Abstract
Aim
First, we investigated whether the exposure to different visual feedback conditions may modulate pain perception by means of visual induced analgesia in patients with chronic migraine. Second, to comprehend the way emotional face expressions could induce visual analgesia, we evaluated the degree of identification with the four experimental conditions.
Methods
In a 1 × 4 within-subject study design, 38 female chronic migraine patients were exposed to different visual stimuli – positive face, neutral face, negative face, and control (white screen) – during a migraine attack. Visual stimuli were presented 3 times in a randomized order (each condition lasted 40 seconds). Migraine pain ratings and identification scores were assessed immediately after the observation of each visual condition.
Results
We observed a significant difference in pain ratings between the positive (median: 30, 95% CI 26.69 to 38.20) and the negative (median: 30, 95% CI 33.09 to 44.13) (z = −4.46, p < 0.0001) facial expressions or the neutral facial expression (median: 30, 95% CI 31.89 to 42.41) (z = 3.41, p < 0.001). Participants identified more with the neutral face condition than with the other conditions.
Conclusions
Observation of a positive emotional face resulted sufficient to modulate pain perception possibly via the mediation of emotion regulation for positive emotions. This study paves the way for the integration of new cognitive behavioural interventions based on the adoption of visual induced analgesia to further control pain perception in chronic migraine patients.
Introduction
Migraine is one of the most prevalent and disabling neurological disorders worldwide (1). It is characterized by recurrent attacks, which are often accompanied by nausea, vomiting, photophobia, and/or phonophobia (2). Current consensus is that migraine is an episodic, recurrent, genetically determined dysfunction of brain excitability that leads to the activation and sensitization of the trigeminovascular pain pathway (3). In most patients, migraine is present in less than 15 days/month (episodic migraine), but in a small, though clinically relevant portion of sufferers, migraine may acquire a negative evolution with headache being present of 15 or more days/month, the so-called chronic migraine (CM) (2).
Though the precise pathophysiological mechanisms of migraine are largely unknown, a relevant number of studies suggest a critical role of the thalamus in pain processing (4). Indeed, electrophysiological studies have shown a specific electroencephalographic (EEG) pattern, called thalamocortical dysrhythmia (TCD) (5) in many chronic pain conditions (6), including migraine (7). TCD is characterized by a hyperpolarization of the thalamic neurons, a low-frequency resonant recurrent interaction between thalamic and cortical neurons, and the presence of an edge effect (8). Thalamocortical (TC) pathways are important, given their role in the perception and integration of both the sensory and the emotional qualities of pain (9). Moreover, TCD has also been observed in patients suffering from complex regional pain syndrome type I, leading to a reduced cortical representation of the painful body part in the primary and secondary somatosensory cortex (SI, SII) (10). As a result of such cortical representational changes, patients with some types of chronic pain may present with internal body image (BI) distortions of the painful part of the body (11–13). Along these lines, in a preliminary study, it has been demonstrated that by reducing BI distortions via medical care (aesthetic treatments), it is possible to reduce depressive symptomatology and pain perception in CM with acute medication overuse (MO) (14). Even if interesting, these data should be considered with caution given that they cannot be generalized to the whole CM population.
To date, a large amount of pharmacological and non-pharmacological treatments have been considered for reducing pain perception in CM. Among non-pharmacological approaches, there are novel and alterative possibilities such as “visual feedback” techniques through which it is possible to determine visually induced analgesia by regulating emotions or the affective state, or modulating body perception (15–17). In fact, by observing others’ facial expressions during social interaction it is possible to effectively modulate one’s own pain perception (18). Along these lines, it has been shown that the observation of smiling (16) or of relieved (19) facial expressions may induce pain relief in healthy subjects submitted to experimental pain. Indeed, observing negative facial expressions induces abnormal gamma oscillatory activation in patients with chronic migraine, furthermore, these are negatively correlated with depressive symptom severity and migraine attack frequency (20). Furthermore, another study showed that migraine patients were hypersensitive to negative visual stimuli (21). In detail, in this study the authors found that negative affective pictures elicited stronger brain networks activation, which included the bilateral cerebellum anterior lobe/culmen, the bilateral lingual gyri, the bilateral precuneus, and the left cuneus, than neutral affective pictures in patients with migraine compared to a healthy control group (21).
The association existing between affective/emotional state and pain perception is particularly interesting and critical in CM considering the frequent comorbidity of CM and psychiatric disorders (22) such as anxiety and depression. In this regard, it is also known that most patients with CM tend to present with alexithymia (23), which is a personality trait characterized by a cognitive-affective disturbance that impacts the way individuals experience and express their emotions, internal states, and feelings (24,25). Indeed, it has been suggested that alexithymia is associated with mental disorders and negative emotions in particular (23).
In light of these considerations, in the present study we had two main goals. First, we investigated whether it is possible to modulate pain perception in CM patients through the observation of different emotional facial expressions (positive, neutral, and negative) and of a control condition (white screen). Second, for the purpose of comprehending the way emotional facial expressions could induce visual analgesia, we investigated the degree of identification with the four experimental conditions. In order to further understand the mechanisms behind these two goals, we also explored the possible associations existing between patients’ emotional/affective state at baseline with pain responses and levels of identifications. We hypothesized that the emotional condition associated to the observation of the positive face would decrease pain perception in patients with CM. We also expected that patients would identify more with those experimental conditions involving emotional faces instead of the white screen control condition.
Methods
Participants and study design
This study was conducted from January 2019 to December 2020 at the Pavia Headache Science and Neurorehabilitation Centre (a tertiary referral centre) of the Mondino Foundation in Pavia, Italy. The final study population comprised 38 Caucasian female patients with CM. Only females were included in this study because of the reported differences on pain perception between males and females (26). Inclusion criteria were: (a) 18–65 years of age, (b) fulfilment of the ICHD-3 criteria (27) for CM. Exclusion criteria were: (a) dementia, (b) previous diagnosis of psychosis, and (c) intellectual disability, based on medical records. An expert neurologist verified the eligibility criteria during the recruitment process based on history, headache diaries, and neurological evaluation. The study was approved by the local Ethics Committee and written informed consent was obtained from all patients. This study was carried out according to the Declaration of Helsinki (28).
Patients who fulfilled inclusion/exclusion criteria signed a written informed consent and were instructed to contact the Center at the onset of the next migraine attack to plan the experimental session in the subsequent 24 hours. All patients received the advice to avoid any abortive medication until the study visit. On the experimental day, patients who had a VAS score between 30 and 70 completed a baseline psychological evaluation. For all patients without a VAS score between 30 and 70 or who took an abortive medication the visit was postponed to the next migraine attack. The psychological evaluation lasted approximately 30-40 minutes, for the collection of the following variables:
presence/absence of mood (major depressive disorder, dysthymia, bipolar disorder I/II) and anxiety (agoraphobia, generalized anxiety disorder, panic disorder, specific phobia, social phobia, post-traumatic stress disorder, separation anxiety disorder) disorders based on Diagnostic and Statistical Manual of Mental Disorders criteria (29) via a clinical interview; headache-pain level with the visual-analogue scale (VAS) (0-100) (30); anxious and depressive symptomatology using the Hospital Anxiety and Depression Scale (HADS) (31); positive and negative affective state using the Positive and Negative Affect Schedule (PANAS) (32); body image perception using the Body Image questionnaire (BIQ) (33); alexithymic traits with the Toronto Alexithymia scale (TAS-20) (34); and headache-related disability with the Migraine Disability Assessment questionnaire (35).
Once the psychological evaluation was over, patients started the experimental session. This was a within-subjects study, with 1 × 4 factorial design (see Figure 1). Patients were exposed to the four different visual conditions (see Experimental session section) and 1) immediately after, rated the corresponding pain levels with the VAS and 2) reported their level of identification with the observed visual condition on a Likert scale (36), ranging from −3 (no identification at all) to 3 (total identification). Both these assessment scales (i.e., pain rating and identification question) were displayed in the centre of the computer screen for three seconds consecutively. Participants’ ratings were promptly annotated by the experimenter.

Experimental session
During the experimental session, patients were comfortably seated in front of a computer and were asked to relax and fixate on the center of the screen placed in front of them. The background of the computer screen was black throughout the experiment. The experiment consisted of observing a video clip composed of three blocks lasting eight and a half minutes. Each block consisted of four experimental conditions presenting the three different emotional faces (positive, neutral, negative) and the white screen (see Visual stimuli section) presented in a counterbalanced order. Each experimental condition lasted 40 seconds. The whole experimental session lasted between 15–20 minutes.
Visual stimuli
Emotional facial expressions (see Figure 2) and the white screen condition were displayed in a video clip created with a video editing software (Wondershare™ Filmora). Emotional facial expressions were selected from the Karolinska Directed Emotional Faces (KDEF) database (37). The KDEF is a set of 4,900 pictures of human facial expressions, created at the Karolinska Institutet, Stockholm (for more details, see www.facialstimuli.com). The set contains pictures of 70 individuals (35 females and 35 males) displaying seven different emotional expressions (neutral, happy, angry, sad, disgusted, surprised, and fearful). Each expression is viewed from five different angles (full left profile, half left profile, straight, half-right profile, and full right profile) and was photographed in two sessions. The pictures are colour photographs. The models were Caucasian amateur actors (mean age, 25 years; range 20–30 years) who were instructed to pose with the different expressions. In this study, we chose only three emotional expressions: positive (positive emotion), neutral (neutral emotion), and disgusted (negative emotion), from three females, viewed from a straight profile. We conducted a short preliminary study with healthy subjects to select images more able to express happiness, neutral, or disgusted emotion for this study.

Experimental visual conditions. The experimental visual conditions consisted of three emotional faces (positive, neutral, negative) and one white screen control condition.
Statistical analysis
The primary outcome measure was pain rating assessed with the 100-point VAS, while secondary outcome measures were the identification question score and the psychological variables assessed at T0. All statistical tests were performed in Stata 13 (StataCorp LP, College Station, TX, USA). This was a mixed-effects design, with fixed-effects in ‘condition’, emotional face (positive, neutral, negative) and white screen, and with random effects over the individual subjects. We analyzed differences in pain ratings across conditions and differences in pain ratings from baseline (delta VAS = post condition minus baseline VAS scoring) with a multilevel mixed-effects linear regression test (the “mixed” function in Stata). Furthermore, in order to observe differences between emotional faces, we ran a pairwise comparison with the Scheffe test for multiple comparisons. In order to observe differences in the identification question scores across conditions, we used multilevel mixed-effect ordered logistic regression test (the “meologit” function in Stata). Moreover, Spearman’s correlation tests were used to evaluate associations between patients’ psychological state at T0 with pain levels and identification question in the four experimental conditions. Statistical significance was considered p < 0.05 for two-tailed calculation.
The sample size calculation was based on the primary outcome (VAS) and a previous study (38) obtaining a baseline VAS (0–10) value of 8.8 ± 1.7. We did not expect a significant difference between exposure to white screen and neutral expression; whereas we expected a VAS decrease (7.9 ± 1.7) in the positive emotion condition and an increase (9.7 ± 1.7) in the negative emotion condition. To this end, a total sample size of 38 patients was considered sufficient to ensure 80% statistical power and 95% confidence.
Results
Participants’ characteristics
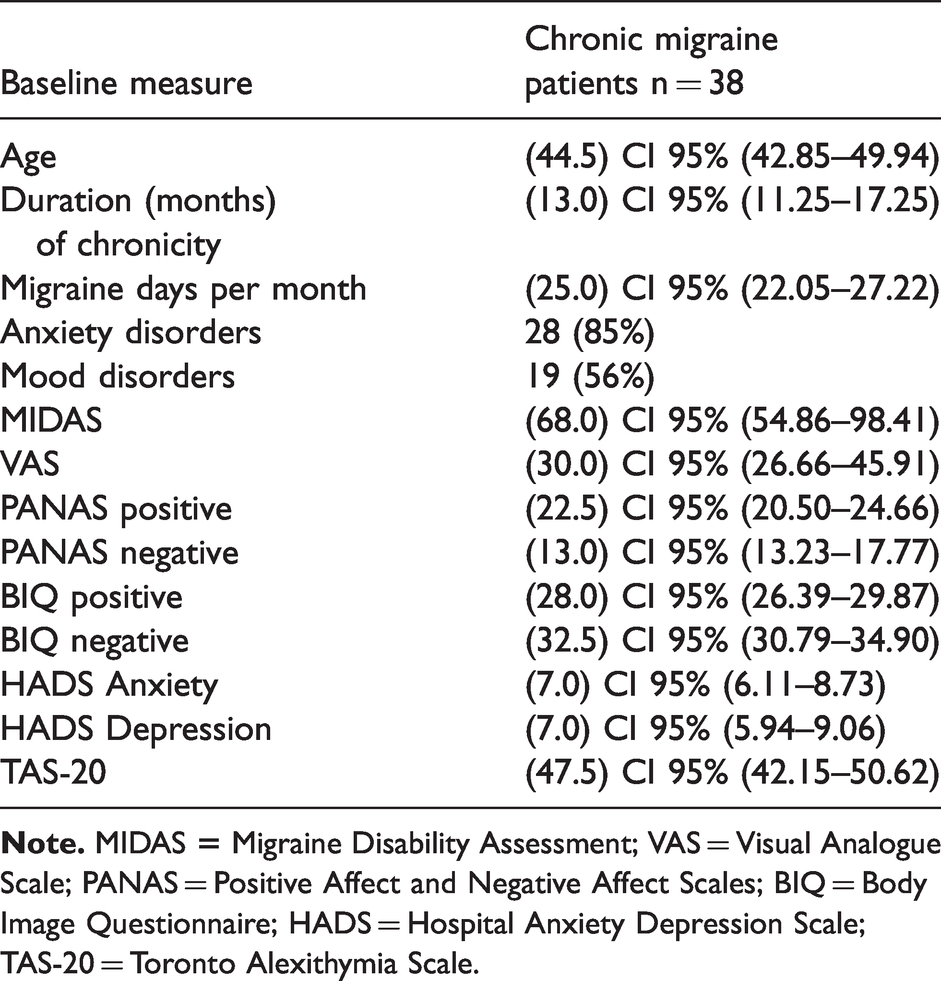
Thirty-eight female patients with chronic migraine were included in this study. Patients’ characteristics at baseline are shown in Table 1. Overall, at T0, more than half of the patients tested positive for anxiety (85%) and mood (56%) disorders and had high levels of anxious and depressive symptomatology at the HADS. However, no relationship was found between the reported pain ratings (VAS) during the experimental session and the scores reported for anxiety (rs =−0.23, p = 0.16) and depression (rs = −0.18, p = 0.28) at the HADS. Patients had low scores in the positive and negative PANAS subscales, while, interestingly, they presented significantly higher scores in negative BIQ items compared to positive BIQ items (t = −3.54, p < 0.001). Further, patients presented high levels of disability at the MIDAS. Overall, patients presented non-significant scores for alexithymia in the TAS-20.
Summary of baseline characteristics of chronic migraine patients. Data are presented as “(median), 95% CI”, and/or “absolute value (percentage)”.
Positive emotional face decreases pain ratings
Pain ratings were significantly lower in the positive emotional face condition compared to the negative emotional face (z = −4.46, 95% IC: 10.04 to −2.31, p < 0.001) and to the neutral emotional face conditions (z = 3.41, 95% IC: 0.84 to 8.58, p < 0.001) (see Figure 3A); whereas no differences were observed between the positive emotional face and the white screen control condition (z = −1.95, 95% IC: −0.38 to 7.35, p = 0.285). As evident from Figure 3B, the positive emotional face condition significantly decreased pain ratings from T0 compared to the negative emotional face (z = −4.46, 95% IC: 10.04 to −2.31, p < 0.0001) and to the neutral emotional face (z = 3.41, 95% IC: 0.84 to 8.58, p < 0.001) conditions, while no significant changes from baseline were observed with the white screen control condition (z = −1.95, 95% IC: −0.38 to 7.35, p = 0.285).

(a) Positive face condition decreased pain ratings compared to the negative and neutral face condition. Boxplots show median lines (horizontal lines), interquartile ranges (IQR;boxes) data outside 1.5 × IQR (whiskers). (b) Positive face condition decreased pain ratings from baseline. Graph shows mean difference between pain ratings during the experimental session and the baseline assessment before the start of the experimental session. Error bars show confidence interval values (highest and lowest values). **p < 0.01 ***p < 0.001.
Patients’ identified more with emotional faces than with the white screen
The degrees of identification with the visual stimuli significantly differed in the four experimental conditions (z = −2.28, p = 0.021). The patients reported a significantly lower degree of identification with the white screen condition with respect to the negative (z = −2.92, 95% IC: −1.37 to −0.029, p < 0.05), positive (z = −4.59, 95% IC: −1.78 to −0.43, p < 0.0001), and neutral (z = −6.23, 95% IC: −2.17 to −0.83, p < 0.0001) emotional face conditions. Moreover, the patients identified significantly more with the neutral face compared with the negative face (z = 3.32, 95% IC: 0.12 to 1.47, p < 0.05) (see Figure 4A).

(a) Patients identified more with the neutral face condition and with the three emotional faces stimuli compared to the white screen condition. Pain rating difference from baseline variates depending on the visual stimuli condition. There is a negative relationship between pain rating difference from baseline (Delta VAS) and the identification scoring under the (b) positive face condition, and a positive relationship under the (c) negative face condition. Boxplots show medians (horizontal lines), interquartile ranges (IQR; boxes) data outside 1.5 × IQR (whiskers) and outliers (o). *p < 0.05 **p < 0.01 ***p < 0.001.
Associations between pain rating changes, identification question scoring and patients’ psychological state at baseline
As regards the association between pain rating changes and identification questions, the results showed that, during the experimental sessions, the higher the identification scoring with the negative face condition the higher the pain rating difference from baseline (rs = 0.29, p < 0.01), and the lower the identification scoring with the positive face condition the higher the pain rating difference from baseline (rs = −0.31, p < 0.01). However, no relationship was found between the identification scoring with the neutral face (rs = 0.17, p = 0.07) and white screen (rs = −0.05, p = 0.61) conditions with pain rating difference from T0 (see Figure 4B and C).
A significant positive correlation resulted between the negative affective state (negative PANAS) of the patients and the pain ratings reported (VAS) during the experimental session (rs = 0.32, p = 0.05) (Figure 5A). Moreover, the results showed a significant positive relationship between the levels of alexithymia (TAS-20) at baseline and the identification scoring with the neutral emotional face condition (rs = 0.79, p < 0.05). However, no significant correlations were observed for the negative (rs = 0.07, p = 0.88), positive (rs = 0.07, p = 0.88), and white screen condition (rs = 0.20, p = 0.58) (see Figure 5B). No other significant correlations were observed between pain rating changes, identification question scoring, and psychological variables collected at T0 (p > 0.05).

Relationship of the emotional state of the patients at baseline with pain rating and identification questions scoring during the experimental session. (a) There is a positive relationship between pain rating during the experimental session and the negative affective state of the patients at baseline. (b) High scores in the TAS_20 scale at baseline are positively related with higher scores in the neutral face identification question. **p < 0.01.
Discussion
Two main findings can be deduced from this study. First, the observation that a positive emotional face expression can decrease pain perception in patients with CM. Second, patients with CM identified more with face emotional conditions (positive, negative, and neutral) compared with the white screen control condition.
As regards the former, the positive emotional face condition was the only one that effectively reduced pain ratings from T0. This phenomenon is in keeping with the crucial role in pain modulation of specific representations of human physical emotional content, via the mediation of automatic reactions of social learning and empathy (39,40). According to this view, subjective experience of acute pain in healthy subjects can be increased by the observation of scenes of physical pain (40); whereas unpleasant pictures of the same emotional level, but without physical content, have no effect on pain perception (40). Interestingly, visual induced analgesia was reported following social observation via video replay as well as after live face-to-face social observation (19). In contrast to this, one study found that even though the observation of facial expressions while inducing experimental pain in healthy subjects enhances placebo analgesia, this was not correlated with personality traits like empathy and behavioral activation/inhibition (16). One reason for this discrepancy could be that the authors here associated facial expressions to colored visual stimuli (green and red) (16), which could have impacted the attentional modulation of pain (41,42). Hence, these results (16) could be mediated more by attention than by an automatic reaction of empathy. From a neurobiological point of view, the effects of human physical emotional content on pain perception have been associated with activity changes in the right hemisphere without involving low-level somatosensory but associative cortices, including the temporo-occipital junction, the temporal pole, and the dorso-lateral prefrontal cortex (DLPFC) (40). These cortical regions are involved in higher-order cognitive, emotional, and memory processes. Moreover, the right hemisphere is related to the assessment of emotional stimuli (43). Specifically, the right temporal pole has been associated with the encoding of emotional stimulus, such as fearful faces (44). However, other investigations reported that negative emotional states induced by looking at emotional faces, the sound of unpleasant music, or the smell of unpleasant odors altered pain-evoked cortical activation consistently in the anterior cingulate cortex (ACC) (45). Indeed, the activation of the ACC has proved crucial to pain unpleasantness (45). Regarding the attentional modulation of pain, evidence showed that pain-evoked activity was modulated by attention only in the insula and in the primary somatosensory cortex (S1), which is consistent with the role of these regions in pain perception (45). Then, even though both attentional and emotional factors modulate pain perception, the nature and the neurobiological mechanisms differ. Here, we postulate that the positive emotional content induced by the positive face expression of the experimental video decreased pain perception. Neuroimaging data are needed to further support our findings.
Patients identified more with the three emotional face conditions compared with the white screen condition, as is evident from Figure 4A. In other fields of research it has been shown that pain perception can be shaped by social modelling in humans beings (19). Specifically, greater levels of placebo analgesia occurred in subjects with higher levels of empathy when observing placebo analgesic responses in a live observation situation (19). In fact, unpleasant pictures not showing physical emotional pain had no effect on pain perception (40), which could be due to the lack of personal identification with the observed stimulus. In this frame, it is interesting to note that our patients identified more with the emotional face conditions (positive, negative, and neutral) than with the control white screen condition, which suggests a certain degree of identification with emotional stimuli. However, they identified more with the neutral emotional facial expression when compared with the positive or negative emotional faces. When considering that our patients were characterized by high levels of anxious and mood symptomatology, as shown from the clinical interview and from the HADS responses, we can hypothesize that the preferential identification with the neutral face may be ascribed to the difficulty in identifying and describing emotions characterizing CM (23,46). Moreover, the fact that patients that presented higher levels of alexithymia at baseline identified more with the neutral emotional face condition also support such hypothesis.
Interestingly, those patients who reported higher identification scores during the observation of the positive face condition, reported higher pain changes (lower pain ratings) from baseline in these conditions, and the opposite effect happened with the negative face condition, where higher identification scores with the negative face were related to higher reported pain ratings from baseline (Figure 5). Such a finding seems to suggest that the visual induced analgesic response following the observation of emotional stimuli might be mediated by social learning and empathy processes toward the positive emotional facial expression.
There are some limitations that should be noted. First, the study was a within-subjects design in which participants served as their own control. Second, we did not collect any functional measure of activity changes in the pain modulatory system. Third, an evaluation of affective/emotional changes after the exposure to the experimental conditions could have shed light on the possibility of enhancing patients’ emotional state. Fourth, we did not test the white screen condition for photophobia and unpleasantness during or after the experimental session; however, none of the patients reported sensation of unpleasantness during or after the experimental session. Even though we assessed some emotional variables during the baseline assessment, other subjective and environmental conditions that may influence pain perception were not evaluated. This should be taken into consideration for future investigations. Hence, for all these reasons, larger studies are needed in order to fully understand to what extent visual placebo analgesia can be effective in modulating pain perception.
In conclusion, to the best of our knowledge, this is the first study evaluating the effects of a positive emotional face visual stimulus in reducing pain perception in CM. CM is a very challenging condition from a therapeutic point of view. Evidence suggests the usefulness of a multidisciplinary approach (47) in the management of difficult-to-treat primary headaches. The present findings could pave the way to the definition and testing of non-pharmacological therapies mediated by visual analgesia techniques induced by emotions and social observation in the management of CM.
Clinical implications
A positive emotional face condition feedback decreased pain ratings in chronic migraine. The positive emotional face condition was the only one in which decreased pain ratings relative to baseline were observed. Emotional regulation techniques through visual feedback or social observation should be further investigated for pain modulation in patients with chronic migraine.
Footnotes
Data availability statement
The datasets presented in this study can be found in online repositories. Repository/repositories and accession number(s): [Zenodo; 10.5281/zenodo.4808069].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a grant from the Italian Ministry of Health to IRCCS Mondino Foundation, Pavia (Current Research 2017–2019).
