Abstract
Abstract
Objective
To evaluate the efficacy of high-frequency repetitive transcranial magnetic stimulation over dorsolateral prefrontal cortex as a migraine prevention by conducting a systematic review and meta-analysis.
Background
The efficacy of high-frequency repetitive transcranial magnetic stimulation over dorsolateral prefrontal cortex as preventive migraine treatment remains debatable.
Methods
PubMed, Scopus, CINAHL, CENTRAL, and BioMed Central databases were searched from their inception until December 2020. Randomised trials comparing high-frequency repetitive transcranial magnetic stimulation over dorsolateral prefrontal cortex with sham for migraine prevention were included. The risk of bias was assessed using the Cochrane guidelines. Headache days, pain intensity, acute medication intake, and disability were extracted as study outcomes and the mean difference with a random-effects model was used to determine the effect size.
Results
Meta-analysis revealed that high-frequency repetitive transcranial magnetic stimulation over dorsolateral prefrontal cortex significantly reduced acute medication intake (Mean Difference = 9.78, p = 0.02, 95%CI: 1.60, 17.96, p = 0.02) and functional disability (Mean Difference = 8.00, p < 0.05, 95%CI: 4.21, 11.79). However, no differences were found in headache days and pain intensity reduction, although there was a slight trend favouring high-frequency repetitive transcranial magnetic stimulation.
Conclusion
High-frequency repetitive transcranial magnetic stimulation over dorsolateral prefrontal cortex may be effective in reducing acute medication intake and disability. However, more studies are needed to strengthen this preliminary evidence.
Keywords
Introduction
Migraine is a common disorder that primarily impacts women and young adults. It is one of the leading causes of disability across the globe (1–3). Currently, pharmacological drugs are the default treatment for acute and preventive migraine treatments (4). However, non-pharmacological migraine treatments, including neuromodulation therapies that have fewer side effects, are being continuously explored. High frequency repetitive transcranial magnetic stimulation (HF-rTMS) is a neuromodulation therapy that relies on non-invasive brain stimulation. Moreover, it is an interesting candidate as a migraine prevention therapy. This method induces cortical excitability by generating high-frequency (>5 Hz) magnetic pulses at short intervals (5,6). It is mainly used for treatment-resistant depression (7), in addition to several other psychiatric and neurological disorders, including anxiety, chronic pain, and stroke (8–13).
The stimulation site differs in accordance with specific target protocols for the condition requiring treatment. For example, the treatment site for depression is often the left dorsolateral prefrontal cortex (DLPFC) (13). Recent neuroimaging research into major depression revealed that rTMS augmented the regional function in the left DLPFC and the average functional connectivity density of the central executive network. Furthermore, rTMS can alter the entire neural network and is not just limited to regional areas (14). In addition, functional connectivity between striatum and left DLPFC may have the ability to predict treatment responses in cases of depression (15).
In the preventive treatment of migraine, HF-rTMS is a promising option due to its safety and efficacy in relation to the treatment of depression and chronic pain, which are common comorbidities associated with migraine (5). The mechanism by which rTMS relieves migraine or headache pain has not been entirely established. Previous research suggested that DLPFC stimulation could alter glutamate/glutamine levels (16) and induce dopamine release (17). Furthermore, rTMS may be able to modulate thalamocortical signals (18) and normalise brain excitability in migraine patients (19). Randomised controlled trials in clinical contexts have demonstrated that HF-rTMS reduces migraine attack frequency and pain intensity, in addition to improving the functional disability of migraine patients when compared to placebos (20–23).
Unfortunately, different rTMS protocols and stimulation sites were employed in these studies, Hence, the optimal standard HF-rTMS protocols for migraines could not be established. Therefore, by systematically analysing the HF-rTMS studies according to a specific protocol, evidence for the most reliable and practical HF-rTMS protocol could be determined. For example, a recent systematic review of HF-rTMS application over motor cortex in migraine cases (24) suggested that HF-rTMS may be an effective preventive treatment with a good safety profile, despite the fact that the protocols and outcomes adopted in the included studies varied. However, no systematic review or meta-analysis has yet been conducted in relation to DLPFC. Therefore, the effectiveness and safety of HF-rTMS at the DLPFC remain unclear. In order to address this gap in current scholarship, the current review seeks to evaluate the efficacy, safety, tolerability, and functional ability of HF-rTMS at DLPFC as a preventative therapy in cases of episodic and chronic migraine in comparison to sham treatments. Specifically, the results from randomised control trials (RCTs) are examined.
Methods
Study design and search strategy
This review was conducted in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (25). The review protocol was registered with PROSPERO (Registration number: CRD42020220636) and has been published recently (26).
Five electronic data sources were consulted, namely: Scopus, Cumulative Index to Nursing and Allied Health Literature (CINAHL) Plus, PubMed, Cochrane Central Register of Controlled Trials (CENTRAL) and Biomed Central. The search period comprised the inception of the databases until 31st December 2020. In addition, the following keyword and search term strategy was employed: rTMS OR “repetitive transcranial magnetic stimulation” AND headache*, rTMS OR “repetitive transcranial magnetic stimulation” AND migraine*, rTMS OR “repetitive transcranial magnetic stimulation” AND hemicran* and rTMS OR “repetitive transcranial magnetic stimulation” AND “migraine disorders.” Moreover, a backwards and forwards criteria search was performed, and two trial registries were also consulted, namely: the World Health Organization trial registry and clinicaltrial.gov.
Eligibility criteria
The following eligibility criteria were applied in accordance with the PICO (Patient, Intervention, Comparison and Outcome) strategy.
Outcomes (O)
Exclusion criteria
Only full-text articles published in the English language were selected for inclusion in this review. Study protocols and conference proceedings were excluded. Studies that did not examine migraines as a primary condition and non-DLPFC stimulation areas, such as the motor cortex, were also excluded.
Study selection, data extraction, and the risk of bias assessment
All citations for the articles were downloaded into Endnote software. Title and abstract searching were independently performed by two reviewers (NIMS and NAM). Relevant articles were selected, and full texts were retrieved for further evaluation. Disagreements were discussed with a third reviewer (WAWS). Data extraction was conducted independently by two authors (NIMS and NAM). Essential data, such as first author, publication year, study location, number of participants, treatment sessions, treatment duration, frequency of rTMS, stimulation intensity, type of rTMS coil, type of sham, principal findings, and adverse effects were extracted from all the studies. Study authors were contacted for additional relevant information during the data extraction process.
Four reviewers (AMAR, AHKYK, WCL and JB) assessed the risk of bias in each study using the Version 2 of the Cochrane risk-of-bias tool for Randomised trials (RoB 2). Bias in each domain was assessed in accordance with the written articles and information supplied by study authors. The risk of bias was classified as low risk, high risk, or of some concern based on the algorithm applied in each domain.
Data synthesis
We reported our data using mean difference (MD) ± standard deviation (SD). The MD of all primary outcome measures refers to the mean differences, which gauge the absolute differences between the mean values of two groups. This can be calculated by subtracting the mean of post-interventional measurements from the corresponding mean of baseline measurements. Furthermore, the MD of secondary outcome measurements (functional disability) refers to the mean difference between two groups, which was calculated by subtracting the mean of the baseline measurement from the corresponding mean of post-interventional measurements. The SD of the endpoint measurements was used if the SD of the corresponding MD could not be calculated or was missing. For trials that included sham interventions, HF-rTMS at DLPFC, and several other interventions were used (27). Data was only calculated only sham interventions and HF-rTMS at DLPFC.
When comparable data was available from multiple studies, the findings were combined. In addition, evaluations were performed twice. On the first occasion, per-protocol (PP) analysis was used when total patient numbers were combined in accordance with the total number of patients randomised in the studies. The second time involved the employment of intention-to-treat (ITT) analysis wherein total patient numbers were combined in accordance with the total number of patients that were randomised in studies. Cochrane Collaboration’s software program Review Manager V.5.4.1 was adopted for the meta-analysis, wherein a random-effect model was employed. In order to evaluate heterogeneity, I2 was employed, wherein a p-value of less than 0.05 was regarded as significant.
Results
Search results
The detailed process of study selection is presented in the PRISMA flow diagram in Figure 1. In total, 1576 articles were identified. Once the duplicate articles (n = 767) were removed, only 809 articles were available for independent screening (by NIMS and NAM) through title and abstract selection. Subsequently, twenty-nine articles were deemed eligible for retrieval. Of the included studies, fourteen were then excluded due to non-relevant data and another six were excluded because the full article was not available. In addition, two studies were excluded because they comprised conference abstracts (28,29), one study was excluded because it was a study protocol (30), and another was omitted because it was not published in the English language (31).
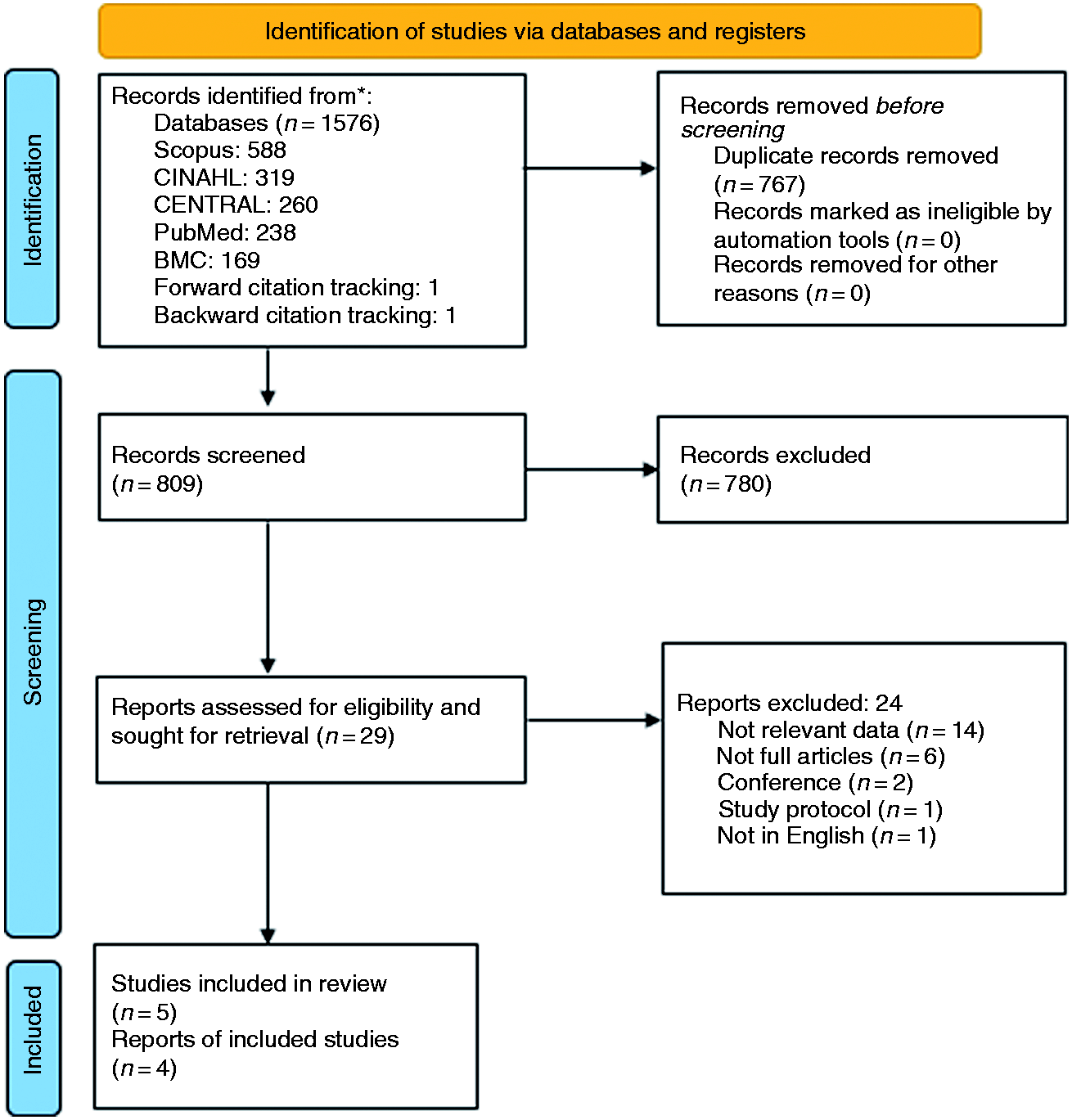
PRISMA 2020 flow diagram.
Study characteristics
The baseline characteristics are presented in Table 1. A total of five sham controlled trials conducted between 2004 and 2020 were included in the systematic review. In addition, 143 participants were included in the intention to treat (ITT) analysis and only 128 participants were included in the per protocol (PP) analysis. The mean age of participants was 34.7 years, with a range from 34.4 to 47.0 years across the five studies. Whilst one study did not provide information about participant genders (33), the overall figure for the review indicates that 82.7% of the participants were women. Study durations averaged three months, the range extending from two months to four months.
Basic baseline characteristics of the included studies.

Data are presented as mean ± SD. BDI: Beck Depression Index, EG: Experimental group, ITT: Intention-to-treat, NA: Not available, NPRS: Numeric Pain Rating Scale, MIDAS: Migraine Disability Assessment, PP: Per Protocol, SG: Sham group, STAI: State Anxiety Inventory.
The included studies comprised research from four nations. Specifically, two were conducted in Italy (32,33), one originated in Brazil (34), one was conducted in Bulgaria (27), and one was from Egypt (20). Four studies consisted of research trials into chronic migraines (27,32–34). Only one study involved episodic migraines (20). Headache days, pain intensity, medication intake, and functional disabilities were adopted as outcome measurements in 80% of studies, whereas headache frequency was recorded in just 40% of the studies.
Intervention characteristics
Active HF-rTMS vs sham
Table 2 presents a summary of the therapeutic sessions and study environments for all the included studies.
Details of the study settings and treatment sessions of the reviewed studies.

All five trials involved left DLPFC stimulation using figure-of-eight coils for active interventions with high frequencies ranging from 5–20 Hz, with a mode of 20 Hz. The rTMS sessions averaged 11 session days, ranging from 5 to 23 days. Three of the studies delivered the sessions in five consecutive days, while the other two studies delivered them on alternate days. The mean pulses used were 820 pulses, ranging from 200 to 1600, over an average of 19 trains, ranging from 1 to 40, with 30-s intertrain intervals. The motor threshold used ranged from 70% to 110%. Only one study used an authentic sham stimulator (33), while the other four studies used the angle manipulation stimulation method, wherein the coil angle differed from the angle in the active rTMS stimulation. All the studies reported outcomes at baseline followed by monthly time-points with all studies having at least one month time-point measurement after intervention. Four studies ascertained the exact placement of the electromagnetic coil with the use of the 5 cm method where a point was located 5 cm in front of the hand motor hotspot, while one study did not mention its localisation method (27).
Treatment efficacy
Three of the five studies included in the systematic review presented positive findings that backed the use of HF-rTMS at DLPFC. One of these three studies exhibited a low bias risk. The meta-analysis encompassed three different outcome measures that could reflect the treatment efficacy. Thus, the mean difference of headache days, pain intensity, and medication intake between the pre-intervention and post-intervention stages. However, one study was excluded (33) because it provided no information about numbers or proportions of patients in the control (sham) group and the intervention (rTMS) group when ITT analysis was employed (33).
When headache days were concerned, one study had no available data for this outcome measurement due to its data being reported in headache frequency instead of headache days, thus it had to be excluded (32).
One study provided no information about the number of headache days, although it did present information about headache frequency. Hence, this study was excluded (32). Meta-analysis of four eligible studies (n = 117) in PP analysis revealed that there was a small positive effect favouring HF-rTMS when compared to sham stimulation. Thus, the application of the DLPFC did not significantly change the number of headache days (MD 0.10, p = 0.96, 95%CI, −4.13, 4.32) and had a considerable statistical heterogeneity (p < 0.05, 75% I2 index) (see Figure 2a). Moreover, when ITT was performed (see Figure 2b), the meta-analysis of three studies (n = 116) yielded similar results to the PP analysis (MD 0.59, p = 0.81, 95%CI, −4.14, 5.33). This translates into the potential clinical significance of HF-rTMS in relation to migraine prevention treatments. However, an analysis of the data that is currently available revealed that it was not statistically significant and markedly heterogeneous.

(a) Presents a meta-analysis showing the rTMS vs sham efficacy based on reduction of headache days using PP analysis, while and (b) meta-analysis was measured using ITT analysis.
When pain intensity is employed as another primary outcome measure, both PP analysis (MD 0.43, p = 0.31, 95%CI, −0.40, 1.27) (see Figure 3a) and ITT analysis (MD 0.42, p = 0.30, 95%CI, −0.37, 1.20) (see Figure 3b) were not statistically significant. The findings for medication intake revealed that HF-rTMS significantly reduced medication consumption compared to sham medication when analysed using PP analysis (MD = 9.78, p = 0.02, 95%CI, 1.60, 17.96) (see Figure 4a). However, in accordance with ITT analysis, the result was not statistically significant (MD = 6.68, p = 0.07, 95%CI, −0.54, 13.89, p = 0.07) (see Figure 4b).

Efficacy was measured in accordance with reductions in pain intensity. (a) showed rTMS efficacy vs sham efficacy using PP analysis and (b) presents the measurements arising from ITT analysis.

The efficacy was calculated using reductions in medication intake. (a) showed the efficacy measured using PP analysis, whereas and (b) presents the calculation results using ITT analysis.
Quality of life and functional disability
Quality of life and disability comprised a secondary outcome measurement. Four out of five trials had examined the quality of life of the trial participants using patient-reported outcome instruments. Two of the trials (28,33) used the Migraine Disability Survey (MIDAS), while another two trials (20,27) used the Headache Impact Test-6 (HIT-6). However, the studies that adopted the MIDAS measurement used this at different assessment times. Hence, it was not possible to combine these results or conduct a meta-analysis.
The results from another two trials that had measured functional disabilities using the HIT-6 questionnaire were then amalgamated for meta-analysis. In both meta-analyses, the two trials included were homogeneous and the results from the PP analysis (MD 8.00, p < 0.05, 95%CI, 4.21, 11.79) (see Figure 5a) and the ITT analysis (MD 8.00, p < 0.05, 95%CI, 4.37, 11.63) (see Figure 5b) revealed a statistically significant improvement after rTMS treatment. Hence, it is possible to conclude that rTMS at DLPFC lessens the burden associated with migraines by improving functional ability of the trial participants.

Quality of life and disability were measured using a reduction of HIT-6. (a) presents the meta-analysis that was conducted with the use of PP analysis and (b) encompasses the results of the meta-analysis that were calculated using ITT analysis.
Treatment tolerability and safety
HF-rTMS at DLPFC was generally well-tolerated, with 128 of 143 total participants completing the scheduled treatment. Thus, the overall discontinuation rate was 10.5%. Two studies reported that there were no adverse effects associated with the treatment. Three studies reported the presence of adverse effects which were recorded as mild to moderate. These are outlined in Table 3. Moreover, the effects did not have any serious outcomes, such as seizures, hospitalisations, or fatalities.
Reported adverse events and drop-out.

EG: Experimental group, SG: Sham group.
Risk of bias of included studies
All five domains of risk of bias were examined across five studies. Potential bias can arise from randomization processes, deviations from intended interventions, missing outcome data, outcomes measurements, and the selection of reported results. The current study focused on the effect of assignment to intervention in respect of the risk of bias assessment in this review. The results for the risk of bias assessment of the five randomised trials are summarised in Figure 6.
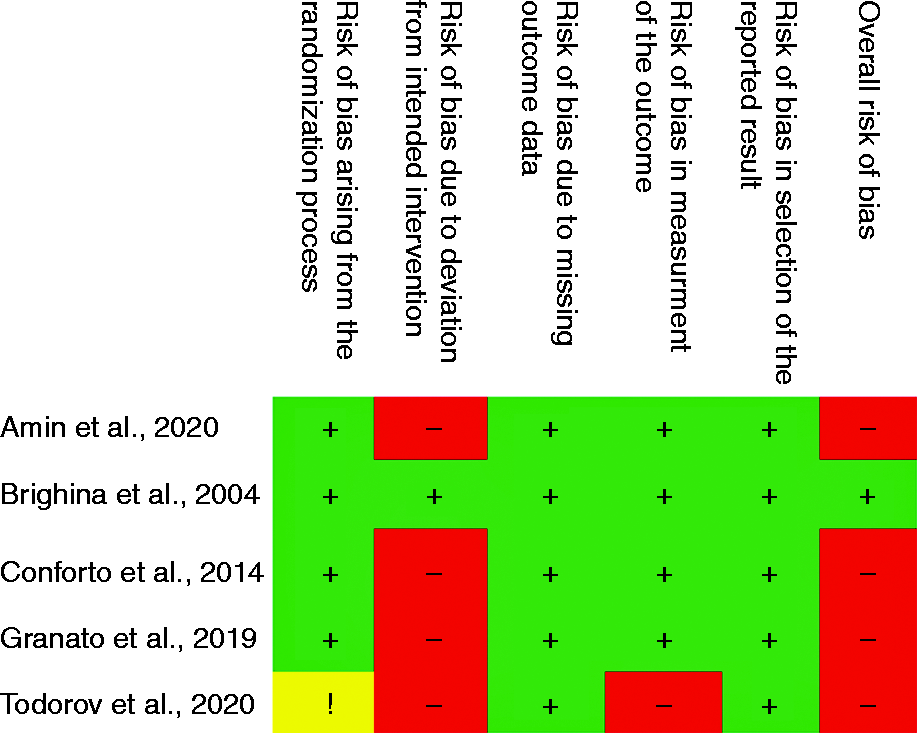
Risk of bias assessment of the reviewed studies using Cochrane Risk of Bias 2 tools.
Bias arising from the randomisation process were low for four of the double-blinded randomised trials and one of the studies which is an open-label randomised trial had raised some concerns, therefore is deemed of high risk of bias (27). Specifically, this study did not provide information about sequence allocation concealment. In relation to the risk of bias in deviation from the intended intervention, only one trial presented with a low risk of bias (32). The remaining four studies had a high risk of bias. This was primarily due to the per-protocol analysis. In a similar vein, most of the included studies were deemed to have a low risk of bias arising from missing outcome data. In the fourth domain, only one study had a high risk of bias, again due to open-label study design (27). Moreover, with the risk of bias in relation to the selection of the reported result, all studies had a low risk of bias. When the overall risk of bias was calculated, four trials had a high risk and only one study (32) had a low risk of overall bias.
Discussion
This review appraised the efficacy and safety of HF-rTMS applications in relation to the DLPFC for migraine preventive treatments, thereby suggesting that there is evidence that HF-rTMS over DLPFC reduced either the intake of acute medication or the presence of disability among patients with episodic and chronic migraine. However, it does not reduce the number of headache days or pain intensity. One possible explanation may be that HF-rTMS improves anxiety related to migraine attacks via the modulation of nucleus accumbens (NAc) and the regulation of glutamate and dopamine levels, which may reduce the intake of acute medications and improve the quality of life (15–19). It would be interesting if data on the presence of psychiatric comorbidities as a confounding factor for the results of their trials were available. However, the trials did not measure psychiatric comorbidities as confounding factors. Most of the studies excluded participants with psychiatric comorbidities, and two of the studies failed to examine psychiatric aspects through specific questionnaire items (28,33).
An additional outcome measure in the initial review protocol was employed to gauge quality of life (26) because most RCTs had included it after the IHS guidelines recommended its inclusion. In addition, disability was employed as a secondary outcome in the clinical trials. Although both MIDAS and HIT-6 administered in the included RCTs were validated questionnaires that were recommended for evaluation of quality of life and functional disability (35–37), only two studies using HIT-6 with similar time-points could be included. Nonetheless, the positive result was significant and accorded with large effects. With regards to safety and tolerability, HF-rTMS at DLPFC was found to be safe and well-tolerated in relation to neuromodulation therapy for migraines. The overall discontinuation rate was low, and all the reported adverse events were non-life threatening. The common adverse events to be reported were all reversible and short lived. These included headaches, dizziness, and sleepiness.
Unlike this current review, which is specific at HF-rTMS at DLPFC in relation to migraines, previous systematic reviews and meta-analyses (39–41) included broad neuromodulation treatments for headaches and migraines. A previous meta-analysis conducted in 2016 (39) demonstrated that TMS was not effective when compared with shams. Another meta-analysis in 2017 (40) showed that TMS was significantly effective as a treatment for acute migraines. However, the review concluded that the rTMS protocols used were too heterogeneous and the result also had statistical heterogeneity. Moreover, a more recent systematic review revealed that rTMS was more promising than tDCS and recommended that HF- rTMS either at DLPFC or motor cortex, should be investigated further (41).
This review is the first meta-analysis to exhibit significant improvements in acute medication intakes and disability with the HF-rTMS neuromodulation treatment at DLPFC among migraine patients. This indicates that there is evidence that it can be employed as an effective migraine prevention treatment for some patient groups. However, these findings should be considered cautiously because there is high statistical heterogeneity. Moreover, most studies were associated with a high risk of bias. The limited quality of the analysed outcomes and statistical insignificance could be due to the clinical and methodological diversity of the available studies, in addition to the small study numbers. For example, one study (32) failed to include headache days and pain intensity. Hence, it was excluded from the analysis. Similarly, in the ITT analysis of medication intake, one study (33) was excluded on the grounds that it might produce non-significant findings (p = 0.07).
An important methodological diversity factor identified among the studies is the type of sham intervention used. Only one study used an rTMS sham stimulator (33), whilst others used the angle manipulation method as the blinding method or placebo. This poses a problem because the ideal sham should be able to copy the look, sound, and feel of active HF-rTMS without the presence of a significant electromagnetic field. Sham is indeed a pertinent issue in rTMS studies. The blinding of both subjects and examiners means that potential experimental and subject bias may arise. All subjects were typically provided with a patient information sheet detailing the potential experience to which they might be exposed during the trial, including clicking sounds and skin vibratory sensations. As a result, those who received the suboptimal sham rTMS may believe that they were not receiving any treatment and/or they were not in the treatment group.
Patients who had received TMS in the past might guess the treatment correctly, while those who were unfamiliar with TMS are usually unable to guess the right treatment (38). Therefore, it is ideal to only include TMS-naïve subjects in rTMS studies. However, only one study included in the meta-analysis had adopted this approach (20). In addition to subject and experimental bias, the issue with the tilting of the active TMS coil introduced the possibility of unwanted significant brain stimulation (38). This is exemplified by either the nocebo effect or the occurrence of adverse events, irrespective of the fact that the sham group that was observed in two studies used this technique, which might be due to the presence of an electromagnetic field suitable for brain stimulation.
The current study has limitations, the first of which is that the results were acquired from a small sample size, even after all the participants had been combined. This limits the analysis. For example, by evaluating migraines in a homogeneous fashion despite the awareness that there was not enough statistical power to differentiate between major clinical subgroups, such as episodic and chronic migraines, in addition to migraines without or without auras or depression. Furthermore, the use of unstandardised rTMS protocols for migraine prevention with resultant equivocal outcomes of comparable protocols (39) also comprises a limitation. While it is tempting to overemphasise the importance of attribute factors, such as the lack of treatment fidelity, this may perhaps reflect the heterogeneity of migraineurs in terms of neuroanatomical and neurophysiological differences as present in certain neuropsychiatric disorders, such as major depressive disorders, where up to 30% of patients are non-compliant with multimodal treatments (42).
The strengths of this analysis include a focused clinical question, the prospective registered protocol, the thorough search strategy, and a review process, in addition to the fact that no previous reviews have specifically addressed the clinical question we sought to appraise. Although the included studies were clinically and methodologically diverse, meta-analyses were able to show that HF-rTMS over DLPFC significantly reduced the acute medication intake and improved quality of life and functional ability. Future studies should address the heterogeneity and the lack of standardisation in the methodology of previous studies, in addition to formulating research questions to identify the profile of rTMS treatment responders and the duration of remission after effective treatments.
Conclusion
To conclude, HF-rTMS at DLPFC appears to be beneficial in relation to the reduction of acute medication intake and migraine disability. Moreover, it exhibits a good safety profile and tolerability. Therefore, this approach should be included in attempts to seek effective migraine prevention therapies in order to address the issues of acute medication intake and major migraine disability. However, it must be acknowledged that any evidence is in the preliminary stages. Thus, future research into the effectiveness of HF-rTMS in migraine treatments should include more expansive sample sizes.
Clinical Implications
This review is the first meta-analysis to exhibit significant improvements in acute medication intakes and disability with the HF-rTMS neuromodulation treatment at DLPFC among migraine patients. The limited quality of the analysed outcomes and statistical insignificance could be due to the clinical and methodological diversity of the available studies, in addition to the small study numbers. The strengths of this analysis include a focused clinical question, the prospective registered protocol, the thorough search strategy, and a review process, in addition to the fact that no previous reviews have specifically addressed the clinical question we sought to appraise. Although the included studies were clinically and methodologically diverse, meta-analyses were able to show that HF-rTMS over DLPFC significantly reduced the acute medication intake and improved quality of life and functional ability.
Footnotes
Author contributions
Data availability statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Research Management Centre, Universiti Putra Malaysia (Grant Number GPB/2017/9585500).
