with five exons and four introns (8,9). The mature PRL hormone has 199 amino acids arranged in loops and alpha helixes (10). Three molecular domains highlight the difference between PRL and growth-hormone: 1) the N-terminal loop is in contact with the first alpha-helix, 2) PRL lacks a minihelix in the loop between the first and second alpha-helix, and 3) PRL has a shorter loop between alpha-helix two and three and without a perpendicular minihelix in this loop (11) (Figure 1). The vast majority (80–90%) of circulating PRL is monomeric, while approximately 10% is dimeric (“Big PRL”) and 5% is multimeric (“Big Big PLR”) (12–14). “Big PLR” and “Big Big PLR” have low receptor binding affinity and, thus, decreased bioactivity (12,13). This might explain why elevated total serum PRL (macroprolactinoma) with increased concentration of dimeric and multimeric PRL, does not affect pituitary-gonad function (15).

Prolactin (PRL) structure. The core of human PRL molecule is made up by four major alphahelices that wind slightly around each other through three disulfide bonds. The PRL molecule varies from growth-hormone in three minor areas: 1) the N-terminal loop is in contact with the first alpha-helix, 2) PRL has not a minihelix in the loop between the first and second alpha-helix, and 3) PRL also has a shorter loop between alpha-helix two and three and lacking a perpendicular minihelix in this loop [11].
Prolactin receptor (PRLR) belongs to the class one cytokine receptor superfamily encoded by a gene located on chromosome 5, and it is closely related to the growth hormone receptor (16). The structure is a non-tyrosine, single-pass transmembrane chain with three major domains: the extracellular, transmembrane, and intracellular domain with two conserved regions termed box one and two (16). PRLR has three isoforms which only differ in the length and sequence of the intracellular domain. The long (PRLR-L) and intermediate (PRLR-I) isoforms have box one and two binding domains, whereas the short (PRLR-S) isoform has box one but lacks box two binding domain (Figure 2).

Prolactin receptor (PRLR) structure. (a) The PRLR is a non-tyrosine, single-pass transmembrane receptor that is composed of three major domains extracellular (EC), transmembrane (TM), and intracellular (IC) domains. When PRL is not present, the PRLR is inactive due to the arrangement of the kinase domains in JAK2 kinase. Binding of PRL induces dimerization of the PRLR and hereby activation of JAK2 kinase and (b) PRLR is found in three isoforms. The structural variations are observed in the IC domain. The long (PRLR-L) and intermediate (PRLR-I) isoforms include two binding domains, known as box 1 and 2 binding domains, whereas the short (PRLR-S) isoform only has the box 1 binding domain [16].
The binding of PRL to its receptor induces dimerization and stimulates numerous kinases, including proto-oncogene tyrosine-protein (src), Janus (Jak) and Ras/Raf/MAP kinases. This leads to phosphorylation of the cytoplasmatic signal transducer and activator of transcription-5 (STAT-5) proteins which then act as transcription factors regulating genes involved in proliferation, differentiation, and survival of cells (15,17). The finding that PRL induced fast signals within one minute after application indicated that PRL also might regulate ion channels (18–20) (Figure 3). PRL-induced currents are composed of separate low- and high-voltage components. Inhibition of big conductance potassium (BK) channel by paxilline reduced PRL-induced high voltage components, revealing that PRL signaling is in part dependent on BK channel activation (21). Moreover, PRL modulated peripheral transient receptor potential cation channel subfamily V member 1 (TRPV1), a channel also known as the capsaicin receptor, which is linked with nociception and cooperates with other TRP channels in the detection of noxious stimuli (22).

The main prolactin receptor (PRLR) kinase pathway. When prolactin (PRL) binds, dimerization of the PRLR activates Janus kinase (JAK2) which phosphorylates tyrosine residues in the intracellular domains of PRLR. Phosphorylation of tyrosine residues mediates the binding and phosphorylation of signal transducer and activator of transcription-5 (STAT-5) proteins [74]. Phosphorylated STAT proteins dimerize and translocate to the nucleus where they regulate transcription of genes involved in proliferation, differentiation, and survival of cells [15; 75].
Regulation of PRL secretion
The systemic factors affecting PRL homeostasis are called PRL-releasing factors (PRFs) and PRL-inhibiting factors (PIFs) (Table 1). During a day, PRL level peaks 14 times, particularly after sleep onset and 30 minutes after a meal, and each climax last approximately one hour (23–26). During pregnancy, PRL level increases continuously and reaches a tenfold elevation due to high estrogen level (27), which also causes lactotroph cell hyperplasia (28–30). High PRL level stimulates tuberoinfundibular dopamine (TIDA) neurons located in the hypothalamic arcuate nucleus to release dopamine which inhibits PRL secretion through dopamine 2 (D2) receptors in a short-loop feedback manner (31,32). This negative feedback mechanism explains hyperprolactinemia observed when using dopamine inhibitors (33–36) or when D2 receptors genes are knocked out (37–40). During late pregnancy and lactation, dopamine synthesis is blocked, meaning that PRL secretion is disinhibited, and PRL level remains high after birth (41,42). Noteworthy, the cycle of PRL and dopamine is dependent on the nonselective, Ca2+-permeable, and receptor-operated cation channel, denoted as transient receptor potential channel (Trpc5), expressed in dopaminergic neurons. Trpc5 is critical for mediating PRL signaling, and Trpc5 deficiency leads to profound hypoprolactinemia. Thus, Trpc5 is a major determinant of hypothalamic PRL regulation and defines the functional properties of dopaminergic neurons that plays a central role in PRL homeostasis (40).
Prolactin releasing and inhibiting factors.
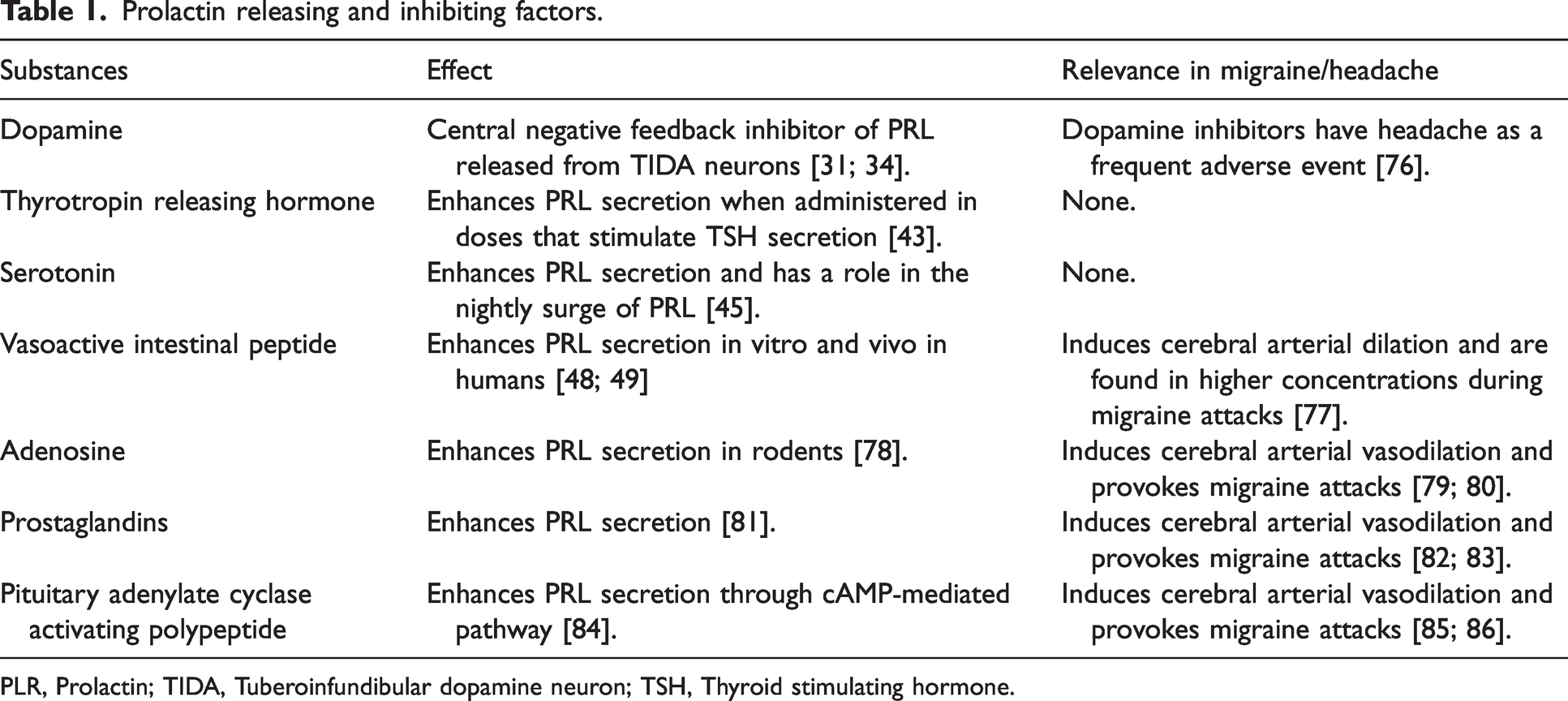
PLR, Prolactin; TIDA, Tuberoinfundibular dopamine neuron; TSH, Thyroid stimulating hormone.
Thyrotropin-releasing hormone (TRH) is an indirect PRF which stimulates PRL release through thyrotropin stimulating hormone (TSH). Individuals with primary hypothyroidism and thus high levels of serum TSH developed hyperprolactinemia, and treatment with levothyroxine reversed it (12,13,43,44). Serotonin is a PRF (45), and serotonin antagonists inhibit the nightly increase of PRL level (46,47). Vasoactive intestinal peptide (VIP) is another important PRF stimulating PRL synthesis and release in vitro as well as in vivo in humans (Table 1) (48,49).
Method
We searched PubMed and EMBASE for articles on PRL in relation to headache and migraine. The search was conducted on 6 August 2022, and the search string was “(“prolactin “[All Fields] OR “PRL”[All Fields] OR “hyperprolactinemia” [All Fields] OR “macroprolactinemia” [All Fields] OR “hypoprolactinemia” [All Fields] AND “Migraine” [All Fields] OR “Headache”[All Fields] OR “head pain”[All Fields]) OR “trigeminal pain pathway” [All Fields])”.
Selection criteria and study inclusion
An a priori systematic review protocol was developed. The full protocol can be obtained from the corresponding author upon request. We followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) reporting guidelines and the recommendations from the Cochrane Collaboration (50). The population, intervention, comparison, outcome, and study design (PICOS) approach were chosen as follows: study design, sample characteristics of the sample, intervention, comparator and outcomes.
After removing duplicates, two investigators (HG and VK) independently screened articles to confirm suitability for this review. References in the included studies were also screened, and relevant studies were included. Disagreements between the investigators were resolved through discussion. If the conflict remained, a third investigator (MMK.) had the final verdict. Studies were restricted to the English language and were excluded if they had a nonexperimental methodology (e.g., case reports, editorials, reviews, and meta-analyses) or their full text was not available. After extracting, obtaining, and screening the titles and abstracts for experimental preclinical and clinical studies assessing the involvement of PRL in trigeminal pain pathway and associated neuropeptides including calcitonin gene-related peptide (CGRP) and pituitary adenylate cyclase-activating polypeptide-38 (PACAP38), the full texts of the selected articles were obtained. Titles and abstracts lacking sufficient information regarding inclusion criteria were also obtained as full texts.
For each included study, the following data were extracted: demographic information (title, authors, and journals), study design, characteristics of the sample (animal/human, sex, and size), intervention, technique, substances, and outcomes. The review protocol for preclinical and clinical studies is identical.
Results
The database search identified 828 citations without duplicates. An additional 13 studies were included through a manual search of identified primary articles. A total of 841 studies were screened by title and abstract, 73 studies underwent full-text screening. Of these, 34 studies were included: 15 pre-clinical and 19 clinical (Figure 4 and Table 2). Pre-clinical findings are in press in Headache: The Journal of Head and Face Pain. Clinical studies investigated headache outcomes of pituitary adenomas based on PRL levels, intrasellar pressure, size of pituitary tumor, transsphenoidal surgery, and treatment with D2 receptor agonists (Table 2). The consensus of these studies was that PRL secreting pituitary adenomas (prolactinomas) caused headache (51–55) and stimulation of PRL release by TRH provoked migraine attacks (52). Furthermore, headache incidence was higher in patients with prolactinomas compared to nonfunctioning pituitary adenomas (54) and lowering PRL levels with D2 receptor agonist relieved headache in hyperprolactinemic patients (56).

Flow chart of the search strategy.
Clinical studies, design, and findings.

Reporting of adenoma type, headache, and location of headache.
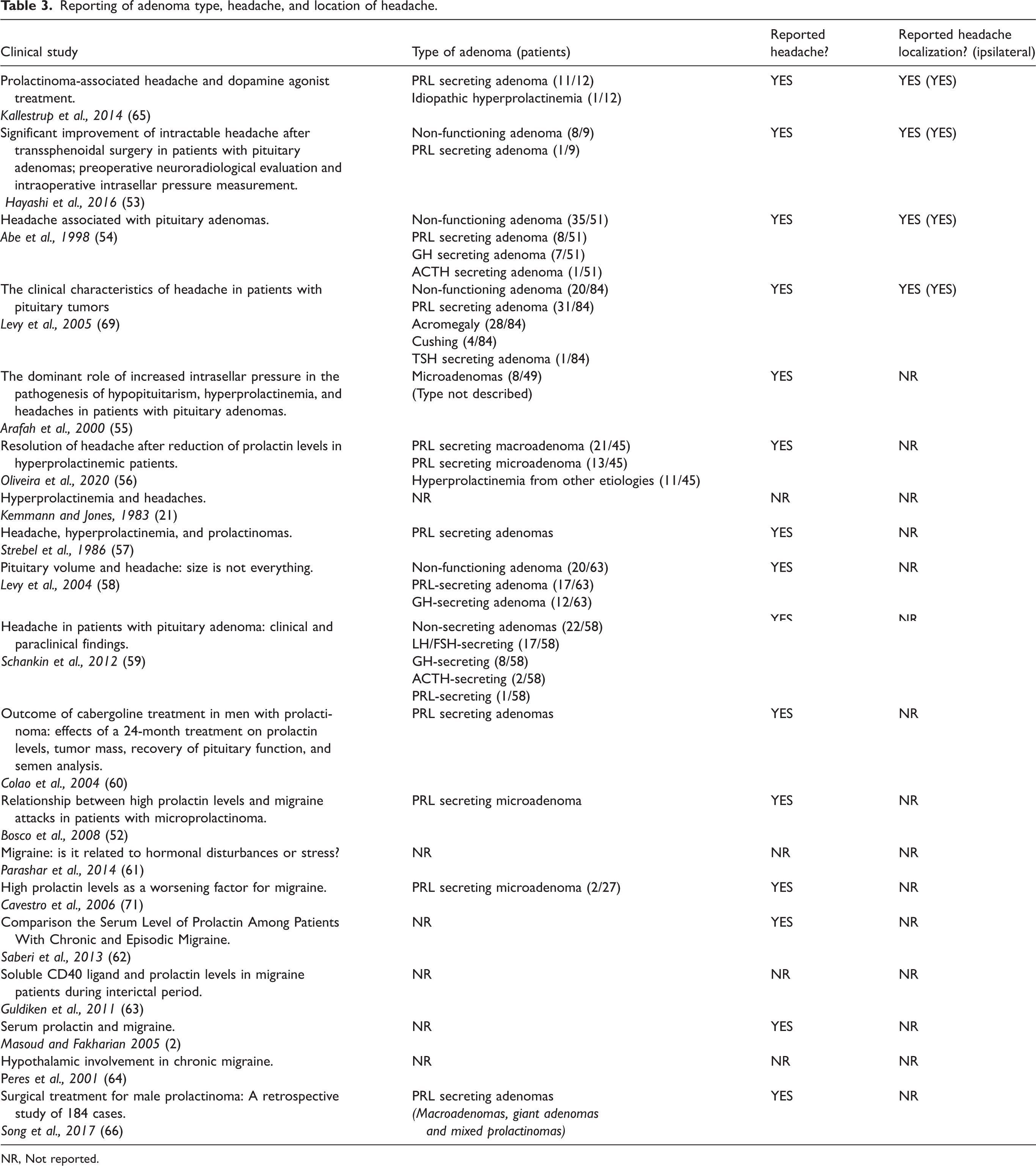
NR, Not reported.
Narrative summaries of clinical studies
Hayashi and colleagues (53) assessed 180 patients with pituitary adenoma. Nine patients had intractable headache as the main complaint: eight with non- functioning and one with PRL-secreting adenoma. All tumor patients underwent transsphenoidal surgery for pituitary adenoma and reported complete or substantial resolution of headache. Intrasellar pressure was significantly higher in the nine patients with headache compared to control patients.
Abe and colleagues (54) assessed 51 patients with pituitary adenoma. Nineteen (13 women and six men) of these patients had headache. Patients with PRL-secreting adenoma (11 of 19, 58%) had more prevalent headache compared to patients with non-functioning adenoma. After transsphenoidal surgery for pituitary adenoma, 14 of the 19 patients (74%) reported substantial resolution of headache.
Arafah and colleagues (55) assessed 49 patients with pituitary adenoma and reported that PRL levels is positively correlated with high intrasellar pressure but the tumor size was not correlated to PRL levels or intrasellar pressure. The authors concluded that increased intrasellar pressure might be the major mechanism contributing to the development of hyperprolactinemia.
Oliveira and colleagues (56) included in a longitudinal study 69 patients with PRL-secreting adenoma. Of these, 45 (65%) patients reported headache. After treatment with dopamine agonist, 33 (75%) of 43 (two were excluded because of missing evaluation of PRL level) of patients reported complete or partial resolution of headache which was correlated with reduction of PRL level. These 33 patients included 20 with migraine, six with tension-type headache and seven with non-classified headache.
Kemmann and Jones (21) showed that 46 women with hyperprolactinemia but no pituitary enlargement had significantly more headache episodes compared to 56 healthy women. The authors failed to find relationship between PRL levels and frequency or severity of headache episodes.
Strebel and colleagues (57) conducted a population follow-up study between 1973 and 1979 of 469 women with hyperprolactinemia. Of these, 212 were subsequently diagnosed as having a prolactinoma. Headache was four times more frequent in the presence of an adenoma than in its absence. This finding remained unchanged after adjustment for several factors including PRL levels and age. The authors concluded that headache is likely to be induced by the space-occupying mass effect of an adenoma.
Levy and colleagues (58) prospectively evaluated 63 patients after being diagnosed with pituitary adenoma. Headache was reported by 70% (44 of 63 patients), and the highest headache scores were seen in the PRL- and GH-secreting adenoma. The authors found no association between headache and pituitary volume or cavernous sinus invasion.
Schankin and colleagues (59) prospectively evaluated 58 patients after being diagnosed with pituitary adenoma. Headache was reported by 41% (24 of 58 patients); the headache fulfilled the criteria of tension-type headache (n = 11), migraine (n = 7), tension-type headache and migraine (n = 3), cluster headache (n = 1) and unclassified headache (n = 2).
Colao and colleagues (60) investigated the outcome of 24 months of dopamine agonist treatment on PRL level in 10 men with micro- (<1.0 cm in diameter)- and 41 men with macroprolactinoma (≥1.0 cm). Before treatment, two of 10 (20%) patients and 14 of 41 (34%) patients reported headache. After 24 months of therapy, PRL levels were normalized in eight (80%) patients with micro- and in 31 (76%) patients with macroprolactinoma, and headache disappeared in 50% of patients with micro- and in 83% patients with macroprolactinoma.
Bosco and colleagues (52) recruited 29 patients (group A: 17 patients diagnosed with migraine; and group B: 12 patients diagnosed with tension type headache) with microprolactinoma. PRL levels measured during migraine attacks were significantly higher in ten of 16 (59%) participants of group A and in one participant (8%) of group B compared to baseline. In five of the ten participants of group A, TRH induced PRL release and caused severe migraine attacks which were successfully treated with dopamine agonist.
Parashar and colleagues (61) measured serum PRL levels during the second day of the menstrual cycle and found significantly higher PRL levels in 20 migraine without aura patients compared to 20 age-matched healthy controls.
Cavestro and colleagues (71) showed that seven women of 27 patients (one man and 26 women) with chronic migraine had high serum PRL levels compared to standardized laboratory values, and treatment with dopamine agonist cabergoline improved their headache and migraine attacks became episodic.
Saberi and colleagues (62) included 114 women with migraine and reported that the presence of hyperprolactinemia and serum PRL levels were higher in women with chronic migraine compared to women with episodic migraine.
Guldiken and colleagues (63) reported no difference in serum PRL levels measured in 50 female migraine patients (with and without aura) during interictal period compared to 25 age-matched healthy female controls.
Masoud and Fakharian (2) reported that serum PRL levels from 37 migraine patients during migraine attacks were significantly lower compared to not age-matched 37 healthy controlled. The authors did not report if the healthy controls had any headache during PRL measurement and did not compare ictal to interictal PRL levels in migraine patients.
Peres and colleagues (64) reported that nocturnal PRL peaks (determined every hour for 12 hours) were lower in 17 patients with chronic migraine compared to nine age- and sex-matched healthy controls.
Kallestrup and colleagues (65) included 11 patients with prolactinoma and one patient with idiopathic hyperprolactinemia presenting with headache. The authors found a significant ipsilateral location of tumor mass and reported headache symptoms, a significant headache relief after treatment with dopamine agonist, and no significant association between headache relief and tumor shrinkage or normalization of serum PRL level. Whether headache relief is correlated with reduction of serum PRL level is not reported.
Song and colleagues (66) reviewed 184 male patients with prolactinoma in a retrospective study. They found that 55.9% of the patients reported headache and 87% reported symptom relief after transsphenoidal surgery for pituitary prolactinoma.
Levy and colleagues (69) investigated the clinical characteristics of 84 patients – both male and female patients – with pituitary tumors and troublesome headache, in which 46% was chronic and 30% episodic migraine patients. 37% of the tumors were prolactinomas. 60% reported strictly unilateral headache. Interestingly, cavernous sinus invasion was present in 21% of the cases, and the headache was located ipsilateral to the invasion. The patients were treated with either surgery (hypophysectomy), somatostatin analogues or dopamine agonists.
Discussion
The present review indicated a possible involvement of PRL and PRLR in the complex molecular interplay leading to headache and migraine. Clinical studies reported headache as a frequent symptom in patients with prolactinoma and high PRL levels in migraine patients. These results are strengthened by studies reporting preventive headache and migraine effects by dopamine agonists in patients with hyperprolactinemia. D2 receptor antagonists including metoclopramide and domperidone relief migraine pain. Therefore, D2 receptor antagonists are recommended on the treatment of acute migraine attacks (67,68). D2 receptor agonists, however, relieve migraine pain in the presence of prolactinoma (56,69). Further studies with control groups investigating the correlation of pain relief and PRL level are needed to draw a conclusion on PRL involvement in treatment response.
The significant association between lateralization of headache and adenoma (65), and the finding that intrasellar pressure was significantly higher in patients with headache compared to patients without headache indicate that pituitary tumor-associated headache might simply be a structural problem. However, 1) there was no significant association between headache relief and tumor shrinkage after treatment with a dopamine agonist, 2) patients with PRL-secreting adenoma had more prevalent headache compared to patients with non-functioning adenoma, 3) patients with hyperprolactinemia but no pituitary enlargement had significantly more headache episodes compared to healthy controls. Additionally, one study reported that headache scores were significantly higher in the PRL- and GH-secreting adenoma compared to other adenomas (69), and there was no association between headache and pituitary volume or any compartment of cavernous sinus invasion (58).
Serum PRL levels was positively correlated with high intrasellar pressure, but the tumor size was not correlated to PRL levels or intrasellar pressure. Kruse and colleagues (70), however, showed no difference in adenoma volume, intrasellar pressure and suprasellar extension between patients with elevated PRL levels compared to patients with normal PRL level. Studies investigated correlation between serum PRL levels and headache reported conflicting results. One study reported an increase of serum PRL levels during migraine attacks compared to baseline (52). Serum PRL levels during the menstrual cycle were significantly higher in migraine without aura patients compared to age-matched healthy controls (61). Moreover, the presence of hyperprolactinemia and serum PRL levels were higher in women with chronic migraine compared to women with episodic migraine (62). Cavestro and colleagues (71) showed that seven women of 27 patients (one man and 26 women) with chronic migraine had high serum PRL levels compared to standardized laboratory values, and treatment with dopamine agonist cabergoline improved their headache and migraine attacks became episodic. These findings implicate PLR in chronification of migraine. However, other studies reported no difference in serum PRL levels in migraine patients (with and without aura) during interictal period (63), and even significantly lower serum PRL levels in migraine patients during migraine attack compared to age-matched healthy controls (2). Peres and colleagues (64) reported that nocturnal PRL peaks (determined every hour for 12 hours) were lower in 17 patients with chronic migraine compared to nine age- and sex-matched healthy controls. Different methodological approaches, particularly, not differentiating in migraine types might explain this discrepancy. A systematic review and a meta-analysis of available studies measuring serum PRL levels included 460 migraine patients and 429 healthy controls and reported that serum PRL levels were higher in migraine patients compared to healthy controls (72).
Serum PRL levels follow a circadian rhythm and underlay a tight control by the hypothalamus, which is believed to be involved in the onset of migraine attacks since premonitory symptoms in migraine, including food craving and mood disturbances suggest a transient hypothalamic dysfunction and perturbations in the hypothalamic–pituitary–gonadal (HPG).
PRL seems to exert its influence by activation of the peripheral trigeminal pain pathway; however, the exact signaling cascades are yet to be elucidated (Figure 5). PRLR isoforms PRLR-L and PRLR-S are expressed in sensory neurons in the trigeminal pain pathway. In contrast to PRLR-L, stimulation of PRLR-S induced excitability of sensory neurons. Therefore, it seems that PRLR-S is involved in migraine attack initiation, while activation of PRLR-L might block migraine attacks. Since the PRLR in humans is distinct from rodents (the PRLR in human is also activated other hormones, including GH) these preclinical data needs to be translated before making the case that PRLR-S in a potential target for the treatment of migraine (73).

Distribution of PRL and PRLRs in the trigeminal pain pathway.
Conclusion
Clinical research indicates that PRL and its receptors might be involved in migraine pathophysiology. These findings move the field a step closer to a molecular classification of a complex condition with diverse clinical manifestations. Further randomized and placebo-controlled clinical studies targeting PRL signaling are needed to further clarify influences of PRL in migraine attack initiation and to possibly shape a sex-based treatment of migraine.
Key Findings
Serum prolactin levels were found to be higher in individuals with migraine compared to healthy controls. Prolactinomas (prolactin-secreting pituitary adenomas) were correlated with higher incidence of headache in otherwise healthy individuals and migraine attacks in individuals with migraine. Further randomized and placebo-controlled clinical studies targeting prolactin signaling are needed to further clarify influences of PRL in migraine attack initiation.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank NIH for funding professor Dussor for his work on prolactin (NIH grant number NS 104200), the Research Fund of Rigshospitalet (E-23327-04) and the Lundbeck Foundation Professor Grant (R310-2018-3711).
