Abstract
Abstract
Objectives
To assess changes in mechanical sensitivity of the pericranial muscles in healthy individuals after a local anesthetic block of the temporalis tendon. In addition, to assess, if experimentally induced temporalis tendon pain, can lead to an increase in mechanical sensitivity of the pericranial muscles and reports of headache.
Methods
40 healthy participants were recruited for this randomized, double-blinded, controlled experiment, and were randomly injected with mepivacaine and isotonic saline into the dominant-side temporalis tendon in two different sessions, and either nerve growth factor (n = 20) or isotonic saline (n = 20) in a third session. Mechanical sensitivity was assessed in the temporalis, masseter, and trapezius muscles as well as in the temporalis tendon, on the dominant side, before and 10 minutes after each injection, and in a fourth session two days after the third session. Pain drawings and headache diaries were kept for 30 days after the final session to register any developing pain or headache.
Results
Mepivacaine injection into the temporalis tendon caused a significant decrease in mechanical sensitivity in the temporal tendon (−54.5%) and the masseter (−15.4%) muscle (P < 0.05) but not the temporalis (−12.1%) and trapezius muscles (−12.7%) (P > 0.05). Nerve growth factor injection into the temporalis tendon caused a significant increase in mechanical sensitivity in the tendon (+15.4%) and masseter muscle (+14.4%) (P < 0.05) but not the temporalis (+2.8%) or trapezius muscles (+3.1%) (P > 0.05). A significant increase was found for headache frequency in the first seven days (P < 0.05) after nerve growth factor injection compared to after isotonic saline injection, but not intensity (P > 0.05).
Conclusion
These findings suggest that the therapeutic effect of temporalis tendon anesthetic injections on facial pain and headaches are most likely not only due to a direct effect of the local anesthetic on the temporalis tendon but rather to a more generalized block of the nerves in the area. In addition, the temporal tendon may contribute to the pathophysiological processes of headache.
Introduction
Temporal tendonitis has been implicated in disorders such as headache, orofacial pain (1–3), and even neck pain. However, the clinical presentation of temporalis tendon pain is not well understood as most of what is known about temporal tendonitis comes from case reports (2,3). It has been suggested that pain may transfer from the temporalis tendon to nearby structures, such as the inner ear, eyes, and maxillary and mandibular teeth (4,5). To this date, no study has systematically assessed the characteristics of temporalis tendon pain or their relation to headache disorders.
Nerve growth factor (NGF) is a neurotrophic protein and has been shown to modulate nociception by increasing the release of mediators, such as histamine, bradykinin, ATP, etc. from inflammatory cells (6). Several studies have reported that NGF induces long-lasting mechanical hyperalgesia in different muscles (7–11). However, no study has assessed if NGF-injection can induce temporalis tendon sensitization, and as such, it would be interesting to determine whether injection of NGF into the temporalis tendon could increase the mechanical sensitivity of the temporalis tendon and in consequence induce long-term pain and/or headache.
Anesthetic injections into the temporalis tendon have been reported as a treatment for facial pain and chronic headaches that are accompanied by temporal tendonitis (3,12). However, to our knowledge, no study has been published assessing the effectiveness of this treatment approach and the mechanisms behind this treatment option remain unexplained. The most common explanation for the empirical efficacy of the temporalis tendon injection is that the source of pain is the temporalis tendon and as such targeting this structure with local anesthetic will reduce pain intensity and frequency. An alternative hypothesis is that since the temporalis tendon is in close proximity to other structures (13,14), specifically the branches of the trigeminal nerve, and because the temporalis tendon is surrounded by soft tissue, the analgesic fluid can easily spread out and affect other nerves such as the masseteric and temporal nerves causing a nerve block. Reports in the literature of a temporalis tendon injection aborting headaches, including migraine seem to support this hypothesis (15).
The objective of this study was to assess, in healthy individuals, changes in mechanical sensitivity of pericranial muscles after a local anesthetic block of the temporalis tendon. In addition, to assess if an injection of NGF into the temporalis tendon can induce temporalis tendon sensitization, and as a consequence lead to an increase in headache frequency and mechanical sensitivity of the pericranial muscles.
Materials and methods
Participants
A total of 40 healthy participants (14 men, 26 women) with a mean age ± SD of 23.6 ± 4.2 and 24.2 ± 3.9 years, respectively, participated in the study. Participants were recruited from flyers placed around Aarhus University campus, the internet group pages of Aarhus Internationals and an internet volunteer recruiting system, forsoegsperson.com. All participants in the study were screened for temporomandibular disorders (TMD) in accordance with the Diagnostic Criteria for TMD (DC/TMD) (16). None of the participants were diagnosed with any painful TMD or other orofacial pain and were not taking any regular medication except for birth control. Furthermore, participants were not included in the study if they reported any kind of headache, with the exception of one headache day in the last month and no more than 11 headaches in the last year. All participants were given a verbal and written explanation of the study and written informed consent was obtained from all participants prior to inclusion in the study. The study was approved by the Central Denmark Region Research Ethics Committee (no.: 1-10-71-30-20) and conducted in accordance with the Declaration of Helsinki. The sample size was estimated that with 40 individuals in the cross-over part of the study we would be able to detect a 25% difference in mechanical sensitivity between injected substances with the risk of type I and II errors being set at 5% and 20%, respectively.
Study design
This study was performed in a randomized, double blind, controlled, design (Figure 1A). All participants participated in four separate sessions. In the first two sessions, participants were randomly injected with either 3% mepivacaine or isotonic saline (IS), as a control, into the temporalis tendon after a baseline assessment of the mechanical sensitivity of the temporalis, masseter and trapezius muscles, and temporalis tendon. Ten minutes after the injection was administered, the mechanical sensitivity of the above-mentioned muscles and tendon was assessed again. In the third session, participants were randomly divided into two groups. One group had an injection of NGF (5 µg in 0.2 mL) into the temporalis tendon while the other group had an injection of IS as a control. Mechanical sensitivity was assessed before, 10 minutes after and two days after the injection, at the fourth session. All injections and assessments were performed on the participant’s dominant side. Before leaving the laboratory in the third session, participants were asked to keep a 30-day headache diary and pain drawing and were instructed on how to fill out the diary. The interval between the first two sessions, where participants received either mepivacaine or IS, was two days. The interval between the second and third sessions was between two and seven days. The same examiner (SY) performed all the experimental procedures and assessments at the Department of Dentistry and Oral Health, Aarhus University. The preparation and randomization of the injections were performed by a colleague who was not involved in the assessments. Participants and the examiner were all blinded to the injections in each session and the applied forces. Data were blinded to the examiner until the whole study was finished.

(a) Timeline of study. (b) Graphical representation of the assessed sites (black dots) in the temporalis, masseter and trapezius muscles and the temporalis tendon.
Temporalis tendon injections
Localization of the temporalis tendon by intraoral single-digit palpation was done according to the DC/TMD recommendations (16). Based on the anatomy of the temporalis tendon, the injected site was located with the left index finger of the examiner (SY) following the anterior border of the mandibular ramus intraorally almost to the zygomatic arch level on the dominant side of each participant. Before each injection, the syringe was checked for bubbles and the volume of the substance was confirmed. A 27-G, 40 mm needle was used to administer the substance slowly (20–30 s) into the temporalis tendon after aspiration. Participants were also asked what substance they thought had been injected (mepivacaine or IS) after each session.
Mechanical sensitivity assessment
Mechanical sensitivity was assessed with three palpometers (Palpeter®, Sunstar Suisse SA, Route de Pallatex 15 1163 Etoy, Switzerland) (18), 0.5, 1 and 2 kg, that were applied to the temporalis, masseter and trapezius muscles and temporalis tendon (Figure 1B). For repeatability of the mechanical sensitivity assessment, an individual template was made using a transparent sheet for each participant to reproduce the palpation sites at each of the four sessions. The anterior-posterior and superior-inferior borders of the temporalis and masseter muscles were confirmed by palpation during teeth clenching from the participant. These borders were used as landmarks to divide these two muscles into nine sites (three vertical and three horizontal sites). As for the trapezius muscle, three sites were assessed; one site was on the dominant side of the 7th cervical spine protrusion, one site was on the dorsal side of the acromion, the other site was on the midpoint of the line of these two sites above. The assessment was done with the muscles relaxed. As for temporalis tendon, the assessment site was located in the same way as described above for the temporalis tendon injections and the palpation was done with a custom-made, finger-shaped extension applied to the palpometer. For each muscle, the palpometer was applied to each site (nine sites for temporalis and masseter muscles, three sites for trapezius muscle and temporalis tendon) for two seconds as recommended in the DC/TMD (16) and all structures were assessed two times. Each assessment was done with an interval of at least 10 seconds that allowed the participant to rate the sensations following palpation as well as to prevent temporal summation of the assessed muscles. The order of the assessed muscles and the applied forces was randomized using randomization.com and the second round of assessments was only done after all muscles had been palpated a first time. After each palpation participants were asked to rate mechanical pressure/pain sensitivity on a 0-50-100 scale with 0 being ‘no sensation whatsoever’, 1 to 49 being ‘sensation of pressure but the stimulus is not painful’, 50 being ‘barely painful’ and 100 being ‘the most painful imaginable’ (9,18–20).
Headache diary and pain drawing
All participants were asked to fill out a thirty-day headache diary and pain drawing after the third session. As for the headache diary, participants noted duration, intensity, and associated symptoms of the headache as well as any medication they had taken to abort or relieve the headache (21). As for the pain drawing, participants were asked to draw their perceived areas of pain on an anatomical map that included the lateral, frontal, and intra-oral projections of the head (8,22). All the areas above the orbital-meatal line were analyzed as headache pain regions.
MRI assessment
To assess if the substances injected into the temporalis tendon were confined to the temporalis tendon region a volunteer was asked to participate in an MRI assessment that was done pre- and post- injection into the temporalis tendon. 2 mL of IS was administered into the temporalis tendon via a needle protected IV cannula (BD Venflon™ Pro Safety, 18G, 32mm, Becton Dickinson Infusion Therapy AB, SE-251 06 Helsingborg, Sweden) connected with a catheter (B Braun Original-Perfusor® Line, B. Braun Melsungen AG 34209 Melsungen, Germany). Data were acquired using a 3T MRI system (Siemens, Skyra, Germany) and the standard head coil was used for the imaging. High-resolution, three-dimensional (3D), T2-weighted anatomical images were acquired, covering the entire brain, and including both the right and left temporal region. A 3D SPACE sequence with the following parameters was used: field of view 248 × 256 × 156 mm; voxel dimensions: 1.33 × 1.78 × 1.3 mm; echo time: 357 ms; repetition time: 2500 ms; scan time 37 secs. The sequence was scanned before injection of IS and then repeated four times after the injection to follow the distribution of IS.
Statistical analyses
Data were assessed for normality by using Q-Q plots and all data were found to be normally distributed. The primary outcome parameters for this study were changes in mechanical sensitivity before and after anesthetics injection, and before and after NGF injection. To ascertain differences in mechanical sensitivity for the different structures, before and after mepivacaine or IS injection, a 4-way repeated measures analysis of variance (ANOVA) was used with the following factors: structure (temporalis muscle, masseter muscle, trapezius muscle and temporalis tendon), session (mepivacaine, IS), time (before and after injection) and force (0.5, 1.0 and 2.0 kg). To assess differences in mechanical sensitivity before and after NGF or IS injection a 4-way repeated measures ANOVA was used with the factors: group (NGF, IS), structure (temporalis muscle, masseter muscle, trapezius muscle and temporalis tendon), time (before injection, 10 minutes after injection and two days after injection) and force (0.5, 1.0 and 2.0 kg). P-values of less than 0.05 were considered significant for the main factors and interactions. In addition, Tukey’s HSD (Turkey’s honestly significant difference) tests were used for post hoc comparisons. Unpaired t-tests for independent samples were used to test for differences in age of different genders, headache frequency, average intensity, headache index (defined as average intensity × frequency) and pain drawing areas between the IS group and the NGF group. Headache frequency was assessed for the whole 30 days but also for the first 7 days. This was done because the effect has been shown to last up to seven days (23). Chi-square test was used to test for difference in genders between the IS group and the NGF group. To calculate pain areas, the pain drawings were digitized and areas assessed in mm2 using the software image J (National Institutes of Health, USA), similarly to a previous study (8). P-values of less than 0.05 were considered significant for the unpaired t-tests. All statistical analyses were performed in Statistica Statistics for Windows (Version 13, TIBCO Software Inc.).
Results
Mechanical sensitivity before and after mepivacaine and IS injection
A significant difference was found for the factors structure (F (3, 117) = 184.69, P < 0.05, ANOVA), session (F (1, 39) = 65.13, P < 0.05, ANOVA), time (F (1, 39) = 102.55, P < 0.05, ANOVA), and force (F (2, 78) = 529.38, P < 0.05, ANOVA) as well as the interactions of session × structure (F (3, 117) = 124.02, P < 0.05, ANOVA), session × force (F (2, 78) = 13.31, P < 0.05, ANOVA), session × time (F (1, 39) = 169.72, P < 0.05, ANOVA), session × structure × time (F (3, 117) = 116.71, P < 0.05, ANOVA), session × structure × force (F (6, 234) = 14.03, P < 0.05, ANOVA), session ×time ×force and structure × session × time × force (F (6, 234) = 10.91, P < 0.05, ANOVA). Regarding the interaction structure × session × time × force, the post hoc analysis revealed a significant mechanical sensitivity decrease in the temporalis tendon after the mepivacaine injection when compared to before the injection (P < 0.05, Tukey’s HSD) which was not seen after the IS injection (P > 0.05, Tukey’s HSD). This significant difference was present for all three forces applied (P < 0.05, Tukey’s HSD) (Figure 2). In addition, a significant decrease in mechanical sensitivity of the temporal tendon was seen for the local anesthetic when compared to the isotonic saline after injection and this was valid for all three forces used. (P < 0.05, Tukey’s HSD). For the masseter muscle, there was a significant decrease in mechanical sensitivity after mepivacaine injection for 1 and 2 kg (P < 0.05, Tukey’s HSD) but not 0.5 kg (P > 0.05, Tukey’s HSD) (Figure 2). There were no significant changes found in the temporalis muscle or the trapezius muscle (P > 0.05, Tukey’s HSD). Finally, participants were able to correctly identify which substance they had injected (mepivacaine and IS) in both sessions 80% of the time.
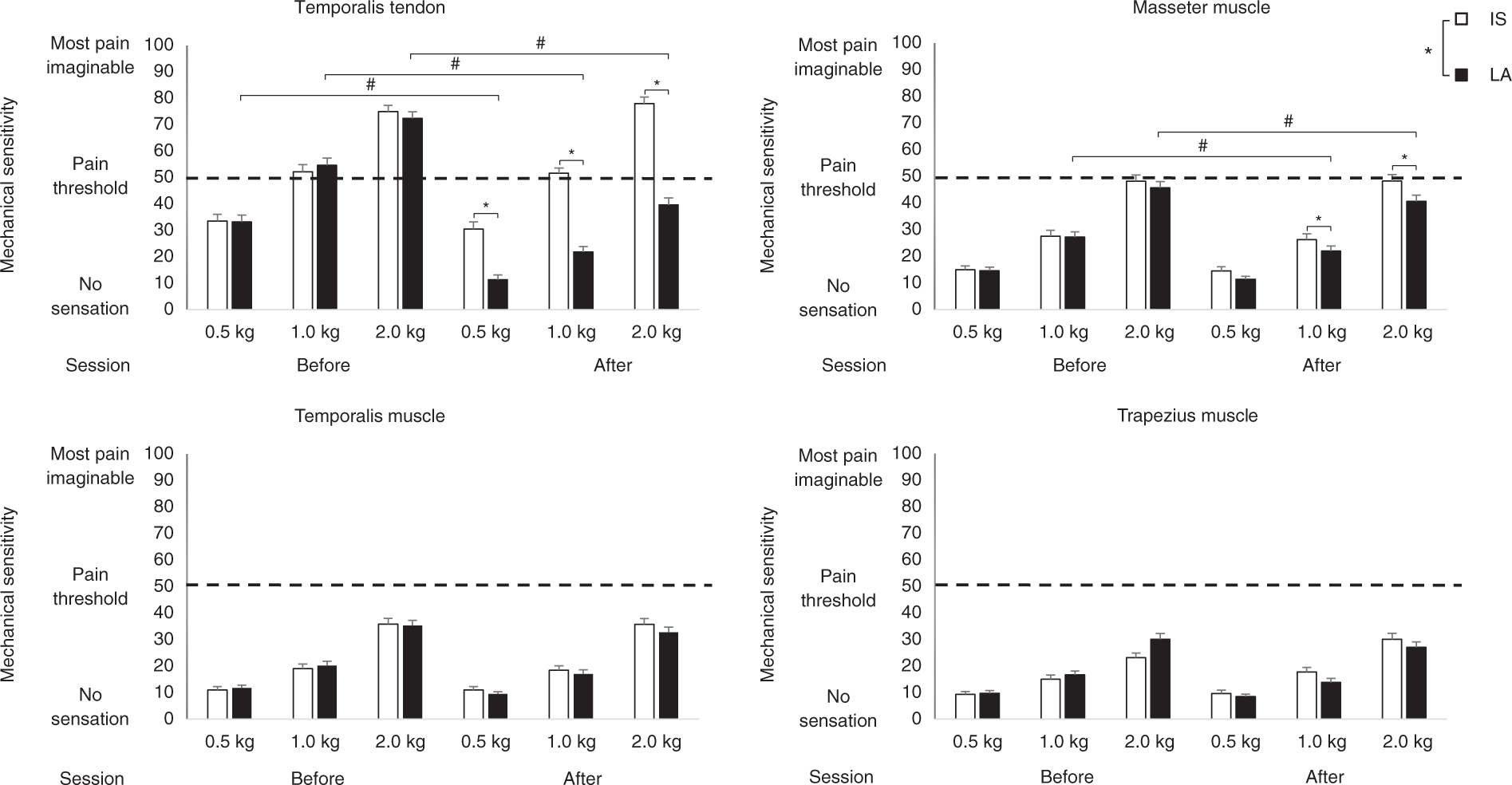
Mechanical sensitivity scores (n = 40) when palpated with 0.5, 1.0 and 2.0 kg on the dominant side for the temporalis, masseter, trapezius muscles and the temporalis tendon before and after IS injection and mepivacaine injection. All figures show mean values and SE. There was a significant decrease in mechanical sensitivity after mepivacaine injection in the masseter muscle and the temporalis tendon (# P < 0.05). Significant difference was also shown between mepivacaine and IS group after injection session in the masseter muscles and the temporalis tendon (*P < 0.05).
Mechanical sensitivity comparison between the NGF group and the control group
A significant difference was found for the main factors structure (F (3, 114) = 184.60, P < 0.05, ANOVA), time (F (2, 76) = 4.87, P < 0.05, ANOVA), and force (F (2, 76) = 570.25, P < 0.05, ANOVA) as well as the interactions of group × time (F (2, 76) = 9.05, P < 0.05, ANOVA), group × structure (F (3, 114) = 3.09, P < 0.05, ANOVA) and group × structure × time (F (6, 228) = 6.70, P < 0.05, ANOVA). The post hoc analysis revealed a significant increase in mechanical sensitivity for the masseter muscle and the temporalis tendon two days after the NGF injection (P < 0.05, Tukey’s HSD) which was not seen for the IS injection (P > 0.05, Tukey’s HSD) (Figure 3). There were no significant changes found in the temporalis muscle or the trapezius muscle (P > 0.05, Tukey’s HSD).

Mechanical sensitivity scores (n = 40) when palpated with 0.5, 1.0 and 2.0 kg on the dominant sides for the temporalis, masseter, trapezius muscles and the temporalis tendon before and after injection, and two days after IS injection and NGF injection. All figures show mean values and SE. There was a significant increase in mechanical sensitivity two days after NGF injection comparing to post-injection in the masseter muscle (# P < 0.05). Significant difference was also shown in MS two days after NGF injection session comparing to pre- and post-injection in the temporalis tendon (# P < 0.05).
Headache frequency and intensity comparison between the control group and the NGF group
The demographic information for participants in the IS group and the NGF group is shown in Table 1. No significant difference was found in age of males and females between groups (P < 0.05, t-test). No significant difference was found in genders between two groups. (P < 0.05, Chi-square test). In the NGF group, 16 out of 20 participants experienced at least one headache episode in the 30-day follow-up period accounting for 89 headache episodes. Of these, 54 episodes occurred in the first seven days after injection. In the IS group 14 out of 20 participants experienced at least one headache episode during the 30-day follow-up period accounting for 54 headache episodes of which 22 episodes happened within seven days of the injection (Figure 4A). The characteristics of the headaches can be seen in Table 2. There was no significant difference in headache frequency between the NGF and IS groups (P > 0.05, Tukey’s HSD). A significant difference was seen between the NGF and the IS groups with a mean value ± SD of 2.7 ± 2.8 and 1.2 ± 1.8 respectively in regard to headache frequency (P < 0.05, t-test) in the first seven days following injection. Moreover, no significant difference was seen between the NGF and IS groups for headache intensity or the headache index either in the first 7 days or in total (P > 0.05, t-test).
Demographic data for the IS group and the NGF group. The table shows the age and gender of different groups.

IS: isotonic saline; NGF: nerve growth factor; M: male; F: female.

(a) Headache frequency in the first seven days following injection. (b) Headache frequency in the 30 days following injection. The cluster of dots represents the distribution of headache days of each individual in both groups. Significant different was found in headache frequency in participants after NGF injection comparing to participants after IS injection in the first seven days. (* P < 0.05). Non-significant difference was found in the comparison of 30-day headache frequency (P > 0.05).
Headache characteristics. The table shows the percentages of different headache characteristics recorded in the 30-day headache diary.

IS: isotonic saline; NGF: nerve growth factor.
Pain drawings
There was no significant difference between the NGF (2.69 ± 4.45) and the IS groups (2.53 ± 5.38) in regards to pain areas (P > 0.05, t-test). The location of pain areas can be seen in Figure 5.
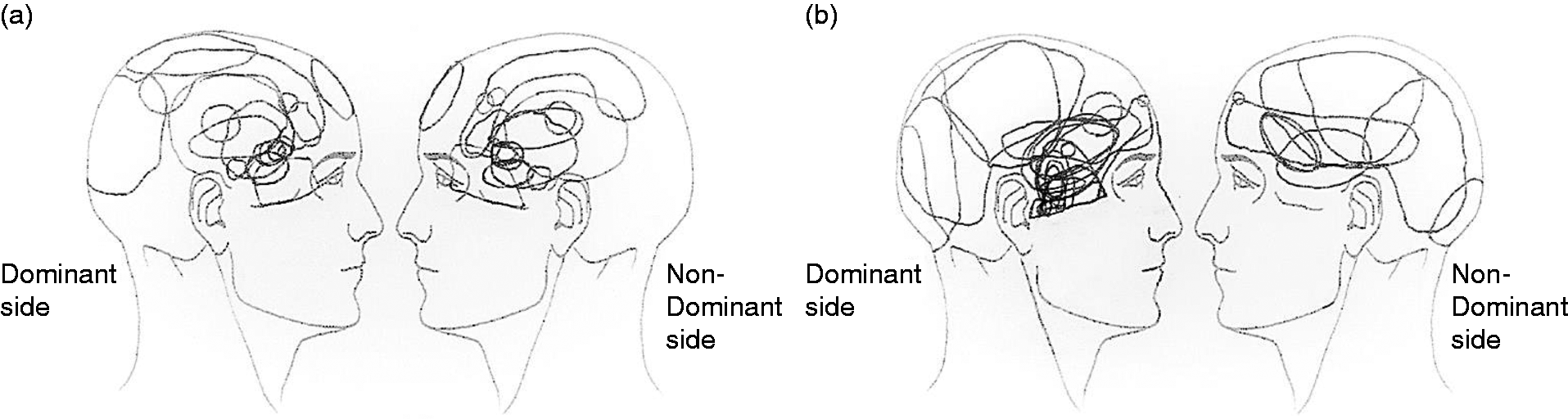
Headache drawings. Headache area in the dominant and non- dominant sides after (a) IS injection or (b) NGF injection. There was no significant difference between the headache areas in both groups on both sides (P > 0.05).
MRI assessment
The images in Figure 6 show that the substance was injected into the temporalis tendon but was not confined to the temporalis tendon due to a significant spread to the nearby structures.

MRI assessment. The images are shown in the axial, sagittal and coronal plane. (a) pre-injection images; (b) post-injection images; (c) post-injection images with reference line. Red square: The injected substance is shown spreading to regions adjacent to the temporalis tendon.
Discussion
The main findings of this study were that: a) anesthetic injection into the temporalis tendon caused a decrease of mechanical sensitivity in the temporalis tendon and masseter muscle, but not in the temporalis or trapezius muscle; b) NGF injection into the temporalis tendon caused an increase of mechanical sensitivity in the temporalis tendon and masseter muscle, but not in the temporalis or trapezius muscle; c) NGF injection into the temporalis tendon led to an increase in headache frequency in the first seven days following injection.
Mechanical sensitivity after local anesthetic
Following mepivacaine injection into the temporalis tendon, mechanical sensitivity was decreased in the masseter muscle and temporalis tendon, but not in the temporalis or trapezius muscle. In addition, it was shown that the mechanical sensitivity decrease was much bigger for the temporalis tendon than for the masseter muscle (54.5% vs. 15.4%). It can be inferred that this difference was due to the local anesthetic being directly injected into the temporalis tendon area. On the other hand, the only way the local anesthetic injected into the temporalis tendon could have decreased the mechanical sensitivity of the masseter muscle would be indirectly, such as through a nerve block. Since it is known that in the temporalis tendon area there are many nerve branches and vessels, including the mandibular nerve (24), it is likely that the anesthetic spread out, as seen in the MRI images (Figure 6), and affected the mandibular nerve. However, this does not explain why the mechanical sensitivity of the temporalis muscle was not affected. What may explain this is the fact that the mandibular nerve splits in an area close to the site of injection of the temporalis tendon into the masseteric nerve, which innervates the masseter muscle (25,26) and as such, this could explain why the masseter muscle was affected but the temporalis muscle was not. Another reason could be that by only waiting 10 minutes before the post-injection assessment not enough time was allowed for the local anesthetic to have an effect on the temporalis branch of the mandibular nerve. This seems likely as there was a decrease in mechanical sensitivity of the temporalis muscle of around 12%. The idea of a nerve block being responsible for the decreased mechanical sensitivity in the masseter muscle is further reinforced by the fact that the entire muscle decreased in mechanical sensitivity rather than just the areas closest to the injection site. The clinical implications of these results are that effects on facial pain and headache that are observed following a temporalis tendon injection are most likely due to both a direct effect on the temporalis tendon but perhaps more importantly due to a nerve block of parts of the mandibular nerve such as what might occur with a greater occipital nerve block (27–29). Empirically, this hypothesis is in accordance with patient reports in the clinic, as well as the reports of some of the participants in this study that reported feeling the side of their face where they received the injection very differently from the non-injected side.
Mechanical sensitivity after NGF injection
Mechanical sensitivity was found to be increased in the masseter muscle and temporalis tendon, but not the temporalis or trapezius muscle, two days after an NGF injection into the temporalis tendon. In general, it was shown that NGF caused mechanical sensitization of the temporalis tendon at the same level as has been seen in previous studies where the masseter was injected with NGF (7,23,30). Regarding the increased mechanical sensitivity of the masseter muscle, we propose two reasons that may explain this. The first is that similarly to what occurred for the local anesthetic, there might have been some NGF spillover from the tendon into nerve structures that may have caused the increased sensitization of the masseter muscle. However, due to the small amount of liquid (0.2 ml) we do not think this is the most likely explanation. Another explanation could be that the prolonged nociceptive input caused by the NGF injection into the temporalis tendon may have led to lowering of mechanical pain thresholds within the trigeminal dorsal horn (31) and this in turn may have led to an increase in mechanical sensitivity of the masseter muscle. The reason why these central changes did not affect the temporalis and trapezius muscles are unknown but may be because the mechanical sensitivity of both of these muscles, as assessed in this study, were not within the painful range and as such unaffected by the lowering of mechanical pain thresholds. In terms of mechanical pain thresholds, mechanical sensitivity of temporalis tendon before and after NGF injection was much beyond the limit compared to other muscles. As such, the clinical significance shown two days after NGF injection was not apparent though it was statistically significant.
Headache development following NGF injection
In this study no difference was found between the NGF and IS groups in the 30 days following the injections. However, when concentrating on the first seven days a significant difference was seen between the two groups with more headache days occurring in the NGF group. The reasons for this are likely to be explained by the fact that the sensitization caused by the NGF lasts around seven days (8,22). As such, it could be thought that the headaches would fall under the category of headache attributed to temporomandibular disorders (TMD). However, many of the headaches that developed in the first seven days were described as not being modified by jaw function and bilateral rather than unilateral. In addition, some of the headaches were reported to be accompanied by nausea and aggravated by routine physical activity which would indicate more primary headache qualities than those of headache attributed to TMD. As such, it could be speculated the temporalis tendon may contribute to patients experiencing headaches in two different ways. The first would be a direct relationship, in that the patients would perceive the temporalis tendon pain as a headache, or that the temporalis tendon pain could refer to the temple region. The second would be an indirect form in which the pain caused by the NGF in the temporalis tendon could facilitate the occurrence of primary headaches such as tension-type headache and migraine through central mechanisms. As mentioned in the introduction, temporal tendonitis pain may radiate or transfer to various locations, such as the temple and eye regions (2). When this occurs, it may be difficult for patients to describe their pain as temporalis tendon pain and likely will describe it as a headache, which may lead to difficulties in ascertaining the source of pain. If clinicians are increasingly aware that temporalis tendon pain may lead to headaches, a more effective and targeted pain management can be implemented.
In this study a high percentage of participants also developed headaches following IS injection. The reasons for this could be related the mechanical trauma caused by the injection or it could have been that participants were more aware of the headaches once they were asked to fill out a headache diary.
Limitations
The main limitation of this study is that it was difficult to completely blind which injection, local anesthetic or IS, was received by the participant as 80% of the participants were able to guess correctly which injection they received. However, participants had no a priori knowledge of the goal of the study and it seems unlikely that they would be biased into reporting a decrease in mechanical sensitivity for the masseter muscle but not the temporalis or trapezius muscles. Nonetheless, this should be taken into consideration when analyzing the results of this study and for future studies.
It could be argued that another limitation is that there was no run-in headache diary for healthy participants which can lead to high incidence of headaches after injection. Even though participants could only be included in the study if they reported less than 12 headaches in the last year and only one in the last month, a recall bias may have occurred with participants having more headaches than reported.
Conclusion
In summary, it was shown that anesthetic injection into the temporalis tendon led to decreased mechanical sensitivity of both the temporalis tendon and the masseter muscle. This suggests that the therapeutic effect of this type of injection could be more related to that of a nerve block rather than a local effect at the tendon level. Furthermore, NGF injection into the temporalis tendon induced temporalis tendon sensitization which led to an increase in the mechanical sensitivity in the masseter muscle and the temporalis tendon, and caused increased headache frequency in the first 7 days following injection. These findings support the idea that the temporalis tendon may contribute to the pathophysiological processes of headache.
Key Findings
Mepivacaine injection into the temporalis tendon caused a significant decrease in mechanical sensitivity in the temporal tendon and the masseter muscle. NGF injection into the temporalis tendon caused a significant increase in mechanical sensitivity in the tendon and masseter muscle. A significant difference was found for headache frequency in the first seven days after NGF injection compared to after IS injection, but not headache intensity. The therapeutic effect of temporalis tendon anesthetic injections on facial pain and headaches are most likely not only due to a direct effect of the local anesthetic on the temporalis tendon but rather to a more generalized block of the nerves in the area. The temporal tendon may contribute to the pathophysiological processes of headache.
Footnotes
Acknowledgements
We would like to acknowledge all the participants. We thank to Bente Haugsted for the help with the randomization and preparation of the IS and NGF injections, we thank to Steffen Ringgaard for the help with the MRI assessment and we also thank to Nicole Renner for providing the graphical presentation of assessed muscles and tendon. We would also like to show our gratitude to Rubens Spin-Neto for providing insight and expertise on the MRI assessment analysis.
Declaration of conflicting interests
The authors declare they have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Danish Dental Association.
