Abstract
The nosology of migraine premonitory (PS) and resolution (RS) symptoms was studied in 100 migraineurs consulting their general physician. They were asked to fill in, for three attacks, a PS and RS questionnaire. ‘True’ PS/RS were those experienced the day before (or the day after) the headache had started only if they were not present in a questionnaire completed in a pain-free period. True PS and RS were experienced by 84± and 80±, respectively, of subjects for the first attack. The mean and range (per patient) of PS were 6.8 and 0–21 and of RS 4.7 and 0–15. Anxiety, phonophobia, irritability, unhappiness and yawning were the commonest PS, whereas asthenia, tiredness, somnolence and concentration difficulties were the most common RS. Gender, age and Migraine Disability Assessment scores did not influence PS and RS. Both PS and RS were more frequent in migraine with aura subjects. Patients on preventatives showed a decreased frequency of PS and, to a lesser degree, of RS. Severity of headache was associated with a higher frequency of RS. Individual RS and especially PS were quite consistent after three attacks. Almost two-thirds of the symptoms were noticed in at least two out of three attacks, while more than a half of PS and more than a quarter of RS repeated in three out of three attacks. In conclusion, around 80± of unselected migraineurs experience RS and PS. Migraine with aura and severe pain are risk factors for experiencing PS and RS, while preventatives were protective, especially for PS.
Introduction
Migraine is a frequent disorder characterized by attacks of unilateral, pulsating headache often accompanied by a constellation of non-headache symptoms, of which nausea, phono- and photophobia are an established part of the International Headache Society (IHS) criteria for migraine diagnosis. Leaving migraine aura aside, non-headache symptoms have been associated with the three stages of the attack: the premonitory symptoms (PS), the headache and the resolution symptoms (RS) phases.
Little attention has been paid to the variety of PS preceding the headache phase (1–8), while RS, though clinically recognized (9), remain almost unstudied. In the 1980s, Blau published the first results of a clinical assessment of the PS (1, 2). He studied 50 patients who had only migraine without any additional symptoms, such as tension-type headache. He found that 17 patients had early PS, such as mood changes, somnolence, changed mental state, unusual appetite or fluid disregulations from 1 h to 1 day before the attack. He proposed the term ‘complete migraine’ for those migraine patients also experiencing PS. Isler has since 1970 evaluated the answers of 100 migraine patients to the specific queries about prodromes in his questionnaire. He found that 65 patients had mentioned prodromes which were different from focal aura symptoms, occurring from within minutes to within several days before the headache (4). In 1985, Waelkens described the characteristic warning symptoms in 49 selected migraine patients with ‘complete migraine’ (3). Fewer than 10% of his patients spontaneously reported the existence of PS symptoms. He distinguished evolutive symptoms appearing from a few minutes up to a few hours before the headache and including visual aura from the non-evolutive early warning symptoms beginning up to 48 h before the attack, which he found in 88% of his patients. A 30-mg dose of domperidone, but not an equivalent dose of cisapride, a non-antidopaminergic stimulant of gastrointestinal motility, taken at the first appearance of the PS prevented two-thirds of the attacks in a double-blind, placebo-controlled crossover study (10, 11). The question of whether PS accurately predict headache, with relevant implications in the pathophysiology and management of migraine, was addressed in 120 patients who reported PS that they believed predicted migraine headache in at least two out of three attacks. Using electronic diaries, patients correctly predicted 72% of migraine headaches (12). Finally, a retrospective review of the frequency of PS in 893 headache patients seen consecutively at a tertiary Headache Clinic showed that 39.2% of 627 IHS migraine patients had PS (8).
The heterogeneous studies reviewed above do not allow us to define the actual frequency of PS and especially of RS. In addition, it is far from clear how clinically to classify PS and RS and their nature and pattern. These vary considerably among the different studies regarding both the clinical pattern of these symptoms and their number, which has ranged from 59 to 21 potential PS (1–8). One crucial point is that many of these symptoms are experienced outside of the attacks and none of these studies has recorded the background to these symptoms interictally. As an example, estimates for fatigue in the general population are high, around 35% (13–15). In the only study analysing migraine postdromes, a total of 255 RS were reported by 40 migraineurs who were asked to complete a questionnaire the day after the attack (16).
To sum up, the few studies which have examined PS and RS in migraine have been flawed for many methodological reasons, such as retrospective data collection, absence of diaries, scarcity of patients, heterogeneous classification or by the lack of interictal testing. Our study was undertaken to clarify the nosology of PS and RS in the standard migraine patient.
Patients and methods
Sampling
This study was carried out in the Health Area of Camargo-Astillero, Cantabria, Spain. Our objective was to study PS and RS in 100 consecutive subjects spontaneously consulting their general practitioners (GPs) in this Health Area due to migraine headache. To be included in this study subjects had to give their informed consent, be above 14 years old and meet IHS criteria for migraine (without or with typical aura) (17). They could use any of the available symptomatic treatments for migraine headache. They were asked not to modify preventive treatment during the study. Those patients suffering from either tension-type headache or migraine for more than 15 days per month or suffering from any other active systemic or mental disorder which could theoretically interfere with the results were excluded. The project was approved by the local ethics committee, started in January 2001 and concluded in June 2003.
Interviews and Premonitory/Resolution Symptoms Questionnaire
All subjects attending the Health Centre due to headache were personally seen by a physician experienced in headache diagnosis (Fig. 1). Patients meeting IHS criteria for migraine diagnosis as well as the other inclusion/exclusion criteria were interviewed in depth regarding sociodemographic variables, medical antecedents and headache characteristics. They received a general and neurological physical examination. Blood sampling, neuroimaging or ophthalmological check-up were electively planned. As a general measure of their migraine severity, all patients were asked to complete the Migraine Disability Assessment (MIDAS) test (18). In this interview, and after the phases of migraine were explained, we carefully taught migraine patients to fill in a detailed questionnaire. This was an ad hoc questionnaire containing a total of 28 symptoms which have been described as possible migraine PS or RS. We arbitrarily arranged these symptoms into four groups: neuropsychiatric, sensory, digestive and general symptoms. Patients were asked to complete this questionnaire (referred to the previous day) after at least three headache-free days, with the following day also being headache-free. They had to visit the Health Centre again to confirm they had filled in the questionnaire without problems, to resolve possible doubts and check that they had been headache free at least 2 days before and after the day to which the questionnaire referred. This visit was repeated as many times as necessary until we were sure that the patient was familiar with the questionnaire and that it had been completed within the required headache-free period.

Summary of study methodology. IHS, International Headache Society; HA, headache; PS, premonitory symptoms; RS, resolution symptoms.
The patient was seen again at the Health Centre twice, 2–5 days after having the first migraine attack and finally after having experienced the second and third migraine attacks. In these two visits, and for each attack, the patient was asked to fill in the PS and RS questionnaire (always referring to the 24 h before and after the beginning and resolution of migraine headache) and a standard headache questionnaire with the items for the diagnosis of migraine following the IHS Classification. The PS and RS questionnaire results were taken into account only if the accompanying attack fulfilled the IHS criteria for migraine with or without aura diagnosis. Attacks in which pain recurred within 48 h were not included in the final analysis.
Statistics
Statistical analyses were carried out with the SPSS 11.5 program (SPSS Inc., Chicago, IL, USA). Proportions (categorical data) were compared using the χ2 test or, when this was not applicable, Fisher's exact test. Mann–Whitney or t-tests were used for the analysis of continuous data.
Results
Patients and clinical data
A total of 221 subjects spontaneously consulted their GPs in the Health Centre due to headache during the period of this study. There were 144 patients (65.2%) who met IHS migraine criteria. The remaining 77 were diagnosed as having episodic tension-type headache (n = 62), chronic tension-type headache (n = 12), cluster headache (n = 2) and headache attributed to temporal arteritis (n = 1). A total of 100 migraine patients (82 women and 18 men) fulfilling inclusion/exclusion criteria participated in this survey (Fig. 1). Their mean age was 39.3 years (range 15–65). Eighty-five were diagnosed as having migraine without aura attacks and 15 met diagnostic criteria for migraine with typical aura. Thirty-six had a MIDAS score <10, 30 between 11 and 20 and 34 scored >20. Forty-eight patients took different antimigraine preventive treatments during the study period (amitriptyline 23, β-blockers 19, flunarizine 2, topiramate 2 and fluoxetine 2). Regarding symptomatic treatment, 48 were using simple analgesics and/or non-steroidal anti-inflammatory drugs, 30 triptans, 16 ergotamine-containing medications and the remaining six opioids. All 100 completed one questionnaire outside of a migraine attack and at least one more questionnaire during a migraine attack. Sixty patients completed two further questionnaires for the second and third migraine attacks.
Premonitory symptoms
General results
Ninety-two migraine subjects experienced at least one PS the day before headache had begun for the first attack (total number of symptoms 960, range 0–21, mean 9.6, median 10). Ninety-four patients noticed at least one of the potential PS when they filled in the questionnaire during a headache-free period. We decided to consider true PS (or RS) those experienced by the patient the day before (or the day after) the headache had started only if they were not present in the questionnaire completed in the pain-free period. Having discounted these headache-free symptoms, true PS were experienced by a total of 84 subjects for the first attack. The total number of PS was 680 (mean 6.8, median 6.5, range 0–20). The distribution of PS is shown in Fig. 2 and Table 1. The most common individual true PS were anxiety (46%), phonophobia (44%), irritability (42%), unhappiness and yawning (40%) and asthenia (38%). Neuropsychiatric (82%) were the most frequent, followed by sensory (65%), general (63%) and digestive symptoms (58%).

Numerical distribution of premonitory symptoms (▪) and resolution symptoms (□).
Distribution of true premonitory symptoms (PS) and resolution symptoms (RS) and frequency of interictal symptoms in 100 migraine patients

Influence of demographic and clinical variables
The frequency and distribution of true PS, in general, by groups and by individual symptoms, did not significantly differ between males and females. In order to test for a possible influence of age on PS, we analysed these in the 52 patients >40 (mean 6.4, range 0–15) vs. in those 48 <40 years (mean 7.2, range 0–20). The frequency and distribution of true PS did not differ either in general or by symptoms, even numerically, between the two age groups.
Globally, true PS were more frequent in patients experiencing migraine with aura episodes (Table 2). Regarding individual symptoms, differences were significant for photophobia (60 vs. 33%, P = 0.04) and nausea (67 vs. 17%, P < 0.001). Unhappiness (53% in migraine with aura vs. 37% in migraine without aura), anxiety (53% vs. 45%), yawning (47% vs. 39%) and phonophobia (60% vs. 42%) were numerically more frequent in migraine with aura patients, while constipation (7% vs. 24%) and passing urine frequently (0% vs. 12%) were more frequent in migraine without aura.
Influence of several clinical variables in premonitory symptoms (PS) and resolution symptoms (RS)
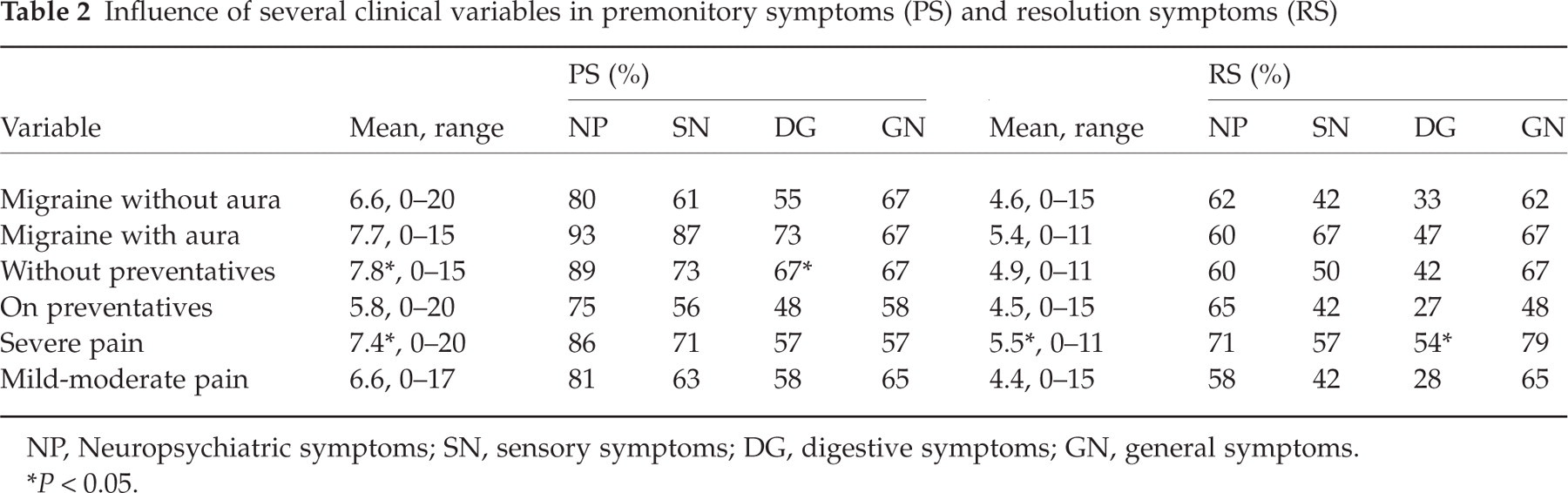
NP, Neuropsychiatric symptoms; SN, sensory symptoms; DG, digestive symptoms; GN, general symptoms.
P < 0.05.
The number of true PS in those 48 patients taking preventive treatment for migraine was significantly lower (P = 0.027) than in those who were not using preventatives (Fig. 3 and Table 2). Regarding individual PS, there were only three symptoms which were numerically more frequent in those patients on preventatives. The three symptoms were of digestive origin: flatulence (29% vs. 15%), anorexia (19% vs. 21%) and abdominal pain (15% vs. 8%).

Percentage of premonitory symptoms in patients taking preventatives compared with those not on preventatives. ∗P < 0.05.
Regarding the relationship between the severity of the migraine attack and the frequency and distribution of PS, there was no significant difference in the frequency of PS in patients suffering from severe pain (n = 28 patients) compared with those patients with mild–moderate pain (n = 72 patients) (Table 2). Anxiety (54% vs. 43%), insomnia (36% vs. 24%), photophobia (50% vs. 32%), phonophobia (54% vs. 40%), focusing difficulties (39% vs. 21%) and anorexia (39% vs. 13%, P = 0.003) were more frequent in attacks with severe pain, whereas somnolence (29% vs. 38%) and tiredness (25% vs. 33%) were less frequent in attacks with severe pain.
No clear increase was dectable in the frequency of PS in patients with a higher disability compared with those with lower MIDAS grades.
PS consistency
The consistency of PS was analysed in the 60 patients who had completed the questionnaire for three attacks. We analysed consistency only for those PS which were present in at least one attack in more than 25% of patients. In these patients, PS consistency in at least two out of three migraine attacks ranged from 25 to 83.3% (mean 63.4%) and in three out of three attacks ranged from 5.9 to 53.3% (mean 29.8%). The consistency for those PS seen in at least 25% of the patients in one attack is illustrated in Fig. 4. Concentration difficulties, unhappiness, anxiety and yawning were the most consistent PS.

Consistency of premonitory symptoms seen in at least 25% of the patients in one attack. □, Three out of three; ▪, two out of three.
Resolution symptoms
General data
Some 86 subjects experienced at least one RS the day after headache had begun for the first attack (total number of RS 661, mean 6.6, median 6, range 0–23). Eighty migraine subjects noticed, in the first attack, at least one true RS the day after headache had ceased. The total number of true RS was 471 (mean 4.7, median 4, range 0–15). The distribution of RS is shown in Fig. 2 and Table 1. The most common individual RS were asthenia (55%), tiredness (46%), somnolence (29%), concentration difficulties (28%), phonophobia (27%), photophobia (26%), unhappiness (26%) and yawning (24%). General RS (69%) were the most frequent, followed by neuropsychiatric (62%), sensory (46%) and digestive (35%) symptoms.
Influence of demographic and clinical variables
The frequency and distribution of true RS, in general, by groups and by individual symptoms, did not significantly differ between males and females. The number of RS for those patients >40 (mean 5.4, range 0–13) was not significantly different from that for patients <40 years old (mean 4.1, median 4, range 0–15). Neuropsychiatric (63% vs. 62%), sensory (54% vs. 39%), digestive (38% vs. 33%) and general (60% vs. 65%) RS were numerically more frequent in subjects >40 than in those <40 years. Additionally, each individual RS was numerically more frequent for subjects >40 years. These differences, however, were not significant.
Globally, true RS tended to be more frequent in patients experiencing migraine with aura episodes compared with those having migraine without aura (Table 2). The greatest differences favouring migraine with aura were seen for photophobia (47% vs. 22%, P = 0.04), phonophobia (47% vs. 24%, P = 0.06), abdominal pain (20% vs. 4%, P < 0.01) and diarrhoea (13 vs. 1%, P = 0.01).
The number of true RS in those patients on preventatives was slightly lower than in those who were not using preventive treatment (Table 2). Somnolence (35% vs. 23%) and concentration difficulties (31% vs. 25%) were the only two RS which were more frequent in patients on preventive treatment. There was no relationship between RS frequency and profile and the different types of acute medication taken in the attacks.
RS were significantly more frequent in those patients experiencing severe pain compared with those with slight–moderate pain (Table 2 and Fig. 5). RS, mainly neuropsychiatric (64% for MIDAS scores 0–10 vs. 76% for MIDAS scores >20) and sensory (53% vs. 42%), tended to increase slightly with higher MIDAS scores. There were no significant differences, however, for the general or group distribution of RS for the three MIDAS levels.

Percentage of resolution symptoms in patients with severe pain compared with patients with slight–moderate pain. ∗P < 0.05.
RS consistency
The consistency of true RS was analysed in the 60 patients who completed the questionnaire for three attacks. For those RS which were present in at least one attack in more than 25% of patients, RS consistency in at least two out of three migraine attacks ranged from 40 to 81.1% (mean 60.5%) and in three out of three attacks ranged from 0 to 48.6% (mean 26.4%). The consistency for those RS seen at least 25% of the patients in one attack is illustrated in Fig. 6. Tiredness, asthenia and somnolence were the most consistent RS.

Consistency of resolution symptoms seen in at least 25% of the patients in one attack. □, Three out of three; ▪, two out of three.
Discussion
The present study is the first nosographic analysis of migraine PS and RS in a group of non-selected migraineurs after the appearance of the revised IHS Classification. Key methodological aspect of our study was that, to be considered as true PS or true RS, we carefully discounted, in the same patient, the symptoms experienced by these subjects when they were headache-free. No previous studies had recorded the background prevalence of these symptoms interictally, which could be methodologically compared to offering the results of a clinical trial without taking into account a placebo arm. This approach turned out to be crucial when interpreting the final results, as the frequency of some interictal symptoms was in fact relevant. As shown in Table 1, symptoms such as asthenia, fatigue, anxiety, speech difficulties, yawning or irritability were referred to by 15% or more of our patients in a headache-free period. The questionnaire, comprising 28 items (see Table 1), included the PS symptoms already and consistently described in the literature in migraine patients (1–6, 12). In order to avoid repetition and to increase the clarity of results, we made an effort not to include: items with the same meaning (e.g. headache tension, headache tightness), anecdotal PS (e.g. irritated by collar, aversion from menstruation) or untrue PS (e.g. pain above the eyes, typical aura), which has been one of the reasons for the variability of the results among previous studies. Collection of exact information about migraine PS and RS, however, is complicated by different kinds of bias. Recording may be biased by the individual's difficulties in describing important characteristics, especially if the patient is confused or sleeps during the attack, and towards the more severe or recent episodes. The present data were collected using a complete and easy to answer questionnaire, but are limited by the memory of the participants. In this regard and to avoid including interictal and not true PS, we decided to consider PS/RS only those experienced by the patient the 24 h before/after the headache had started. Even though PS have been described several days before the headache phase, most PS have been shown to occur up to 24 h before the aura/headache phases appear (1–4). This was confirmed in the prospective electronic diary study, where most headaches were predicted within 24 h, in spite of a 72-h limit for PS (12).
This study shows that both PS and RS are very prevalent and numerous in migraine attacks. Even after discounting headache-free symptoms and considering the 24-h period, PS and RS were noticed in 84% and 80% of the attacks, respectively. The average was seven PS and five RS per migraine attack (1, 16). The prevalence of RS is largely unknown, while the prevalence of PS in previous studies has ranged from 7% to 88%, depending largely on study methodology (1–8, 12). Even though the symptoms were collected after 24 h and not exactly prospectively with electronic diaries, the high frequency of the symptoms found here probably reflects our improved methodology, with specific and immediate data collection, a clear and easy questionnaire and experienced together with personalized interview. Anxiety, phonophobia, irritability, unhappiness and yawning were the most common PS, being noticed in between almost one-half and more than one-third of the attacks, whereas asthenia and tiredness and, to a lesser degree, somnolence and concentration difficulties were the most common RS. We arbitrarily grouped the individual symptoms into four categories: neuropsychiatric (encompassing ‘pure’ psychiatric symptoms), sensory (symptoms due to ‘neurological’ hypersensitivity), digestive and general. Neuropsychiatric (82%) were the most frequent PS, while general (69%) were the most frequent RS.
These data illustrate differences in the clinical profile of RS compared with PS, probably reflecting different pathophysiological mechanisms, at least for some of these symptoms. Of interest is the high proportion of symptoms typically associated with the headache phase, such as nausea, phono- and photophobia, in the premonitory phenomenon continuing through to the resolution phase. Assuming that PS are an integral part of a migraine attack and taking into account the PS profile, these results agree with the proposal that an attack probably begins with a rather diffuse cerebral disturbance, which spreads to the hypothalamus or to the brainstem dopaminergic nuclei (19, 20) and carries on through the headache and into the resolution phase, giving clinical weight to evidence of electrophysiological changes beginning 24 h before the headache. Neurophysiological techniques have shown interictal loss of cognitive habituation which increases continuously during the migraine interval until sudden normalization on the first day of the attack (21). Other studies have shown an increase in early contingent negative variation amplitude the day before an attack (22, 23) which is associated with changes in EEG power spectrum from 4 days before the attack (24). This increasing interictal abnormality in cortical hyperexcitability may reflect a neurophysiological readiness to generate an attack and an increasing susceptibility of the migrainous brain to precipitating factors. A further study has shown normalization of visual and auditory evoked potentials just before and during migraine attacks compared with interictally, which may represent the brain's attempt to abort the impending attacks by activation of appropriate sensory-modulating systems (25).
Individual RS, and especially PS, were quite consistent after three attacks. Almost two-thirds of the symptoms were noticed in at least two out of the three attacks, while more than one-half and a quarter of PS and RS, respectively, repeated in three out of three attacks. This high consistency agrees with previous data obtained in more selected migraine patients (12) and may allow us to investigate in more detail the therapeutic potential for the management of migraine in the premonitory phase. Non-randomized trials have suggested that naratriptan and dopamine antagonists may be effective in preventing the development of migraine attacks when given during the PS phase. There is also evidence that the dopamine antagonist domperidone can prevent migraine, but only if taken at least 6 h before the expected attack (3, 26, 27). The type of symptoms reported may also give us some insight into migraine pathophysiology. For example, excessive yawning, which is associated with dopamine release (28), was one of the most common and consistent PS, which again suggests that dopaminergic mechanisms, possibly involving brainstem nuclei, may play a role in the premonitory phase.
With regard to the influence of clinical and demographic variables, gender, age, severity of headache and MIDAS scores did not clearly influence the frequency or the profile of PS. Even though the global numbers were not significant due to the low proportion of aura attacks in this series, migraine with aura patients experienced more PS and RS than migraine without aura patients. Interestingly, the individual symptoms which were significantly more frequent in migraine with aura patients were those usually associated with the headache phase, such as nausea and phono- and photophobia, raising the question as to when ‘headache phase’ really begins. Other authors have noticed that some patients added voluntarily ‘headache of different quality or severity’ as a PS although they had been instructed to record only non-headache symptoms (12). They considered that headache evolves from the PS phase over a variable period, with full-blown migraine headache finally developing when a critical physiological threshold is reached. It seems logical to propose that patients with migraine with aura attacks experience more relevant CNS changes and cross this physiological threshold more easily and intensely compared with migraine without aura subjects. Future studies with a greater and more balanced number of migraine with and without aura patients are therefore necessary. Of interest is the finding that patients on preventatives showed a significantly decreased frequency of PS, and to a lesser degree of RS, compared with those not on these medications. This suggests that preventatives, known to reduce the frequency and sometimes the intensity of the headache phase, are also able to reduce the non-headache symptoms and that these medications can reduce in some way the CNS activation occurring before the headache phase. In line with this, the use of preventatives has been shown to revert the increased intensity dependence of auditory evoked potentials seen in migraineurs (29). Finally, severity of headache was significantly associated with a higher frequency of RS, which suggests that, together with specific non-headache symptoms beginning before and remaining after the headache through to the resolution phase, at least some of the RS may be non-specific transient sequels of a severe, full-blown migraine attack.
