Abstract
Objective
To assess if repeated intramuscular injections of nerve growth factor into the temporalis and masseter muscles increase mechanical sensitivity and entropy scores. Furthermore, to investigate if increased mechanical sensitivity would lead to increased prevalence of referred pain in the studied individuals. Finally, if increased muscle sensitization would lead to an increase in number of headache days during the experimental period.
Methods
The present double-blind, randomized placebo-controlled study recruited 16 healthy participants who were injected with nerve growth-factor, on 2 days, into the masseter and temporalis muscles and isotonic saline on the contralateral side. Mechanical sensitivity was assessed at seven different time-points (total of 21 days) by application of three different forces to 15 different sites of both muscles. Participants were asked after each force application if they experienced referred pain and were asked to keep a headache diary during the experimental period.
Results
In summary, a) repeated intramuscular injections of nerve-growth-factor caused an increase in mechanical sensitivity for the masseter but not the temporalis muscle, and an increase in entropy scores when compared to the isotonic saline side. b) Both referred pain frequency and number of headache days were not increased following nerve-growth-factor injections.
Conclusions
These findings support the idea that mechanical sensitization in the masseter and temporalis muscles differs following injections of nerve growth factor. Furthermore, referred pain and headache frequency do not seem to be related to nerve growth factor sensitization in this model. These findings support the idea that in healthy individuals referred pain may be an epiphenomenon of the muscle in response to noxious input.
Introduction
Pericranial tenderness (PCT) has been shown to be increased in patients with tension-type headache (TTH) when compared to controls (1,2). Furthermore, the degree of tenderness is significantly related to headache frequency, intensity, and treatment outcome (3,4). This suggests that PCT is of importance in the development of TTH. Studies have attempted to reproduce PCT both in the cervical and masticatory muscle groups through injections of algogenic substances (5,6), static muscle contraction (7) and clenching tasks (8,9). Despite these efforts, the pain and tenderness caused by these experimental models does not last longer than 24 hours, which makes the assessment of how headaches develop over time difficult.
Referred pain (RP) has also been shown to be present more frequently in patients with TTH when compared to controls (10). Furthermore, the referral patterns in these patients have been shown to reproduce their habitual headache pattern (10). It is thought that RP is due to both peripheral and central mechanisms leading to unmasking of new synapses that converge in the central nervous system (11). Furthermore, the unmasking of new synapses is thought to be a result of expanded receptive fields, and this can occur following noxious stimulation of muscle afferents (12).
Nerve growth factor (NGF) has been shown to be involved in the development of muscle hyperalgesia (13,14). It does so by exciting high threshold mechanical muscle afferents (15) and increasing receptive fields (16). Furthermore, it has been shown that trkA (17) and TRPV1 (18,19) are the main receptors involved in mediating this mechanical hyperalgesia. Previous studies have shown that repeated injections of NGF cause long-lasting sensitization, up to 10 days, and expanded pain areas in the tibialis muscle (20), but no such data exists for the pericranial muscles. It would, as such, be interesting to assess whether repeated injections of NGF into the temporalis and masseter muscles would cause the same long-term sensitization and increased receptive field activity as seen in previous studies.
It has been proposed that the spatial distribution of muscle pain as a variable should be considered to characterize different diagnoses such as temporomandibular disorders (21). This is because the degree of spatial diversity of mechanical sensitivity within the craniofacial muscles may serve as a measure to distinguish between healthy individuals and individuals with myalgia and/or headache. With this in mind we applied a variation on the concept of Shanon's entropy (22) to determine the diversity of mechanical sensitivity values obtained from a systematic evaluation of the masseter and temporalis muscles before and after injections of NGF. When assessing diversity in this way, the higher the entropy value, the more diversity, and the lower the entropy value, the less diversity is present.
The aims of this study were to assess whether repeated intramuscular injections of NGF into the temporalis and masseter muscles would increase mechanical sensitivity and entropy scores in both muscles. Furthermore, it aimed to investigate whether increased mechanical sensitivity would lead to increased prevalence of RP in the studied individuals, and finally, whether increased sensitization of the evaluated muscles would lead to an increase in the number of headache days during the experimental period.
Material and methods
Participants
A total of 16 healthy participants (five women and 11 men) with a mean age of 28.1 (±11.4) years participated in this study. These participants were recruited from flyers posted around the campus at the University of Aarhus. All participants in this study were unaware of RP patterns for the head, face and neck. Participants were screened for TMD in accordance with the Diagnostic Criteria for TMD (DC/TMD) (23). None of the participants reported or were diagnosed with any painful TMD or other orofacial pain, and were not taking any regular medication. Furthermore, participants were not included in the study if they reported any kind of headache, with the exception of one TTH headache day in the past 4 weeks. All participants were given a verbal and written explanation of the study, and written informed consent was obtained from all participants prior to inclusion in the study. The study was approved by the Central Denmark Region Research Ethics Committee (1-10-72-286-14) and conducted in accordance with the Declaration of Helsinki. A sample size estimate suggested that with 16 participants we would be able to detect a 25% difference in means with α = 0.05, and β = 0.2, and an intra-individual variability of 25%.
Study design
The study was performed in a randomized, double-blind, placebo-controlled manner. Both left and right masseter and anterior temporalis muscles received two injections each of either NGF or isotonic saline (IS) as a control. The side (left or right) and the injected substance (NGF or IS) were randomized for the first injection using the random number generator at randomization.com. The injections were repeated in an identical manner in the second injection session. This meant that eight participants were injected with NGF on the right side and IS on the left and that eight participants were injected with IS on the right side and NGF on the left side. Injections were done in the following order for all participants for both sessions: Right temporalis, right masseter, left temporalis, and left masseter. Assessments were performed on five separate days. Injections were given on the first two assessment days. In each of these two first days, two assessments were performed; injections were given immediately after the first assessment of the day. Assessments were done at baseline (D0S1), 3 hours after the first injection (D0S2), 2 days after baseline, before the second injection (D2S1), 3 hours after the second injection (D2S2), 7 days after baseline (D7), 14 days after baseline (D14) and 21 days after baseline (D21). The evaluated parameters and when they were assessed can be seen in Figure 1(a).
Timeline of study and site numbers. Study design timeline (a). Graphical representation of the way the masseter and temporalis muscles were divided into 15 sites (b).
The same examiner performed all assessments and experimental procedures at the Department of Dentistry and Oral Health, Aarhus University. The preparation and randomization of the NGF and IS injections was done by a staff member that was not involved in the assessments. Both the examiner and participants were blinded to the experimental conditions, including the side in which NGF was injected, and data was unblinded after all data was collected.
Assessment sites
Fifteen sites in both the masseter and temporalis muscles were selected for assessment of muscle palpation sensitivity and cutaneous mechanical sensitivity. The sites were reproduced with the use of an individual plastic template created for each side and individual. The 15 sites were identified by outlining the borders of the masseter and temporalis muscles and then dividing them in a five horizontal by three vertical grid for the masseter muscle and a three horizontal by five vertical grid for the temporalis (Figure 1(b)).
NGF and isotonic saline injections
Based on previous studies (13,20,24) the NGF was given as a bolus injection (5 µg of NGF in 0.2 ml) to both the masseter and anterior temporalis muscle on one side to all 16 individuals. A 0.2 ml buffered IS bolus was injected into the contralateral masseter and anterior temporalis muscles. For the masseter muscle, all injections were given in site 8, and all injections into the temporalis muscle were given in site 14 (Figure 1(b)).
Measurement outcomes
Mechanical sensitivity
Cutaneous sensitivity was assessed by applying a 512 nm Von Frey hair to each of the 15 sites in the masseter and temporalis muscles. After each stimulus, participants were asked to rate the perceived intensity on a 0–50–100 numeric rating scale (NRS) where 0 is “no sensation”, 1–49 is a “sensation of pressure”, 50 is “just barely painful” and 100 the “most pain imaginable” (25).
Mechanical sensitivity was assessed with three palpometers (26) that applied 0.5, 1 or 2 kg of pressure. For each assessment, the palpometer applied pressure to the muscle site for 5 seconds to assess possible referral patterns (23). The assessment was done with the muscles relaxed and on both sides. The order in which the palpometers were used was randomized using randomization.com. For each palpation, participants were asked to rate mechanical pressure/pain sensitivity (MPS) on the same 0–50–100 NRS (25). Finally, following each palpation, they were asked to report if they experienced RP during the palpation. For this, the participants were told to let the examiner know if they had experienced pain that was not only under the palpometer. If yes, they were asked to point to which areas they had felt the pain, and the examiner would assess if this was outside of the border of the palpated muscle as per the DC/TMD protocol (23). Each assessment was done with an interval of at least 10 seconds that allowed the participant to rate the sensations following palpation as well as to prevent temporal summation of the assessed muscles.
Entropy of mechanical pressure/pain sensitivity scores
Entropy is a measure of diversity of values. Entropy for a particular experimental condition with a set of M possible outcomes is highest if all values have maximum diversity, whereas the minimum value of entropy is zero if all outcomes are equal. In the context of the 15 sites, entropy indicates the degree of diversity of the 15 MPS values for each muscle, with higher entropy values corresponding to more diverse MPS values; that is, greater heterogeneity over the grid. The entropy was calculated in Excel from the MPS of the 15 sites within each muscle and for each assessment. The maximum entropy possible for this set up was 2.708 (22).
Pain drawing
At the beginning of each of these assessments, participants were asked to draw their perceived areas of pain since the last appointment on a figure of the head. These pain areas were digitized and calculations were done in mm2 using the image J software, similarly to a previous study (14).
Assessment of headaches
Before enrolment in the study, participants were asked if they had experienced any headache in the last 30 days according to the DC/TMD (23). If they answered no, they were enrolled in the study. If they answered yes, they were asked how many headache they had experienced in the last month. If they answered more than 1 they were excluded from the study, and 1 or less they were enrolled in the study. Participants were asked to fill out a headache diary during the experimental period, where they were to report how many headache days they had and any kind of medication they took to abort the headache. The headache impact test (HIT-6) is a questionnaire that measures several headache-related quality of life domains such as pain, social functioning, and psychological distress (27). The description of how to apply the questionnaire can be found elsewhere (28). Participants were asked to fill out the HIT-6 on D0S1, D2S1, D7, D14 and D21.
Statistics
Average values of the 15 sites were used to assess MPS and cutaneous sensitivity values. Normality of these values was assessed through Q–Q plots, and data was found to be normally distributed. Values for MPS, cutaneous sensitivity and entropy were analyzed as the difference from baseline (D0S1). All other factors were analyzed as absolute values, and these are the levels of significance presented in the figures. MPS and entropy were analyzed by 4-way repeated measures ANOVA with the following factors: Time (D0S1, D0S2, D2S1, D2S2, D7, D14 and 21), muscle (masseter, temporalis), substance (NGF, IS) and force (0.5, 1 and 2 kg). Cutaneous sensitivity was analyzed by 3-way repeated measures ANOVA with the following factors: Time (D0S1, D0S2, D2S1, D2S2, D7, D14 and 21), muscle (masseter, temporalis), and substance (NGF, IS). Baseline differences for MPS, entropy and cutaneous sensitivity were analyzed in the same way, but without time as a factor. Pain drawings were analyzed by 3-way repeated measures ANOVA with time, substance, and muscle as factors. HIT-6 scores were analyzed by 1-way repeated measures ANOVA with time as a factor. Tukey post-hoc tests were used to adjust for multiple comparisons. Headache frequency difference from baseline was assessed by paired t-Test. Mcnemar's test for paired data was used to analyze the difference in number of sessions with and without RP. For all tests, the significance level was set at p < 0.05. Mean absolute ( ± standard error (SE)) values are reported in the figures.
Results
Mechanical sensitivity
Cutaneous sensitivity
Baseline differences were assessed by using a 2-way repeated measures ANOVA and differences from baseline were assessed with a 3-way repeated measures ANOVA with a Tukey post-hoc test. Cutaneous sensitivity showed no baseline differences for any of the analyzed factors (p > 0.22). ANOVA showed no significant differences for any of the analyzed factors when compared to baseline (p > 0.064).
Mechanical pressure/pain sensitivity
Baseline differences were assessed by using a 3-way repeated measures ANOVA and differences from baseline were assessed with a 4-way repeated measures ANOVA with a Tukey post-hoc test. At baseline, there were no significant differences in MPS between the NGF side and IS side for both the masseter and temporalis muscles (p = 0.22) (Figure 2(b)). Conversely, at baseline, the masseter muscle presented with higher MPS scores than the temporalis muscle (p = 0.005) (Figure 2(b)).
Mechanical pressure/pain sensitivity scores on the NGF and IS sides for the masseter and the temporalis muscles. All figures show mean values and Figure 2(a) and 2(b) show SE. Baseline differences were assessed using a 3-way repeated measures ANOVA, and differences from baseline were assessed using a 4-way repeated measures ANOVA with a Tukey post-hoc test. Bar graphs show NRS scores MPS scores (a) for the masseter and temporalis muscles when palpated with 0.5, 1 and 2 kg on the NGF and IS sides (*p < 0.05). Differences over time in MPS scores (b) are shown for the masseter and temporalis muscles (*p < 0.05) indicating significant differences between the NGF and IS side, and (#p < 0.05) indicating differences between D0S1 and the other assessment days.
Significant differences in MPS were found for the following factors: Time, substance, and force (p < 0.002). Interactions between time × muscle, time × substance, muscle × substance, time × force, muscle × force, time × muscle × substance and time × muscle × force (p < 0.016) were also significant. Post-hoc analyses showed a higher increase in MPS on the NGF side when compared to the IS side (p < 0.002), and a significant increase when comparing the 2 kg to the 0.5 and 1.0 kg force (p < 0.001) (Figure 2(b)). Furthermore, a significant increase in MPS was noted for the masseter muscle between the NGF side and the IS side (p < 0.001), but this difference was not present for the temporalis muscle (p = 0.43) (Figure 2(a)). Moreover, for the masseter muscle, there were higher MPS scores on the NGF injected side on D0S2, D2S1, D2S2 and D7 (p < 0.001) when compared to D0S1 (Figure 2(b)). Finally, there were significantly higher MPS scores on the NGF side when compared to the IS side on D0S2, D2S1, D2S2, D7 and D14 (p < 0.002) (Figure 2(b)).
Entropy of mechanical pressure/pain sensitivity
Baseline differences were assessed by using a 3-way repeated measures ANOVA and differences from baseline were assessed with a 4-way repeated measures ANOVA with a Tukey post-hoc test. At baseline, there were no significant differences in entropy between the NGF side and the IS side for both the masseter and temporalis muscles (p = 0.69). Furthermore, there were no significant differences between the masseter and the temporalis muscles (p = 0.086).
Entropy scores showed a significant difference for time, substance, and force (p < 0.045) as well as for interactions between time × substance, time × force and muscle × force (p < 0.004). Post-hoc analyses showed an overall significant increase in entropy on the NGF side when compared to the IS side (p < 0.001). Finally, there was a significant overall increase in entropy when comparing 2 kg with the 0.5 kg force (p < 0.038) (Figure 3(a)).
Entropy scores. All figures show mean values, and Figure 3(a) shows SE. Baseline differences were assessed using a 3-way repeated measures ANOVA, and differences from baseline were assessed with a 5-way repeated measures ANOVA with a Tukey post-hoc test. Bar graph shows entropy scores for the masseter and temporalis muscles when palpated with 0.5, 1 and 2 kg on the NGF and IS sides (*p < 0.05) (a). Entropy scores shown over time for the masseter muscle (b). Significant differences between the NGF and IS side are shown for 0.5 kg ($p < 0.05) and 1 kg (#p < 0.05) of force. (*p < 0.001) shows differences between D0S1 and other assessment days on the NGF side (b).
For the masseter muscle, there was a significant difference for time, substance, and force (p < 0.005) as well as for the interactions time × substance, time × force, substance × force and time × substance × force (p < 0.033). Post-hoc analyses showed that there was a significant increase of entropy on the NGF side when compared to the IS side (p < 0.005) (Figure 3(a)). Furthermore, there was a significant difference between the NGF and IS side for 0.5 kg and 1 kg (p < 0.049) but not for the 2 kg force (p = 0.99) (Figure 3(b)). Moreover, there were significant differences on the NGF side between D0S1 and D0S2, D2S1, D2S2 and D7 (p < 0.001) (Figure 3(b)). Finally, significant differences were seen between the NGF and IS side when assessing with 0.5 kg on D0S2, D2S1, D2S2, D7 and D14 (p < 0.023) and D7 and D14 (p < 0.025) when assessing with 1 kg of force (Figure 3(b)).
For the temporalis muscle, there was a significant difference for time, substance (p < 0.019) and for the interaction time × substance (p < 0.001). Post-hoc analyses showed that there were significantly higher entropy changes for the NGF side when compared to the IS side (p < 0.002) (Figure 3(a)).
Referred pain
Referred pain outcomes.
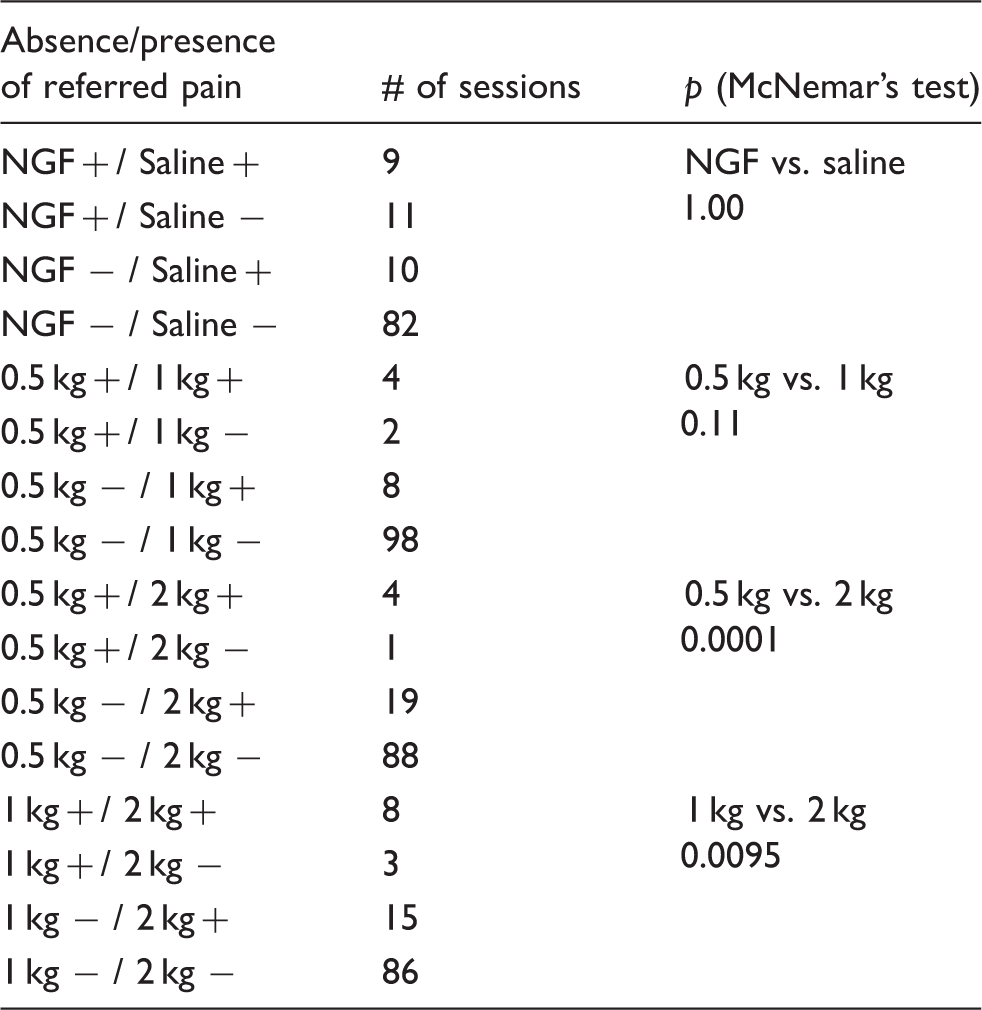
Note. + = referred pain was elicited; − = referred pain was not elicited. There was no significant difference in the presence of RP between the NGF and the IS side (p = 1.00, Mcnemar's test). There was, however, a significantly higher number of participants with RP when using the 2 kg of force when compared to both 0.5 and 1 kg (p < 0.001, Mcnemar's test).
Pain drawings
Differences from baseline were assessed with a 3-way repeated measures ANOVA and a Tukey post-hoc test. The analysis showed a significant difference in areas of perceived pain for time and substance (p < 0.004) as well as for the interaction between time × substance (p < 0.001) (Figure 4). The post-hoc analyses showed that the NGF side had a significantly bigger pain drawing area when compared to the IS side (p < 0.004) (Figure 4 and Figure 5). It also showed a bigger area on D2S1, D2S2 and D7 (p < 0.001) when compared to D0S1 on the NGF side, but no differences on the IS side (Figure 4). Also, when comparing the NFG and IS sides there were significant differences at D2S1 and D2S2 (p < 0.001), with the NGF side presenting with bigger pain areas (Figure 4 and Figure 5).
Pain drawing area. All figures show mean values, and error bars show SE. Differences from baseline were assessed with a repeated measures ANOVA with a Tukey post-hoc test. Pain drawings showed a significant difference on the NGF side when compared to D0S1 (*p < 0.001) and between the NGF and the IS side ($p < 0.001).
Assessment of headaches
No significant difference in number of headaches was found between baseline and the duration of the study (p = 0.06). No significant differences were noted for time (p = 0.86) for the HIT-6. All headaches that developed during the study period were described as an unspecific headache with a pressing quality and without migrainous symptoms such as nausea, vomiting, photophobia or phonophobia accompanying the headaches.
Discussion
The main findings of this study were: a) intramuscular injections of NGF into the masseter but not the temporalis muscle increased MPS when compared to IS injections; b) NGF injections did not cause an increase in the number of headache days; c) there was no difference in prevalence of RP between the NGF and the IS sides.
Mechanical pressure/pain sensitivity
Masseter muscle MPS scores at baseline were increased when compared to the temporalis muscle. These MPS differences have been shown before (29,30) and they could be due to different morphological and anatomical characteristics that have been previously discussed (31).
MPS scores were also increased on the NGF side when compared to the IS side for the masseter muscle. This has been previously shown for MPS in the masseter muscle (13,14). Surprisingly, there were no differences in MPS scores for the temporalis muscle when comparing the NGF and the IS sides. Increased MPS to NGF has been shown to be caused through both peripheral (32,33) and central mechanisms (34,35). More specifically, trkA (17) and TRPV1 (18,19) are the main receptors involved in mediating this mechanical hyperalgesia. It is unknown if their expression differs between the masseter and temporalis muscles. If a difference exists, it could not only explain the findings of our study, but also why some studies have reported pain on palpation to be more common in the masseter than the temporalis muscle in both temporomandibular disorders (36) and TTH patients (3).
Interestingly, for both muscles, there were no significant differences between the NGF and IS side when taking force into account. This indicates that for this experimental model there is no difference in palpating the muscles with 0.5, 1 or 2 kg when assessing MPS.
Assessment of headaches
In this study, no difference was found between the number of headaches at baseline and the number of headaches that developed after the NGF injections. On the other hand, 3 out of 14 participants in this study had a significant increase in the number of headaches compared to baseline, roughly 21% of the participants (Figure 6). This percentage is in accordance with previous studies that provoked headache in healthy individuals through a clenching task (8,9) and trapezius static contraction (7). This seems to indicate that a subgroup of individuals that do not usually suffer from headaches are prone to develop one, given a noxious stimulus. The reasons for this are unknown, but may be related to the individual pain modulation profile of the participants.
Referred pain
Three of the 16 healthy participants reported RP to mechanical stimulation before injections. RP has been reported in healthy individuals following stimulation with noxious electric stimuli (37) and injections of algogenic substances (38), but to our knowledge it has not been reported during palpation of a non-painful muscle. This finding is important, as RP is normally seen as a pathological occurrence that is elicited through activation of trigger points (39). The idea that RP may be an epiphenomenon of the muscle in response to noxious input in healthy individuals (40) is therefore strengthened by this finding.
No difference was found in incidence of RP between the NGF and the IS side. This was surprising, since it was expected that the prolonged and profound sensitization caused by the NGF would lead to increases in RP. Conversely, there was a higher incidence of RP when palpating with 2 kg compared to 0.5 and 1 kg. Even though all three forces produced MPS scores above the pain threshold, the 2 kg was the only force that on average produced MPS scores around the pain threshold (Figure 2). It could then be thought that, regardless of injected substance, what increases the chances of eliciting RP through palpation is reaching or exceeding the pain threshold. Similar results have been shown after injections of hypertonic saline (41). Furthermore, animal studies have shown that noxious stimuli can cause an increase in receptive fields, making it possible to activate latent 2nd order wide-dynamic range neurons (12). This is important because, as shown in this study, palpating with 2 kg can evoke pain in healthy individuals and cause an increase in chances of eliciting RP, regardless of the presence of muscle pathology.
Other findings
Cutaneous sensitivity
No differences were found between the NGF and IS side; as such, the skin can be excluded as a contributor to changes in mechanical sensitivity reported in this study. This has been shown before when injecting into the muscle, and is most likely due to the depth of injection (20). It can also be excluded as a contributor to differences in mechanical sensitivity between the masseter and the temporalis muscles reported in the literature (29) and in our study.
Entropy of mechanical pressure/pain sensitivity
The low entropy scores in this study ( < 1.4/2.708) suggest that MPS scores following experimentally-induced muscle sensitization are quite uniform. Regardless, both muscles had significantly increased entropy scores on the NGF side when compared to IS. This is in accordance with a previous study that showed that injections of an algogenic substance, in this case glutamate, caused an increase in entropy of MPS scores (22). Previous studies have shown that there seems to be less diversity in distribution of MPS within the temporalis in TTH patients when compared to controls (42), but no difference between episodic TTH and chronic TTH (43). This is different from what we found in this study, as the NGF side was more diverse than the saline side. This could be because, in these studies, the assessed sites were analyzed individually and not as a measure of diversity such as entropy, and because the whole of the temporalis muscle was not assessed.
Finally, for the masseter muscle, the difference between the NGF and IS side could only be noted if applying 0.5 or 1 kg of force but not 2 kg. This is also in agreement with previous findings (22). The reason for this could be that MPS scores were already quite high and diverse when applying the 2 kg force before injections and, following NGF injections, the 2 kg stimulus did not cause further increases in diversity that allowed for changes in entropy. This was not seen for the temporalis muscle and could be due to lower MPS scores of the temporalis muscle before injections. This suggests that to obtain a true picture of the diversity of MPS of the masseter muscle a force of no more than 1 kg should be used, in order to avoid ceiling effects in the assessment procedure.
Pain drawings
When looking at both muscles together, there was a bigger pain area for the NGF side when compared to the IS side (Figure 5). This increase lasted to D2S2, which is less then what has been reported for three injections of NGF into the tibialis anterior muscle (20). To our knowledge, previous studies of NGF injections into masticatory muscles have not used this method to evaluate pain areas and, as such, there is no available data to compare with. However, since the purpose of experimental pain studies are to mimic pain conditions, it is of interest to assess pain in the same way it would be assessed in the clinic, which is what we have done for the pain drawings.
Participants perceived areas of pain in the different sessions. The left side corresponds to the NGF side and right side corresponds to the IS side. Number of headache days for each individual participant. The number of headache days within the last 30 days is represented in white, and the number of headache days during the experimental period is represented in grey. X axis numbers represent each individual participant.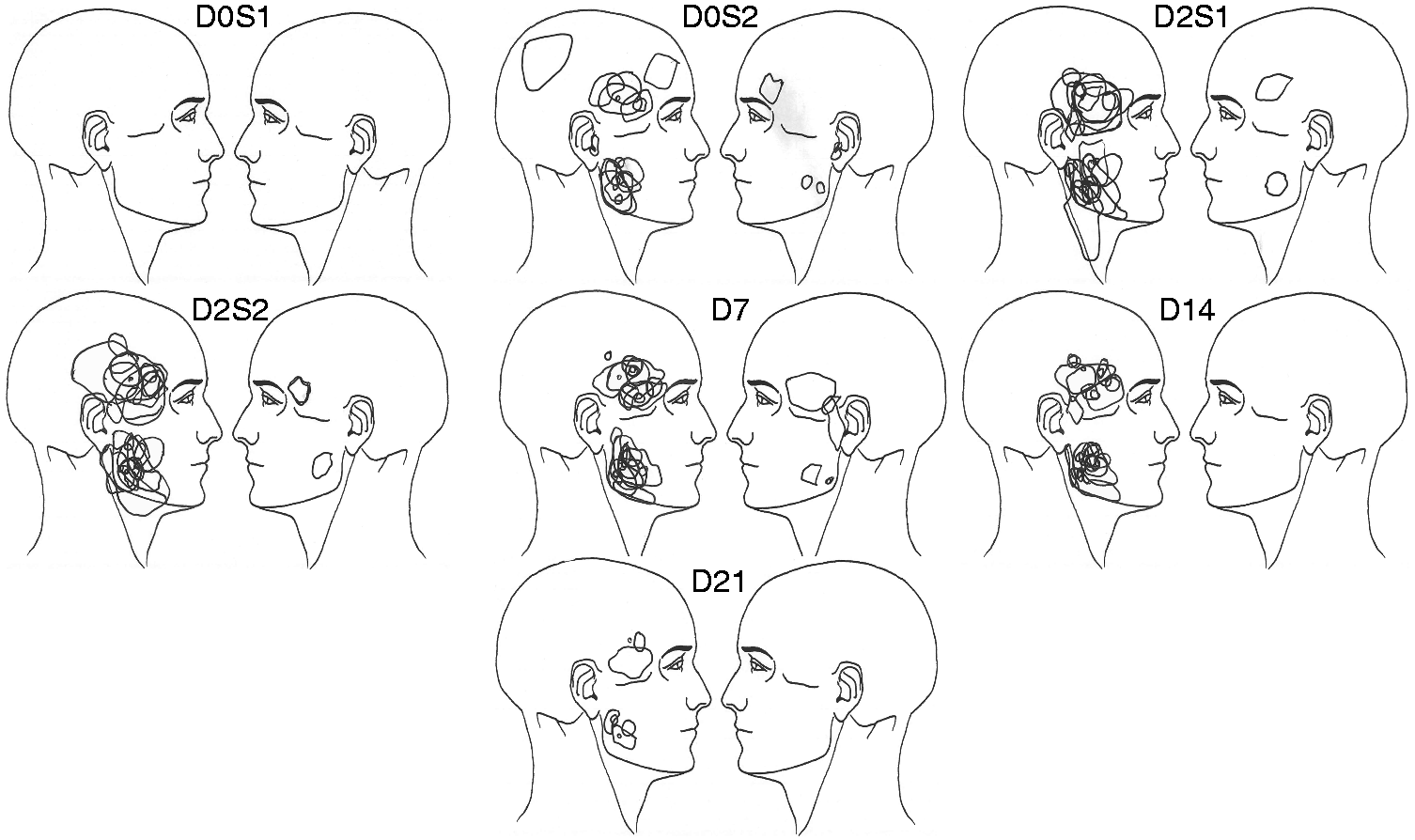

Special considerations and limitations
It needs to be acknowledged that even though we used a NGF-evoked pain model that mimics some characteristics of pericranial muscle pain (14), we cannot directly extrapolate these results to TTH and myalgic TMD patients as the assessed variables may present differently in chronic pain patients.
The fact that participants received bilateral injections does not allow us to discard that the NGF-sensitized side may have influenced the control side, as it has been shown that unilateral nociceptive input to the masseter muscle may, to some degree, influence the contralateral side (44,45). However, robust contralateral changes in somatosensory function are usually not present in human experimental models of orofacial pain (46).
Conclusion
In summary, it was shown that repeated intramuscular injections of NGF caused an increase in MPS (for the masseter but not the temporalis) and entropy scores when compared to the IS side. Despite this, an increase in the presence of RP following palpation was not seen, nor was an increase in number of headache days. These findings support the idea that in healthy individuals RP may be an epiphenomenon of the muscle in response to noxious input.
Footnotes
Article highlights
Repeated intramuscular NGF injections caused mechanical sensitization in the masseter muscle but not the temporalis muscle when compared to IS injections.
An increase in the number of headache days was not seen following NGF injections to the masseter and temporalis muscle.
An increase in RP following palpation was not seen despite mechanical sensitization of the masseter muscle.
Acknowledgements
We would like to thank Bente Haugsted for the help with the randomization of the NGF and IS injections.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
