Abstract
Introduction
The development of novel pharmacotherapies for the acute treatment of migraine has been challenging due in large part to a poor understanding of the pathogenesis of this disease. While there has been much debate over the years about whether migraine is neurogenic or vascular in origin, the current standard pharmacotherapies were developed when migraine was considered to be a vascular, rather than a neurological, disorder. A prominent concept has been that vasodilatation of cranial blood vessels is the genesis of the pain of migraine (1). Hence, it was reasoned that constriction of dilated cerebral blood vessels would alleviate migraine; thus, the strategy was developed to search for molecules with potent cerebrovasoconstrictor properties in animal experiments. This culminated in the development of sumatriptan, which was later defined as a serotonin (5-HT) 5-HT1B/1D receptor agonist (2–4). The vasoconstrictor activity is believed to be conferred via activation of the 5-HT1B receptor located on vascular smooth muscle (5). Subsequently, additional indole-based molecules, selected for their activity as 5-HT1B/1D receptor agonists, progressed to the market for the acute (abortive) treatment of migraine, and these are collectively referred to as the triptan class of antimigraine drugs.
The triptans are currently the class of choice for abortive migraine pharmacotherapy, but there are concerns about potential coronary vasoconstriction/vasospasm and a high incidence of symptoms of chest discomfort. Such effects and the fact that these compounds are contra-indicated for patients with symptoms of coronary artery disease have limited the patient population for the triptans and have underscored the need for novel migraine therapies without cardiovascular liabilities (6).
An alternative to the vascular hypothesis of migraine is the neurogenic hypothesis, whereby dilatation of cerebral and extracerebral blood vessels is considered a secondary mechanism in migraine pathogenesis. The primary cause of the pain is hypothesized to be the result of neural events that increase transmission of noxious sensory information through the trigeminal system (for reviews, see Goadsby et al. (6) and Charles (7)). Targeting this system provides the possibility of alleviating migraine pain without vasoconstriction.
In addition to their direct vasoconstrictor activity, triptans may also alleviate migraine through neural mechanisms, such as inhibition of neurotransmitter release, including calcitonin gene-related peptide (CGRP), from trigeminal neurons via presynaptic 5-HT receptor activation (8). Since 5-HT1F receptors are expressed on trigeminal neurons, and sumatriptan has modest affinity for 5-HT1F receptors, it was speculated that selective 5-HT1F receptor agonists might possess antimigraine efficacy (9). Hence, the guinea pig dural plasma protein extravasation model of migraine was used to evaluate this possibility using a variety of serotonergic agonists, including triptans (10). In this model, there was no correlation of agonist potency to inhibit dural plasma protein extravasation with either 5-HT1B or 5-HT1D receptor affinity. However, there was a highly significant correlation with 5-HT1F receptor affinity. Unlike triptans, compounds that selectively activated 5-HT1F receptors did not affect blood vessel diameter or contractility (11–13). Thus, 5-HT1F receptor agonists represented a promising and unique new antimigraine mechanism within the neurally acting anti-migraine agent (NAAMA) class.
Binding affinity of lasmiditan at human 5-HT receptor subtypes: comparison with LY334370
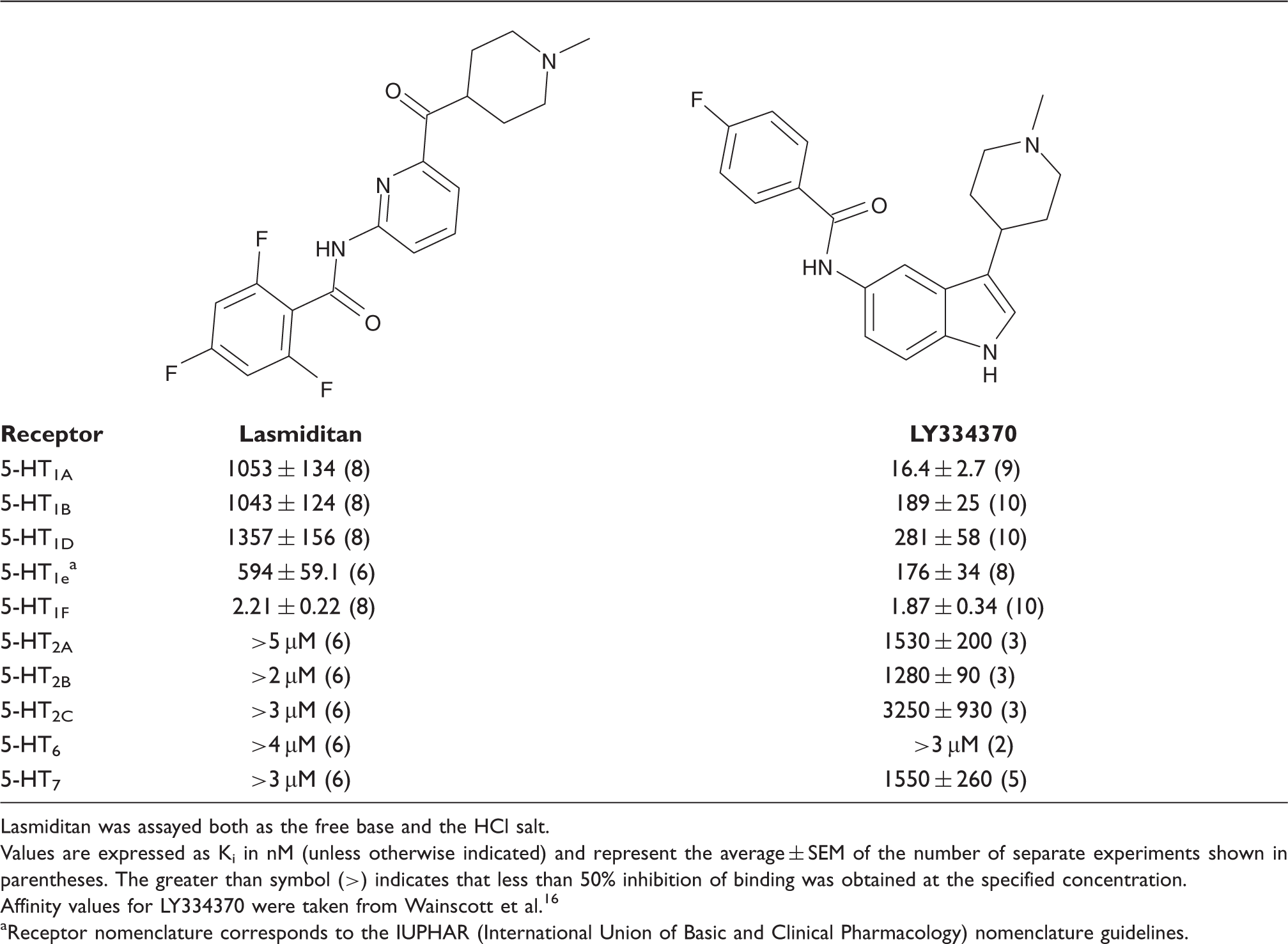
Lasmiditan was assayed both as the free base and the HCl salt.
Values are expressed as Ki in nM (unless otherwise indicated) and represent the average ± SEM of the number of separate experiments shown in parentheses. The greater than symbol (>) indicates that less than 50% inhibition of binding was obtained at the specified concentration.
Affinity values for LY334370 were taken from Wainscott et al. 16
Receptor nomenclature corresponds to the IUPHAR (International Union of Basic and Clinical Pharmacology) nomenclature guidelines.
Based on the promising results with LY334370, an effort was undertaken to develop an agonist with even greater 5-HT1F receptor selectivity and greater structural differentiation from the triptans. This led to lasmiditan (also known as COL-144, formerly LY573144), a highly selective 5-HT1F receptor agonist from a structurally novel pyridinoyl-piperidine chemical series.
Materials and methods
Binding assays
Standard radioligand-binding assays were used to evaluate the selectivity and affinity of lasmiditan across a variety of receptors. For the serotonin (5-HT) receptor subtype assays membranes from cells expressing cloned human versions of the receptors were used. These assays have previously been described in detail (17,18). Broad screening of lasmiditan across a panel of 52 binding assays covering a mix of receptors, ion channels and other binding sites was carried out at NovaScreen (Hanover, MD, USA).
[35S]-GTPγS assays
Membrane preparation
Assays were performed with cells expressing the cloned human 5-HT1A, 5-HT1B, 5-HT1D, 5-HT1e or 5-HT1F receptor. On the assay day, an aliquot of cells (stored at −70°C) was thawed and resuspended in 50 mM Tris-HCl, pH 7.4, and centrifuged at 39,800
Binding assay
The methods for the 5-HT1A, 5-HT1B, 5-HT1D, 5-HT1e or 5-HT1F receptor [35S]-GTPγS binding assays are modified from previously described assays (19–21), and adapted to an SPA (scintillation proximity assay) format. Incubations were performed in a total volume of 200 µl in 96-well assay plates. [35S]-GTPγS and guanosine-5′-diphosphate (GDP) in assay buffer (MgCl2, NaCl, EGTA in Tris-HCl, pH 7.4; 50 µl) were added to 50 µl of test compounds diluted in water. WGA (wheat germ agglutinin) beads (Amersham Pharmacia Biotech Inc., Piscataway, NJ, USA) for SPA in assay buffer (50 µl) were then added. Membrane homogenate (50 µl) from cells expressing the cloned human 5-HT1A, 5-HT1B, 5-HT1D, 5-HT1e or 5-HT1F receptor in assay buffer was added, and the plates were covered with sealing tape (PerkinElmer Wallac, Inc., Gaithersburg, MD, USA) and allowed to incubate at room temperature for 2 h. The final concentrations of MgCl2, NaCl, EGTA, GDP, [35S]-GTPγS, and Tris were 3 mM, 120 mM, 0.2 mM, 10 µM, approximately 0.3 nM, and 50 mM, respectively. The plates were then centrifuged at approximately 200
Data analysis
Using GraphPad Prism software (22), non-linear regression analysis was performed on the concentration-response curves (generating EC50 and Emax values for stimulation of [35S]-GTPγS binding) using a four-parameter logistic equation described by De Lean et al. (23). Efficacy (Emax) values, determined by the non-linear regression analysis, for the selected compounds were expressed as the percentage of [35S]-GTPγS binding relative to the response produced by 10 µM 5-HT(5-HT1A, 5-HT1B, 5-HT1e or 5-HT1F) or 1 µM 5-HT(5-HT1D) which was run as a standard with each concentration-response curve.
Saphenous vein contractility
The vascular contractile effects of compounds were measured
Trigeminal ganglion stimulation
Dural extravasation
The dural extravasation assay was modified from that previously described by Johnson and Phebus (25). Male Wistar rats from Harlan Laboratories (250–350 g), fasted overnight, were dosed with test compound by oral gavage (2 ml/kg) 45 min prior to administration of anaesthesia (sodium pentobarbital, 65 mg/kg i.p.) and placement in a stereotaxic frame for implantation of stimulating electrodes. Prior to stimulation, the femoral vein was exposed and a 20 mg/kg dose of FITC-labelled bovine serum albumin (FITC-BSA) was injected intravenously as a marker of protein extravasation. The left trigeminal ganglion was stimulated for 3 min at a current density of 1.0 mA (5 Hz, 4 ms pulse duration) by a Model S48 electrical stimulator with PSIU6 photoelectric isolation unit (Grass-Telefactor). Fifteen minutes following stimulation, the animals were exsanguinated with saline. The exsanguination also rinsed residual FITC-BSA out of the blood vessels. The dural tissues were collected from both hemispheres, and the amount of fluorescence due to extravasation of the FITC-BSA was determined spectrophotometrically in the dural tissue from each hemisphere. The extravasation induced by electrical stimulation of the trigeminal ganglion was an ipsilateral effect (i.e. occurs only on the side of the dura in which the trigeminal ganglion was stimulated). This allowed the other (unstimulated) half of the dura to be used as a control. The extravasation ratio (i.e. the ratio of the amount of extravasation in the dura from the stimulated side compared to the unstimulated side) was calculated. Animals dosed with vehicle alone or an ineffective dose of the test compound will have an extravasation ratio of approximately 2.2, while totally effective treatments will result in a ratio of approximately 1.
c-fos assay
The measurement of trigeminal nerve-stimulated c-fos production in the nucleus caudalis was based on the method previously described (26). Rats were pretreated with drug or vehicle, anaesthetised with sodium pentobarbital (65 mg/kg i.p.) and placed in a stereotaxic apparatus where their trigeminal nerves were electrically stimulated unilaterally. This procedure is identical to that performed in the neurogenic dural inflammation model with the exception that the stimulation current was 10-fold higher. Ninety minutes after stimulation, when c-fos expression in spinal and brain stem neurons would be expected to peak, animals were sacrificed and their brains perfused with 4% paraformaldehyde. In a multiday process, brain sections were exposed to an antibody directed against Fos protein. This antibody binds to Fos protein in the brain section and is subsequently localised using a secondary antibody and peroxidase staining. When brain sections were examined under a microscope, neuronal cell bodies, stained with Fos protein-like immunoreactivity, were present in areas of the rostral spinal cord and brainstem known to receive nociceptive input from trigeminal afferents. The number of cells stained for Fos protein was greater ipsilateral to the electrical stimulation than contralateral, as would be expected since the trigeminal nerve projects ipsilaterally to the CNS. The numbers of c-fos positive cells on the stimulated side were compared in vehicle and compound-pretreated animals. A treatment that inhibits neurotransmitter release from the central branches of trigeminal sensory neurons decreases the number of labelled cells ipsilateral to the stimulation. This is taken as evidence that the test compound inhibited trigeminal-mediated nociceptive neurotransmission to the CNS and should reduce migraine pain. LY334370 fumarate (10 µg/kg, p.o.) was used as a positive control. Lasmiditan.HCl or its vehicle was administered orally ∼1 h before trigeminal nerve stimulation. The number of central c-fos expressing neurons in sections of nucleus caudalis was quantified, with image analysis by a blind observer, on both the stimulated and non-stimulated sides. In vehicle pretreated animals, the number of nucleus caudalis cells demonstrating Fos protein-like immunoreactivity was greater on the stimulated side as compared to the contralateral, non-stimulated side. There were, however, some stained cells in the nucleus caudalis on the non-stimulated side. The c-fos expression seen on the non-stimulated side most likely arose from non-specific effects, including the stress of stereotaxic surgery. In the analysis, c-fos expressing cells from the non-stimulated side were subtracted from the stimulated side so that the final counts reflect cells specific to the trigeminal nerve stimulation. Statistical comparisons were performed using analysis of variance followed by a
Materials
Lasmiditan (2,4,6-trifluoro-
Results
To test the 5-HT1F receptor hypothesis of migraine treatment, minimising the potential for triptan-like side-effects including 5-HT1B receptor-mediated vasoconstriction, a focused effort was made to develop an agonist that was not only highly selective for the 5-HT1F receptor, but also structurally distinct from the triptans. It was also desired to develop a molecule that had even greater 5-HT1F receptor selectivity compared to the first-generation compound LY334370.
5-HT1F receptor selectivity profile in vitro
Lasmiditan (2,4,6-trifluoro- The chemical structure of lasmiditan is distinct from triptans.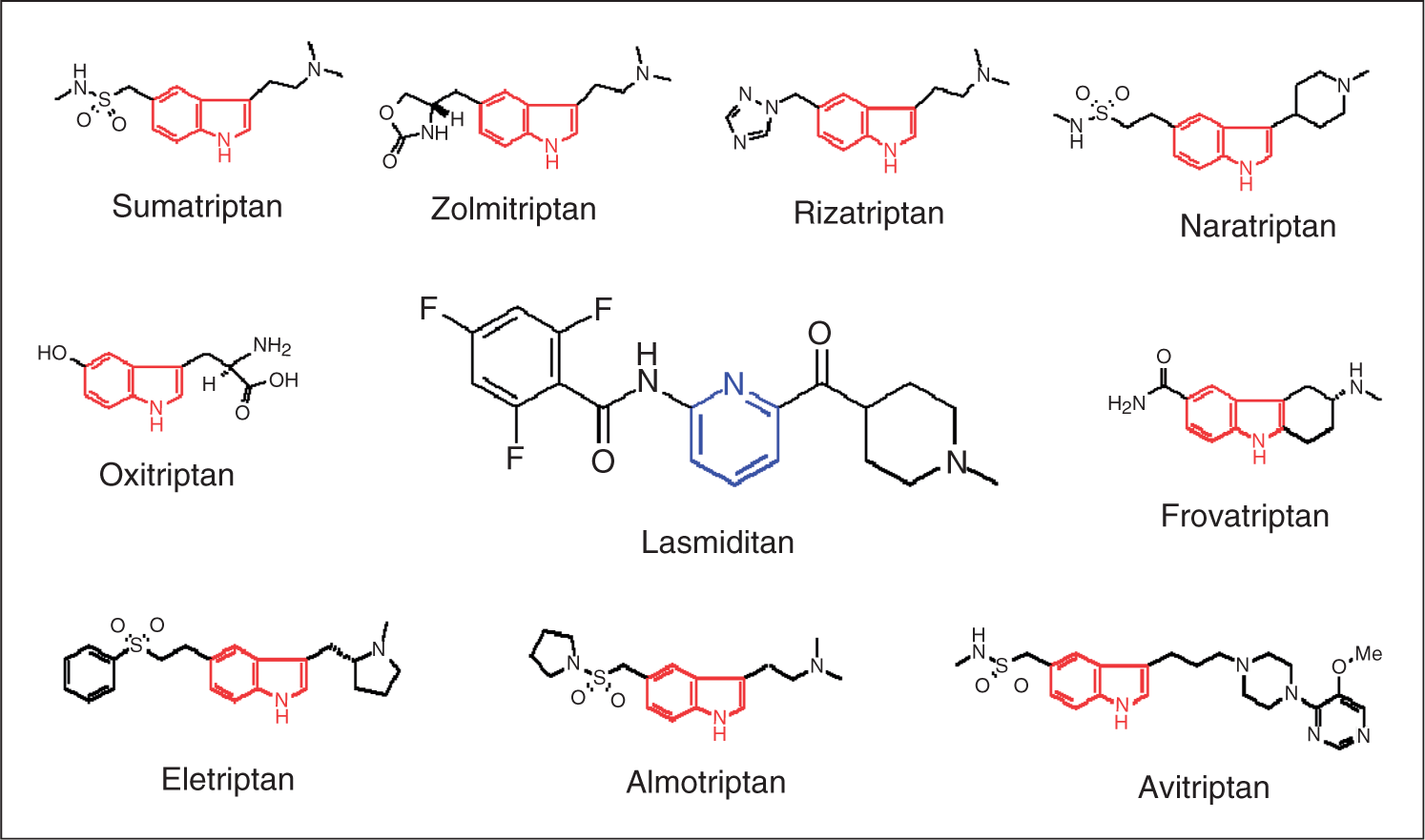
When lasmiditan was examined across a panel of human recombinant 5-HT receptor subtypes (Table 1), it showed great selectivity for the 5-HT1F receptor relative to other 5-HT receptors, including the other members of the 5-HT1 receptor family. It also showed enhanced selectivity compared to LY334370 (Table 1). In particular, the 5-HT1A receptor affinity was much reduced (Ki value increased) for lasmiditan compared to LY334370. 5-HT1B and 5-HT1D Ki values were also increased (affinity decreased) compared to LY334370. Thus, the selectivity of lasmiditan for the 5-HT1F receptor was considerably improved relative to other serotonergic receptors and to LY334370.
To assess overall selectivity further, lasmiditan was tested at NovaScreen (Hanover, MD, USA) across a panel of 52 binding assays covering a mix of receptors, ion channels and other binding sites. At a concentration of 1 µM, lasmiditan produced less than 50% inhibition of binding in 51 of these assays. The one exception was the benzodiazepine binding site on the GABAA receptor where 1 µM lasmiditan produced 67% inhibition of binding. In-house evaluation, using inhibition of [3H]-flunitrazepam binding, gave a Ki value for lasmiditan of 191 ± 33 nM (mean ± SEM determined from three different experiments, each run in duplicate).
Binding affinity of lasmiditan at other monoamine receptors: Ki values

The apparent Ki for each receptor was determined from at least two separate experiments. The greater than symbol (>) indicates that less than 50% inhibition of binding was obtained at the specified concentration, and thus, an accurate Ki value could not be determined.
These values are taken from Table 1 and are included here for comparison since both 5-HT2A and 5-HT2B receptors are known vasoconstrictor receptors in certain vascular beds.
5-HT1F receptor agonist efficacy
Lasmiditan was also evaluated for its functional activity Stimulation of [35S]-GTPγS binding in cells expressing the cloned human 5-HT receptors (A) 5-HT1B, (B) 5-HT1D and (C) 5-HT1F. Each point represents the mean ± SEM from the number of experiments given in parentheses. Potency and efficacy at 5-HT1 receptors of lasmiditan compared to selected triptans measured by EC50, concentration of compound that produces 50% of its maximal response. Emax, maximal response of the compound expressed as a percentage of the response produced by 10 µM 5-HT (5-HT1A, 5-HT1B, 5-HT1e, and 5-HT1F receptor assays) or 1.0 µM 5-HT (5-HT1D receptor assay). ND indicates that the compound was so impotent at this receptor that neither EC50 nor Emax could be determined with confidence. Therefore the efficacy is given as the percentage stimulation (%E) at the highest concentration of compound tested relative to the Emax produced by a maximal concentration of 5-HT. Each value is given as the mean ± SEM for the number of separate experiments given in parentheses.

Vasoconstrictor activity
The vasoconstrictor potential of lasmiditan was assessed using rabbit saphenous vein rings, a surrogate assay for human coronary artery vasoconstrictor liability (27). As can be seen in Figure 3, lasmiditan in concentrations up to 100 µM produced no discernable contractions under conditions where sumatriptan produced approximately 50% of the maximal KCl contraction across the same concentration range.
Lack of contractive effect of lasmiditan on rabbit saphenous vein. Points are mean values, and vertical lines represent the SEM for the number of tissues indicated in parentheses.
Efficacy in in vivo migraine models
Lasmiditan was evaluated in two Inhibition of trigeminal stimulation-induced dural plasma protein extravasation in rats 1 h after oral administration of lasmiditan or rizatriptan. Saline vehicle pretreatment resulted in extravasation ratios of approximately 2.2, while values of 1 indicate complete inhibition of extravasation. Statistical analysis was performed by comparison to the saline control group using Student’s 
In the second model, anaesthetized rats received electrical stimulation of the left trigeminal ganglion, the animals were sacrificed 90 min later, and the brainstems processed for c-fos immunoreactivity in the trigeminal nucleus caudalis. As can be seen in Figure 5, lasmiditan and LY334370 inhibited trigeminal stimulation-induced c-fos expression in the nucleus caudalis of rats 1 h after oral administration.
Effect of lasmiditan and LY334370 on trigeminal stimulation-induced c-fos expression in the nucleus caudalis of rats 1 h after oral administration. For each animal, 10–15 sections were analyzed to provide an average number of stained cells per section. Each bar represents the mean ± SEM for the number of stained cells for each treatment group (
Discussion
Lasmiditan fulfilled the goals of developing a molecule that: (i) was structurally unique compared to existing classes of acute antimigraine compounds; and (ii) showed greater 5-HT1F receptor selectivity than any existing molecules. Lasmiditan satisfied the criterion of being structurally different by providing a unique pyridinoyl-piperidine scaffold not found in any other class of antimigraine compounds and by being devoid of the indole moiety that is at the core of all the triptans. It is likely that these critical structural modifications from the triptans confer the unique pharmacological attributes seen with lasmiditan.
Sumatriptan, the first of the triptans, was developed for the treatment of migraine headache based on its ability to constrict cerebral blood vessels (3). This activity, which was later attributed to direct activation of 5-HT1B receptors expressed on vascular smooth muscle (5,28), is the basis for the activity of all of the triptans (29). However, concerns have been raised regarding potential adverse effects of the activation of 5-HT1B receptors in peripheral blood vessels, especially the coronary artery (29,30). In addition, the triptans can produce a relatively high incidence of neck, jaw, and chest symptoms (as high as 40%) that can limit tolerability and limit patient compliance (31).
An alternative approach to alleviate migraine is to affect the neuronal release of pain sensitising agents such as CGRP by targeting 5-HT1F receptors expressed on trigeminal neurons (9,32). Initially, Johnson et al. (10) found that 5-HT1F receptor affinity correlated with the ability of a range of 5-HT receptor agonists to inhibit dural plasma protein extravasation in the guinea pig. Subsequent clinical findings with the 5-HT1F prototypic receptor agonist LY334370 strengthened the hypothesis that selective 5-HT1F receptor agonists could provide efficacy in the treatment of migraine without the potential for causing vasoconstriction (14). Thus, a selective 5-HT1F receptor agonist would have a potential safety advantage over the triptans, which are contra-indicated in patients with cardiovascular disease. Thus, as an inhibitory presynaptic receptor, 5-HT1F receptor activation is well positioned to provide an alternative mechanism to alleviate migraine symptoms without vasoconstriction.
Lasmiditan met the goal of developing a highly selective 5-HT1F receptor agonist. It showed excellent overall selectivity when compared against a panel of over 50 receptors, ion channels, and transporters, using radioligand-binding techniques. Of particular significance was the low cross-reactivity of lasmiditan with other members of the 5-HT1 receptor family, since all of these receptors share significant structural homology. It was especially important to minimise interactions with 5-HT1A, 5-HT1B and 5-HT1D receptors in order to avoid potential concerns expressed for the first generation 5-HT1F receptor-selective agonist LY334370. Lasmiditan showed good selectivity based on binding affinity with greater than 450-fold higher affinity at the 5-HT1F receptor than 5-HT1A, 5-HT1B and 5-HT1D receptors. Overall, lasmiditan was at least 270-fold selective compared to all of the binding sites evaluated, with the exception of the benzodiazepine binding site of the GABAA receptor. In this case, lasmiditan still showed approximately 85-fold selectivity. It is not known whether there is any functional correlate of this binding affinity. Importantly, lasmiditan had no significant affinity across a panel of monoaminergic receptor subtypes that are known to regulate vascular tone.
In addition to the
Lasmiditan was orally active in two
Conclusions
Lasmiditan is a high-affinity, highly selective 5-HT1F receptor agonist that shows potent efficacy after oral administration in animal models used to test antimigraine compounds. Its chemical structure and pharmacological profile clearly distinguish it from the triptans. The potency and selectivity of lasmiditan make it ideally suited to test the 5-HT1F receptor hypothesis of migraine therapy definitively. The efficacy of lasmiditan to alleviate acute migraine clinically is described in the accompanying manuscript by Ferrari et al. (33).
