Abstract
Background
It has been suggested that patients with migraine have a higher risk of stroke. Despite considerable research on this topic in younger populations, a clear answer is still lacking for older individuals. We studied the association between migraine and the risk of stroke in a middle-aged and elderly population.
Methods
Within the ongoing prospective population-based Rotterdam Study, the presence of migraine was assessed using a validated questionnaire in a structured interview between 2006 and 2011, which formed the baseline. The association between migraine and the risk of stroke was analyzed using Cox proportional-hazards models with adjustments for age, sex, and cardiometabolic risk factors.
Results
A total of 6925 (mean age 65.7 ± 11.3 years, 57.8% females) stroke-free participants were included. At baseline, 1030 (14.9%) participants had lifetime history of migraine. During a median follow-up of 6.2 years, 195 participants developed a stroke (163 ischemic stroke). Analyzing the association between migraine and stroke, we found a hazard ratio of 1.44 with a 95% confidence interval of 0.96–2.15. The results were similar for the ischemic stroke (HR 1.50, CI: 0.97–2.32).
Conclusion
Our data suggested an association between migraine and the risk of stroke in a middle-aged and elderly population, but this was not statistically significant.
Introduction
Migraine is suggested to be related to cardiovascular disease events. Although the exact pathophysiology linking these conditions is unclear, suggested mechanisms include endothelial dysfunction, systemic vasculopathy, and hypercoagulability (1). A recent meta-analysis that included several case-control and cohort studies suggested that migraine is linked with stroke (adjusted pooled Hazard Ratio [HR] 1.62, 95% Confidence Interval [CI] 1.44–1.83) (2). Despite its large sample size and robust methodology, specific considerations should be taken into account. Firstly, there were considerable heterogeneity in the results (I2 = 95.8%; p < 0.001) and methodology of the included studies. In the literature, most studies rely on either self-report or medical history for identifying stroke cases without imaging confirmation. Likewise, mostly self-report questionnaires or physician diagnoses from health records are used for migraine assessment. Secondly, some studies focusing on older (>50 years) participants (3,4) were excluded from the meta-analysis, limiting the possibility to generalize these results to the middle-aged and elderly population.
There is only a limited number of population-based studies investigating migraine and stroke association in elderly (aged ≥50 years) participants (3–9) and the results are inconclusive. Some studies reported an increased stroke risk in older migraine patients with statistically significant hazard ratios in the range of 1.20–1.51 (3,7), while many others demonstrated no statistically significant associations (4,5,8,9). Some studies reported an association only in female migraine patients (6), patients who are also current smokers (9) or who have late-onset (after age 50) migraine with aura (5).
In the present study, we determined the association between migraine and the risk of stroke (and ischemic stroke) in the middle-aged and elderly population using comprehensive migraine and stroke ascertainment.
Materials and methods
Study setting and population
This study was embedded in the Rotterdam Study, a prospective population-based cohort study among middle-aged and elderly community residents of Ommoord, Rotterdam, the Netherlands (10). The original cohort of the study started in 1990, followed by an expansion cohort in 2000, and both cohorts included participants ≥55 years old. In 2006, the Rotterdam Study expanded with another cohort that included participants ≥45 years old. From all three cohorts, a total of 7266 participants were interviewed to determine their migraine status. Among these, 279 had a history of stroke at the time of migraine interview, and 62 did not provide informed consent for follow-up, resulting in 6925 participants with both migraine and stroke information available (see Figure 1 for a flowchart of participant inclusion). Lifetime migraine prevalence was assessed between 2006 and 2011 in a home interview or over the telephone (n = 634, due to logistic reasons). Participants were followed for incident stroke, starting from migraine interview until: a stroke; death; last date when they were known to be stroke-free; 1 January 2016 (end of follow-up), whichever came first.
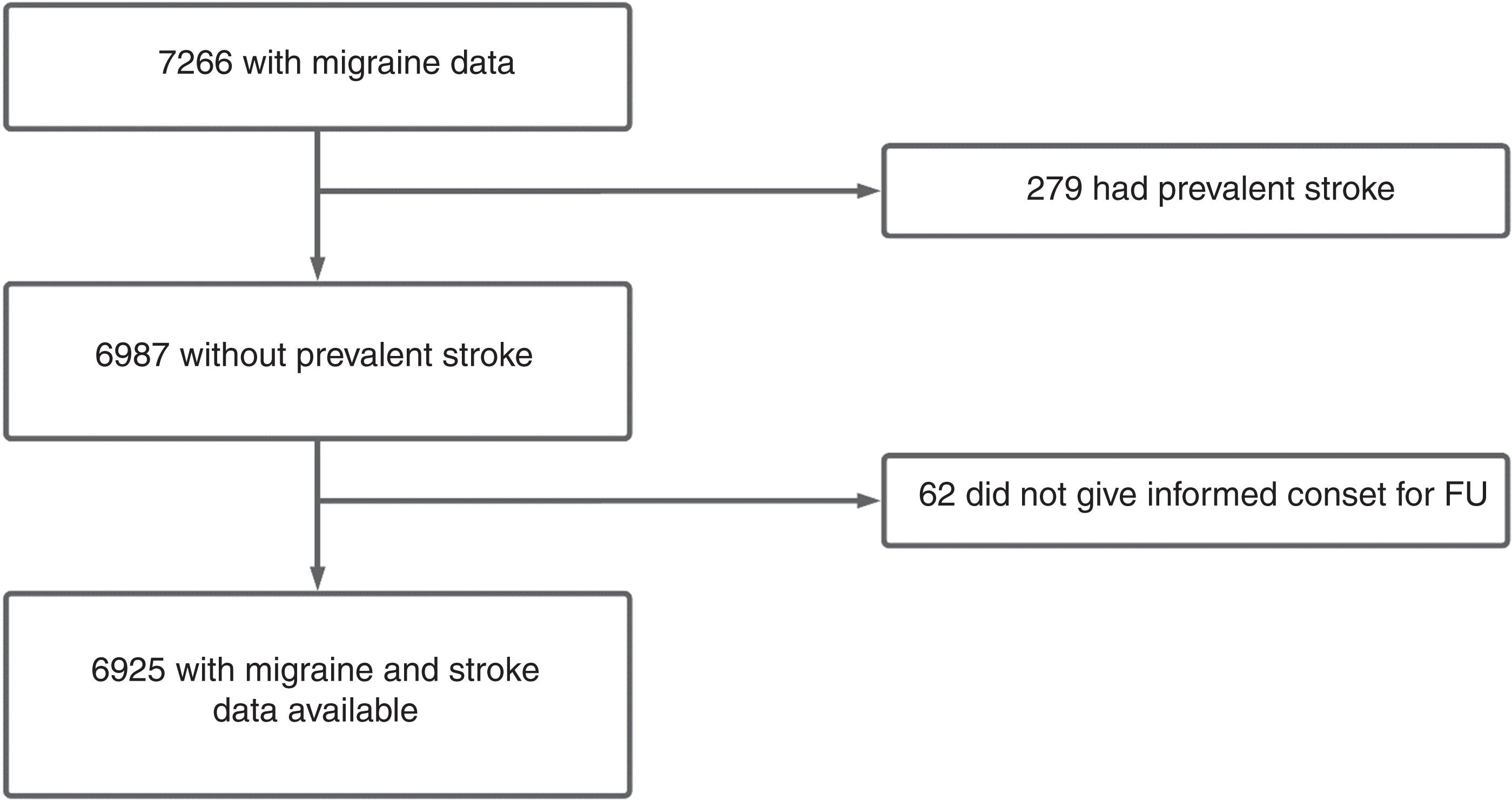
Flowchart for the selection of study participants.FU: Follow-up.
The Rotterdam Study has been approved by the Medical Ethics Committee of the Erasmus MC (registration number MEC 02.1015) and by the Dutch Ministry of Health, Welfare and Sport (Population Screening Act WBO, license number 1071272-159521-PG). The Rotterdam Study Personal Registration Data collection is filed with the Erasmus MC Data Protection Officer under registration number EMC1712001. The Rotterdam Study has been entered into the Netherlands National Trial Register (NTR; www.trialregister.nl) and into the WHO International Clinical Trials Registry Platform (ICTRP; https://apps.who.int/trialsearch/) under shared catalogue number NTR6831. All participants provided written informed consent to participate in the study and to have their information obtained from treating physicians.
Assessment of migraine
Migraine was assessed during a structured interview with a questionnaire based on the International Classification of Headache Disorders, second edition (ICHD-2) criteria (11). Items were modified from the questionnaire validated for use in the Genetic Epidemiology of Migraine (GEM) study in Leiden which has a sensitivity of 0.93 and a specificity of 0.36 (12). The following diagnostic criteria were applied to assess the lifetime occurrence of migraine headache attacks: having at least five attacks of severe pain lasting 4–72 hours (when untreated) with a minimum of two of the four characteristics (unilateral location, pulsating quality, severe pain intensity, and worsening by, or causing avoidance of, routine physical activity), and any of the two characteristics (nausea, photophobia/phonophobia), with these symptoms not being attributed to another disorder. All participants who met the criteria for lifetime history of migraine were categorized as persons with migraine. Migraine with aura was defined as meeting all the criteria for migraine without aura and reporting headache attacks with visual, sensory, or language-related aura symptoms that lasted between 5–60 minutes. Furthermore, to differentiate the history of migraine from current migraine at the time of the baseline interview, we dichotomized individuals as having active migraine (the last attack occurred <12 months ago) or non-active migraine (the last attack occurred >12 months ago). Finally, probable migraine was defined as meeting all but one criterion for migraine without aura.
The questionnaire was slightly modified from ICHD-2 criteria; only participants who had reported a history of severely (instead of moderately) painful headaches in the first question received the rest of the questions (i.e., the first question served as a screening tool). See online Supplementary Table S1 for a detailed list of the criteria.
Assessment of stroke
Stroke was defined according to the World Health Organization (WHO) criteria as a syndrome of rapidly developing symptoms, with a vascular cause of focal or general disturbance in cerebral functioning, which lasts more than 24 hours or leads to mortality (13). Initially, stroke history was determined at baseline in the interview by a qualified physician and verified from medical records. Next, participants were continuously monitored for incident strokes by automatically linking the study database with files from nursing homes and general practitioners. Medical files of participants who moved out of the study district were also checked continuously. Additionally, imaging reports were obtained from hospital records to ascertain stroke subtypes as ischemic or hemorrhagic. When this information was insufficient to discern between these types, then it was categorized as ‘unspecified’. Subarachnoid hemorrhages caused by ruptured aneurysms were not classified as stroke events. Finally, the information gathered on potential strokes was assessed and classified by research physicians and verified by two experienced stroke neurologists.
Other measurements
Covariables were selected based on their biologically plausible role in migraine etiology or risk of stroke or both (14). Based on the latest literature review; body mass index (15,16), smoking (17,18), educational level (17,19) and physical activity (20,21) were considered as common causes of migraine and stroke. Furthermore, research shows coronary heart disease (22,23) and hypertension (23,24) are possible mediators on the pathway from migraine to stroke. On the other hand, the role of hypercholesterolemia (23,25), alcohol use (23,26), and diabetes mellitus (23,27) in the migraine-stroke relationship is still debated. They could either be common causes of migraine and stroke, or mediators. See Figure 2 for a directed acyclic graph showing the causal framework underlying our analysis.
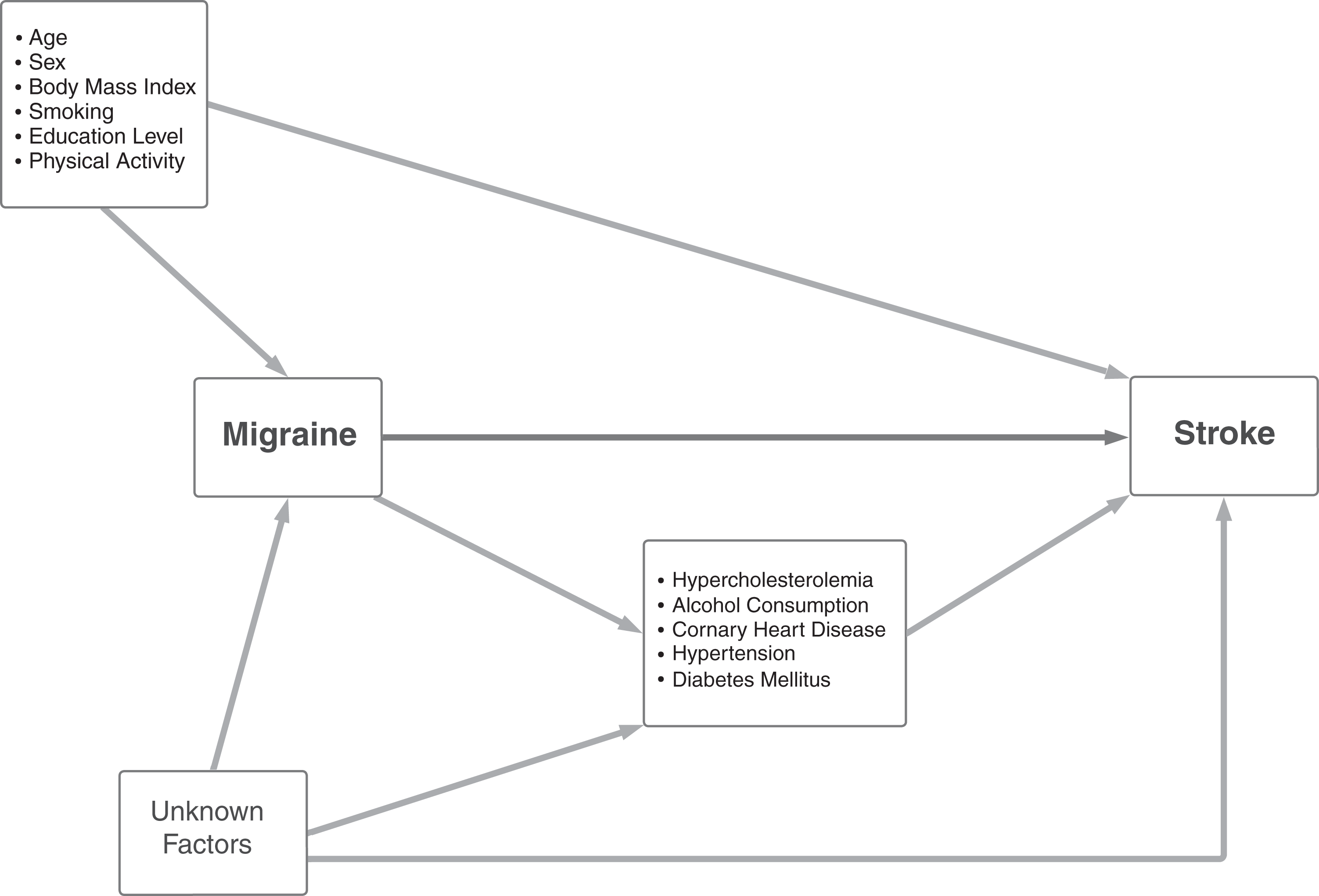
Directed acyclic graph of the postulated causal framework underlying the migraine-stroke association.
Detailed information on variables was collected via interview, physical examination, and blood sampling. Firstly, the cardiometabolic variables that were collected via physical examination and blood sampling were: body mass index (weight in kg divided by height in m2); hypertension (blood pressure systolic ≥140 mm Hg, diastolic ≥90 mm Hg or treated) (28); hypercholesterolemia (total cholesterol ≥6.2 mmol/L or treated) (28); and diabetes mellitus (fasting glycaemia ≥7.0 mmol/L or treated). Secondly, the following variables were measured via questionnaires: education level (primary; lower/intermediate education; intermediate vocational or higher general education; and higher vocational education or university), smoking (never, former and current smoker), physical activity (metabolic equivalent of a task in hours per week) (29) history of coronary heart disease (myocardial infarction or revascularization) and alcohol use (grams/day).
Statistical analyses
To determine the effect of migraine on the risk of stroke, we applied the following strategy. First, Kaplan-Meier survival curves were estimated to compare the probability of stroke-free survival in people with and without migraine. Second, we constructed three multivariable Cox regression models to investigate the association of migraine with the risk of stroke. These included a crude model (model 1), a model adjusted for age and sex (model 2), and a model adjusted for the following covariables; age, sex, educational level, smoking status, body mass index and physical activity (model 3). Importantly, some of the aforementioned variables (hypertension, hypercholesterolemia, coronary heart disease history, alcohol use and diabetes mellitus) are debated to be potential mediators in the migraine-stroke relationship. In the case they are mediators, adjusting for these variables may introduce collider bias. Therefore, we excluded these variables from the adjustment set. Third, we repeated these analyses with ischemic stroke as the outcome. Fourth, we combined definite and probable migraine diagnoses as exposure and repeated the analyses with any and ischemic stroke as the outcome.
Proportionality assumptions for Cox Models were checked visually via log-minus-log plots and Schoenfeld residuals test. No variable was violating the proportional hazards assumption. IBM SPSS (Chicago, IL, USA) version 25 and R version 4.0.5 (R core team, Vienna, Austria) for Windows were used for the analyses. P-values <0.05 were considered to be statistically significant. Missing values among covariables were imputed using multiple imputations by chained equation based on all variables but migraine and stroke, with 5 imputations and 40 iterations using “mice” package (version 3.13.0) in R (30).
Results
Table 1 shows the baseline characteristics of the study population. The mean age of the population was 65.7 (±11.3) years. A total of 1030 individuals (14.9%) met the criteria for lifetime migraine. Among participants with migraine, 402 (39.0%) had active migraine and 210 (20.4%) migraine with aura (see Table 2). Highest prevalence observed between ages 55–64 for males (7.9 per 100) and 65–74 for females (22.9 per 100; see Table 3 for migraine prevalence per age group and sex). Follow-up was complete for 95.3% of potential person-years. During a median of 6.2 years (maximum 9.9 years) follow-up, 195 persons developed a stroke. Among the stroke events, 163 (84%) were ischemic, 23 (11%) were hemorrhagic, and 9 (5%) were classified as unspecified stroke.
Baseline Characteristics of the study population.
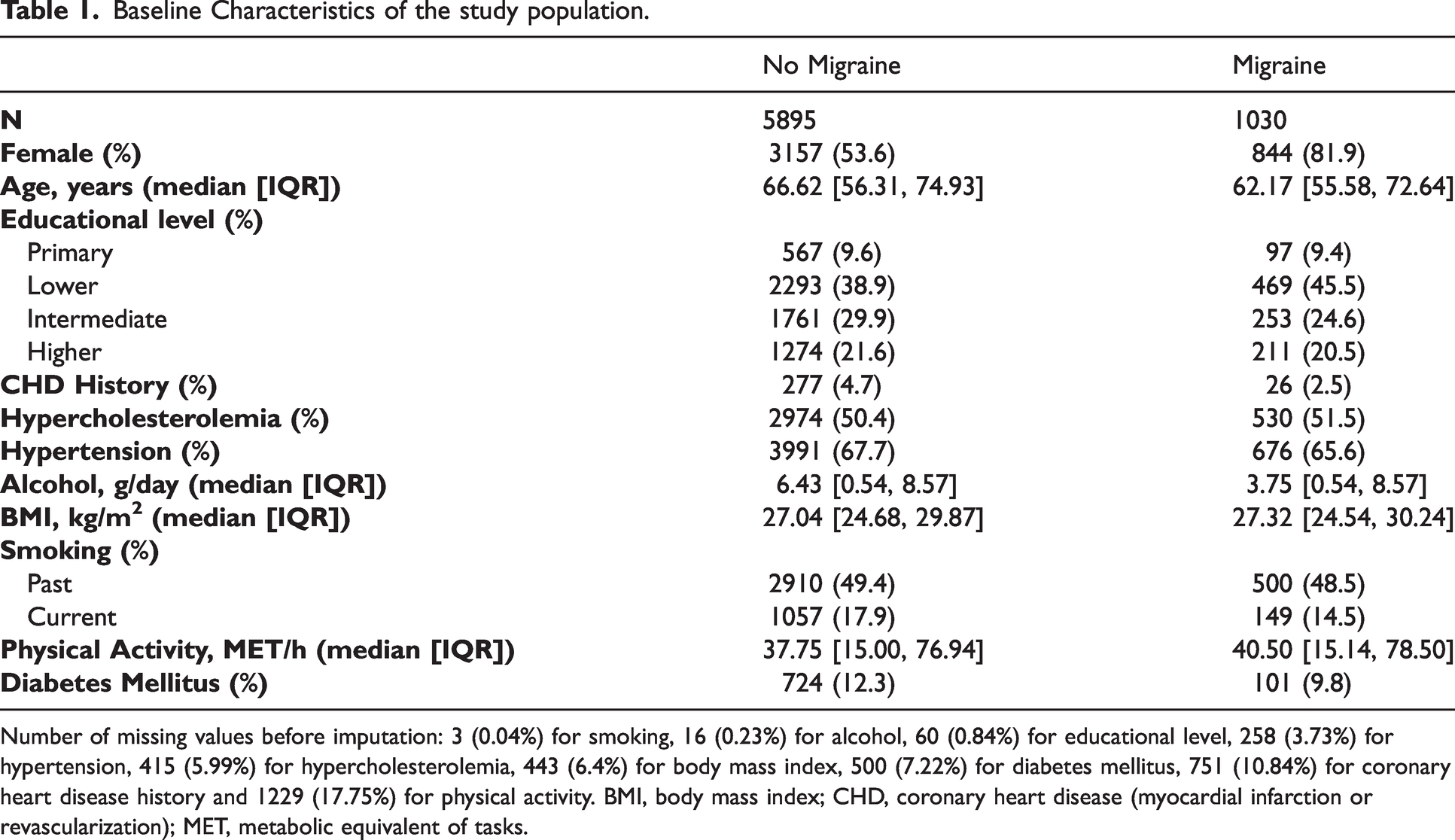
Number of missing values before imputation: 3 (0.04%) for smoking, 16 (0.23%) for alcohol, 60 (0.84%) for educational level, 258 (3.73%) for hypertension, 415 (5.99%) for hypercholesterolemia, 443 (6.4%) for body mass index, 500 (7.22%) for diabetes mellitus, 751 (10.84%) for coronary heart disease history and 1229 (17.75%) for physical activity. BMI, body mass index; CHD, coronary heart disease (myocardial infarction or revascularization); MET, metabolic equivalent of tasks.
Migraine subgroup prevalence by sex.
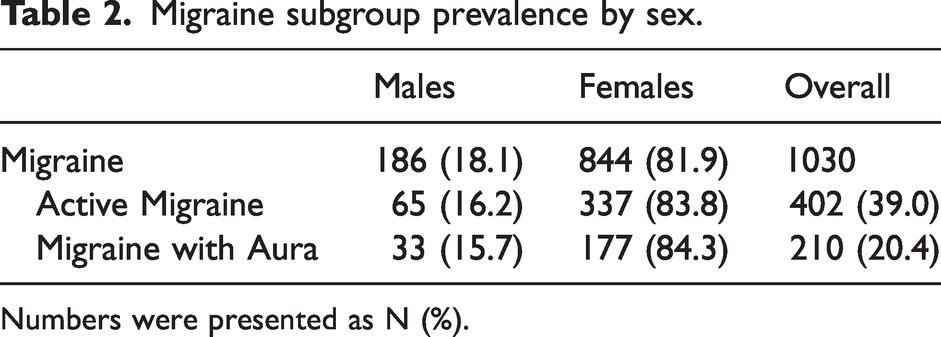
Numbers were presented as N (%).
Lifetime prevalence of migraine by age groups and sex.

Kaplan-Meier estimates showed no significant difference in stroke-free survival probability between individuals with and without migraine (Figure 3). Adjusted Cox regression models indicated a HR of 1.44, (CI: 0.96–2.15, Table 4) for the association between migraine and the risk of stroke. Similarly, adjusted Cox models indicated a HR of 1.50 (CI: 0.97–2.32, Table 4) for the association between migraine and the risk of ischemic stroke. Lastly, we repeated these analyses after combining definite (n = 1030) and probable migraine (n = 290) categories as the exposure. We found smaller effect estimates but results remained not statistically significant (see Supplementary Table S2).
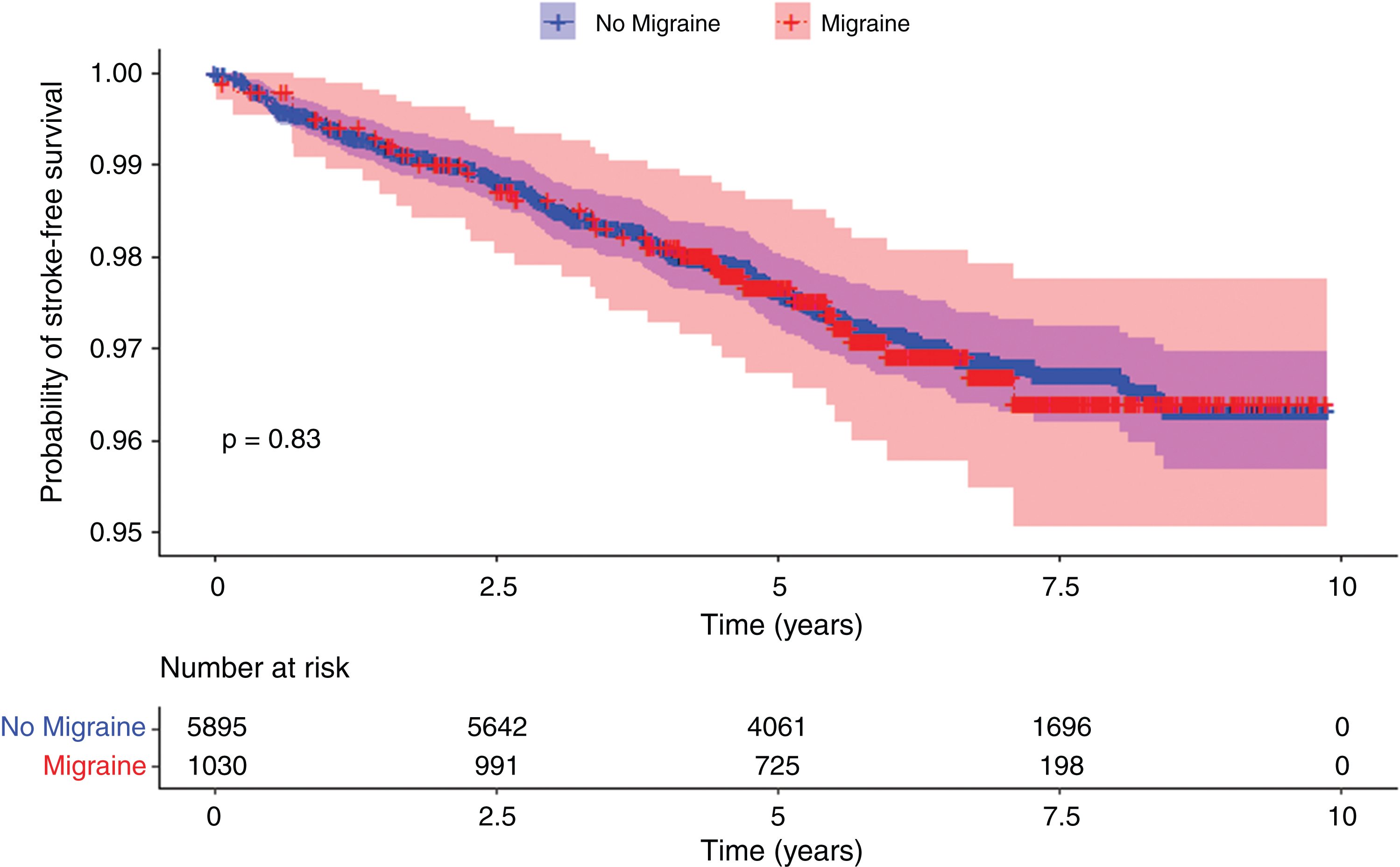
Kaplan-Meier cumulative estimates of the probability of stroke-free survival in participants with migraine or without migraine. Red = participants with migraine. Blue = participants without migraine. Shading represents 95% confidence intervals. p = log-rank test.
Cox regression analyses for the association of migraine with any and ischemic stroke.
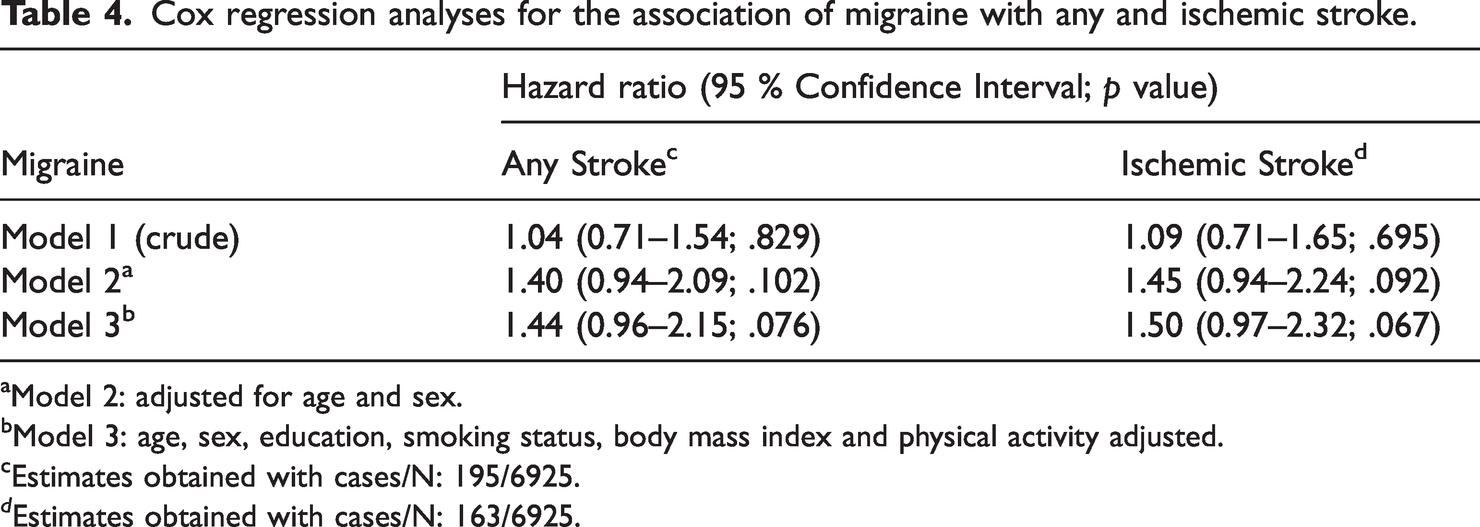
aModel 2: adjusted for age and sex.
bModel 3: age, sex, education, smoking status, body mass index and physical activity adjusted.
cEstimates obtained with cases/N: 195/6925.
d Estimates obtained with cases/N: 163/6925.
Discussion
Our results suggest an association between migraine and the risk of stroke in a middle-aged and elderly population, although this was not statistically significant. Likewise, our data suggested a similar association of migraine and ischemic stroke, albeit again not statistically significant.
Previously, some studies reported an increased stroke risk (3,6,7) in elderly migraine patients, whereas others found no significant association (4,5,8,9). In the studies that reported an overall association, migraine status was ascertained based on recorded diagnostic codes in medical files. Large-scale survey studies indicate that fewer than half of individuals suffering from migraine consult a healthcare professional (31,32). Therefore, studies using diagnosis codes from medical files for migraine ascertainment may not be representative of all migraine patients in the population. In the present study, we also included individuals who were suffering from migraine but had not yet visited a healthcare professional.
In the elderly, a history of migraine may not be a risk factor for stroke (33) except in current smokers (9) and individuals with late-life migraine onset (5). There might be two explanations for these findings. First, it has been suggested that in elderly cohorts, the cardiovascular burden could be too high to detect a difference in the risk of stroke according to migraine status (9). This is in line with another study that reported an association between migraine with aura and ischemic stroke among women only in the lowest Framingham risk score group (34). Second, in the vast majority of migraine patients, symptoms start before the age of 50 (35). Alternatively, migraine may increase the stroke risk exclusively in young people (in a “younger” vascular system) around the symptom onset. This would align with another study in which an association was found between migraine with aura and ischemic stroke only in patients with a late-life (age ≥50) migraine onset (5). Future research should focus on the time period around migraine onset and the resulting processes in the cerebrovascular system.
This study has several strengths. Firstly, our study has a prospective design with a relatively long and virtually complete follow-up. Secondly, we assessed and adjusted our results for a wide range of possible confounders. Thirdly, we used a validated questionnaire for migraine assessment in the general population, increasing the generalizability of our results. Finally, stroke cases were evaluated using a detailed adjudication method, including a consensus meeting and imaging confirmed stroke subtype diagnoses.
There are also some limitations that should be considered when interpreting our results. First, our study could be underpowered due to the relatively low number of stroke events. Second, the criteria used for migraine assessment were slightly modified from the ICHD-2 criteria and we did not have information on headache intensity or headaches other than migraines. Therefore, migraine patients with less than severe headache intensity may have been misclassified as having no migraine, leading to a potential dilution of the strength of the association. However, research shows that headache intensity does not change significantly with older age (36–38). Furthermore, in the literature, it is inconclusive if the potential association between migraine and stroke is modified by headache intensity (33). Therefore, we believe this would not have a prominent effect on our results. Third, migraine with aura is reported to be associated with cardiovascular disease and all-cause mortality (22). Considering the finding that the individuals with migraine were younger on average than the ones without migraine, some of the participants may have died before migraine interview, which may have further diluted the risk estimates. Finally, it would have been interesting to investigate the role of migraine with aura, active migraine and sex on migraine and stroke association separately. Although we measured these variables, event numbers were not sufficient for the subgroup analyses.
Our results warrant the need for future studies to further unravel the possible association between migraine and stroke in the elderly population. Although migraine with aura is an established risk factor for cardiovascular and cerebrovascular events in younger adults (33), little is known about the relative contribution of migraine in the elderly. Specialized clinical guidelines and recommendations are needed for clinicians treating elderly individuals with migraine.
Conclusion
To conclude, we observed an association between migraine and the risk of stroke (overall and ischemic) in the middle-aged and elderly population, but it was not statistically significant.
Public health relevance
Association between migraine and stroke poses a public health burden for the younger population. Whether this is the case for older patients is unknown. In the middle-aged and elderly population, migraine may be associated with a higher risk of overall and ischemic stroke. Future studies are needed to unravel the link between subgroups of migraine and the risk of stroke in the elderly.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221132008 - Supplemental material for Migraine and the risk of stroke in a middle-aged and elderly population: A prospective cohort study
Supplemental material, sj-pdf-1-cep-10.1177_03331024221132008 for Migraine and the risk of stroke in a middle-aged and elderly population: A prospective cohort study by Cevdet Acarsoy, Lana Fani, Linda Al-Hassany, Brian Berghout, Peter J Koudstaal, Antoinette Maassen Van Den Brink, M Kamran Ikram and Daniel Bos in Cephalalgia
Footnotes
Acknowledgements
The authors are grateful to the study participants, the staff from the Rotterdam Study, and the participating general practitioners and pharmacists. Special thanks to Elizabeth A Loehrer and Joyce van den Ende for their contribution to data cleaning.
Author contributions
All authors have made a substantial intellectual contribution to conception and design of the study, acquisition of data (CA, LF, BB, PJK), analysis and interpretation of data (CA, MKI, DB), drafting the article (CA), or drafting a significant portion of the article or figures (CA, LA-H, MKI, DB, AMVDB). All authors approved the final version of the article for publication. All authors had full access to the data in the study and took responsibility for data integrity and accuracy of data analysis.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Antoinette Maassen Van Den Brink received research grants and/or consultation fees from Abbvie/Allergan, Amgen/Novartis, Eli Lilly, Teva and ATI, and was supported by the Dutch Research Council (NWO, Vici Grant 09150181910040).
Ethics statement
The Rotterdam Study has been approved by the Medical Ethics Committee of the Erasmus MC (registration number MEC 02.1015) and by the Dutch Ministry of Health, Welfare and Sport (Population Screening Act WBO, license number 1071272-159521-PG). The Rotterdam Study Personal Registration Data collection is filed with the Erasmus MC Data Protection Officer under registration number EMC1712001. The Rotterdam Study has been entered into the Netherlands National Trial Register (NTR; www.trialregister.nl) and into the WHO International Clinical Trials Registry Platform (ICTRP; ![]() ) under shared catalogue number NL6645/NTR6831. All participants provided written informed consent to participate in the study and to have their information obtained from treating physicians.
) under shared catalogue number NL6645/NTR6831. All participants provided written informed consent to participate in the study and to have their information obtained from treating physicians.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Rotterdam Study is funded by Erasmus Medical Center and Erasmus University, Rotterdam, Netherlands Organization for the Health Research and Development (ZonMw), the Research Institute for Diseases in the Elderly (RIDE), the Ministry of Education, Culture and Science, the Ministry for Health, Welfare and Sports, the European Commission (DG XII), and the Municipality of Rotterdam.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
