Abstract
Objective
Several genetic variants have been found to increase the risk of restless legs syndrome (RLS). The aim of the present study was to determine if these genetic variants were also associated with the comorbidity of RLS and migraine in patients.
Methods
Thirteen single-nucleotide polymorphisms (SNPs) at six RLS risk loci (MEIS1, BTBD9, MAP2K5, PTPRD, TOX3, and an intergenic region on chromosome 2p14) were genotyped in 211 migraine patients with RLS and 781 migraine patients without RLS. Association analyses were performed for the overall cohort, as well as for the subgroups of patients who experienced migraines with and without aura and episodic migraines (EMs) vs. chronic migraines (CMs). In order to verify which genetic markers were potentially related to the incidence of RLS in migraine patients, multivariate regression analyses were also performed.
Results
Among the six tested loci, only MEIS1 was significantly associated with RLS. The most significant SNP of MEIS1, rs2300478, increased the risk of RLS by 1.42-fold in the overall cohort (p = 0.0047). In the subgroup analyses, MEIS1 augmented the risk of RLS only in the patients who experienced EMs (odds ratio (OR) = 1.99, p = 0.0004) and not those experiencing CMs. Multivariate regression analyses further showed that rs2300478 in MEIS1 (OR = 1.39, p = 0.018), a CM diagnosis (OR = 1.52, p = 0.022), and depression (OR = 1.86, p = 0.005) were independent predictors of RLS in migraine.
Conclusions
MEIS1 variants were associated with an increased risk of RLS in migraine patients. It is possible that an imbalance in iron homeostasis and the dopaminergic system may represent a link between RLS incidence and migraines.
Introduction
Restless legs syndrome (RLS) is a disease that is characterized by an imperative urge to move the legs. RLS mostly affects individuals at night and leads to disturbed sleep. The urges are exacerbated by lying down and are accompanied by unpleasant sensations in the lower limbs. Temporary relief is achieved with leg movements (1). The prevalence of RLS is estimated to range from 4% to 29% in the general population (2), yet has been found to affect a greater number of individuals in the populations of Western countries (5%–10%) (3) compared with Asian populations (1%–4%) (4). Currently, the pathophysiology of RLS remains unknown; although a range of comorbidities have been associated with RLS, including renal failure, pregnancy, iron deficiency anemia, migraine, depression, and Parkinson’s disease (1,5).
In several studies, an association between migraines and RLS has been documented (5,6). For example, patients experiencing migraines have exhibited a 1.2-fold to 3.5-fold higher risk of RLS compared to patients who did not report migraines (6,7). In addition, a greater incidence of RLS has been associated with individuals who reported chronic migraines (CMs) compared with those who reported episodic migraines (EMs) (5,8). The incidence of RLS has also been found to positively correlate with the burden of migraine severity as measured by the Migraine Disability Assessment Scale (MIDAS) (8). Dopaminergic system dysfunction has been proposed to be a common pathophysiological mechanism that mediates both migraines and RLS (9). In particular, the A11 dopaminergic nucleus of the dorsal-posterior hypothalamus was previously implicated in an animal model of RLS (10), and also in the tonic inhibition of nociceptive neuronal firing in the trigemino-cervical complex (11).
RLS has strong genetic components with an estimated heritability of 50% (12). Correspondingly, genome-wide association studies (GWAS) in diverse populations of Northern European origin identified six susceptible loci of RLS. These loci included: the BTB domain containing 9 gene (BTBD9), the Meis homeobox 1 gene (MEIS1), the protein tyrosine phosphatase receptor type D gene (PTPRD), the mitogen-activated protein kinase kinase 5/SKI family transcriptional corepressor 1 gene (MAP2K5/SKOR1), the TOX high mobility group box family member 3 gene (TOX3), and an intergenic region on chromosome 2p14 (13–16). Furthermore, up to 80% of the genetic risks of RLS have been attributed to these genetic loci (17). Based on the results of association studies that were conducted in end-stage renal disease (ESRD) patients, variants in BTBD9 and MEIS1 conferred approximately a 1.5-fold risk for RLS in a German population, and were associated with a positive association trend for MAP2K5/SKOR1 and BTBD9 in Greek patients (18). Taken together, these findings suggest that these susceptibility genes and their related pathogenesis may be shared at least partially by both idiopathic and symptomatic RLS.
The aim of the present study was to investigate whether these genetic factors are associated with the comorbidities of RLS and migraines. Significant single-nucleotide polymorphisms (SNPs) of the six RLS susceptible loci previously identified in GWAS (13–16,18) were examined in migraine patients recruited from the general population in Taiwan. Subgroup analyses were also performed to evaluate whether genetic effects are associated with different migraine subtypes (i.e. migraine with or without aura and CMs vs. EMs).
Methods
Participants
Participants were recruited from the headache clinic of Taipei-Veterans General Hospital (Taipei-VGH, in northern Taiwan), the Living Water Neurological Clinic (in southern Taiwan), and the Neurologic Clinic of Lin-Shin Hospital (in middle Taiwan) between April 1, 2010 and September 30, 2011. Patients with symptoms consistent with the diagnoses of migraine (coded 1.1, 1.2, 1.5.1, or 1.6) according to the second edition of the International Classification of Headache Disorders (ICHD-II) (19) were eligible for this study if no secondary headache disorders were identified, except for medication-overuse headaches (MOHs). The diagnosis of CM was defined as a tension-type headache and/or a migraine-quality headache that occurred for ≥15 days per month and had a migraine frequency ≥8 days per month, with this pattern lasting for three months. The remaining patients were diagnosed with EMs (19). The study protocol used was approved by the institutional review board of Taipei-VGH and National Yang-Ming University. Each participating patient signed an informed consent before entering the study, and there was no industry support or involvement in the present study.
Assessment of migraine
All participants completed a standardized form that requested information regarding patients’ demographics and headache profile. The latter included descriptions of headache intensity, frequency, locations, characteristics, analgesics use, and accompanying symptoms. Headache severity and sleep quality were evaluated with a self-administrated MIDAS (20) and the Pittsburgh Sleep Quality Index (PSQI) (21). The five-item MIDAS questionnaire measured headache-related disabilities that had occurred within a three-month period based on time lost from employment or school, household work, and family and social activities because of headaches. Scoring for the MIDAS ranged from 0 to 270 (20). The PSQI evaluated the quality and patterns of sleep for the patients in the past month. It contained 19 self-rated questions from which seven component scores were calculated and summed to provide a global score (21). The component scores ranged from 0 to 3 and the global scores ranged from 0 to 21. Higher scores indicated worse sleep quality.
Assessment of RLS
RLS was diagnosed by using a screening questionnaire that included four essential diagnostic criteria proposed by the International RLS Study Group (IRLSSG) (22). The “yes/no”-type questions addressed: (1) if there were any urges to move the legs that were accompanied by uncomfortable sensations in the legs; (2) if the urge to move or the uncomfortable sensations were partially or totally relieved by leg movement; (3) if the urge to move or the uncomfortable sensations worsened during periods of inactivity that may have included sitting or lying; and (4) if the urge to move, or the uncomfortable sensations, worsened during the early evening or night compared with daytime, or did they occur only in the evening or at night. In addition, the presence or absence of self-reported periodic limb movements in sleep (PLMS) was also queried. The patients subsequently were asked by neurologists to establish whether a diagnosis of RLS was appropriate based on the recommendations of the IRLSSG (22) (i.e. when all four essential criteria were met and other conditions mimicking RLS were excluded).
To obtain a control group (i.e. individuals with a migraine history and without an RLS diagnosis), migraine patients with an equivocal RLS diagnosis (i.e. two or three items of the IRLSSG criteria were fulfilled) or self-reported PLMS were excluded. Secondary RLS patients were also excluded based on the detection of ferritin levels <50 ng/ml, anemia, creatinine levels >1.5 mg/dl, or pregnancy. Figure 1 shows the flowchart of the enrolled and screened patients for the present study.
Flowchart of screened and enrolled participants.
Genotyping
Previous GWAS and replication studies conducted in Caucasian populations identified 13 SNP markers for RLS. These included: four SNPs in MAP2K5 (rs1026732, rs3784709, rs6494696, and rs11635424), three SNPs in BTBD9 (rs7740763, rs9349077, and rs3923809), two SNPs in MEIS1 (rs2300478 and rs12469063), two SNPs in PTPRD (rs1975197 and rs4626664), one SNP in TOX3/BC034767 (rs3104780), and one SNP in the intergenic region on chromosome 2p14 (rs6747572) (13–16,18). Genotyping of these SNPs was performed using iPLEX® Gold primer extension assays. The results were analyzed with a Sequenome® MassARRAY® system (Sequenom, San Diego, CA, USA) by the High-throughput Genome Analysis Core Facility at the Veterans General Hospital Yang-Ming Genome Research Center. Experimental quality control and genotype calls were performed using the MassARRAY® Typer 4.0 software (Sequenom). Among the thirteen SNPs tested, rs1975197 failed at the genotype clustering stage and was not included in the analyses. Additional quality filters for the exclusion of SNPs included call rates below 95% and deviation from Hardy-Weinberg Equilibrium (HWE; p < 0.05). DNA samples were also excluded if the missing genotypes exceeded 10%.
Statistical analysis
Demographic data for the migraine patients with and without RLS were subjected to Chi-square, Fisher’s exact, or t tests. Genotypes were tested for HWE by applying Fisher’s exact test prior to data analysis using PLINK (v1.07) (23). Genotype association tests between migraine patients with and without RLS were performed by using logistic regression analyses with the assumption of an additive genetic model using PLINK software. Odds ratios (ORs) and their 95% confidence intervals (CIs) were also calculated. Permutation tests based on 10,000 replications were performed to correct for the multiple comparisons using PLINK. Association analyses were performed within each subtype category according to aura status and migraine subtype in order to test genetic associations at each SNP. Stepwise multivariate regression analyses were also performed to identify factors that were independently associated with RLS after adjusting for risk factors such as age, gender, migraines with aura status, CM status, and Beck Depression Inventory (BDI) score. Haploview (v4.2) was used to generate a linkage disequilibrium (LD) map (24). The SAS/HAPLOTYPE procedure was used to test for an association between RLS status and haplotype block.
Results
Initially, a total of 1489 migraine patients were enrolled in the present study. Subsequently, 277 patients were excluded based on an equivocal diagnosis of RLS, 42 patients were excluded because of low ferritin levels or anemia, five patients were excluded for creatinine levels being >1.5 mg/dl, and 173 patients were excluded based on self-reported PLMS. Therefore, a total of 992 participants were analyzed in the present study (Figure 1). Of these migraine patients, 211 were diagnosed with RLS and 781 were not.
Demographic data.
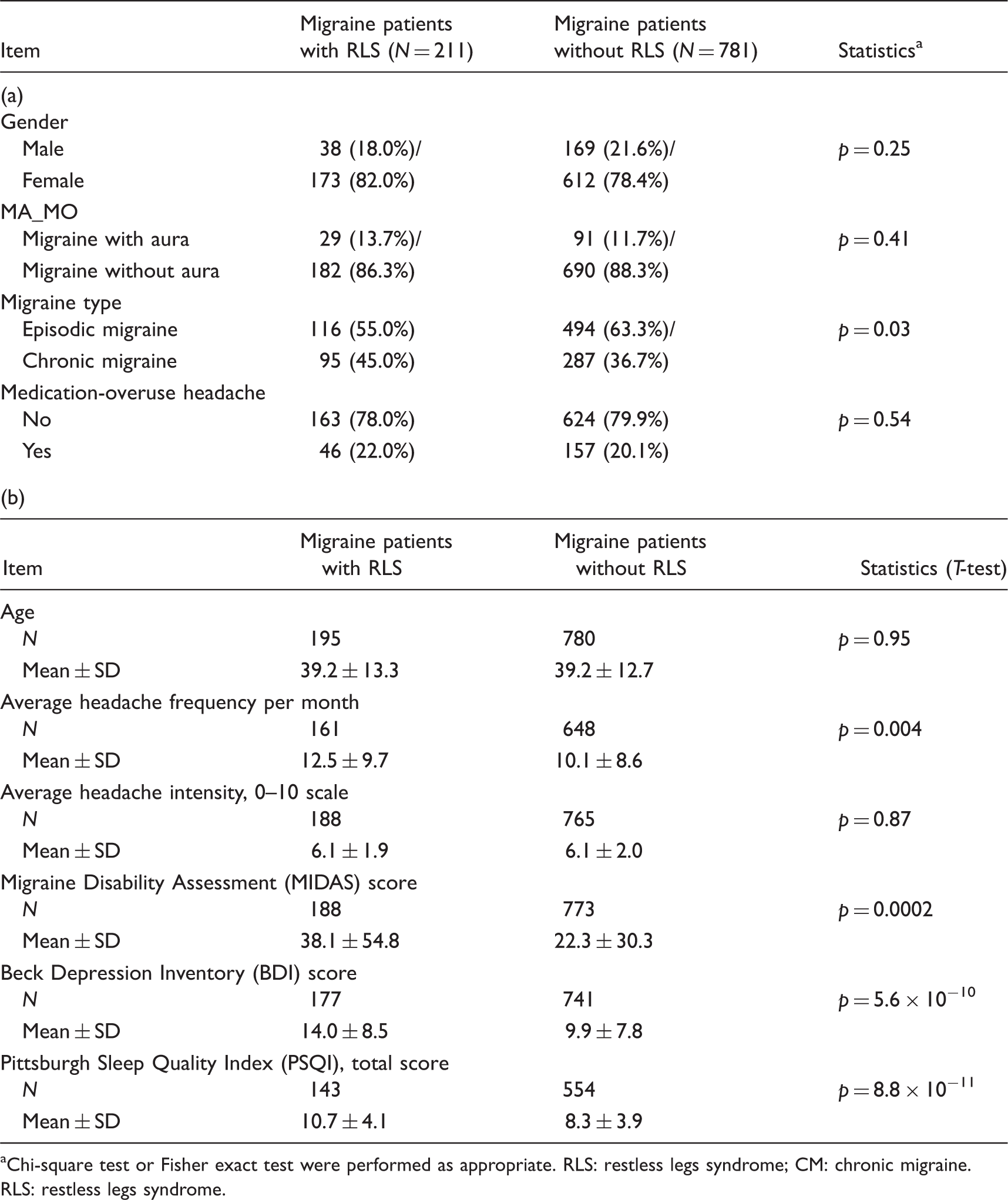
Chi-square test or Fisher exact test were performed as appropriate. RLS: restless legs syndrome; CM: chronic migraine.
RLS: restless legs syndrome.
The associations between candidate genetic variants and restless legs syndrome (RLS).

Empirical p values, ORs and CIs were calculated based on the additive effect model by logistic regression without adjustment for any risk factors. SNP: single-nucleotide polymorphism; Chr: chromosome; OR: odds ratio; CI: confidence interval.
When the participants were stratified according to migraine subtype, rs2300478 in MEIS1 was found to be significantly associated with RLS in migraine patients with aura (OR = 2.95, empirical p value = 0.0022; Figure 3(a)); while only a trend of significance was observed for the migraine patients without aura (OR = 1.29, empirical p value = 0.064). In addition, the T allele of rs2300478 carried an increased risk for RLS in patients with EMs (OR = 1.99, empirical p value = 0.0004; Figure 3(b)), and it was not a risk factor for RLS in individuals affected by CMs (OR = 1.06, empirical p value = 0.72).
Linkage disequilibrium of MEIS1. Subgroup analysis according to migraine subtypes.

Association analysis of haplotypes consisting of rs12469063 and rs2300478 with response to RLS.

RLS: restless legs syndrome.
Multivariate regression analysis.
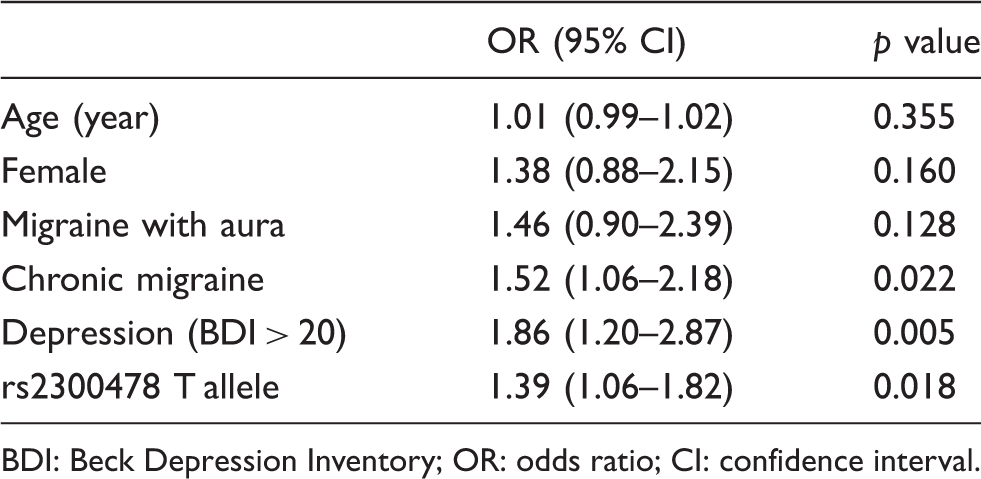
BDI: Beck Depression Inventory; OR: odds ratio; CI: confidence interval.
Discussion
To our knowledge, this is the first study to investigate the genetic contributions of RLS in migraine patients. The present data demonstrate that SNPs in MEIS1 increased the susceptibility of migraine patients to RLS, while SNPs at other susceptible loci (MAP2K5, PTPRD, BTBD9, TOX3, and the intergenic region on Ch2p14) did not. Moreover, the MEIS1 variants increased the risk of RLS in the EM patients, yet did not appear to mediate the same effect in CM patients. In addition to genetic factors, depression and CM were identified as independent and strong predictors of RLS in migraine patients.
The prevalence of RLS differs between Western and Asian populations, and it is possible that the diverse genetic backgrounds that characterize these ethnic groups are a key factor. In Western countries, genetic predisposition to RLS has been delineated in GWAS that have investigated primary RLS (14–16). Specifically, variants in the MEIS1, BTBD9, PTPRD, and MAP2K5/SKOR1 genes have been identified as risk factors of idiopathic RLS. Furthermore, the significant association between these four genes and RLS were successfully replicated in Caucasian populations in the United States and Europe (25,26). In a Korean population, the BTBD9 gene has also been found to increase the risk of primary RLS (27). Moreover, BTBD9 and MEIS1 have been found to be related to RLS in ESRD (18), and in a study of Taiwanese ESRD patients, variants of the PTPRD and TOX3 genes also tended to increase the risk of RLS (28). The current study is the first to explore the potential for genetic predispositions in migraine patients to contribute to RLS. Furthermore, the present data confirm the crucial role of two MEIS1 SNPs (rs2300478 and rs12469063) in the comorbidity of RLS and migraine.
The mechanism responsible for the link between MEIS1 and an increased RLS risk in migraines has not previously been elucidated. However, a haplotype composed of the two significant SNPs identified in the present study (rs2300478 and rs12469063) in MEIS1 has been shown to be related to decreased genetic expression of MEIS1 in the peripheral lymphocytes and brain tissues of RLS patients (29). Furthermore, reduced MEIS1 expression has been found to induce the up-regulation of genes that encode iron transporters and proteins that are responsible for ferritin production (30). Therefore, we hypothesize that the significant associations that were identified among migraine, RLS, and MEIS1 in the present study imply that aberrant iron homeostasis is a common pathogenesis shared by the two diseases.
Previous studies had demonstrated imbalance of iron homeostasis and the dopaminergic system in the migraine patients with RLS. Data from animal models, as well as from biochemical, postmortem, and imaging studies conducted in humans, supported the hypothesis that disruption of iron trafficking in the brain leads to disturbances in the striatal dopaminergic system during RLS (31). For patients who experienced chronic daily headaches that usually developed from EMs without aura, increased iron deposition was detected in the periaqueductal gray area compared with non-migraine controls (32). Brain magnetic resonance imaging also detected increased iron deposition in the basal ganglion and red nucleus of migraine patients (33). Furthermore, fluctuations in dopamine affinity that occur during migraine attacks have been found to be related to the prodrome and associated migraine symptoms (34). Iron is an important co-factor of dopamine, and an imbalance in dopamine levels before, during, or after migraine attack may possibly affect certain migraine patients who have genetic variants, particularly variants in the MEIS1 gene.
The finding that MEIS1 variants confer a risk for RLS in EM patients, and not in CM patients, awaits further consideration. In our multivariate regression analysis, both CM and depression were found to be independent predictors of RLS in addition to the genetic factors. These results are consistent with those of previous studies where the occurrence rate of RLS was found to be two-fold higher in CM participants compared with individuals who experienced EMs (34.3% vs. 16.0%, respectively) (8). In addition, CM patients were frequently affected by comorbidities, such as depression, which further increased the risk of RLS (35). Taken together, these results suggest that genetic factors may play a less important role in CMs, and the comorbidity of the disease itself may have a greater impact.
An important finding of the present study was that the risk alleles identified differed from those reported in previous studies of Caucasians. In the present study, the T allele in rs2300478 and the A allele in rs12469063 were both associated with an increased risk of RLS in migraine patients, while previous studies reported that G alleles in both of these SNPs predispose individuals in the general population to RLS (15,25,26). There are several plausible explanations for these conflicting results. First, magnetic resonance imaging (MRI) studies have detected reduced iron content in the substantia nigra, thalamus, and basal ganglion of primary RLS patients (36). In contrast, an iron overload has been observed in the periaqueductal gray and red nucleus secondary to repeated activation of the nociceptive network in migraine patients (37). Since MEIS1 directly down-regulates several iron-binding proteins including ferritin, transferrin, and divalent metal iron transporter 1 (DMT1) (30), the effects of MEIS1 on iron homeostasis may differently impact RLS between migraine patients and the general population. Second, RLS has been associated with dopamine dysfunction in the hypothalamus A11 nucleus (10), whereas migraines have been related to dopamine receptor hypersensitivity (34). Given that iron is a cofactor that is essential for dopamine synthesis, MEIS1 may also play a role in the dopaminergic system of these two comorbidities. In addition, the difference in MEIS1 risk alleles may be due to distinct minor allele frequencies and LD structures among different ethnic groups. In previous RLS association studies conducted in Korean and Taiwan, the T allele in rs2300478 was found to be overrepresented in RLS patients (27,28). The SNPs rs2300478 and rs12469063 were previously identified as surrogates that tagged the function of a nearby, conserved region within introns 8 and 9 of MEIS1 that exhibited a capacity to regulate MEIS1 expression (29). Thus, further studies are needed to test the relationship between MEIS1 and RLS in migraine patients among different ethnic groups.
There were several limitations associated with the present study. First, there were several migraine prophylactic medications and antidepressants for migraine-comorbid diseases that were taken by the patients in the present cohort, and this may have aggravated RLS symptoms and inflated the associations between RLS and CMs. Unfortunately, information regarding concurrent medication usage was not obtained in the data collected. Second, there was no difference in the distribution of hypertension, diabetes, and depression among the MEIS1 genotypes, suggesting that these comorbid diseases were unlikely to confound the association between the MEIS1 variants and RLS. Furthermore, although MEIS1 rs2300478 was shown to confer a risk to RLS in migraine, our data were insufficient to comment on its association with the severity or frequency of RLS. Third, the number of patients affected by depression was established based on the questionnaires that were administered, rather than on psychiatrist interviews. However, our findings regarding the relationship among depression, migraine, and RLS are in agreement with literature reports (38,39). Fourth, although Bonferroni correction for multiple testing was not used, the significance of both SNPs at MEIS1 persisted following the permutation tests applied. Fifth, the current sample size had limited power to detect a genetic effect with an OR less than two. However, according to a genetic study software for power estimation (http://pngu.mgh.harvard.edu/∼purcell/gpc/), our sample size provided a power of 65% for an OR of 1.40, 84% for an OR of 2.00, and 93% for an OR of 3.00, with an alpha level of 0.05. Finally, recall bias for the frequencies of positive family history in the RLS-negative migraine group may have been present in the current study.
In conclusion, the results of the present study demonstrate that rs2300478 and rs12469063 in MEIS1 significantly increased the susceptibility of migraine patients to RLS in the cohort examined. CMs, depression, and MEIS1 SNPs were also found to be independent predictors for RLS in migraine patients. Since MEIS1 expression levels may modulate the genes that encode iron transporters and proteins involved in ferritin production, imbalances in iron homeostasis and the dopaminergic system may represent plausible explanations regarding the connection between RLS and migraines. Accordingly, further studies are needed to confirm these possible connections, and to investigate whether additional causative genes may be responsible for a shared pathogenesis between RLS and migraine.
Footnotes
Clinical implications
The rs2300478 and rs12469063 in Meis homeobox 1 gene (MEIS1) were associated with a significantly higher susceptibility to restless legs syndrome (RLS) in patients with migraine.
MEIS1 expression levels may modulate genes that encode iron transporters and proteins for ferritin production, thereby suggesting that an imbalance between iron homeostasis and the dopaminergic system may represent the connection between RLS and migraines.
The T allele in rs2300478 and the A allele in rs12469063 in MEIS1 were associated with an increased risk of RLS in patients who experienced migraines, while previous studies have reported that G alleles in both of these SNPs predispose individuals in the general population to RLS.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.-L. Fuh is a member of a scientific advisory board of Novartis and Orient EuroPharma Co. and has received research support from the Taiwan Ministry of Science and Technology and Taipei-Veterans General Hospital. M-Y Chung currently has research grants from the Taiwan Ministry of Science and Technology and Taipei-Veterans General Hospital. S.-J. Wang has served on the advisory boards of Allergan, Daiichi-Sankyo, and Eli-Lilly (Taiwan). He has also received speaking honoraria from the Taiwan branches of MSD, GSK, Allergan, and Pfizer and research grants from the Taiwan Ministry of Science and Technology, Taipei-Veterans General Hospital, Novartis (Taiwan), and Daiichi-Sankyo. The other authors have nothing to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology of Taiwan (NSC 102-2321-B-010-030-, MOST 103-2321-B-010-017-); Taipei Veterans General Hospital (V100E6-001, V101E7-003, VGHUST102-G7-6-1, V103E9-006, V104E9-001); Ministry of Science and Technology Support for the Center for Dynamical Biomarkers and Translational Medicine, National Central University, Taiwan (MOST 103-2911-I-008-001); Academia Sinica (grant no. IBMS-CRC103-P04), Brain Research Center, National Yang-Ming University, Ministry of Health and Welfare (MOHW104-TDU-B-211-113-003); and a grant from the Ministry of Education, Aim for the Top University Plan.
