Abstract
Objectives
It is unclear whether the electrophysiological effects of erenumab, a monoclonal antibody against the calcitonin gene-related peptide receptor, occur only at the periphery of the trigeminal system or centrally and at the cortical level.
Methods
We prospectively enrolled 20 patients with migraine who had failed at least two preventative treatments. We measured the nociceptive blink reflex and non-noxious somatosensory evoked potentials in all participants. The area under the curve and habituation of the second polysynaptic nociceptive blink reflex component (R2) as well as the amplitude and habituation of somatosensory evoked potentials N20-P25 were measured. Electrophysiological data were collected at baseline (T0), 28 days (T1), and 56 days (T2) before each injection of erenumab (70 mg).
Results
Erenumab reduced the patients’ mean monthly headache days, headache intensity, and acute medication intake considerably at T1 and T2 (all p < 0.05). The nociceptive blink reflex area under the curve was considerably lower at T1 and T2 than at baseline without changing the habituation slope. At T2, there was a significant increase in the delayed somatosensory evoked potentials amplitude reduction (habituation) but not in the initial cortical activation.
Conclusion
Our findings showed that erenumab, in addition to its well-known peripheral effects, can induce central effects earlier in the brainstem and later in the cortex. We cannot rule out whether these results are due to a direct effect of erenumab on the central nervous system or an indirect effect secondary to peripheral drug modulation.
Introduction
Calcitonin gene-related peptide (CGRP) is a vasoactive neuropeptide densely expressed in the central and peripheral nervous systems. The role played by CGRP and its receptor is complex and involves the modulation of different functions, including motion, secretion, olfaction, and nociception (1). Given the presence of CGRP in secondary trigeminal neurons in the brainstem, trigeminal ganglia, and peripheral sensory nerve terminals of the trigeminovascular system around cerebral and extracerebral blood vessels, it has been suggested that the CGRP pathway plays a crucial role in migraine attack initiation (2,3).
Anti-CGRP monoclonal antibodies (mAbs) have recently become accessible as a result of animal model research and human trials (4). Together with triptans and gepants, these biological treatments precisely target migraine pathogenesis. Erenumab is the only anti-CGRP receptor antibody currently available, whereas others operate against the molecule itself (5). Because mAbs are large molecules, they are unable to cross the blood-brain barrier in significant amounts. Nonetheless, some researchers have characterized the central effects of monoclonal antibodies, showing deactivation of several central areas such as the hypothalamus through the use of neuroimaging (6), as well as desensitization of pain-related cortical potentials (7), the nociceptive brainstem (8), and spinal (9) reflexes using electrophysiological techniques.
The purpose of this study was to determine whether the neurophysiological effects of preventative therapy with erenumab occur at the cortical and trigeminal system levels. We attempted to address this question by testing a group of patients with episodic or chronic migraine by recording their nociceptive blink reflexes (nBRs), a technique for examining the integrity of the caudal trigeminal nucleus (10), and somatosensory cortical evoked potentials (SSEP) (11) before then 28 and 56 days after erenumab (70 mg, sc) treatment.
Based on the aforementioned studies and previous data showing rapid patient responses to these medications, we hypothesized that erenumab has an electrophysiological effect as early as the first month of treatment, at both the brainstem and cortical levels, and in parallel with a reduction in headache days.
Material and methods
This observational study was conducted between February and December 2020. At this time, mAbs were not yet reimbursed by the National Health Service, but these could be prescribed by selected headache centres that purchased the drug at a nominal cost. We prospectively recruited 22 patients with episodic migraine (MO, N = 5) or chronic migraine (CM, N = 17) who presented to our headache centre. After the initial screening, two patients decided not to participate in the recording sessions; therefore, the full study analysis was performed on 20 patients (15 CM and 5 MO) (Figure 1). Before the recording session, the patients were instructed to fill out a paper headache diary for at least one month. The clinical characteristics collected were history of illness (years), number of days with headache (migraine-like or tension-type-like) (N/month), duration of attacks (h/month), severity of attacks (0–3 visual analogue scale), total number of acute medications taken per month (N/month), number of days with acute medication intake (N/month), and number of days free since the last headache (N) (Table 1). We recruited only patients who did not respond to at least two prophylactic treatments for migraine (episodic and chronic). None of the enrolled patients had undergone prophylactic botulinum toxin treatment. All patients were registered outside the days of migraine. We included only patients who had not received migraine prophylaxis for at least two months. Patients were instructed to continue completing the diary throughout the study period. All patients were informed of the study purpose and provided informed consent. The study protocol (Studio 23.20) was approved by the Lazio 2 Ethics Committee.

STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) enrolment flow chart of study participants.
Neurophysiological procedures
During the recording session, study participants underwent recording of their nociceptive blink reflexes and non-painful somatosensory evoked potentials in random order, using a Digitimer D360 amplifier (band-pass 0.05–2000 Hz, Gain 1000) and CED™ power 1401 digital-to-analog converter (Cambridge Electronic Design Ltd, Cambridge, UK). The study participants were seated on an armchair in a quiet room and instructed to relax with their eyes open. During the entire stimulation session, we regularly checked the participants’ levels of attention and vigilance. The recording sessions were all carried out in the afternoon from 2 pm to 6 pm by two experienced neurophysiologists (FC and GS). Recordings were made at time 0 (T0), 28 days (T1), and 56 days (T2) before each new erenumab administration.
Nociceptive blink reflex (nBR)
Recordings of nBRs were performed following a previously published method (10). Briefly, using percutaneous stimulation of the supraorbital branch of the right trigeminal nerve using a concentric stimulation electrode (12), we tested the sensory (ST) and pain (PT) thresholds. Recording electrodes were placed infraorbitally on the orbicularis muscle of the eye and latero-orbitally (reference) on both sides, with the ground on the left arm. Subsequently, we stimulated the patients by sending a train of three pulses, each with a duration of 0.1 ms, with an intra-pulse interval of 5 ms at a fixed intensity of 1.2 × PT. The interstimulus interval was 30–35 seconds pseudorandom. For each participant, we recorded two blocks of six electromyographic traces of 150 ms duration with an inter-pulse interval of 2 min. The first EMG trace of each block was eliminated to avoid contamination with the startle reflex, and then, five traces per block were averaged. An examiner, blinded to the patients' diagnosis, measured the area under the curve (AUC; µV × ms) of the R2 component of each block, calculated between two cursors positioned between 27 and 87 ms. The degree of habituation of the R2 component was defined as the slope of the linear regression line between the AUC of the first and second block recordings.
Low frequency somatosensory evoked potentials (LF-SSEP)
SSEPs were recorded following previously published methods (11,13). Briefly, recording electrodes were placed at the right Erb point (with reference to the left Erb), at the level of C5 (with reference to Fz), and at the level of the contralateral parietal cortex (C3′, with reference to Fz), with the ground electrode placed on the right arm. The evoked electroencephalographic (EEG) signal was acquired after median nerve stimulation of the right wrist (1.2 × motor threshold, 4.4 Hz repetition rate). For each participant, we acquired 500 traces of 50 milliseconds duration (5000 Hz sampling rate). These were analysed offline and, after automatically removing artifacts, we measured the latencies and amplitudes of the N9 (on Erb), N13 (at C5), and parietal components N20, P25 and N33. Finally, we calculated the degree of habituation by dividing the first 300 non-artifactual traces into three blocks of 100 sweeps and calculating the linear regression line between the N20-P25 amplitudes of the first and second blocks of acquisition.
Somatosensory high frequency oscillations (HFOs)
In accordance with a method published elsewhere (13), after removing the stimulus artifacts, we applied a digital band-pass filter between 450 and 750 Hz (Barlett–Hanning window, 51 coefficients) to the broad-band-averaged signal (300 sweeps). On the offline-filtered signal, we calculated the latency and amplitude of the maximum peak of the oscillation burst preceding (early HFO) and following (late HFO) the low-band N20 peak. According to pharmacological modulation and source localization studies, early HFO reflects thalamocortical activity, and late-HFO primarily reflects cortical activity (14).
Statistical analysis
Sample size calculation was not based on formal statistics but on previous literature (8,15).
Statistical analysis was performed using the SPSS statistical software for Windows, version 26.0. The normal distribution of all data was tested using the Anderson–Darling and/or Kolmogorov–Smirnov tests. Changes in electrophysiological parameters of nBR and SSEP at T1 and T2 after erenumab administration were analyzed using ANOVA and Tukey’s multiple comparisons test or Friedman’s nonparametric test using the Dunn-Bonferroni multiple comparisons test (16).
To test the clinical efficacy of erenumab, we calculated the percentage change in the number of days with headache per month, patients’ severity of headache, and acute medications taken monthly at T1 and T2 compared with T0.
Depending on whether the variables were normally distributed, Pearson’s or Spearman’s correlation tests were performed for the changes in clinical parameters and percentage changes in R2 nBR homolateral and contralateral AUC, as well as slope and SSEP amplitude slope. Statistical significance was set at p ≤ 0.05.
Results
None of the participants dropped out or experienced any side effects related to the drug or its administration.
Clinical features
Demographic and clinical characteristics of enrolled patients.

h: hours; N: number.
Descriptive statistics for clinical variables.

Median and interquartile range (25%–75%); inferential statistics based on ANOVA and Tukey’s method; *Friedman’s tests S, p values.
N: number.

Graphs showing the significant reduction in the number of days with headache (a), the number of acute medications consumed in one month (b), and the severity of headache and (c) before each new administration of erenumab 70 mg. Data are shown as mean ± standard deviation.
Nociception-specific blink reflex (nBR)
Friedman’s test did not reveal a repetition effect for the nBR pain threshold (chi-square = 2.563, p = 0.712). We also did not detect a repeat effect for R2 nBR latency (chi-square = 2.056, p = 0.358). In contrast, Friedman's test revealed a significant repeat effect for the AUC of the first nBR block homolaterally (chi-square = 8.44, p = 0.015), but not contralaterally (chi-square = 5.06, p = 0.08) to the stimulus side (Table 3, Figure 3 and 4). Post hoc tests revealed that the homolateral AUC was significantly reduced at T1 (p = 0.013 vs. T0), and it maintained this reduction at T2 (p = 0.013 vs. T0). We did not detect significant changes in the level of nBR habituation either homolaterally (F = 0.52, p = 0.595) or contralaterally (F = 1.98, p = 0.148) to the stimulus side (Table 3).
Descriptive statistics for nociceptive blink reflex (nBR) parameters.

Median and interquartile range (25%–75%); inferential statistics based on ANOVA and Tukey’s method; *Friedman’s tests S, p values.
AUC: area under the curve; mA: milliAmpere; ms: milliseconds; nBR: nociceptive Blink Reflex.

Area under the curve (AUC) of the R2 component of the nociceptive blink reflex (nBR) over 2 blocks of five sweeps homolaterally at the site of stimulation before (T0) and at 28 and 56 days before each new administration of erenumab 70 mg.
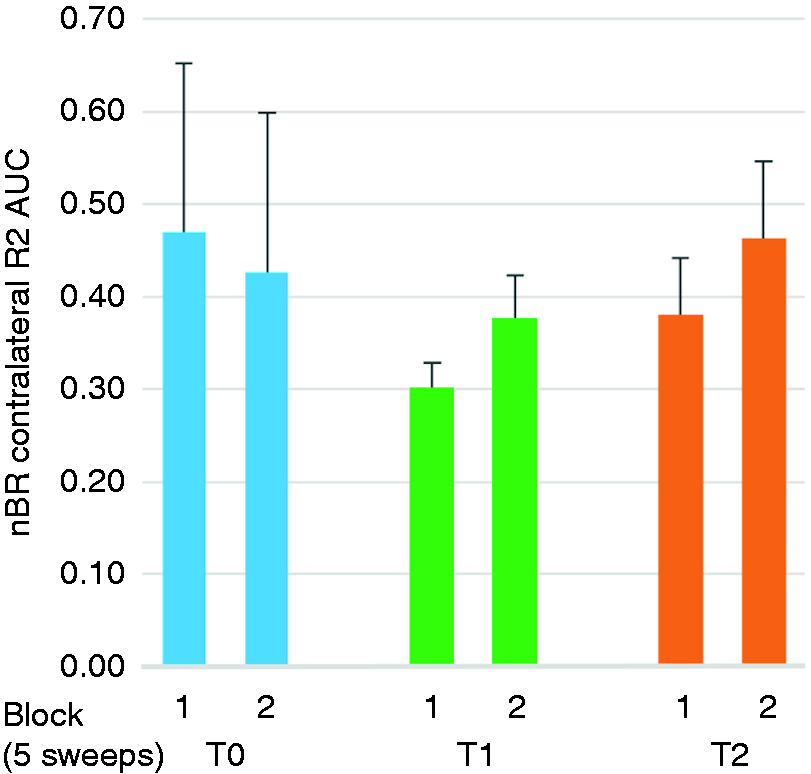
Area under the curve (AUC) of the R2 component of the nociceptive blink reflex (nBR) over 2 blocks of five sweeps contralaterally at the site of stimulation before (T0) and at 28 and 56 days before each new administration of erenumab 70 mg.
Somatosensory low and high frequency evoked potentials
Friedman’s test did not reveal a repetition effect for the SSEP sensory threshold (chi-square = 3.102, p = 0.212). We did not detect a significant repetition effect for any of the SSEP parameters considered (Tables 4 and 5), except for the level of habituation (F = 4.57, p = 0.015). Post-hoc tests revealed that, compared to T0, SSEP habituation was significantly more pronounced at T2 (post-hoc test vs. T0, p = 0.011, Figure 5).
Descriptive statistics for somatosensory evoked potentials parameters.

Median and interquartile range (25%–75%); inferential statistics based on ANOVA and Tukey’s method.
mA: milliAmpere; ms: milliseconds; μV: microVolt; SSEPs: somatosensory evoked potentials; ST: sensory threshold.

Amplitude of the cortical N20-P25 component of the somatosensory evoked potential (SSEP) along three blocks of 100 sweeps before (T0) and at 28 and 56 days before each new administration of erenumab 70 mg. The black solid line indicates the slope of the regression line (habituation) calculated between the three blocks of stimuli.
Descriptive statistics for somatosensory high frequency oscillations parameters.

Median and interquartile range (25%–75%); inferential statistics based on ANOVA and Tukey’s method; *Friedman’s tests S, p values.
HFO: high frequency oscillations; ms: milliseconds; μV: microVolt.
Correlation analyses
The percentage changes in neurophysiological variables calculated at T1 and T2 compared to T0 did not correlate with the percentage reduction in the number of days with headache/month, VAS, and acute medication consumption calculated at T1 and T2 compared to T0 (Table 6, Supplementary Figure 1 and 2).
Correlation analyses between the percentage changes in neurophysiological variables calculated at T1 and T2 compared to T0 with the percentage reduction in the number of days with headache/month, VAS, and acute medications consumption calculated at T1 and T2 compared to T0. We did not find statistically significant correlations.

The table shows Pearson's R's and p's (* = Spearman Rho and p).
AUC: area under the curve; nBR: nociceptive Blink Reflex; SSEP: somatosensory evoked potentials; VAS: visual analogue scale.
Discussion
Our findings show that erenumab, a monoclonal antibody against the CGRP receptor, can alter both pain-related trigeminal brainstem activity, as measured by nBR, and cortical activity obtained in response to non-painful SSEPs. However, these effects occur at different times in migraine patients who have failed to respond to at least two prophylactic treatments. Indeed, there was a significant decrease in the nBR AUC, which evaluated the reactivity of the spinal trigeminal nucleus as early as one month following erenumab delivery. SSEPs, on the other hand, responded differently, changing considerably only at T2 in the form of increased N20-P25 SSEP habituation. According to our findings, there was no link between changes in clinical and neurophysiological variables.
Although monoclonal antibodies against CGRP and its receptor were the first explicitly-developed preventative medicines for episodic and chronic migraine, how and where they operate to achieve this goal is still being debated. Similarly, the long-term effects of these biological therapies on the neurological system remain unknown. Due to their high molecular weight, they have a minimal chance of crossing the blood-brain barrier (BBB), leading to the conclusion that the peripheral sensory terminals of the trigeminovascular system are their preferred targets (1). In rats with an intact BBB, fremanezumab, a mAb against the CGRP molecule, was detected in the dura, sensory, and autonomic trigeminal ganglia, but not in the spinal trigeminal nucleus, thalamus, hypothalamus, or cortex. Fremanezumab was detected in the cortex only in rats with a compromised BBB, especially surrounding the compromised BBB site (17). In addition, radiolabelled galcanezumab, another mAb against the CGRP molecule, was reported to accumulate (although in minimal amounts) within the brain parenchyma (hypothalamus, prefrontal cortex, and cerebellum) and in the peripheral tissue (dura mater, spleen, and trigeminal ganglia) (18). Melo–Carrillo et al., (19) in a study where they electrically and mechanically stimulated the exposed cranial dura and mechanically stimulated the facial skin and cornea, showed decreased activation of high threshold trigeminovascular neurons by dural afferents but not activation of wide dynamic range (WDR) neurons in the skin/cornea of male, but not female, rats up to 4 h after a single intravenous administration of fremanezumab. Nonetheless, in cats, other authors have shown reduced superior sagittal sinus stimulation-induced firing of central trigeminovascular neurons that receive WDR or nociceptive specific input from cutaneous receptive areas on the face up to 30 min after local and intravenous administration of the CGRP receptor antagonist BIBN4096BS (20).
Considering the differences in the stimulation paradigm (stimulation of the superior sagittal sinus or dural afferents) and the different time observation window (up to 4 h after mAb administration) in animal models compared to in our human study, we can hypothesize that persistent and sustained blockade of the CGRP receptor or molecule at the level of peripheral sensory fibres of the trigeminovascular system and its ganglia may prevent impulse transmission to second-order brainstem trigeminal neurons. This prevents sensitization of the trigeminovascular system while also preventing excitatory inputs to third-order trigeminal neurons in the thalamus. This indirect mechanism could explain the early inactivation of the caudal trigeminal nucleus observed during nBR recordings. Nonetheless, we hypothesize that the lack of effect on the thalamocortical early-HFO and the improved habituation to LF-SSEP we also observed may be explained by a similar thalamic inactivation mechanism. In fact, experimental and animal models have shown that spinothalamic-projecting neurons have branches ending in both ventroposterolateral and ventroposteromedial nuclei (21–23), the latter being the site where the trigeminothalamic tract converges. In turn, these thalamic nuclei send inputs to the parietal cortex, processing information from the head (24). The lack of correlation between trigeminal and parietal central electrophysiological responses and clinical improvement in terms of migraine clinical features suggests that the clinical efficacy of monoclonal antibodies is mediated exclusively through peripheral inactivation of the trigeminovascular sensory system. However, this discovery implies that the central mechanisms underlying migraine recurrence are only mitigated by the peripheral impact of mAbs, which would account for the resumption of attacks following mAb withdrawal (25).
While the levels of mAbs that crossed the BBB and were detected in the trigeminal ganglion, hypothalamus, and cortex were relatively low in animal models (18), we cannot rule out the possibility that a central effect contributed to the neurophysiological changes observed. Neurophysiological and neuroimaging studies support this interpretation. As early as one week after erenumab 70 mg administration, de Tommaso et al. (7) observed a decrease in the amplitude of the N1 and N2 components and a slight tendency to increase the N1 habituation of the cortical laser-evoked potential obtained from right and left supraorbital stimulation. Other investigators tested the nociceptive avoidance reflex, a response associated with central spinal sensitization levels, and observed spinal desensitization, as evidenced by an increase in the reflex threshold and temporal summation three months after the first administration of erenumab 70 mg, specifically in patients who responded with at least a 30% reduction in migraine days in the preceding month (9). Additionally, Thiele et al. (8) measured BR using standard surface electrodes in a group of patients with predominantly episodic migraine before and after three months of treatment with monoclonal antibodies against CGRP or its receptor. These authors, in agreement with our findings, also detected a decrease in the ipsilateral R2 AUC following mAb administration, but in contrast to our findings, they also observed a decrease in the R2 AUC and an increase in the R2 habituation coefficient contralateral to the stimulus location. The disparity in the reported results could be explained by the fact that the two studies used different experimental protocols and had clinically distinct patient populations. Indeed, we calculated habituation as the slope of the regression line of the AUC R2 between two blocks of five stimuli with a 35-second interblock interval, rather than between the six sweeps within each of the 10 blocks of six stimuli, as Thiele et al. did. Additionally, we enrolled a greater proportion of patients with chronic migraine compared to episodic migraine.
In a functional MRI study of trigeminal nociceptive stimulation, Ziegeler and colleagues (6) observed a reduction in the activation of the thalamus contralateral to the site of stimulation, the medial temporal gyrus, the right lingual gyrus, the left operculum, and the cerebellum two weeks following the first administration of erenumab 70 mg. Contrary to the results of this study, we did not observe any effect of the drug on thalamocortical activity, most likely because of the distinct stimulus employed in our case, which was not painful.
Considering the clinical studies discussed above, we cannot determine whether the central effects of erenumab, as measured by nBR and SSEPs, are the result of a direct effect on the central nervous system or of an indirect effect secondary to peripheral drug modulation. Intuitively, the sequential recruitment of central structures, with the spinal trigeminal nucleus recruited first and in which the peripheral fibres are more directly connected, followed by the parietal cortex, suggests a long-term neuromodulatory effect on the interconnected network responsible for neurosensory information processing. Furthermore, because we did not observe a direct correlation between drug-induced electrophysiological changes and clinical changes, we believe that the latter were not a result of a reduction in the frequency of headache pain. Finally, because a significant decrease in hypothalamic activation, a so-called migraine generator, was observed only in patients responding to erenumab (6), we cannot rule out an indirect modulatory effect of the hypothalamus on functionally connected brain structures, such as the caudal trigeminal nucleus (26) and the parietal cortex (27).
As in any previous study, our study has some limitations. The first is the absence of a placebo comparison, which makes it impossible to rule out the possibility that the progressive increase in habituation observed with repeated drug administration is due to a learning phenomenon known as “potentiation of habituation” (28). However, there are compelling grounds against this, including the lengthy interval between sessions (28 days) and lack of a similar effect on nBR habituation. Another potential weakness of the study is that all recruited patients had an excellent response to treatment, precluding us from examining whether neurophysiological patterns differ according to different pharmacological responses. Finally, the relatively small sample size and inclusion of episodic and chronic migraine patients in a single group could be another possible limitation of the study. Future studies with adequate sample sizes should investigate whether the electrophysiological effects of mAbs are different in patients with episodic and chronic migraine. Further studies are needed to verify whether the electrophysiological effects are more pronounced at the 140 mg dose of erenumab, as has been observed clinically, especially in episodic migraine (5,29).
Conclusions
In conclusion, we discovered that erenumab can elicit an electrophysiologically-detectable effect in the caudal trigeminal nucleus and parietal cortex. In this context, it would be interesting to investigate whether the neurophysiological effects persist over a longer treatment interval and after erenumab discontinuation. Although preliminary, these findings may serve as a basis for a larger investigation using different types of monoclonal antibodies to gain a better understanding of their mechanisms of action.
Article Highlights
After one month of treatment, erenumab decreased the area under the curve of the nociceptive blink reflex. After two months of treatment, an increase in the degree of habituation of cortical somatosensory evoked potentials was observed. These findings suggest that erenumab may have a central effect.
Supplemental Material
sj-jpg-1-cep-10.1177_03331024221103811 - Supplemental material for The monoclonal CGRP-receptor blocking antibody erenumab has different effects on brainstem and cortical sensory-evoked responses
Supplemental material, sj-jpg-1-cep-10.1177_03331024221103811 for The monoclonal CGRP-receptor blocking antibody erenumab has different effects on brainstem and cortical sensory-evoked responses by Francesco Casillo, Gabriele Sebastianelli, Antonio Di Renzo, Ettore Cioffi, Vincenzo Parisi, Cherubino Di Lorenzo, Mariano Serrao and Gianluca Coppola in Cephalalgia
Supplemental Material
sj-jpg-2-cep-10.1177_03331024221103811 - Supplemental material for The monoclonal CGRP-receptor blocking antibody erenumab has different effects on brainstem and cortical sensory-evoked responses
Supplemental material, sj-jpg-2-cep-10.1177_03331024221103811 for The monoclonal CGRP-receptor blocking antibody erenumab has different effects on brainstem and cortical sensory-evoked responses by Francesco Casillo, Gabriele Sebastianelli, Antonio Di Renzo, Ettore Cioffi, Vincenzo Parisi, Cherubino Di Lorenzo, Mariano Serrao and Gianluca Coppola in Cephalalgia
Footnotes
Acknowledgements
The contribution of the GB Bietti Foundation to this paper was supported by the Italian Ministry of Health and Fondazione Roma.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
