Abstract
Background
The role of the NOTCH3 p.R544C variant, the predominant variant of cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy in multiple East Asian regions, in migraine is unknown.
Methods
Migraine patients (n = 2,884) (2,279F/605M, mean age 38.8 ± 11.7 years), including 324 (11.2%) with migraine with aura, were prospectively enrolled by headache specialists according to the International Classification of Headache Disorders criteria. These patients and 3,502 population controls free of stroke, dementia, and headache were genotyped for NOTCH3 p.R544C by TaqMan genotyping assay or Axiom Genome-Wide TWB 2.0 Array. Clinical manifestations and brain magnetic resonance images were examined and compared between migraine patients with and without NOTCH3 p.R544C.
Results
Thirty-two migraine patients (1.1%) and 36 controls (1.0%) harbored the p.R544C variant, and the percentages were comparable among migraine patients without and with aura, and controls (1.2%, vs. 0.6% vs. 1.0%, p = 0.625). Overall, migraine patients with and without the p.R544C variant had similar percentages of migraine with aura, headache characteristics, frequencies and disabilities. However, those with p.R544C were less likely to have pulsatile headaches (50.0% vs. 68.2%, p = 0.028), and more likely to have moderate to severe white matter hyperintensities in the external capsule (18.8% vs. 1.2%, p = 0.006) and anterior temporal lobe (12.5% vs. 0%, p = 0.008).
Conclusions
Our findings suggest that NOTCH3 p.R544C does not increase the risk of migraine with aura, or migraine as a whole, and generally does not alter clinical manifestations of migraine. The role of NOTCH3 variants, as well as potential influences from ethnicity or modifier genes, in migraine needs to be further clarified.
Introduction
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is an inherited cerebral small vessel disease caused by mutations in the NOTCH3 gene (1,2). Most of the pathogenic NOTCH3 variants result in a loss or gain of a cysteine residue within one of the 34 epidermal growth factor-like repeat (EGFr) domains of the NOTCH3 protein (3), including the p.R544C variant (4). Various clinical presentations have been reported in patients with CADASIL, including ischemic stroke, intracerebral hemorrhage, cognitive dysfunction, psychiatric disorders, and migraine (1,5). However, the correlations between genotypes and phenotypes remain inadequately understood.
Migraine is one of the common manifestations of CADASIL, occurring in 30%-75% of Caucasian patients with CADASIL (6). However, the prevalence of migraine in CADASIL patients is below 6% in some East Asian countries, including China, Korea and Taiwan (7–9), and the figures are not higher than that in the general population (9.1%) (10). In addition, even though the pooled prevalence of migraine among Caucasian CADASIL patients is 43% (11), estimates in some studies (14–18.5%) (3,12) are closer to those in the general population (12–15%) (13,14). It is also widely believed that the association between CADASIL and migraine is stronger in patients with migraine with aura (MA), and the reported proportions of MA among Caucasian CADASIL patients with migraine are usually >80% (11,15–17). The prevailing theory underlying the association between CADASIL and migraine aura is that NOTCH3 mutation-related vascular dysfunction leads to increased susceptibility to cortical spreading depression (11,18). However, only one out of the three CADASIL patients diagnosed with migraine had MA in our prior study (n = 112) (7), and none of the CADASIL patients with migraine or probable migraine reported aura in a Korean series (n = 53) (8). Even though there is a well-established association between migraine and CADASIL in Caucasian populations, whether the association between migraine and CADASIL could vary with different ethnicities or different NOTCH3 genetic variants deserves further elucidation.
CADASIL is an uncommon disease in Caucasians, with an estimated population prevalence of about 2–5 per 100,000 (19–22). Even though it has become evident that cysteine-altering NOTCH3 variants are much more common than previously thought, as demonstrated by recent studies analyzing population databases, i.e., 1.4–2.2 per 1,000 (3,23,24), such a low prevalence still makes it difficult to explore the association between migraine and NOTCH3 pathogenic variants in the general population. On the other hand, cysteine-altering NOTCH3 variants, particularly p.R544C, are relatively common in East Asian populations (22). The p.R544C variant is the predominant variant associated with CADASIL in Taiwan and some other East Asian regions (7,8,25). It has been estimated that the population prevalence of the pathogenic NOTCH3 p.R544C variant is 0.7–0.9% in East Asia, including Taiwan (4,22). In fact, the prevalence of one single genetic variant, i.e., p.R544C, in East Asian populations is much higher than that of all of the NOTCH3 variants taken together in Caucasian populations. Besides, variants in Caucasians are spread across different regions of the NOTCH3 gene, rather than concentrated in a particular region. By taking the unique advantage of the high prevalence of p.R544C in Taiwan, the present study examined the association between p.R544C and migraine at a population scale with the following specific aims: i) determining the prevalence of the NOTCH3 p.R544C variant among a large cohort of migraine patients, ii) comparing the proportions of the p.R544C variant between migraine patients and non-headache population controls, and iii) evaluating the differences in clinical and radiological manifestations between migraine patients with and without the p.R544C variant.
Methods
Migraine patients
Patients were recruited from the Headache Clinic of Taipei Veterans General Hospital, a tertiary medical center providing medical services for veterans and civilians. The initial evaluation involved questionnaire-based interviews by research assistants, followed by face-to-face evaluations by headache specialists. The diagnoses of MA, migraine without aura (MO), and chronic migraine (CM) were based on the criteria of the Second, Three-beta, or Third Edition of the International Classification of Headache Disorders (ICHD-2, ICHD-3β, or ICHD-3) (26,27). Patients with a history of stroke, transient ischemic attack (TIA), or dementia were excluded. Genomic DNA samples were extracted from peripheral white cells collected from the patients.
The present study included two chronologically divided cohorts of migraine patients recruited by the same group of investigators (YFW, SPC, JLF, WTC, KLL, and SJW). All of these patients were recruited for an ongoing migraine genome-wide association study (GWAS) project. Even though the patients were recruited in a veterans’ general hospital, 99.6% of the study participants were civilians. The first cohort consisted of 699 patients who were previously genotyped with a genome-wide array without the single nucleotide polymorphism (SNP) rs201118034 (i.e., NOTCH3 p.R544C variant), and therefore the NOTCH3 p.R544C variant was investigated by a specific TaqMan genotyping assay separately. The second cohort comprised 2,215 individuals, and were genotyped with the Axiom Genome-Wide TWB 2.0 Array (Affymetrix, Santa Clara, CA), which included the rs201118034 SNP.
Questionnaire-based interview
A specifically designed questionnaire was used to collect clinical data from the patients, including demographics, past medical histories, and headache profiles, etc., as part of the routine evaluation for headache patients at their first visit. Headache-related disabilities were measured by using the Migraine Disability Assessment (MIDAS) scale (28). Psychological disturbances were screened by using the Hospital Anxiety and Depression Scale (HADS) (29).
Controls
DNA samples of 3,502 stroke-, dementia-, and headache-free controls were obtained from the Taiwan Biobank, a nationwide research database designed to collect genetic and biomedical information from 200,000 Taiwanese individuals aged between 30 and 70 years from the general population (30,31). The samples and clinical information were coded and delinked from personal identifiers. Only individuals reporting the absence of a history of stroke, dementia, and headache and hemicranias on the questionnaire were included. Genotyping of the NOTCH3 p.R544C variant was performed using a TaqMan genotyping assay.
TaqMan genotyping assay
Genotyping of the NOTCH3 p.R544C variant of the first migraine cohort and population controls was carried out with a TaqMan genotyping assay (C_190521044_10, Applied Biosystems, Foster City, CA) as previously described (4). Polymerase chain reaction (PCR) was performed in 96-well microplates on an ABI 7500 real-time PCR system (Applied Biosystems, Foster City, CA), and the alleles were discriminated by fluorescence detection using System SDS software version 1.2.3 (Applied Biosystems, Foster City, CA). Samples with the p.R544C variant detected by the TaqMan genotyping assay were further confirmed by Sanger sequencing using Big Dye 3.1 dideoxy terminator on an ABI Prism 3700 Genetic Analyzer (Applied Biosystems, Foster City, CA).
Axiom Genome-Wide TWB 2.0 Array
Genotyping in the second cohort was carried out by using the Axiom Genome-Wide TWB 2.0 Array (Affymetrix, Santa Clara, CA), which included 648,611 autosomal SNPs designed for the Taiwan Biobank (30,31), at the National Center for Genomics Medicine, Academia Sinica, Taiwan. The genotyping data of rs201118034 (i.e., the NOTCH3 p.R544C variant) on the Axiom Genome-Wide TWB 2.0 Array were retrieved.
Brain magnetic resonance imaging (MRI)
Brain MRIs were available from 16 of the 32 migraine patients (age 41.6 ± 14.3 years, range 17–65, 12F/4M) (50%) carrying the p.R544C variant, and none of these patients had a history of stroke, hypertension, diabetes, or tobacco use. Subsequently, p.R544C-negative migraine patients, for whom brain MRIs were available, were matched to p.R544C-positive migraine patients at a ratio of 10:1 based on age, sex, migraine diagnosis (MA/MO/CM), and the status of stroke, hypertension, diabetes, or tobacco use. The severity of white matter hyperintensities (WMHs) on fluid-attenuated inversion recovery (FLAIR) or T2-weighted images was rated by using the modified Scheltens scale focusing on the external capsule (EC) and anterior temporal lobe (ATL) as described previously (4,32,33). A score was assigned for each side separately based on the following scale: 0 = absent, 1 = up to five lesions <3 mm, 2 = six or more lesions <3 mm, 3 = up to five lesions 4–10 mm, 4 = six or more lesions 4–10 mm, 5 = one or more lesions >10 mm, and 6 = confluent hyperintensities. Moderate or severe WMHs were defined as a modified Scheltens scale ≥3 on one or both sides (33).
Statistical analysis
Descriptive data are expressed as the mean ± standard deviation or number (n) (percentage). Genotype distributions were tested for Hardy-Weinberg equilibrium by using the chi-square goodness-of-fit test. Continuous variables were compared by using Student’s t test, or the Mann-Whitney U test for non-normally distributed data. Categorical variables were compared by using the Chi-square test or Fisher’s exact test. Comparisons were made between migraine patients and population controls, as well as between migraine patients with and without the p.R544C variant. The Mantel-Haenszel test was used for comparisons of the percentages of p.R544C between migraine patients and controls stratified by age groups. Logistic regression modeling was carried out to estimate the odds ratios (ORs) and the 95% confidence intervals (CIs) for having migraine in individuals carrying the p.R544C variant. Statistical analysis was carried out by using IBM SPSS Statistics for Windows, version 24.0 (IBM Corp., Armonk, NY, USA). Statistical significance was defined as a two-sided p of <0.05.
Ethics approval
The study protocol was approved by the institutional review board of Taipei Veterans General Hospital (VGHTPE IRB No. 2010-06-013IA, 2011-11-002GA), and all of the patients enrolled provided written informed consent before entering the study.
Results
Patient characteristics and headache profiles
In total, 2,884 migraine patients (2279F/605M, mean age 38.8 ± 11.7 years) were recruited, including 324 patients (236F/88M, mean age 36.5 ± 11.5 years) (11.2%) with MA, and 1,026 patients (840F/186M, mean age 41.9 ± 12.9 years) (35.6%) with CM (Table 1). The mean age at migraine onset was 20.3 ± 20.2 years. The average headache frequency was 11.9 ± 9.2 days per month, and the average frequency of acute medication use was 7.1 ± 7.9 days per month.
Clinical profiles of two cohorts of migraine patients.
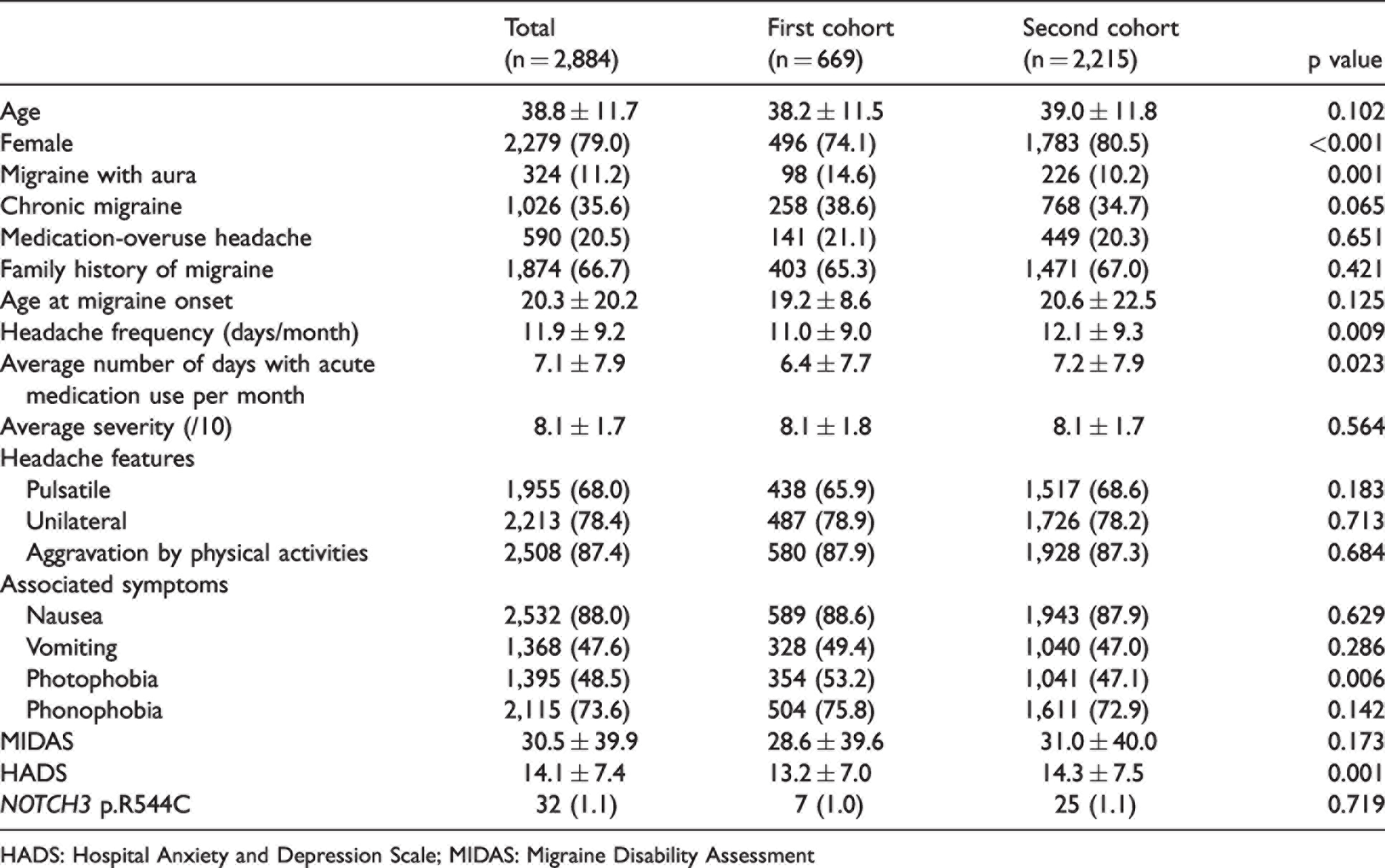
HADS: Hospital Anxiety and Depression Scale; MIDAS: Migraine Disability Assessment
The first cohort consisted of 669 patients (496F/173M, mean age 38.2 ± 11.5 years), including 98 patients with MA (14.6%) and 258 patients with CM (38.6%) (Table 1). The second cohort consisted of 2,215 patients (1783F/432M, mean age 39.0 ± 11.8 years), including 226 patients with MA (10.2%) and 768 patients with CM (34.7%). The demographics and clinical profiles of these two cohorts were similar, although there were slight differences.
Prevalence of NOTCH3 p.R544C in migraine patients and non-headache population controls
In the first cohort (n = 669), 7 patients (5F/2M, mean age 36.7 ± 15.4 years) (1.0%) carried the p.R544C variant, whereas in the second cohort (n = 2,215), the p.R544C variant was present in 25 individuals (21F/4M, mean age 39.0 ± 11.4 years) (1.1%). In total, 32 of the 2,884 migraine patients (1.1%) harbored the p.R544C variant (Table 2), including one patient who was homozygous for the variant. In comparison, 36 of the 3,502 controls (2124F/1378M, mean age 49.8 ± 10.7 years) (1.0%) carried the p.R544C variant, and all of them were heterozygous for the variant. As compared with controls, patients with migraine were younger (p < 0.001) and were more likely to be female (p < 0.001). The percentages of p.R544C were comparable between the migraine patients and controls (p = 0.752, Chi-square test) (Table 2), and the findings were consistent after taking age into consideration (p = 0.976, Mantel-Haenszel test) (Supplemental Table). The proportions of subjects carrying the p.R544C variant were comparable among patients with MO, patients with MA, and controls (1.2%, vs. 0.6% vs. 1.0%, p = 0.625).
Comparisons between migraine patients and controls.
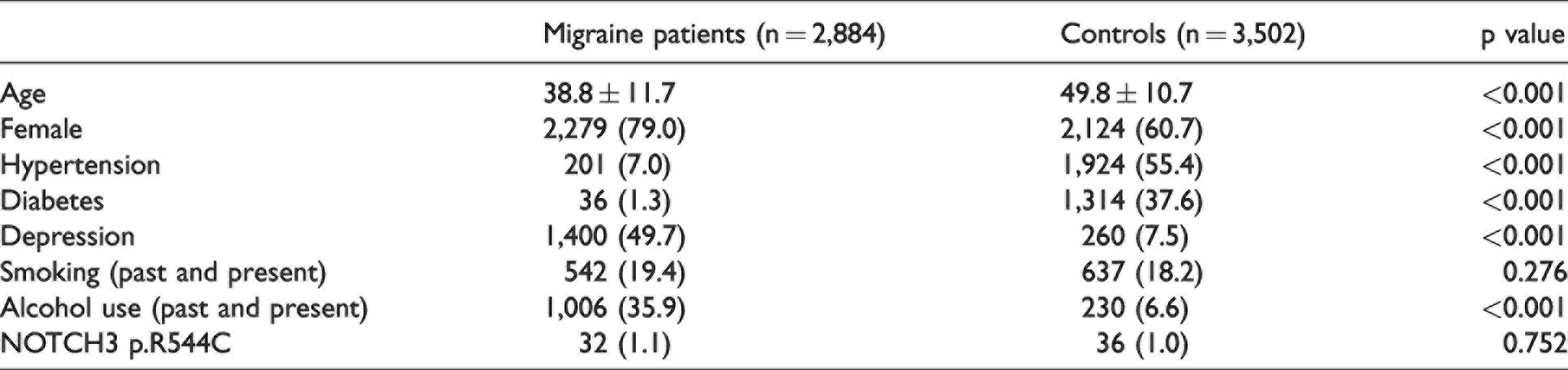
Subsequently, the study participants were stratified by their age (<40/≥40 years). For study participants aged <40, the prevalence of p.R544C was comparable among MO patients, MA patients, and controls (1.3% vs. 1.0% vs. 0.9%, p = 0.751). For those aged ≥40, the percentages of p.R544C were not different among these three groups (1.1% vs. 0% vs. 1.1%, p = 0.542). In addition, harboring the p.R544C variant was not predictive of the presence of migraine (OR = 1.08 [95% CI = 0.67–1.74], p = 0.752), even after controlling for potential confounders, including age, sex, alcohol use, the presence of depression, and family history of migraine (adjusted OR [aOR] = 1.29 [0.56–2.97], p = 0.548). In the entire study population, the presence of the p.R544C variant did not increase the risk of MA (OR=0.56 [0.14–2.31], p = 0.427; aOR = 0.55 [0.13–2.29], p = 0.408). The findings were consistent when patients with MO were excluded (OR = 0.60 [0.14–2.50], p = 0.481; aOR = 0.26 [0.02-4.12], p = 0.341).
Comparisons between patients with and without NOTCH3 p.R544C
The proportions of patients diagnosed with MA (6.3% vs. 11.2%, p = 0.572) or CM (34.4% vs. 35.4%, p = 0.887) were similar between migraine patients with and without the p.R544C variant (Table 3). On the other hand, patients harboring the p.R544C variant were less likely to have pulsatile headaches (50.0% vs. 68.2%, p = 0.028). However, the age, sex, other headache features, associated symptoms, frequencies of headache and acute medication use, headache-related disabilities, and symptoms of anxiety and depression were comparable between the two groups. Among the 16 migraine patients (age 41.6 ± 14.3 years, 12F/4M) carrying p.R544C with brain MRI available, three patients (1F/2M, mean age 56.0 ± 10.0 years) (18.8%) had moderate to severe WMHs on brain MRI, including the only patient who was homozygous for NOTCH3 p.R544C. This patient underwent brain MRI at the age of 42 years, which revealed WMHs rated as 6 in the EC and 3 in the ATL using the modified Scheltens scale (Table 3) (Fig. 1A). MRIs of 160 age- and sex-matched migraine patients without p.R544C were reviewed. Ten (6.3%) and two (1.3%) of them were rated as 1 and 3 in the EC, respectively, on the modified Scheltens scale, and two (1.3%) scored 1 in the ATL. The percentages of moderate to severe WMHs, i.e., ≥3 on the modified Scheltens scale, in the EC (18.8% vs. 1.2%, p = 0.006) and in the ATL (12.5% vs. 0%, p = 0.008) were higher in patients with the p.R544C variant than in those without.
Comparisons between migraine patients with and without the NOTCH3 p.R544C variant.

HADS: Hospital Anxiety and Depression Scale; MIDAS: Migraine Disability Assessment.
aModerate or severe white matter hyperintensities were defined as modified Scheltens scale ≥3 on one or both sides.
bFisher’s exact test.

Distribution of cerebral white matter hyperintensities in the corona radiata, external capsule, and anterior temporal lobe in (A) a migraine patient with homozygous p.R544C variant in NOTCH3, (B) another migraine patient with heterozygous p.R544C variant, and (C) and (D) migraine patients without the p.R544C variant.
Discussion
In the present study, 1.1% of the 2,885 migraine patients harbored the NOTCH3 p.R544C variant, and the findings were consistent in two independent migraine cohorts using different genotyping methodologies, i.e., TaqMan genotyping assay and genome-wide SNP array. The prevalence of individuals with the p.R544C variant was comparable among patients with MO, patients with MA, and population controls, suggesting that NOTCH3 p.R544C is not associated with the risk of migraine in the Taiwanese population. In addition, the percentages of MA and CM were similar between migraine patients with and without the variant. However, migraine patients with the p.R544C variant were less likely to have pulsatile headaches than those without. Otherwise, the headache characteristics, frequencies, and disabilities were similar. Our study failed to provide supportive evidence for an association between the NOTCH3 p.R544C variant and migraine in the Taiwanese population, and the presence of the p.R544C variant did not significantly alter the headache phenotype in migraine patients.
One of the most important strengths of the present study was the sample size, i.e., 2,884 migraine patients and 3,502 controls, which reduced selection bias, and more importantly, gave a more panoramic view and more accurate delineation of the association between the NOTCH3 p.R544C variant and migraine. In fact, considering the low prevalence of p.R544C in the study population, a sufficient sample size is crucial to give adequate statistical power for such a study. In addition, the second cohort replicated the findings from the first cohort, and the percentages of p.R544C were consistent. In contrast, with a few exceptions (17,34,35), most prior studies evaluating the association between migraine and CADASIL, or particular NOTCH3 variants, involved fewer than 150 patients (7,8,11,15,16,36). Besides, our study design allowed a direct comparison of the risks of migraine between individuals with and without the NOTCH3 p.R544C variant, whereas most prior studies were restricted to migraine prevalence or headache features in CADASIL patients (11,35) or in patients with p.R544C (8). In addition, the current study recruited “regular” migraine patients diagnosed by headache specialists, whereas most CADASIL patients in prior studies were recruited due to stroke, dementia, or abnormal brain MRI findings. Patients in those studies were typically in their 50s and had a low headache frequency (8,11,17), e.g., less than once a month in 64.4% of MO patients in one of the largest series (n = 378) (17). Therefore, our findings could have a more practical impact for clinicians treating headache patients.
The frequency of the NOTCH3 p.R544C variant in migraine patients was comparable to that in headache-free controls from the general population. In addition, the presence of p.R544C variant did not increase the risk of migraine, and the clinical presentations in migraine patients with or without the p.R544C variant were similar (Table 3). Interestingly, the percentages of patients with migraine were not significantly different between individuals with and without cysteine-altering NOTCH3 variants in an analysis of the UK Biobank published recently (23). These findings might be suggestive of the lack of an association between pathogenic NOTCH3 variants, including p.R544C, and migraine even in non-Asian populations. In particular, the p.R544C variant is a cysteine-altering NOTCH3 variant, and its pathogenicity has been validated by hundreds of CADASIL patients presenting with stroke, dementia, and extensive WMHs (4,7,37,38).
On the other hand, an association between NOTCH3 variants and MA is commonly reported in Caucasian populations (17,35). Unexpectedly, the present study did not provide supportive evidence of such an association. The prevalence of the p.R544C variant was not different among patients with MO, patients with MA, and controls, and the proportions of MA were comparable between migraine patients with and without the p.R544C variant (Table 3). The finding was in keeping with a Korean series, in which none of the CADASIL patients diagnosed with migraine had aura (8). In addition, in a recently published series involving 118 cases with cysteine-altering NOTCH3 variants and 184 randomly selected controls from the Geisinger DiscovEHR Initiative Cohort, the proportions of MA were similar between cases and controls (4.2% vs. 6.0%, p = 0.61) (3). Also, the proportions of MA in migraine patients were similar between cases (5/22 = 22.7%) and controls (11/51=21.6%) (3), both of which were within the range of those reported in the general population, i.e., 20–40% (39). However, these findings were not consistent with the well-known association between MA and CADASIL or pathogenic NOTCH3 cysteine-altering variants, and more studies are needed for further exploration of these associations.
The mechanisms underlying the discrepancy between the current study and some prior reports are uncertain. The p.R544C variant is located at the boundary of the 13th and 14th EGFr domains of NOTCH3 protein, and could be associated with milder manifestations and a broader phenotypic spectrum than NOTCH3 variants located in the EGFr domains 1–6 (40). However, the prevalence (35.6% vs. 32.6%, p = 0.638) and age of onset (30.6 vs. 26.6 years, p = 0.24) of MA were not different between patients carrying the NOTCH3 variants in EGFr domains 1–6 and those with variants in EGFr domains 7–34 in a large European cohort (n = 664) (40). On the other hand, as the prevalence of migraine is lower in Asians than in Caucasians (10), ethnic differences could play an important role in modifying the clinical manifestations of genetic variants associated with migraine. In addition, individuals harboring NOTCH3 cysteine-altering variants could present with highly variable phenotypes (24,40,41), which could indicate a potential role for modifier genes. According to recently published data from the UK Biobank, although NOTCH3 variants were associated with increased risks of stroke, vascular dementia, and increased WMH volumes, the phenotypic spectrum could range from full-blown CADASIL to nonpenetrance (23,24). Therefore, genetic modifiers could have a substantial impact on the clinical manifestations of individuals carrying NOTCH3 variants. Whether the association between the p.R544C variant and migraine could be modified by other genetic factors related to cortical excitability or ionic homeostasis deserves further study. Even though the presence of the p.R544C variant generally did not alter the headache phenotype in migraine patients, it was associated with a lower percentage of pulsatile headache. Whether this could be attributed to altered endothelial function or autoregulation involved the pathophysiology of CADASIL (42,43) remains to be further clarified.
There are some limitations of this study. First, the migraine patients were younger than the non-headache population controls (mean age 38.8 ± 11.7 vs. 49.8 ± 10.7 years, p < 0.001), and could be too young to develop the full-blown clinical features of CADASIL. However, the present study focused on migraine, rather than cerebral vascular events or dementia, and the migraine patients in the present study were probably not too young considering the age of onset of MA or MO in prior series (17,35,40). In addition, age was taken into account when analyzing the data, and the findings were consistent. Second, recall bias and misclassification bias could compromise the credibility of our findings. The clinical profiles of non-headache controls were based on questionnaires rather than formal medical or neurological evaluation, and it might be possible that some of them could have migraine. Likewise, the presence of comorbidities in migraine patients was based on questionnaires and self-reports, and it could not be excluded that some of them might have a history of clinically silent stroke or TIAs with relatively trivial clinical manifestations. However, such misclassification bias in the controls could only have a limited impact on the present study, as even in migraine patients, the percentage of the p.R544C variant was only 1.1%. In addition, the p.R544C variant is more common in patients with cerebral vascular disease, i.e., 2.1% (n = 17/800) for ischemic stroke and 6.5% (n = 16/245) for small vessel occlusion in our prior series (4), and further exclusion of migraine patients with an unidentified history of stroke or TIA might further decrease the estimated prevalence of the p.R544C variant in migraine patients in the present study. Third, the percentage of MA in migraine was much lower than that reported in Caucasians (39), which could lead to concerns about diagnostic accuracy and generalizability of our findings. Nevertheless, in the present study, MA was seen in 11.2% of migraine patients, which is consistent with that in the general population (12.5%) in Taiwan (44). Fourth, whether there was a family history of CADASIL was not included in the questionnaires for migraine patients or non-headache controls. However, a family history of migraine was taken into consideration in the evaluation of the association between migraine and the p.R544C variant by using logistic regression analysis. Besides, since the study screened p.R544C directly, whether the genetic variant was present would be more important and relevant than whether there was a family history of CADASIL. Finally, WMHs in CADASIL patients typically start to accumulate in their 40s (36), and some of the migraine patients with brain MRIs available in our cohort might have been too young (mean age 41.6 ± 14.3 years) to develop diffuse WMHs. However, the comparisons of brain MRIs were made between age-, sex-, and migraine diagnosis-matched migraine patients with and without the variant. Besides, p.R544C-positive migraine patients had more severe WMHs than those without the p.R544C variant, which is also consistent with the pattern of WMHs in CADASIL.
In conclusion, the current study did not provide supportive evidence for an association between the NOTCH3 p.R544C variant and migraine, including MA. Further studies are needed to clarify the role of NOTCH3 variants, as well as the potential influences from ethnicity or modifier genes, in the pathogenesis of migraine.
Article highlights
The prevalence of the NOTCH3 p.R544C variant is not different between migraine patients and non-headache population controls. The presence of the NOTCH3 p.R544C variant in migraine patients does not significantly alter the clinical manifestations, except for more severe cerebral white matter hyperintensities. Whether the association between migraine and NOTCH3 variants or CADASIL per se could be modified by ethnicity or other genetic factors warrants further study.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221080891 - Supplemental material for Mutation screening and association analysis of NOTCH3 p.R544C in patients with migraine with or without aura
Supplemental material, sj-pdf-1-cep-10.1177_03331024221080891 for Mutation screening and association analysis of NOTCH3 p.R544C in patients with migraine with or without aura by Yen-Feng Wang, Yi-Chu Liao, Yi-Shiang Tzeng, Shih-Pin Chen, Jiing-Feng Lirng, Jong-Ling Fuh, Wei-Ta Chen, Kuan-Lin Lai, Yi-Chung Lee and Shuu-Jiun Wang in Cephalalgia
Footnotes
Acknowledgement
We would like to express our gratitude to the study participants for their help.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: YFW has received honoraria as a speaker from Taiwan branches of Allergan/AbbVie, Eli Lilly, Novartis, Pfizer, Sanofi, UCB, and Viatris, and Orient EuroPharma. He has received research grants from the Taiwan Ministry of Science and Technology, and Taipei Veterans General Hospital. SJW has served on the advisory boards of Daiichi-Sankyo, Eli Lilly and Novartis; has received honoraria as a moderator from Allergan/AbbVie, Pfizer, Eli Lilly, Biogen and Eisai and has been the PI in trials sponsored by Eli Lilly, Novartis, and Allergan/AbbVie. He has received research grants from the Taiwan Minister of Technology and Science (MOST), Brain Research Center, National Yang Ming Chiao Tung University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan, Taipei Veterans General Hospital, Taiwan Headache Society and Taiwan branches of Eli Lilly, Novartis, and Pfizer. YCLiao, YST, SPC, JFL, JLF, WTC, KLL, and YCLee reported no disclosures relevant to the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored in part by Taiwan Ministry of Science and Technology [MOST 109-2314-B-075 -054 and MOST 110-2314-B-075 -041 -MY3 (to YFW), and MOST 104-2314-B-010-015-MY2, MOST106-2321-B-010-009, MOST 107-2321-B-010-001, MOST 108-2321-B-010-014 -MY2, 108-2321-B-010 001, MOST 108-2314-B-010-023-MY3, and MOST-110-2321-B-010-005 (to SJW)]; and Taipei Veterans General Hospital [VGH 108-C-092, VGH 109-C-096, and VGH 110-C-111 (to YFW)]; this work was also supported by the Brain Research Center, National Yang Ming Chiao Tung University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
