Abstract
Objective
To review characteristics and outcomes of all cases of visual snow seen at our institution, with attention to possible triggering events or comorbidities.
Methods
This is a retrospective case series of patients seen at our tertiary care center from January 1994 to January 2020. Charts were reviewed if they contained the term “visual snow”.
Results
Of the 449 charts reviewed, 248 patients described seeing visual snow in part or all of their vision. Thirty-eight reported transient visual snow as their typical migraine aura. Of the remaining 210 patients, 89 were reported to have either an inciting event or contributing comorbidity for their visual snow symptoms, including: Post-concussion (n = 15), dramatic change in migraine or aura (n = 14), post-infection (n = 13), hallucinogen persisting perception disorder (n = 10), ocular abnormalities (n = 7), idiopathic intracranial hypertension (n = 4), neoplastic (n = 1), and posterior cortical atrophy (n = 1). Some patients had partial improvement with benzodiazepines (n = 6), lamotrigine (n = 5), topiramate (n = 3) and acetazolamide (n = 3). Presenting characteristics were similar, but patients with visual snow attributed to an inciting event or contributing comorbidity were more likely to have some improvement in their symptoms by last follow-up compared to spontaneous visual snow (p < .001).
Conclusions
Though most cases of visual snow are spontaneous, potential secondary causes should be recognized by clinicians. Patients who develop visual snow after an inciting event or related to an underlying comorbidity may have a better prognosis than those in whom it develops spontaneously. In select cases, treatment of the suspected underlying cause may significantly alleviate the otherwise typical intractable visual disturbances associated with visual snow.
Introduction
Visual snow (VS) is a symptom described as seeing numerous flickering dots comparable to “TV static”, typically throughout the visual field (1). International Classification of Headache Disorders 3rd edition criteria define patients as having visual snow syndrome (VSS) if they experience persistent VS for longer than 3 months, along with at least two other visual symptoms including: Photophobia, nyctalopia (impaired night vision), palinopsia (after images or trailing of moving objects) and enhanced/excessive entoptic phenomenon (floaters, “self-light” or moving colors in vision, photopsias, or blue-field entoptic phenomena) (1) (Figure 1).

Illustrated examples of symptoms seen in visual snow syndrome. (a) Normal vision; (b) visual snow; (c) palinopsia (both ghost-like afterimages next to the sign and visual trailing behind the car); (d) other frequent or persistent positive phenomena (including increased floaters, photopsia, blue field entopic phenomena, and colored blobs); (e) nyctalopia (impaired night vision); (f) composite illustration showing symptoms that when combined may cause significant disability. From: Metzler AI and Robertson CE. Visual snow syndrome: Proposed criteria, clinical implications, and pathophysiology. Curr Neurol Neurosci Rep 2018; 18: 52; used with permission from Mayo Foundation for Medical Education and Research, all rights reserved.
In the past few years, we have learned more about the epidemiology of VS and VSS through a number of case series and internet surveys (1–5). VSS has a prevalence of around 2% (4) and seems to affect men and women equally, with a mean age of onset of 12.8 ± 13.2 years (2). The symptom of VS may come on gradually or abruptly at any age and for some patients has been present for as long as they can remember. Most describe a spontaneous onset, but a subset report VS as starting abruptly after taking a hallucinogenic drug or following infection or head injury (2). Secondary VS cases, including those related to hallucinogen use, eye pathology, or epilepsy are often excluded from studies on VSS, which could explain why we know less about this subset of patients. The purpose of this study was to review the characteristics and outcome of all cases of VS at our institution, with attention to cases that were attributed to a triggering event or comorbidity.
Methods
Study design
This is a retrospective chart review of patients who presented to our institution between January 1994 and January 2020. This study was approved by our institutional review board (17-010962).
Data collection and analysis
A database engine, Advanced Text Explorer, was used to search clinical records for the term “visual snow”. Four hundred and forty-nine patients were identified by the database. Fifty-nine patients had not consented to research participation, and were excluded prior to their charts being accessed. Charts were reviewed from all medical specialties; however, only patients who were seen by Neurology or Ophthalmology/Neuro-ophthalmology had enough detail about the visual symptoms to be certain of the diagnosis.
To be included, the patient had to use descriptions such as “similar to TV static” or “diffuse grainy/pixelated vision” to describe their visual symptom. If these terms were not present, but if they identified their vision as similar to the computer simulated models of VS, they were also included. As this was a study of all patients with VS, not just patients meeting ICHD-3 criteria for VSS, we did include patients with monocular or partial field VS, and these are discussed in the results. Excluding patients with VS as migraine aura, the majority of our patients (98.5%) had VS described throughout their visual field. If the patient’s description of symptoms involved only “dots” or “spots” or other vague descriptions that could have represented alternative diagnoses, they were excluded, even if the patient or clinician used the term “snow” or “visual snow”.
Cases were reviewed initially by a single author, DM, and final included cases were reviewed by two authors (DM and CER) to ensure agreement that patients met inclusion and exclusion criteria. Ninety-seven patients were excluded after review of the visual symptoms did not match the predefined description of VS. Some of these had no clear diagnosis and some had alternative diagnoses, such as scotomas, visual field defects, persistent aura, photopsias, floaters, and so on. An additional 45 patients had a “visual snow” diagnosis listed by their provider, either in their past medical history or as a referral diagnosis from an outside institution, but were excluded as there was not enough descriptive information available in the chart to confirm the diagnosis (see Figure 2).

Flowchart for excluded patients. Flowchart outlining how patients were excluded from our case series.
Additional details were gathered from cases where the patient and/or their physician attributed the VS to a specific inciting event, systemic illness, structural lesion along the visual pathway or associated comorbidity. These cases were compared to spontaneous cases of VS with regard to VS characteristics, comorbidities, treatment response and patient outcomes. For simplification in our manuscript, instead of referring to these cases as “VS cases attributed to a specific inciting event, illness, lesion, or comorbidity” we assigned these cases the term “secondary VS” and spontaneous cases were referred to as “primary VS”. Though patients and their treating physicians were sometimes quite convinced of the association, it would not be possible to prove causation in this type of study.
Collected demographic data included: Age at symptom onset, duration of follow up, history of migraine, VS characteristics, comorbid diagnoses, treatments and their response, neurologic and ophthalmologic evaluations, and patient outcomes.
Additional details were gathered from cases where the patient and/or their physician attributed the VS to a specific inciting event, systemic illness, structural lesion along the visual pathway or associated comorbidity. These cases were compared to spontaneous cases of VS with regard to VS characteristics, comorbidities, treatment response and patient outcomes. Categorical variables were compared using Fisher’s exact test of independence. Statistical analysis was performed using SPSS 25.0 (IBM Corp., Armonk, NY).
Results
Demographics
Of the 449 charts reviewed, 248 patients met criteria for VS. Thirty-eight reported VS as their typical migraine aura (not included in demographic analysis). Of the remaining 210, 89 were men and 121 were women. Twenty-two (10.5%) patients had VS for “as long as they could remember”. Of the remaining 188 patients, there was a median age at onset of 26.1 ± 14.1 years. The median length of follow up for all patients was 3.6 years after their symptom onset. For those who reported their symptoms present for “as long as they could remember”, an age of three was used to estimate symptom onset. VSS characteristics included: Persistent photophobia (n = 93, 44.3%), nyctalopia (n = 58, 27.6%), palinopsia (n = 104, 49.5%), floaters/blue field entoptic phenomenon (n = 128, 60.9%), self-light (n = 34, 16.2%) and photopsias (n = 50, 23.8%). Additional symptoms included tinnitus (n = 71, 33.8%), brain fog (n = 37, 17.6%), and spells of dissociation/derealization (n = 28, 13.3%). Five patients (2.4%) described a family history of VS, though this was not specifically asked by many interviewers.
Comorbidities included: Migraine with aura (n = 102, 48.6%), migraine without aura (n = 56, 26.7%), anxiety (n = 102, 48.6%), depression (n = 58, 27.6%), postural orthostatic tachycardia syndrome (POTS) (n = 48, 22.9%), frequent or persistent dizziness (n = 28, 13.3%), concussion (n = 23, 11%), attention deficit disorder/attention deficit hyperactivity disorder (ADD/ADHD) (n = 30, 14.3%), fibromyalgia (n = 15, 7.1%) and post-traumatic stress disorder (PTSD) (n = 3, 1.4%).
Testing/imaging
Of the 210 patients, 181 (86.2%) had brain magnetic resonance imaging (MRI) sometimes in combination with MR angiograms or MR venograms, 165 (78.6%) had ophthalmologic exams, 55 (26.2%) had electroencephalograms (EEG), 51 (24.3%) had ocular coherence tomography (OCT), 15 (7.1%) had positron emission tomography ([18F]-FDG PET) brain scans, 28 (13.3%) had paraneoplastic panels, and 18 (8.6%) had visual evoked potentials (VEPs).
Of the 181 cerebrovascular MRIs performed, 23 were read as abnormal. Four showed indirect signs of intracranial hypertension such as an empty sella (n = 3) and bilateral transverse sinus stenosis (n = 2). Two showed white matter lesions consistent with multiple sclerosis. Two patients had pineal cysts, two patients had arachnoid cysts, and one patient had a pars intermedia cyst. Four patients had right hemispheric parenchymal abnormalities including mesial temporal sclerosis (n = 1) or developmental venous anomalies in the temporal lobe (n = 1), thalamus (n = 1), or frontal lobe (n = 1). Three patients had left hemispheric parenchymal abnormalities consisting of a temporo-occipital developmental venous anomaly (n = 1), frontal lobe post-surgical changes (n = 1), or temporal lobe post-surgical changes (n = 1). Two patients had Chiari malformations (11 mm, 9 mm). One patient had midbrain developmental hamartomas. One patient had moderate to severe posterior predominant atrophy involving the bilateral parietal, occipital, and posterior temporal lobes consistent with posterior cortical atrophy (PCA). One patient had extensive subcortical white matter disease considered disproportionate for age.
Of the 165 eye exams, 15 were considered abnormal. Two patients had Fuch’s corneal dystrophy. Three patients had vitreous detachments with subsequent floaters. Two patients had optic nerve abnormalities including bilateral optic neuritis (n = 1) and bilateral, mild optic atrophy (n = 1). One patient had multifocal choroiditis. Seven patients had retinal abnormalities including macular atrophy (n = 2), macular holes (n = 2), nuclear sclerosis (n = 1), central serous retinopathy (n = 1), and vitelliform lesions (n = 1). In total, seven of the 15 abnormal exams were felt to be severe enough that the structural abnormalities could contribute to VS. Two of the 51 OCTs were considered abnormal, both with decreased retinal nerve fiber layers compared to controls, not considered severe enough to contribute to VS. None of the VEPs were abnormal.
Of the 55 EEGs, three were abnormal. One showed generalized slowing. One showed breach rhythm related to a craniotomy. One showed generalized epileptic discharges, maximal in the left posterotemporal region associated with ictal VS in the right visual field.
Of the 15 [18F]-FDG PET CT scans, six were considered abnormal. Two patients had nonspecific, mild generalized cortical hypometabolism. One patient had decreased uptake in the right inferior medial temporal lobe (questioned as a possible artifact). One patient showed possible hypermetabolism of bilateral lingual gyri. One patient had markedly decreased metabolism involving both occipitoparietal lobes with extension to posterolateral temporal lobes consistent with PCA. One patient showed biparietal hypometabolism with borderline bilateral occipital hypometabolism.
Of the 28 paraneoplastic panels, seven were considered abnormal. Two patients had positive anti-retinal antibodies (levels not available). Two patients had positive acetylcholine receptor ganglionic neuronal antibodies (0.03 nmol/L and 0.11 nmol/L). One patient had positive voltage-gated potassium channel antibodies (0.04 nmol/L). One patient had positive N-type calcium channel antibodies (0.18 nmol/L). One patient had positive striational antibodies (titer of 1:960). Four patients were evaluated by Autoimmune Neurology specialists for their positive tests. Ultimately, these specialists felt the positive titers were unlikely to be contributory to the symptoms of VS and that immunosuppressive therapy was not warranted.
Inciting events or possible contributing comorbidities
Of the 210 patients, 89 (42.4%) developed VS either abruptly after a perceived inciting event or related to a comorbid illness (see Table 1). Though causation could not be proven, cases were recorded if the patient or physician attributed the onset of VS to these events or comorbidities. These included: head injury (n = 15, 7.1%), including three that occurred in a delayed fashion 2–12 weeks after injury with other post-concussive symptoms; dramatic change in headache or aura (n = 14, 6.7%) including five with new daily persistent headache and five with their first or most severe migraine or aura; post-infection (n = 13, 6.2%); hallucinogen persisting perceptual disorder (HPPD) (n = 10, 4.8%); medication-related (n = 11, 5.2%); ocular abnormalities (n = 7, 3.3%); hormonal changes (n = 5, 2.4%); idiopathic intracranial hypertension (IIH) (n = 4, 1.9%); epilepsy (n = 3, 1.4%) including one patient with ictal VS in the right hemi-field; new unclassified autoimmune diagnoses (n = 2, 1.0%) of which one improved with prednisone; multiple sclerosis (n = 2, 1.0%); other systemic illness (n = 2, 1.0%) including severe symptomatic hypothyroidism and hypertensive urgency (blood pressure of 190’s systolic, one week post-partum); psychiatric (n = 1, 0.5%) with new onset schizophrenia; neoplastic (n = 1, 0.5%) with left anterior temporal xanthoastrocytoma recurrence; and degenerative (n = 1, 0.5%) involving a patient who developed PCA.
Contributing inciting events/comorbidities and their outcomes.

ADD: attention deficit disorder; DMX: acetazolamide; EMU: epilepsy monitoring unit; IVF: in vitro fertilization; OCP: oral contraceptive; PPI: proton pump inhibitor; TBI: traumatic brain injury; VS: visual snow.
After recording the presence of commonly described VS features (e.g. photophobia, palinopsia, self-light) and select comorbidities, we compared the proportion of patients with either primary VS or secondary (see Table 2). Patients with primary VS were statistically more likely to have anxiety, palinopsia or floaters/blue-field entoptic phenomenon mentioned.
Comorbidities and visual snow characteristics of patients with primary and secondary visual snow.
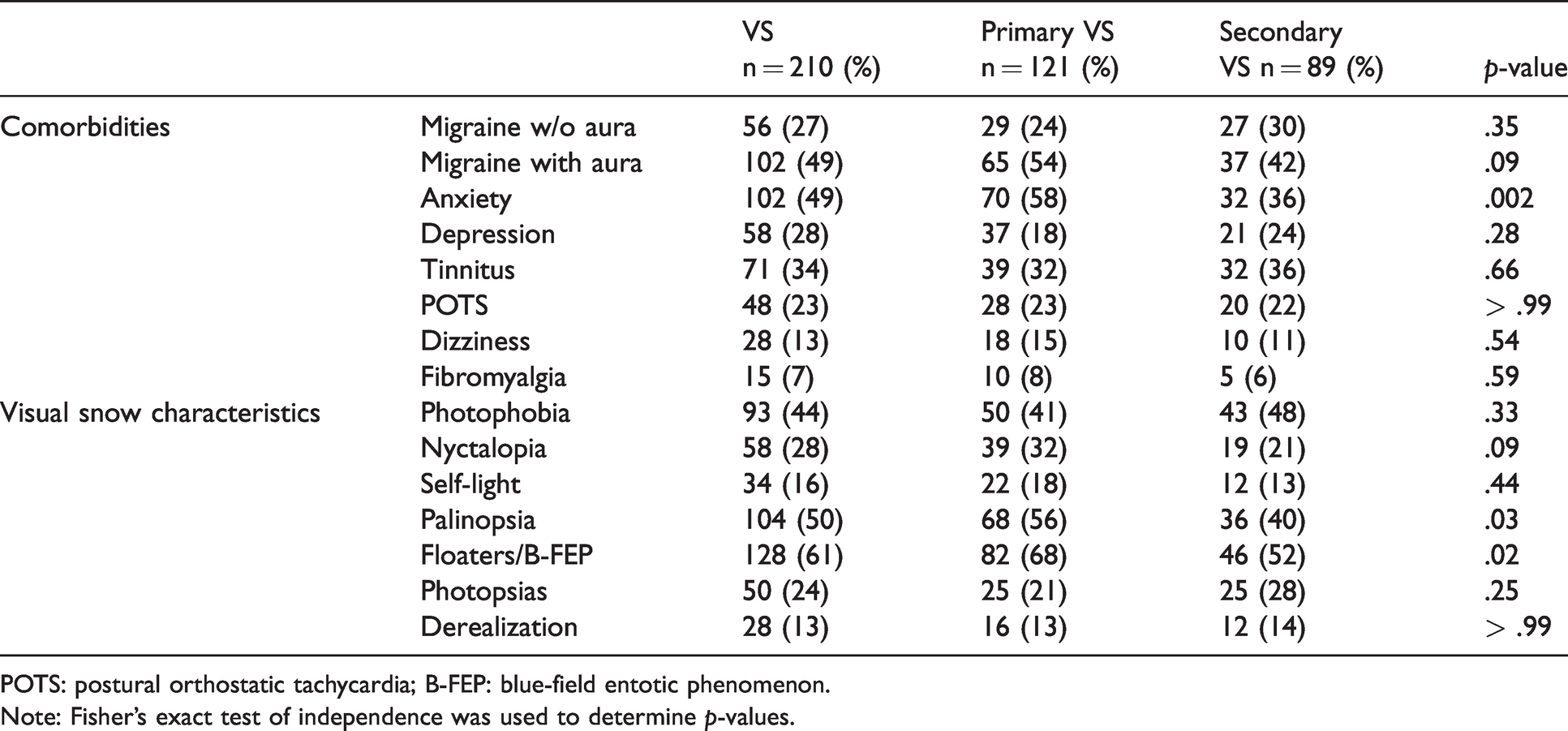
POTS: postural orthostatic tachycardia; B-FEP: blue-field entotic phenomenon.
Note: Fisher’s exact test of independence was used to determine p-values.
Response to treatment
Twenty-three patients had a partial response to medication (see Table 3). A partial response was defined as any improvement in VS symptoms described in follow-up visits or communications. None of the patients in our cohort had complete remission of their symptoms. Information regarding medication treatment was available for 126 patients (60%). Medications associated with the most benefit included: Benzodiazepines (n = 6), lamotrigine (n = 5), topiramate (n = 3), and acetazolamide (n = 3). There were additional medication trials without improvement, including: gabapentin (n = 33), duloxetine (n = 10), valproic acid (n = 10), botulinum toxin (n = 8), and venlafaxine (n = 5), among others.
Improvement of visual snow with medication.

Note: A total of 126 patients (60%) had documented medication trials.
Outcomes
Twenty-seven (30.3%) patients with secondary VS and 10 patients (8.3%) with primary VS reported at least some amount of improvement in their VS symptom by their last follow-up visit. Fisher’s exact test of independence was performed and showed this difference was significant, p < .001.
Factors that improved or worsened VS
Some patients shared factors that made their VS better or worse. Most commonly reported factors that improved VS were: Visual/mental distraction, including visually stimulating environments (n = 3); tinted lenses, details unclear (n = 4); a “good night’s sleep” (n = 2); stress reduction techniques (n = 4); and “visual snow relief” videos (videos of TV static; n = 2).
The most common factors reported by patients to worsen their VS included: Exercise (transient at the moment of exertion; n = 6); caffeine (n = 3); stress (n = 4); marijuana (n = 2); poor sleep quality (n = 2); botulinum toxin (transient on the day of or day after injection; n = 2); various SSRI/SNRI/NDRIs (sertraline (n = 3), venlafaxine (n = 2), bupropion (n = 1), escitalopram (n = 1), fluoxetine (n = 1)); nortriptyline (n = 1); topiramate (n = 1); and verapamil (n = 1).
Select interesting cases from our series
Posterior cortical atrophy (PCA): A 51-year-old woman developed classic VS that she reported as sometimes interfering with reading text. Over 2–3 years, she developed additional difficulty with visual agnosia and visual spatial impairment. Within 3–4 years after onset, she had notable difficulty with construction and calculation, as well as ocular apraxia/optic ataxia. Within seven years after onset, she was “functionally blind”. Brain MRI and [18F]-FDG PET were consistent with PCA. Left temporal lobe neoplasm: A 22-year-old with a history of left anterior temporal pleomorphic xanthoastrocytoma, status-post subtotal resection, presented with new VS throughout her visual field, worse at night. Symptoms did not respond to levetiracetam 500 mg twice daily. Imaging showed an increase in enhancement and mass effect along the previous resection site, consistent with tumor recurrence. After resection of the recurrence and radiation of the region, the VS resolved. Idiopathic intracranial hypertension (IIH): Four patients were diagnosed with IIH-related VS. Two improved with acetazolamide, one improved after a lumbar puncture and followed up locally after that. One resolved with ventriculoperitoneal shunting, but the VS returned 7 months later after a traumatic brain injury. Improvement following thalamic infarct: One patient had a long history of classical persistent VS since at least age 10. At age 25, she developed an unprovoked deep cerebral venous thrombosis with an associated hemorrhagic infarct of the left thalamus. For one week following the infarct, she had resolution of VS (no VS present), followed by a gradual return to baseline symptoms.
Discussion
Thirty-eight patients in our series experienced episodic VS with their migraine attacks, similar to cases recently reported in the literature (6). While this implies that “TV-static” visual phenomena may be seen in the context of migraine aura, persistent VS is thought to represent a different underlying pathophysiology (7,8). Interestingly, worsening of VS during migraine has been seen in previous studies (2,9), and it has been suggested that this may be related to the shared dishabituation common to both migraine and VS (2).
Though the majority of the patients in our series developed VS spontaneously, a significant proportion of our patients (or their physicians) attributed their VS to a secondary cause and/or inciting event. This is consistent with a recently described large web-based survey of VS patients, which described a subset who felt their VS started after trauma, infection, medication, recreational drugs, or a migraine attack (2). While patients who develop VS following recreational drug use are generally classified within the condition hallucinogen persisting perception disorder (HPPD), it is not clear whether this is pathophysiologically different from other secondary forms of VS, such as post-infection or post-traumatic cases.
The pathophysiology underlying VS is not yet understood, but is a subject of great interest. One theory suggests VS is a product of cortical hyperexcitability with dysfunction of higher order visual processing, possibly allowing patients to see normally subthreshold visual stimuli (10). This would seem to be supported by electrophysiologic studies that demonstrate hyperexcitability or reduced habituation of visual cortices (11–13). This may also be supported by functional imaging studies showing increased metabolic activity in parts of the visual cortex, most notably in the right lingual gyrus as well as the left cerebellum (9,14). Visual cortical hypermetabolism has not been the only imaging finding in VS patients, however (15,16). In our series, one of the 15 patients had hypermetabolism in the lingual gyri on [18F]-FDG PET, though in their case it was bilateral. We also had two patients with parietooccipital hypometabolism and one patient with borderline right temporal hypometabolism. Similar to our series, two of the original cases of VS described by Liu and colleagues had evidence of hypoperfusion in the biparietal (n = 1) and bilateral parietooccipital regions (n = 1) (15). A more recent study of VSS patients using [18F]-FDG PET and voxel-based morphometry (VBM) found hypermetabolism near the right lingual and fusiform gyrus, but also noted hypometabolism in the right superior temporal gyrus and left inferior parietal lobe, as well as gray matter volume alterations in the temporal lobe, parietal lobe, and limbic system (16). The authors hypothesized that the changes outside of the visual cortex may explain some of the nonvisual symptoms in VSS, such as tinnitus and cognitive changes (16). Their study, along with cases described in our series and by Liu and colleagues (15), emphasizes the imaging variability that can be seen in VS patients.
A different theory on the pathophysiology of VS emphasizes the thalamic role in visual processing, viewing VS as a type of thalamocortical dysrhythmia, with a functional disconnection of the thalamus (17,18). Though there has been no direct evidence for this, there is growing interest in the role of thalamocortical connections in migraine, visual hallucinations, and tinnitus (17,19–25). Authors describe a situation of dissociated thalamocortical activity, where excessive inhibition of the thalamus creates an effective “thalamic blindness”, with subsequent reduced cortical inhibition allowing hallucinations (26). In this context, it is interesting that one of our cases with spontaneous VS for 15 years noticed resolution of her symptoms for 1 week after a hemorrhagic infarct of the left thalamus. Perhaps the hemorrhage disrupted the excessive thalamic inhibition, allowing a transient return of the excitatory-inhibitory thalamocortical balance.
In our series, the most common inciting event was traumatic brain injury (TBI), with VS often developing in concert with daily headaches and post-concussive symptoms. Like spontaneous VS, it is not clear by what mechanism patients abruptly started seeing VS after their head injury. It seems probable that TBI directly and indirectly damages cortical and subcortical structures that are associated with visual processing. Animal models of TBI have shown altered thalamic myelin integrity (27) and even mild TBI has been shown in some cases to contribute to excitotoxic injury with delayed cell death in the cortex, thalamus, and subcortical white matter tracts (28). Some authors hypothesize that disrupted thalamocortical circuits contribute to some of the symptoms of TBI (27), and in theory this might also contribute to VS symptoms.
Three percent of the patients in our study experienced VS as a symptom of eye pathology. Ocular pathology is a well-known potential cause of positive visual phenomena, with one study showing 22% of patients with retinitis pigmentosa report “TV-static” in their vision (29), and another study showing 23% of patients with macular degeneration and other ocular pathology report multiple visual particles “like rain drops” over everything (30). Bilateral VS has also been reported in Leber’s hereditary optic neuropathy (31). The pathophysiology for these positive visual phenomena is thought to be similar in mechanism to Charles-Bonnet syndrome, where visual deafferentation can give rise to visual release hallucinations; many of these patients may also describe palinopsia, photopsias, and other elementary hallucinations (10,29,30).
Suggested evaluation for VS
Given the possibility of an underlying ocular etiology, it is recommended that all patients presenting with VS undergo an ophthalmologic evaluation (10,17,32,33). Based on our series, we would also recommend a complete neurologic evaluation of patients when they present with symptoms resembling VS (see Figure 3). The neurological history and exam should look for higher order and complex visual deficits (e.g. visual agnosia/prosopagnosia, simultagnosia, constructional apraxia or visual memory) to assess for any neurodegenerative process, particularly in patients who develop VS at an older age. Given that IIH was a potentially treatable cause of VS in our series, we would recommend screening for this by history (e.g. transient visual obscurations, whooshing tinnitus, risk factors) and performing a funduscopic examination for papilledema.

Evaluation of patients with new onset visual snow symptoms. Outline of suggested evaluation of patients with visual snow symptoms including pertinent historical questions, examination findings, and evaluations.
In cases of otherwise unexplained new onset VS, we recommend imaging with an MRI of the brain with gadolinium looking for lesions anywhere along the visual pathway. One of our cases involved neoplasm in the left anterior temporal lobe, presumably contributing to VS through an effect on nearby optic radiations. Interestingly, the VS was described as bilateral and diffuse, and resolved after resection and radiation of the recurrent tumor. MRI of the brain could also look for evidence of demyelinating disease like multiple sclerosis, as this was present in two patients in our series. Atrophy in the occipital and parietal region might also alert the clinician about the possibility of a neuro-degenerative process, though functional imaging may also be needed. Given a recent report of Creutzfeldt-Jakob Disease (CJD) presenting with VS (34), diffusion imaging should be included to evaluate for cortical ribboning. An EEG may be considered, both for changes suggestive of CJD or neurodegenerative processes, as well as to rule out epileptic positive visual phenomena. We did have three patients with VS in the setting of epilepsy; two were felt to be ictal while the third started during a photo-induced seizure and then continued.
Treatment
Our series was not designed to study response to treatment, but we did collect data on medicines tried along with clinical response. As a group, the best clinical outcome was noted in patients with IIH, after addressing the increased pressure with either a lumbar puncture, acetazolamide, or a shunt. A few other cases also improved after removing the aggravating factor (e.g. removal of the temporal neoplasm, discontinuation of the offending drug). Two patients improved after an underlying inflammatory process was addressed: One patient with systemic illness noticed improved VS after starting prednisone, another patient with multifocal choroiditis and inflammatory retinopathy improved after starting adalimumab. In cases of persistent VS, no medicine provided complete relief, but some patients did report mild to significant improvement with lamotrigine, topiramate, or benzodiazepines. There were some patients that also reported improvement with non-medicine strategies, including distraction, tinted lenses, and an internet video called “visual snow relief,” which is a video of TV static that may be analogous to white noise treatment for tinnitus. Similar to other studies (9), patients in the series with concurrent migraine would often notice improvement in their migraine with persistence of their VS.
Strengths and limitations
The biggest strength of this case series lies in its large number of patients. In the past couple of years, we have gained a lot of knowledge about visual snow characteristics through well executed web-based surveys (2). However, our study is the largest series of visual snow patients seen and evaluated in person, with other series containing fewer than 100 patients (5,18). Historical details available in the medical chart, including access to neuroimaging, examination findings, and ophthalmologic studies allowed additional understanding about the potential inciting events and medical or neurological comorbidities that might have contributed to the onset of these patients.
This study had limitations expected in a retrospective review at a tertiary center. We were limited to the information available in the chart, and not all physicians documented the same level of historical detail. For this reason, our study likely underestimates the prevalence of some of the clinical features of VS and accompanying comorbidities. This would be supported by higher numbers of photophobia, palinopsia, and entoptic phenomena and comorbid tinnitus reported in prospective surveys (2). We also had a subset of patients who were seen and evaluated here for their VS, but then returned home. In these cases, our follow-up information was limited only to available outside records or correspondence.
Conclusions
Despite the stereotypic patient presentation, the symptom of VS may be associated with a diverse array of inciting events and medical conditions. While most cases of VS are spontaneous, it is critical for clinicians to rule out possible contributing causes of VS at presentation. Based on our series, patients who develop VS after an inciting event or related to an underlying comorbidity may have a better prognosis than those in whom it develops spontaneously. Furthermore, select cases may be treatable and others may require additional monitoring over time.
Clinical implications
Not all patients presenting with visual snow have visual snow syndrome. When patients present with new onset spontaneous visual snow, the neurological examination should include evaluation for higher order and complex visual deficits such as visual agnosia, simultagnosia or constructional apraxia. When patients present with new onset spontaneous visual snow, clinicians should consider evaluating for underlying contributing and possibly treatable causes, such as idiopathic intracranial hypertension, ocular pathology, lesions along the visual pathway or new medicines/hormones started at the onset of symptoms.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DGM declares no conflict of interest. IG receives honoraria as an author for UpToDate. CER receives honoraria as an author for UpToDate. She has also served on the advisory board for Amgen, Eli-Lily, Biohaven, Impel Neuropharma and Lundbeck.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Publication history
Paper previously presented as a poster presentation at the American Headache Society 62nd Annual Virtual Scientific Meeting, June 2020.
