Abstract
The aim of this study was to test the function of the diffuse noxious inhibitory control system (DNIC) in chronic and episodic migraine, exploring the blink reflex (BR) modifications induced by topical application of capsaicin on the hand. We evaluated 11 migraine without aura (MA) and nine chronic migraine (CM) patients during the not symptomatic phase; they were compared with 14 non-headache subjects (N). The BR was elicited by weak electrical stimuli delivered to the right supraorbital nerve; it was obtained 10 min and 20 min after the application of 1 ml of 3% capsaicin in a cream base (Teofarma) on the skin of the dorsum of the right hand, and 60 min after capsaicin removal. The subjective pain sensation induced by capsaicin was significantly increased in CM with respect to both MA patients and normal subjects; the R2 area was increased in CM patients during capsaicin application, with respect to controls and MA patients, who did not exhibit any reflex alterations. These results may suggest a failure of DNIC and a disturbed control of the trigeminal reflex at the central level, linked with migraine frequency.
Introduction
Deficiencies in diffuse noxious inhibitory controls (DNIC)-like mechanisms have been suggested to be responsible for the development of chronic pain in general (1). The DNIC allows that the activity of pain-signalling neurones in the spinal dorsal horn and in trigeminal nuclei can be inhibited by noxious stimuli applied to body areas far remote from the excitatory fields of these neurones (2, 3). Recently, a reduced effect of remote noxious stimuli on pain threshold tested by electrocutaneous stimuli administered either to the forearm (extra-cranial site) or to the temple (cranial site) was observed in chronic tension type headache, suggesting a deficient DNIC-like pain inhibitory mechanism in this type of chronic headache (4). Another recent study described the effect of heterotopic noxious conditioning stimulation by cold pressure test on the nociceptive flexion RIII reflex in chronic tension type headache and migraine without aura patients, consisting of facilitation rather than inhibition of the reflex response in both types of headache (5). On the basis of these results, a failure of DNIC may be supposed in primary headaches. Chronic migraine (CM), a disabling pathology that in most cases develops from episodic migraine, was recently recognized as a distinct clinical entity (6). The causes of chronicity are largely unclear, although abnormally high iron levels (a marker of disturbed neuronal function) were previously discovered in the midbrain periacqueductal grey (PAG) of patients with CM (7). In addition, changes in cortical processing of pain were found in patients suffering from CM (8).
It is likely that a network of cortical and subcortical structures with modulatory nociceptive and antinociceptive function becomes abnormally activated in a migraine attack or even between attacks (9). The PAG is the centre of the most powerful antinociceptive neuromodulator system in the brain, but the dorsal part of the caudal medulla is an important link in feedback loops determining the diffuse noxious inhibitory control (DNIC) that selectively inhibits action of nociceptive neurones located in the nucleus of the descending trigeminal tract by remote noxious stimuli (10, 11). To the best of our knowledge, the function of DNIC has not been previously tested in chronic migraine patients.
The blink reflex (BR) is a trigeminofacial brainstem reflex that is usually elicited by electrical stimulation of the supraorbital nerve. The R2 component of the blink reflex is mediated by wide dynamic range neurones (WDR) of the spinal trigeminal nucleus (12). The electrically elicited R2 is quite entirely mediated by low-threshold A-beta afferents (13), and it does not appear the best procedure to test trigeminal nociception, in comparison with other more specific methods, such as corneal reflex (14), laser blink reflex (15) and nociceptive blink reflex (16). Remote noxious stimuli suppress the activity in WDR neurones via activation of diffuse noxious inhibitory controls (DNIC) (10), whereas low-threshold mechanoreceptive neurones (LTM) are unaffected (2). Remote painful heat stimulus obtained by Aδ fibres stimulation significantly suppressed the R2 component of BR in healthy controls, while the R1 remained unchanged (17); furthermore, the influence of remote Aδ–C fibres stimulation on the R2 components of the blink reflex, elicited by innocuous electrical stimuli, is an available method to test the function of DNIC.
Capsaicin is the active component of hot peppers and, when applied on the skin, it causes pain mostly by stimulation of C nociceptors (18); the aim of this study was to examine the function of DNIC in episodic and chronic migraine and healthy controls, testing the effects of remote noxious stimuli induced by local application of capsaicin on the BR.
Materials and methods
Twenty outpatients attending the Headache Centre of the Neurological Clinic of Bari University in Italy were enrolled in the study. Eleven patients were affected by migraine without aura (MA) (cod. 1.1) according to the International Classification of Headache Disorders–II criteria (6) and nine patients suffered from chronic migraine (CM) (cod. 1.5.1) (6). All subjects underwent a standardized interview, as well as a clinical neurological and psychiatric examination. Patients with general medical, neurological or psychiatric diseases, according to DSM-IV (19), were excluded from the study. No patients were taking psychoactive drugs or prophylactic treatment for headache at the time of the study, for the low frequency of headache in the case of MA or the recent diagnosis of CM. Considering the diagnosis of CM (cod. 1.5.1.) (6), which may be rarely applied in clinical practice (20), we usually follow a clinical trial to fulfil all the criteria, according to ICHD-II (6). After the first approach to our centre, all patients are generally requested to monitor, using a diary for 3 months, the frequency of headache and of symptomatic therapy intake. The patients reporting a frequency of migraine and analgesic or triptans intake >15 days/month are usually advised to discontinue overuse after a brief admission. After the drug overuse withdrawal has been obtained, the frequency of headache is monitored again over 2 months and only patients with persisting migraine frequency >15 days/month are diagnosed as CM patients (cod. 1.5.1.). All selected headache sufferers were asked to attend the BR recording session (see below) in a pain-free state since at least 72 h. Moreover, five out of the nine CM patients were admitted after a shorter interval since the last attack, with a time range from 51 to 63 h (mean 58.3 ± 2.8). The occurrence of the first migraine attack after the experimental task was defined in all cases, and patients who experienced headache in the 24 h following the recording session were excluded. The MA patients were eight females and three males, aged 27–54 years (mean age 40.5 ± 7.4), the CM patients were five females and four males, aged 26–55 (mean age 47.2 ± 9.6). The frequency of headache (days with headache/month) was computed for the last 2 months; it was 5.25 ± 4.2 days with headache/month in the MA group and 22.8 ± 1.5 in the CM group. The duration of illness was on average 20.4 ± 9.7 years in the MA group and 20.5 ± 9.7 years in CM patients. Fourteen non-headache subjects, with no concomitant general, neurological or psychiatric disease, served as controls; they were four males and 10 females, aged 26–55 years (mean 41.4 ± 3.5). The age did not differ significantly across groups (
All subjects selected for the study gave their informed consent prior to their inclusion, according to the 1964 Declaration of Helsinki; the study was ethically approved by the Neurological and Psychiatric Sciences Department of Bari University.
For recording the BR the subject lay supine on a bed with the eyes lightly closed. The procedure was performed according to Ellrich and Treede (17). The right supraorbital nerve was percutaneously stimulated by conventional electrodes at the supraorbital foramen utilizing square wave pulses with a duration of 200 ìs. We chose to limit the study to a right sided stimulation in order to avoid a lengthy, uncomfortable procedure and because none of the selected migraineurs had a higher attack prevalence on one side. The individual thresholds for detection (DT), pain (PT), R1 (R1th) and R2 (R2th) were determined on the right side by applying two series of electrical pulses with increasing stimulus intensity using increments of 2 mA. The reflex thresholds were the minimum intensities evoking a response that could be discriminated from baseline activity. The stimulus intensity was adjusted to a level below the pain threshold and below five times the individual detection threshold to avoid any activation of nociceptive afferents (16). The interstimulus interval (ISI) was at least 10 s. The series were separated by at least 60 s. Two series of 10 stimuli were delivered in basal conditions and 10 min and 20 min after the application of capsaicin, which was removed after about 25 min; the recording was also carried out 1 h after capsaicin removal. Pain was induced by 3 ml of 3% capsaicin in a cream base (Teofarma), which was applied topically by means of a cotton swab on the skin of the right hand dorsum (radial territory), within an area of 6 cm2. Subjects were asked to rate the subjective pain sensation induced by 10 min and 20 min capsaicin application, using a 0–100 visual analogue scale (VAS) in which 0 indicated no pain in white, increasing in a gradual scale of reds to 100, which indicated the worst possible pain.
EMG signals were simultaneously recorded from both orbicularis oculi muscles by surface electrodes with the active electrode over the mid lower lid and the reference at the temple. Signals were filtered with a 50–1600 Hz bandpass and stored on a biopotential analyser (Micromed System Plus; Micromed, Mogliano Veneto, Italy; http://www.micromed-it.com). A ground electrode was fixed at the middle forehead.
The 10 trials of each stimulus series were rectified and averaged off-line; in addition, in each case the two series recorded in basal condition (before capsaicin and after capsaicin removal) and 10 min and 20 min after capsaicin application were averaged. Areas under the curves (AUC) of R1 in a time window from 9 to 24 ms and of R2 in a time window from 27 to 87 ms were calculated (16, 19). Onset latencies of R1 and R2 were determined in the single responses, and the mean latency was computed in each case for the different conditions. AUC and duration of R1, R2 and R2c during capsaicin were expressed as percentage of AUC and duration obtained in basal condition.
The BR features in the pre-capsaicin and 1 h after capsaicin removal were compared in the single groups by the paired Student's t-test. The detection threshold, pain threshold, the VAS values and the BR features in basal condition, the R1 and R2 AUC and duration rate of reduction induced by capsaicin, were compared employing one-way
The clinical and BR features were correlated within the migraine sample, by the Pearson correlation test. The SPSS version 11.0 for Windows was employed.
Results
BR features in basal condition
The detection threshold of electrical stimulus was on average 2.1 ± 0.1 mA in control subjects, 2.4 ± 0.9 mA in MA patients and 1.7 ± 1.1 mA in CM patients (F = 1.64, NS). The R2 threshold was on average 2.8 ± 0.8 mA in controls, 3 ± 0.9 mA in MA patients and 2.5 ± 1.1 mA in CM patients (F = 1.56, NS). The pain threshold was 17.5 ± 2.5 mA in controls, 18.1 ± 1.5 mA in MA patients and 15.5 ± 2.8 mA in CM patients (F = 1.91, NS). Comparing the control series before capsaicin and after capsaicin removal, the BR features did not differ significantly in any group (Table 1), thus the control series were averaged for further analysis.
Blink reflex features in the pre-capsaicin condition and 1 h after capsaicin removal. Results of Student's t-test for paired in the single groups: NS
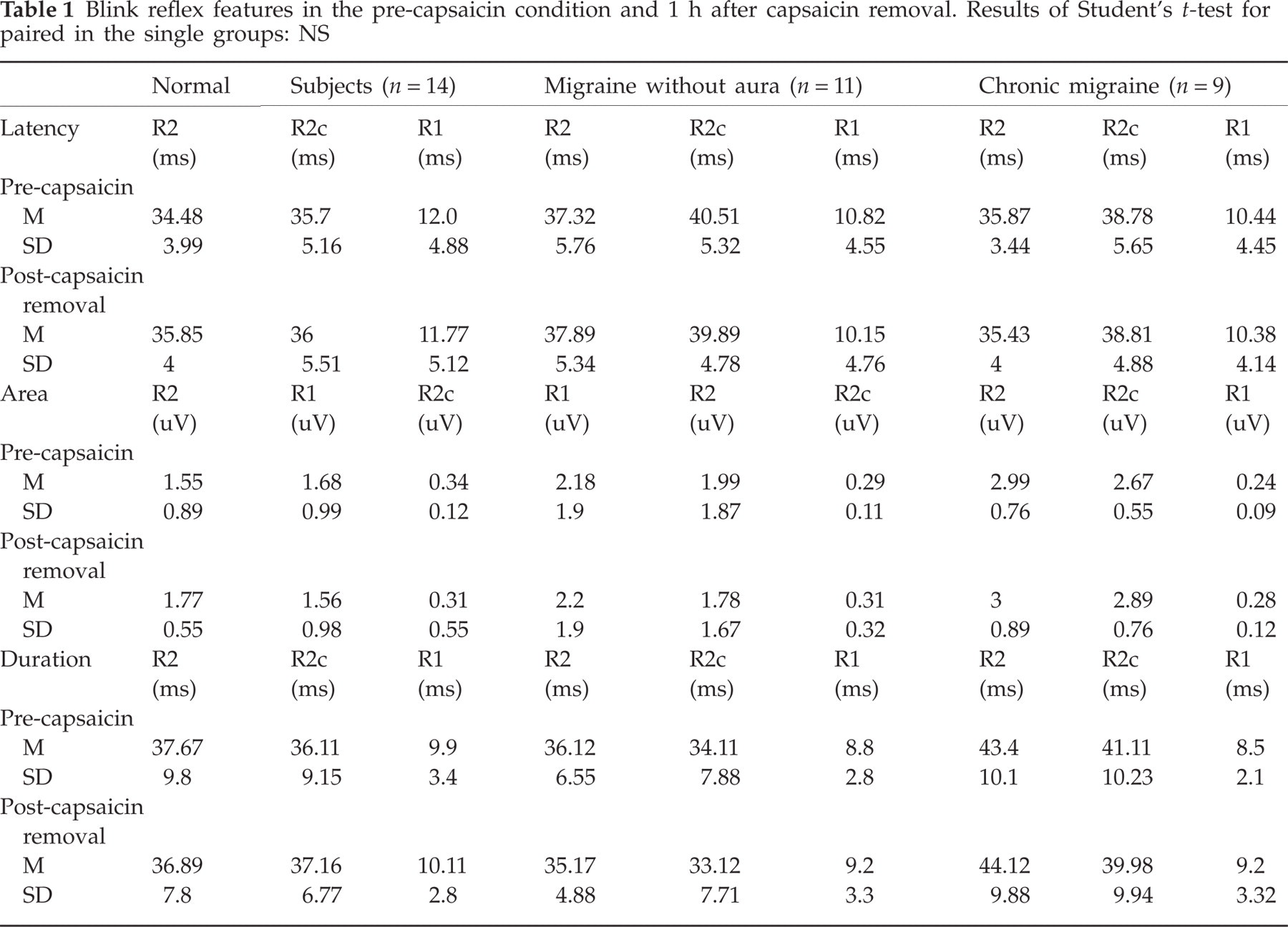
At the intensity level agreed for the experimental trial, which was in all cases four times above the DT and one level below the PT, all subjects exhibited R1 and ipsilateral R2 and controlateral R2 (R2c). In basal conditions, the latencies of R1 and R2 did not differ across groups (R1, F = 1.56, NS; R2, F = 0.4, NS) (Table 2); the R1 AUC was also similar in the three groups (F = 1.56, NS); the R2 and R2c AUC were not significantly different across groups (R2 AUC, F = 1.6, NS; R2c AUC, F = 1.11, NS) (Table 3).
Mean values and standard deviations of blink reflex components latencies. The results of the Bonferroni test, employed within the single groups across the three conditions, are shown: ∗basal vs 10 m caps, basal vs 20 m caps: P < 0.05

Mean values and standard deviations of R2 and R2c area and duration. The results of the Bonferroni test, employed within the single groups across the three conditions, are shown: ∗basal vs 10 min caps, basal vs 20 min caps: P < 0.05

Effects of capsaicin: spontaneous pain
A clear sensation of pain started after 500 ± 12.5 s across patients and controls and it was described by all subjects as a burning sensation. The VAS induced by 10 min and 20 min capsaicin application, was significantly increased in CM patients compared with both normal subjects and controls (VAS 10 min, F = 12.9, P < 0.0001; VAS 20 min, F = 7.9, P < 0.01) (Figure 1).

Mean values and standard deviations of subjective pain induced by capsaicin expressed by 0–100 VAS. The results of the Bonferroni test are shown: CM vs N∗∗∗, P < 0.0001; CM vs MA§§§, P < 0.0001.
BR latencies
In the control group, the R2 latency was not significantly modified during capsaicin application (F 2.53, NS) (Table 2). A significant latency prolongation, was detectable for the controlateral R2 after 10 min and 20 min capsaicin application (F 4.32, P < 0.05) (Table 2). The R1 latency was unchanged during capsaicin application in non-migraine subjects (F = 0.39, NS) (Table 2). In both migraine without aura and chronic migraine groups, the R2, R2c and R1 latencies were unchanged across the three conditions (MA group: R2, F = 0.19, NS; R2c, F = 0.2, NS; R1, F = 0.6, NS; CM group: R2, F = 0.19, NS; R2c, F = 0.35, NS; R1, F = 0.42, NS) (Table 2). The comparison of the R1, R2 and R2c latencies, considering the condition × cases as factors, was not significant (R2, F = 1.34, NS; R2c, F = 1.69, NS; R1, F = 0.51, NS).
R1 AUC and duration
The
The R1 duration was not modified by capsaicin application in any group.
R2 AUC and duration
In controls, there was a significant reduction of R2 AUC after capsaicin application (F = 3.55, P < 0.05) (Table 3). In migraine patients the R2 AUC was not significantly different across the three conditions (F = 3, NS) (Table 3). The R2 AUC was significantly changed across the three conditions in CM patients, for an increase induced by capsaicin application (F = 4.41, P < 0.05). The rate between the R2 AUC computed after 10 min capsaicin application and in basal condition was significantly different across groups, though the Bonferroni test was not significant (R2, F = 3.52, P < 0.05) (Figure 2a). The rate between the R2 AUC computed after 20 min capsaicin application vs. basal condition was significantly enhanced in CM patients with respect to controls and MA patients (R2, F = 5.4, P < 0.01) (Figures 2a and 3). The R2 duration was also significantly reduced by capsaicin in controls (F = 3.44, P < 0.05), but it was not significantly changed in MA (F = 3.12, NS) and CM groups (F = 3.32, NS) (Table 3). The R2 duration rate between 10 min capsaicin vs. basal condition was not significantly different across groups (F = 1.47, NS), but a significant difference emerged after 20 min capsaicin application, for a trend towards an R2 duration increase in CM patients, with respect to controls (F = 5.91, P < 0.01) (Figure 2b).

(a) Effect of capsaicin on R2 and R2c AUC. The results of the Bonferroni test are shown: CM vs. N∗ and CM vs M§, P < 0.05. (b) Effect of capsaicin on R2 and R2c duration. The results of the Bonferroni test are shown: CM vs N∗, P < 0.05.

An example of BR for one representative healthy subject, female, 27 years old, and one chronic migraine (CM) patient, female, 30 years old, in which there was a very clear increment of R2 and R2c AUC and duration during capsaicin application. Each signal resulted from the average of two series of 10 stimuli.
R2c AUC and duration
The R2c AUC was significantly reduced during capsaicin application in controls (F = 3.77, P < 0.05) (Table 3). In migraine patients no significant changes of R2c AUC was evident across the different conditions (F = 1.41, NS) (Table 3). In chronic migraine, the R2c AUC was significantly enhanced during capsaicin application (F = 4.11, P < 0.05) (Table 3). The rate between the R2c AUC computed after 20 min capsaicin application vs. basal condition was significantly enhanced in CM patients with respect to controls and MA patients (R2c, F = 4.2, P < 0.05) (Figures 2a and 3). The R2c significantly changed in duration in normal subjects across the different conditions (F = 3.56, P < 0.05), while it did not show significant changes in the MA group (F = 2.99, NS) and CM group (F = 2.13, NS) (Table 3). The R2c duration rate was not significantly different across groups (R2c duration rate between basal and 10 min capsaicin application, F = 1.69, NS; R2c duration rate between basal and 20 min capsaicin application, F = 2.79, NS) (Figures 2b and 3).
Correlation between clinical and BR features
Considering all the migraine patients together, the rate between the R2 duration after 20 min capsaicin application and basal condition was significantly correlated with the frequency of headache (Pearson correlation test 0.57, P < 0.05); the correlation between headache frequency and rate of R2 AUC reduction after 20 min capsaicin application approached statistical significance (Pearson correlation test 0.49, NS).
Discussion
The results of this study showed that in normal subjects the remote C fibres activation by local capsaicin reduced R2 duration and area, and this effect was evident after 20 min of capsaicin application, when the subjective pain sensation was higher. The time of appearance of capsaicin-induced pain may be coherent with the extension of capsaicin application and with its concentration (21, 22). The rates of R2 suppression were in line with previous studies (12, 17). The R1 was unchanged after capsaicin. These results could help to confirm that the R2 is mediated by wide dynamic range (WDR) neurones of the spinal trigeminal nucleus. Remote noxious stimuli suppress the activity in WDR neurones via activation of DNIC, whereas low-threshold mechanoreceptive (LTM) neurones are unaffected. A moderate increase of R2 latency was previously observed during remote painful stimulation (17); in our normal series it did not reach statistical significance, though capsaicin induced a significant prolongation of R2c latency. In episodic and chronic migraine patients, capsaicin did not induce any change on BR latencies. In basal conditions, migraine without aura and chronic migraine patients showed R1 and R2 features like normal subjects, according to a previous study (23). Patients with chronic migraine exhibited an increase of subjective pain sensation to capsaicin, with respect to both controls and episodic migraine, while the pain threshold to electrical stimulus was only slightly and not significantly reduced with respect to controls. The reason for this phenomenon may be a stronger activation of C afferents at the peripheral level or an amplification of pain at the central level. Considering that patients were free from pain during the recording session and that the site of capsaicin application was outside the zones eventually sensitized for migraine (24), the second hypothesis may be more reliable. Previous investigations failed to detect increased pain sensitivity in extra-cephalic zones during the asymptomatic phases of episodic migraine (25), so our finding of increased sensitivity to extra-cephalic capsaicin application may be a feature of chronic migraine. Despite the noxious remote stimulation, in CM the R2 AUC and duration were paradoxically enhanced instead of reduced, while in episodic migraine the remote noxious stimulation did not cause a significant R2 AUC and duration suppression. In CM an increment of both ipsilateral and controlateral R2 was evident after 20 min capsaicin application. In a recent study about the effect of cold pressure test on RIII reflex, a facilitation instead of inhibition of the reflex response was evident in migraine patients with respect to controls (5). In our migraine without aura group, no significant effect was induced by capsaicin on R2 AUC and duration, thus suggesting a latent deficit of noxious inhibitory system, which is evident in patients with higher headache frequency.
In this scenario, a failure of DNIC may be supposed in chronic, more than in episodic, migraine, as a feature linked with chronic pain rather than with migraine in per se. The correlation between the entity of R2 duration modification and the frequency of headache in the migraine patients supported this hypothesis. The failure of DNIC did not explain in per se the phenomenon of BR increase during remote noxious stimulation. We know that DNIC control is mainly subtended by the subnucleus reticularis dorsalis in the reticular bulbar formation. It is an important link in feedback loops that regulate spinal and trigeminal outflow and spino-reticular-thalamic circuits involved in pain transmission (12). The thalamic nuclei connected with the reticular bulbar formation project to the cortical limbic structures, which are involved in the affective and motor reaction associated with pain (12), so it may be supposed that the inhibitory control is exerted directly on the trigeminal nucleus and on the limbic cortex devoted to pain elaboration. The R2 component of the BR is largely influenced by suprasegmental control: selective attention to the stimulus, a state of tension and fear modulates the size and habituation of the R2 response in different ways (26). In a previous study, an abnormal R2 modulation by alerting was observed in migraine (27). We can suppose that in CM the facilitation instead of the inhibition of trigeminal-facial response during remote noxious stimulation may be induced by both a failure of DNIC control at the trigeminal and cortical level and a disturbed modulation of trigeminal reflex responses at the cortical level. This phenomenon could hardly be attributed to analgesic overuse, because patients had discontinued analgesic symptomatic treatment for at least 2 months; rather it seems a feature linked with the severity of migraine and chronic pain (1). A complex dysfunction of the cortical and subcortical structures with modulatory nociceptive and antinociceptive function may predispose to chronic migraine. The PAG (7) and limbic cortex (8) showed functional changes in chronic migraine; our study provides evidence that the reticular bulbar structures are involved in the general dysfunction of pain-control mechanisms. The occurrence of a migraine attack in such situations may result in prolongation of headache and sensitization phenomena, leading to analgesic over-use, which in turn could worsen the mechanisms of pain control.
Acknowledgements
The study was supported by ‘Progetto Integrato per la Salvaguardia della Salute dell'Uomo' Fondazione Cassa di Risparmio, Bari, Italy.
