Abstract
Aim
Migraine, in particular with aura, has been associated with an increased risk for ischemic stroke and coronary heart disease. The underlying mechanism is unknown. In a cross-sectional case control study we investigated whether an enhanced risk of atherosclerosis in migraineurs explains this increased cardiovascular risk.
Methods
Subjects were participants from the population-based Erasmus Rucphen Family study. Atherosclerosis was assessed in 360 migraineurs (209 without aura and 151 with aura) and 617 subjects without migraine or severe headache. Atherosclerosis was quantified by intima media thickness, pulse wave velocity and ankle-brachial index.
Results
Migraineurs, especially with aura, were found more likely to smoke, have diabetes or a modestly decreased HDL-cholesterol. No differences were found for the atherosclerosis parameters.
Conclusion
In this large population-based study, migraineurs have no increased risk of atherosclerosis. Therefore, enhanced atherosclerosis is an unlikely explanation for the increased cardiovascular risk seen in migraineurs.
Introduction
Migraine is a prevalent neurovascular brain disorder that is characterized by recurrent disabling attacks of headache associated with nausea, vomiting, and photo- and phonophobia (migraine without aura) (MO); in one-third of migraineurs, attacks may be preceded by transient focal neurological aura symptoms (migraine with aura) (MA) (1). Both neuronal as well as vascular mechanisms have been implicated. There is observational, prospective and imaging evidence that migraine increases the risk of ischemic stroke almost two fold (2–4). Patients may also be at increased risk for major cardiovascular disease, including coronary heart disease and peripheral artery disease (2,5,6). This suggests that vascular dysfunction in migraineurs is not restricted to the cerebral vasculature, but is also systemically present.
The underlying mechanism for the association between migraine and ischemic cardiovascular disease is unknown. An adverse cardiovascular risk profile and associated atherosclerosis has been suggested. Some studies found an unfavorable cardiovascular risk profile, in particular for MA patients (5,7,8), while other studies did not find any (9) or only a modest association (10). Framingham Risk Scores for coronary heart disease (FRS-CHD) were elevated in migraineurs in most (5,7,8,11), but not all studies (9,12). None of the studies reported on the Framingham Risk Score for ischemic stroke (FRS-Stroke) in migraine patients, probably because the electrocardiogram (ECG) data that are necessary to calculate this risk score were not available.
The atherosclerotic process, one of the strongest risk factors for stroke and coronary heart disease, can be assessed by means of non-invasive preclinical functional and structural markers of changes in the vessel wall (13). In the present study, we investigated atherosclerosis in a large group of migraine patients from the Erasmus Rucphen Family (ERF) study.
Methods
Study population
We performed a case-control study nested within the population-based ERF study. The ERF study is a family-based cross-sectional study in a genetically isolated community in the Southwest of the Netherlands (14). The study population includes 3465 individuals who were ascertained for genetic-epidemiological studies based on their genealogical background (i.e. not selected on phenotypes of interest). All individuals 18 years or older were invited to participate. For this epidemiological study we did not use genetic or genealogical data, but the genetic and environmental homogeneity of the population can be an advantage. The study was approved by the Medical Ethical Committee of the Erasmus Medical Centre Rotterdam and all participants gave written informed consent.
Migraine patients and controls
Migraineurs were identified using a three-stage screening procedure assessing lifetime occurrence of migraine, which was previously validated in a population-based study (15) and which was based on the second edition of the International Classification of Headache Disorders (ICHD-II) (1). The sensitivity of this case-finding procedure was 0.93 and the specificity was 0.36. The positive predictive value was 0.65 and the negative predictive value was 0.91. Thus the screening procedure picked up many false-positives and missed some true-positives (15). Only patients with “definite” MA and MO are included. Patients with probable migraine are excluded from the study.
Details on the migraine case-finding procedure have been published before (16). In brief, in the first stage, participants were asked to fill out five screening questions on headache and aura symptoms. Then, screen-positives received a detailed questionnaire on headache and aura symptoms. Finally, screen-positives were interviewed by telephone for further clarification of their answers. Subjects who were screen-positive but did not return the extensive headache questionnaire and subjects who could not be screened because they did not (completely) fill out the screening questions were directly contacted by telephone. Telephone interviews were performed by the principal study physician (A.H.S.), who is experienced in diagnosing migraine patients, and by well-trained medical students supervised by A.H.S. A final diagnosis was made only after the telephone interview and in consultation with a neurologist specializing in headache (G.M.T.).
The control group consisted of subjects who 1) did not report severe headache in their lifetime (only headache up to pain severity score ≤4 on a scale from 0 to 10); 2) did not report visual aura symptoms; 3) had never been diagnosed with migraine by a physician; and 4) never used specific antimigraine medication. Thus, subjects with mild headache in combination with visual aura are excluded from the control group. To exclude false-negative control cases, we used an even more conservative definition for non-migraineurs than in our previous population-based study, as from this study we know that about 10% of migraineurs are missed using this screening procedure (15).
Assessment of cardiovascular parameters
Participants (n = 3465) were asked to attend the research center located within the community. Extensive clinical examinations were performed, including the collection of fasting blood samples, anthropometric measurements, cardiovascular assessments, and personal interviews by physicians. Participants were also asked to fill out questionnaires assessing various variables, including symptoms of headache and their level of education. The interview included questions on cigarette smoking status, alcohol consumption, and medical history. Alcohol consumption was measured in units per week and was categorized as <28 consumptions per week and ≥28 consumptions per week. Current smoking was categorized as yes or no. Participants took all prescription medication to the research center, and antihypertensive treatment and the use of oral contraceptives (OAC) was verified by a physician. Height and weight were measured with the participant in light underclothing and body mass index (kg/m2) was computed. An ECG was performed, and each was scored by an experienced cardiologist. Blood pressure was measured twice on the right arm in a sitting position after at least 5 minutes of rest using an automated device (OMRON 711, Automatic IS; Vernon Hills, IL, USA). The average of the two measures was used for the brachial blood pressure in the analyses. Plasma concentrations of triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C) and total cholesterol (TC) were determined according to standard procedures, as described previously (14). Diabetes was defined as the use of blood glucose-lowering medication or sober glucose levels of ≥7 mmol/l or both. Total plasma adiponectin was analyzed with the Human Adiponectin radioimmunoassay (RIA) kit (catalog number: HADP-61HK) of Linco Research (St. Charles, MO, USA). Total plasma C-reactive protein (CRP) was analyzed with the US C-reactive protein enzyme-linked immunosorbent assay (ELISA) (catalog number: DSL-10-42100) of Diagnostic Systems Laboratories Inc. (Webster, TX, USA). All measurements were performed according to the manufacturers’ protocol. Plasma CRP levels showed a high level of kurtosis. Therefore, upper plasma CRP levels exceeding three times the standard deviation of the mean were removed from further analyses.
We used the FRS-CHD (17) and FRS-Stroke (18) to summarize the data. The FRS-CHD estimates the 10-year probability of CHD, defined as myocardial infarction or CHD death. The score assigns points for age, HDL-C, TC, smoking status, and systolic blood pressure stratified by sex and treatment for hypertension. The FRS-Stroke estimates the 10-year probability of stroke. The score assigns points for age, systolic blood pressure, treatment for hypertension, history of diabetes mellitus, smoking status, cardiovascular disease (i.e. history of myocardial infarction, angina pectoris, coronary insufficiency, intermittent claudication or congestive heart failure), and atrial fibrillation and left ventricular hypertrophy on ECG, all stratified by sex. The individual points for both the FRS-CHD and FRS-Stroke scores are summed and converted into 10-year risk percentages.
Functional and structural vascular assessment
The atherosclerotic process can be assessed by means of non-invasive preclinical functional and structural markers of changes in the vessel wall, including intima media thickness (IMT), pulse wave velocity (PWV, measure of arterial stiffness), and ankle-brachial index (ABI) (13). These measures correlate with central (IMT, PWV) or peripheral (ABI) atherosclerosis (13). IMT was measured by ultrasonography with a 7.5-MHz linear array transducer (ATL Ultra-Mark IV; Advanced Technological Laboratories, Bothell, WA, USA) of the left and right common carotid artery. The maximum carotid IMT was determined as the mean of the maximum IMT of near and far wall of both common carotid arteries (14). Carotid-femoral PWV was measured by means of an automatic Complior SP device with subjects in the supine position. The time delay between the rapid upstroke from the base point of simultaneously recorded pulse wave curves in the carotid and the femoral arteries was assessed, and the distance between the carotid and the femoral arteries was measured over the surface of the body with a tape measure. PWV was calculated as the ratio between the distance traveled by the pulse wave and the time delay and expressed in meters per second (14). Ankle blood pressure was measured in both the posterior tibial arteries by using an 8-MHz continuous Doppler probe. Blood pressure was measured twice in the right arm in the sitting position. The ABI was calculated for each leg by dividing the ankle systolic pressure by the mean brachial systolic pressure. The lowest of the two ABI values was used in the analysis. Patients with ABI >1.4 were excluded from the analysis as this indicates non-compressible vessels (19).
Statistical methods
Analyses were performed separately for all migraine, MO and MA. A two-sided p value of alpha <0.05 was considered significant. General characteristics were compared between groups using a Student’s t test for age and a Chi-square test for sex and education. Means for continuous variables and proportions for dichotomous variables were calculated for cardiovascular risk parameters. Means of cardiovascular parameters were compared according to migraine status using analyses of covariance, adjusted for age, sex and education. Mean PWV, IMT and ABI were corrected for age, sex, education, HDL-C and smoking. Proportions were compared by binary logistic regression with the risk factor as the outcome (dependent) variable and migraine status as the independent variable, adjusted for age, sex and education. Odds ratios for having an FRS-CHD or FRS-Stroke >10% were calculated using logistic regression adjusted for age, sex and education. We had 80% power to detect a difference of 0.23 m/s for PWV, 0.03 mm for IMT and 0.016 for ABI. These power calculations are based on the number of participants with data available for that parameter.
Analyses were performed using SPSS version 16.0 (SPSS, Chicago, IL, USA). Significant results were corrected for inbreeding using the SOLAR 2.1.2 software package (Southwest Foundation for Biomedical Research, San Antonio, TX, USA), which is necessary because subjects are part of a genetic isolate. The absolute numbers of cases and controls with diabetes was too small to correct for inbreeding. The coefficient of inbreeding per individual was calculated based on available genealogical information using PEDIG software. The inbreeding coefficient indicates, for each person in ERF, the probability that two alleles at a given locus in an individual are identical by descent and thus is a measure for relatedness. Subjects of the ERF population are the living descendants of 22 couples with at least six children baptized in the community church between 1850 and 1900. Thus, they are part of a large extended pedigree and therefore are more related than individuals from the general population. In isolated populations such as ERF, genetic drift may lead to an increased risk of allele frequencies, which may (falsely) increase associations between traits. Correction for relatedness prevents this and better reflects the association in the general population.
Results
Recruitment of migraine cases and controls
Results on ascertainment of migraine cases and controls have been described elsewhere and are depicted in the flowchart (Figure 1) (18). In total, 3465 subjects participated in the study, from whom we recruited 360 migraine patients (151 MA, 209 MO) and 617 control subjects. Comparing subjects included (n = 977) versus those not included (n = 2488) in the present study showed that included subjects were significantly younger (p < 0.001) and higher educated (p = 0.03) compared with those not included, but there was no difference with regard to gender (p = 0.20).
Flowchart of ascertainment of migraine cases and controls in the Erasmus Rucphen Family study.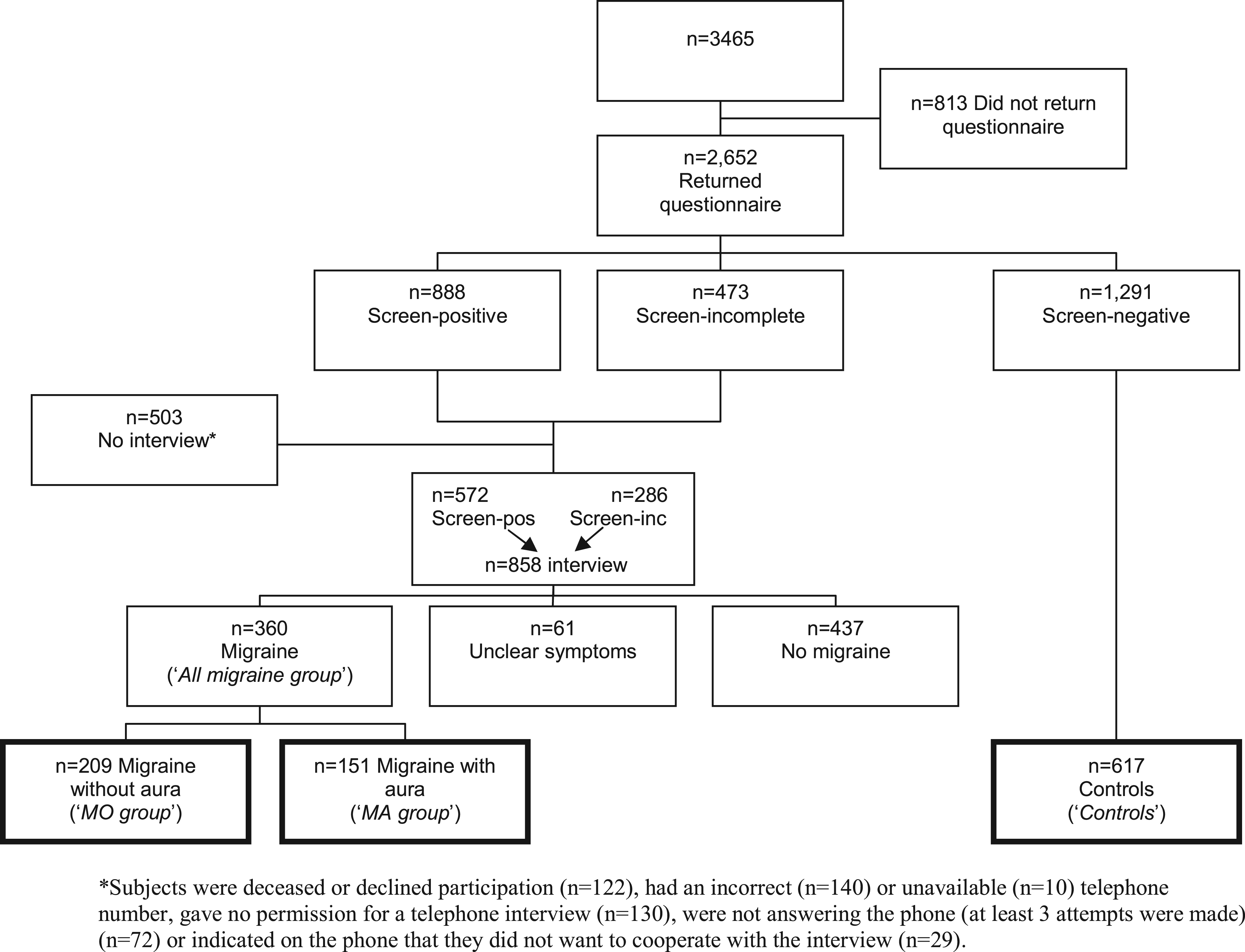
Cardiovascular parameters
Baseline characteristics of the study population by migraine subtype.
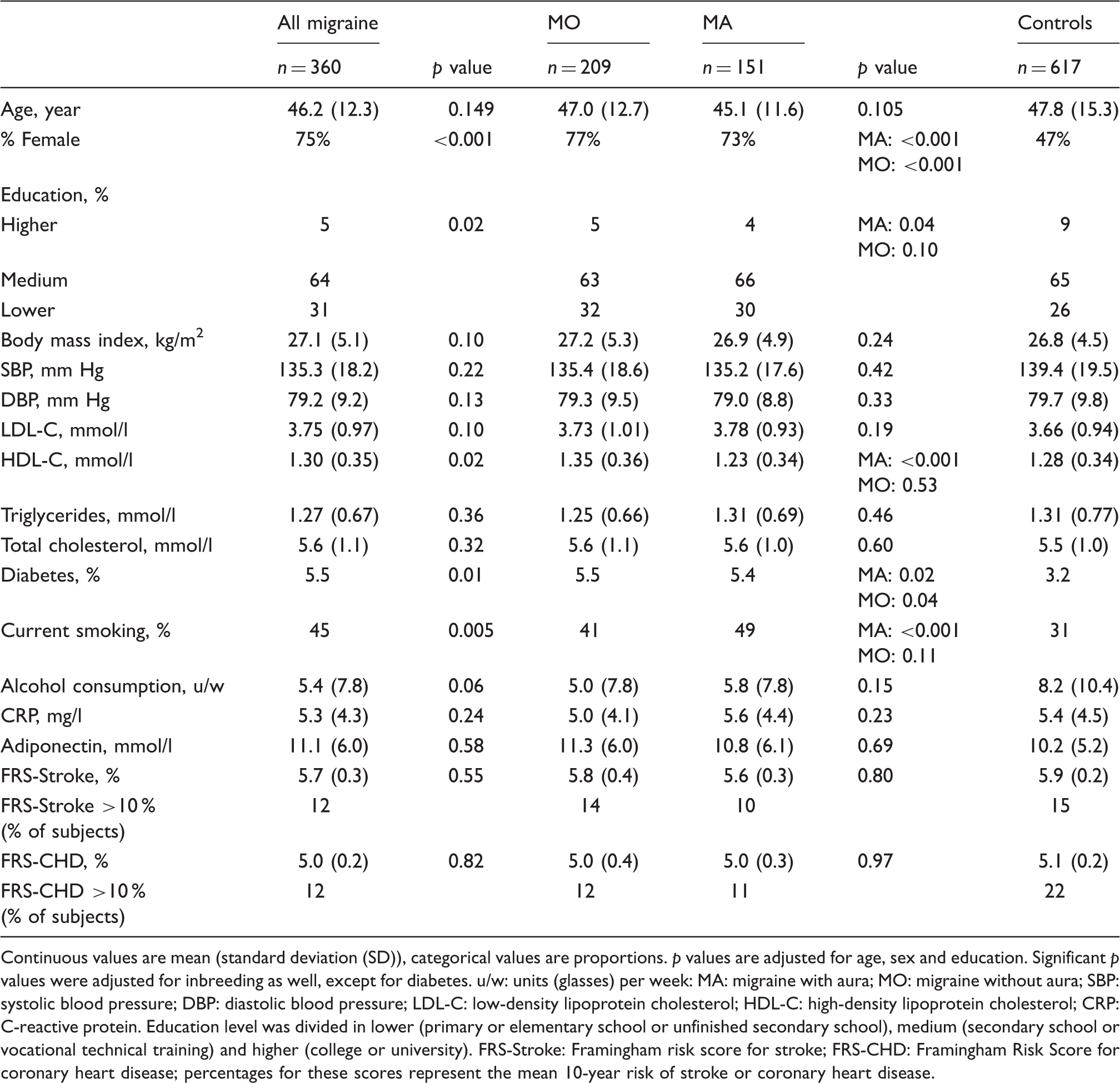
Continuous values are mean (standard deviation (SD)), categorical values are proportions. p values are adjusted for age, sex and education. Significant p values were adjusted for inbreeding as well, except for diabetes. u/w: units (glasses) per week: MA: migraine with aura; MO: migraine without aura; SBP: systolic blood pressure; DBP: diastolic blood pressure; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; CRP: C-reactive protein. Education level was divided in lower (primary or elementary school or unfinished secondary school), medium (secondary school or vocational technical training) and higher (college or university). FRS-Stroke: Framingham risk score for stroke; FRS-CHD: Framingham Risk Score for coronary heart disease; percentages for these scores represent the mean 10-year risk of stroke or coronary heart disease.
Functional and structural measures for atherosclerosis
Functional and structural measures of atherosclerosis in migraine patients versus controls.
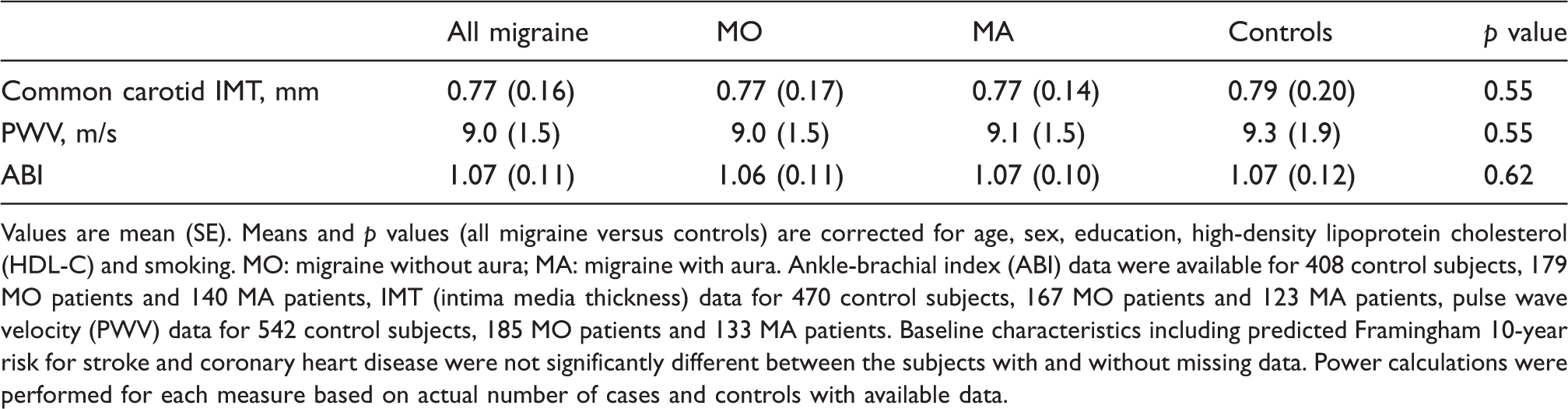
Values are mean (SE). Means and p values (all migraine versus controls) are corrected for age, sex, education, high-density lipoprotein cholesterol (HDL-C) and smoking. MO: migraine without aura; MA: migraine with aura. Ankle-brachial index (ABI) data were available for 408 control subjects, 179 MO patients and 140 MA patients, IMT (intima media thickness) data for 470 control subjects, 167 MO patients and 123 MA patients, pulse wave velocity (PWV) data for 542 control subjects, 185 MO patients and 133 MA patients. Baseline characteristics including predicted Framingham 10-year risk for stroke and coronary heart disease were not significantly different between the subjects with and without missing data. Power calculations were performed for each measure based on actual number of cases and controls with available data.
Discussion
The aim of this study was to investigate whether migraine patients are at increased risk for atherosclerosis. Our main finding is that atherosclerosis, assessed by three complementary noninvasive measures, is no more prevalent in migraineurs than in controls.
Several studies assessed atherosclerosis in migraine patients using comparable functional and structural markers, but with contradictory results. One clinic-based study found a small decrease in ABI in migraine patients compared to controls (20). In line with our findings, another larger clinic-based study found no difference (21). Three studies on PWV found increased values in migraine patients but were clinic based and excluded patients with known cardiovascular risk factors (21–23), which hampers comparison of results with our population-based study, where no selection for migraine or cardiovascular disease risk was made. Seven studies measured IMT in migraine patients with conflicting results (9,23–27). Comparison between these studies is hampered by different case selection methods, such as clinic based (23,25–28) versus population based (9,24), and exclusion of participants with known cardiovascular risk factors (23,25–28) versus correction for these risk factors (9,24). The two largest population-based studies (9,24), with more than 100 migraine patients (unselected with regard to cardiovascular risk factors), found slightly decreased values of IMT, which is in line with our findings.
Compared to previous studies, our study has important strengths. We assessed the largest group of migraine patients thus far, including a large group of MA patients (n ≥ 120). Three complementary measurements that quantified both central and peripheral atherosclerosis were used. Migraine diagnoses were made after a telephone interview in consultation with a neurologist and according to criteria of the International Headache Society. Our sample was unselected for a particular disease phenotype, and included a broad age range of adults (18–87 years), thus preventing ascertainment bias. Participants were from a population-based study with a relatively homogenous genetic and environmental background. Finally, data on a large number of cardiovascular risk factors were available and allowed us to calculate the FRS-CHD as well as the FRS-Stroke.
Our study has some limitations. First, we were unable to assess directly the mediating effect of atherosclerosis in migraineurs with a cardiovascular event because of the size of the study population (i.e. ≤10 migraineurs had a myocardial infarction or stroke). Second, data on functional and structural measures for atherosclerosis were missing for a substantial proportion of cases and controls. We do not think this affected our outcome since power calculations were based on the available number of participants and showed a reasonable power to detect differences in mean values. Moreover, baseline characteristics were not significantly different between subjects with and without missing data, making selection bias unlikely. Third, we were unable to assess the effect of migraine attack frequency. This would have been interesting as the association of migraine with cardiovascular disease varies by attack frequency (29). Fourth, IMT and ABI are indicative of clinical atherosclerotic disease and perhaps limit conclusions on variability in the normal range. However, we have also included a measure that measures subclinical arterial stiffness (PWV). Last, as our study population has a homogeneous genetic and environmental background, future studies are needed to assess to what extent our findings can be replicated in other populations of migraine patients. Although our study has a larger percentage of MA, we feel this is not a disadvantage of our study as the risk for cardiovascular events is highest among MA cases.
We assessed the FRS-Stroke in migraineurs and showed no increased odds for a higher score, which suggests that traditional cardiovascular stroke predictors are not relevant in the migraine-stroke association. This is in line with our data on atherosclerosis parameters. In addition, several previous observations support this conclusion. First, in most prospective cohort studies the increased risk of ischemic stroke appeared independent from traditional cardiovascular risk factors (30) with the exception of use of OAC and smoking in women. Second, population-based magnetic resonance imaging (MRI) studies showed no difference in cardiovascular risk factors between migraineurs with and without posterior circulation territory infarct-like lesions and white matter lesions (31,32). Third, the Women’s Health Study showed an increased risk for myocardial infarction in migraine patients with a high FRS-CHD, while increased stroke risk was observed among migraine patients with a low FRS-CHD (11). This supports the idea that the mechanisms underlying the migraine-stroke association are different from atherosclerosis.
Based on these data, it seems unlikely that the higher risk of cerebro- and cardiovascular disease in migraineurs is mediated by atherosclerosis, although it might be possible that the process of atherosclerosis plays a role on a subclinical level with endothelial dysfunction as a presumed early marker (33). Larger, preferably prospective studies are necessary to further clarify the role of atherosclerosis in incident vascular events in migraineurs. Potential other explanations for the migraine-stroke relationship include the association of migraine with specific etiologies of stroke (i.e. micro emboli caused by patent foramen ovale), the association with a pro-inflammatory, pro-coagulatory state, the use of vasoconstrictive drugs in migraineurs or the (genetic) lowered threshold for spreading depression leading to either MA or ischemia (34). The latter hypothesis is supported by recent data in migraine (familial hemiplegic migraine (FHM)1 CaV2.1) transgenic mice that, after transient ischemia, developed earlier onset of anoxic depolarization and more frequent peri-infarct depolarization resulting in larger infarcts and worse neurological outcomes compared to wild-type mice (35). These data suggest that enhanced susceptibility to ischemic depolarization akin to spreading depression predisposes migraineurs to infarction during mild ischemic events, thereby increasing the stroke risk. In humans, MA is associated with strokes with good functional outcome (36), which seems contradictory to these mouse data. However, disability might be related to the size and type of the infarcts rather than of their specific underlying mechanism and thus does not rule out susceptibility to spreading depression as a causal factor in humans as well. More translational studies are needed to provide more insight in the migraine-stroke relationship and to develop prophylactic treatment strategies to prevent cerebro- and cardiovascular events in migraine patients.
Footnotes
Clinical implications
In this large population-based study migraineurs have no increased risk of atherosclerosis. Therefore enhanced atherosclerosis is an unlikely explanation for the increased cardiovascular risk seen in migraineurs.
Funding
This work was supported by grants from the Netherlands Organization for Scientific Research (NWO) (903-52-291, M.D.F. and R.R.F.; VICI 918.56.602, M.D.F.; 907-00-217, G.M.T.; 920-03-473, A.H.S.; VIDI 91711319, G.M.T.); the European Community (EC) (EUROHEAD, LSHM-CT-2004-504837, M.D.F.); and the Centre for Medical Systems Biology (CMSB) in the framework of the Netherlands Genomics Initiative (NGI) (M.D.F., R.R.F., and A.M.J.M.v.d.M). They had no role in the design or conduct of the study.
Conflict of interest
A.H.S. reports independent research support from NWO. A.C.J.W.J. serves on the editorial boards of Plos ONE, Medical Decision Making, and Public Health Genomics. M.D.F. reports grants and consultancy/industry support from Almirall, Coherex, Colucid, Eisai, GlaxoSmithKline, Linde, MAP, Medtronic, Menarini, Merck, Minster, Pfizer, and St Jude, and independent support from the Netherlands Organization for Scientific Research (NWO). C.M.v.D. reports no disclosures. G.M.T. reports grants and consultancy/industry support from Merck, Janssen-Cilag, and Almirall, and independent support from NWO. C.M.W., Y.S.A., B.A.O., R.R.F., and A.M.J.M.v.d.M. declare no conflict of interest.
