Abstract
Visual snow syndrome (VSS) manifests as continuous, fine–grained visual static that is often accompanied by other visual symptoms. Its frequent association with migraine, particularly migraine with aura (MwA), has prompted debate regarding a shared pathogenic substrate. To interrogate this relationship, we performed a narrative review of clinical, neuroimaging and electrophysiological studies on VSS and MwA. The clinical picture of VSS is a persistent phenomenon that does not fluctuate with the migraine cycle and shows no response to therapeutics established to be useful in migraine. Moreover, structural and functional neuroimaging in VSS consistently demonstrates selective abnormalities within primary visual, salience and attentional networks, paralleled by distinctive evidence of glutamatergic dysregulation and impaired top–down suppression in electrophysiological recordings. Collectively, the available evidence supports VSS as a discrete disorder marked by aberrant salience assignment and impaired sensory gating, with clinical features and pathophysiology that are separate from those of MwA. While features such as shared serotonergic dysregulation, involvement of comparable cortical territories and high comorbidity suggest overlap between MwA and VSS, these similarities are likely better attributed to a shared predisposition for increased cortical excitability than to a single nosological entity. Future research aiming to characterize further network abnormalities in VSS will be pivotal for guiding the development of targeted therapies.
This is a visual representation of the abstract.
Introduction
Visual snow (VS) is a persistent visual disturbance characterized by the perception of continuous snow or flickering dots that affect the entire visual field. Patients describe this visual phenomenon as resembling the static noise of an untuned television (1). VS is not necessarily a stand-alone symptom because it can accompanied by additional visual symptoms in about two-thirds of individuals (2,3). Examination of a clinical cohort supported the concept of a syndrome, leading to the definition of visual snow syndrome (VSS) as a complex chronic disorder with proper diagnostic criteria in 2014 (2). According to the current criteria, at least two additional visual symptoms should be present along with VS to diagnose the full syndrome (2), including palinopsia, nyctalopia (i.e. impaired night vision), enhanced entoptic phenomena and photophobia (4). Although the first description of patients with symptoms resembling VSS dates back to 1995 (1), some of the symptoms possibly involved in VSS can be found in 19th century medical literature (5). The exact prevalence of VS and VSS is currently unknown. In the UK, an estimated prevalence was found to be around 3.7% and 2.2%, respectively (3), while, in Italy, the prevalence of VSS seems to be around 1% (6).
Since its first description, VSS has been considered a possible phenotypic variant of aura (1). This is mainly a result of the high co-occurrence between the two diseases, with a prevalence of around 57–72% for migraine without aura (MwoA) and 27–37% for migraine with aura (MwA) in VSS patients (2,7). Notably, the prevalence of MwA within VSS cohorts appears to be in line with its occurrence in the general population (8), implying that part of their overlap may reflect chance or referral bias. Comorbid migraine can worsen the additional visual symptoms and tinnitus, but VSS severity does not parallel with the migraine cycle (9) Although the exact neurophysiological mechanism for VSS is unknown, some similarities may aid in understanding disease mechanisms. VSS has been hypothesized to be linked with dysfunctional sensory processing and altered occipital cortex excitability (10), a common denominator also in migraine aura pathophysiology (11). Perhaps unsurprisingly, given photophobia in VSS, areas of the brain with increased metabolic activity demonstrated with 18F-deoxyglucose PET in VSS patients (9) are similar to those reported using blood flow methods in migraine patients experiencing photophobia during attacks (12).
Furthermore, Schankin et al. (2) provided evidence in favour of VSS as a distinct and unique visual disorder separate from persistent migraine aura. Although a few cases of episodic VS occurring exclusively during migraine attacks have been reported (13), VSS is a long-standing and continuous presentation of visual disturbance, which contrasts with the episodic nature of migraine aura (14–16). Additionally, the aura by itself is not linked to a more severe phenotype of VSS (9) and standard migraine treatments are unhelpful for VSS (15). Moreover, it should be noted that VSS is often accompanied by other perceptual disturbances such as tinnitus, reported in up to 75% of the cases (7,17), reflecting an involvement that goes beyond purely visual domains (18), and MwA may also present with negative visual symptoms (19) or even no visual symptoms at all (20). Nevertheless, the connection between VSS and migraine aura remains unclear.
In this narrative review, we first show the patient perspective on perceived differences between aura and VSS (Figure 1). We then discuss the clinical perspective by describing the mechanisms of each additional visual symptom of VSS and its presence in MwA. Finally, we focus on the neurophysiological and neuroimaging evidence (pathophysiological perspective, as summarized in Figure 2 and Table 1) by highlighting similarities and differences with migraine aura. Understanding this intricate relationship may inspire and guide future studies that can help reveal the underlying mechanisms of VSS.

Clinical differences between visual snow and visual aura.
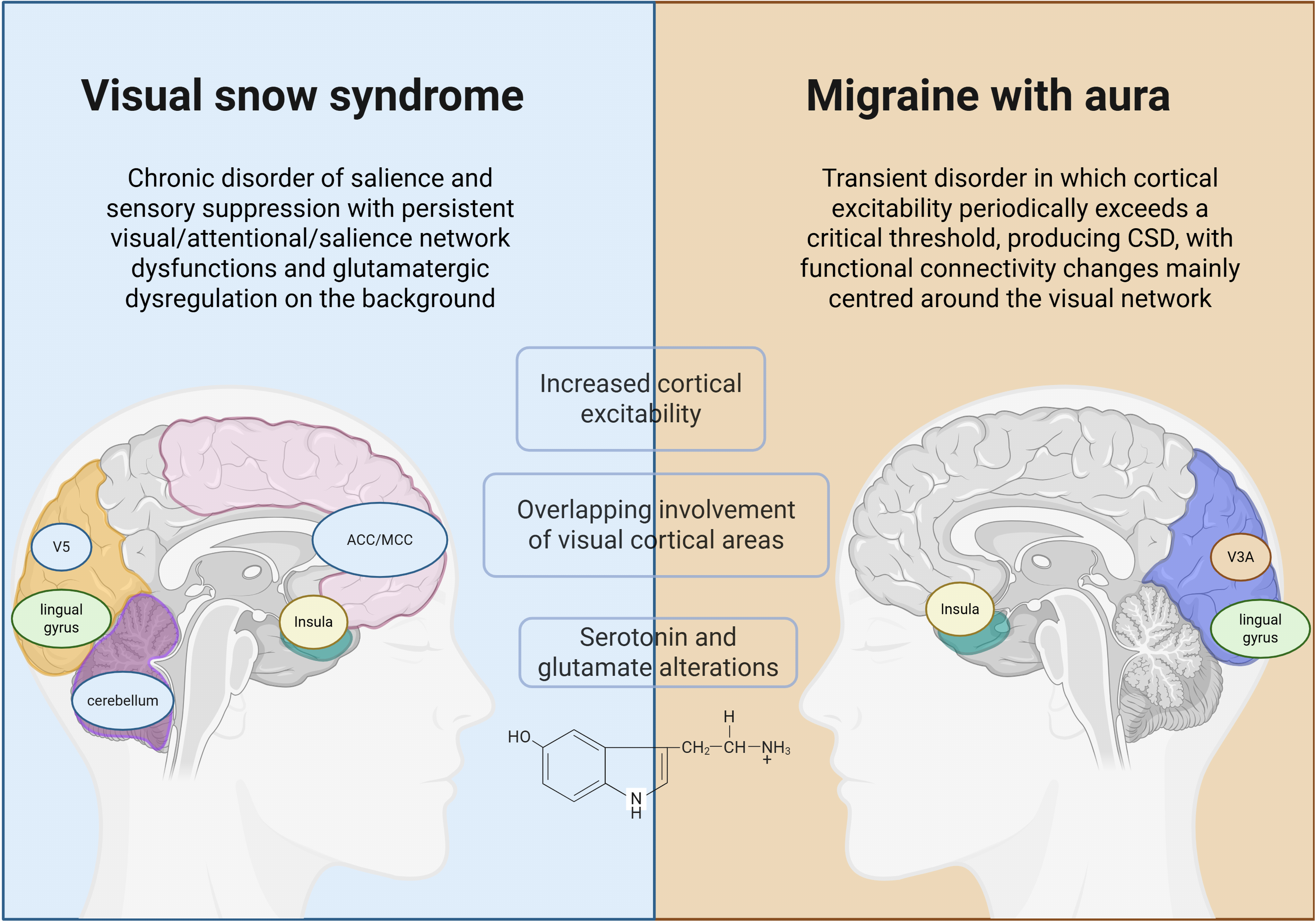
Pathophysiological perspective on visual snow syndrome and migraine with aura. CSD = cortical spreading depression.
Comparison of visual snow syndrome (VSS) and migraine with aura (MwA) by neuroimaging and neurophysiological studies.

Methods
This is a narrative literature review. A literature search was conducted on the PubMed database (by SB, VS and GS independently) on 31 March 2025. We searched for original research publications on VS, VSS and MwA in regard to clinical presentation, as well as structural and functional neuroimaging and neurophysiology, using the following terms: “visual snow”, “migraine” and “aura” in combination with the terms “photophobia”, “nyctalopia”, “palinopsia”, “enhanced entoptic phenomena”, “neuroimaging”, “neurophysiology”, “EEG”, “TMS” and “visual evoked potentials”. Additionally, we selected manuscripts already known to ourselves.
Original articles were preferentially selected, while reviews were selected when space did not allow a more comprehensive discussion. The senior investigators (PJG, CJS and FP) revised the final selection of the articles.
We asked our patients with both VSS and MwA to provide a brief description of their symptoms. We selected for inclusion in the present paper the most detailed and thorough description that we received. Consent on the sharing of clinical details included in the patient perspective was obtained in writing from the patient.
Patient perspective
My VSS symptoms started suddenly – one morning in the fall of 2012, I woke up as usual and looked down at the carpet on the floor. To my surprise, I noticed thousands of flickering dots across the surface. I wasn’t experiencing any headaches or other unusual symptoms. I got up and kept staring at the carpet and the rest of the room. The dots were more visible against the dark-coloured carpet than the lighter-coloured walls. They weren’t fine but rather moderately sized. The flickering was most intense upon immediately waking up but would ease slightly after a few minutes and become a bit finer, smaller in size. However, they never completely disappeared. Over the following months, the symptoms persisted. The flickering was still most intense in the morning and was more noticeable against darker backgrounds. I began to notice new visual phenomena: patterned clothes with fine lines appeared to float, red and blue colours on fabric sometimes seemed to flash, and I saw the bright, squiggly dots that appeared when looking at the sky. I also noticed waves of colourful clouds when my eyes were closed at night, afterimages, and geometric patterns when I closed my eyes firmly for a few minutes. My perception of the world began to change, almost as if everything slowed down. The best way I can describe it is that my vision dropped from 60 frames per second to 30. Also, I’m very sensitive to light: the world is easier for me to look at when it's cloudy, which is why I’ve grown to prefer winter. On the other hand, bright and sunny days are difficult–sunglasses or yellow-tinted lenses are essential. Since childhood, I’ve also experienced migraine attacks, though rarely–perhaps once a year or every two years, and always without aura. However, I do vividly remember my first aura episode, in 2010. I was sitting at the computer, scrolling through social media, when I suddenly saw zigzag lines. The zigzag lines rapidly intensified and within five minutes overtook my entire field of vision. I vomited and then experienced the worst headache I’d ever had. Since then, I’ve experienced several similar episodes. These aura symptoms are completely different from my VSS. Aura usually manifests as either visual aura in the form of zigzag lines, or a sensory aura as a wave of numbness, and they’re episodic – whereas VSS is constant. Interestingly, I’ve noticed that after a migraine with visual aura, there's a period of visual clarity where my VSS symptoms become barely noticeable. Colours appear more vibrant, vision seems to return to 60fps, and my thought processes feel clearer. This improvement lasts only for a few hours after the migraine ends. It's easy for me to distinguish between VSS and aura – the former is constant; the latter only occurs during or around migraine attacks. (M, 30 years old, male)
Clinical perspective
Visual snow
VS is defined as the perception of innumerable, dynamically flickering dots that fill the entire visual field, akin to the static of a badly-tuned analogue television (2). VS is the pathognomonic and defining symptom of VSS (2); however, it can also present as an episodic manifestation of other neurological and ophthalmological conditions (62) including stroke (63–65), migraine (13,66) and retinal disturbances (67). It is important to promptly recognize these secondary forms of VS as they usually require appropriate and timely treatment (68). In particular, key “red–flag” indicators of secondary VS include a sudden new-onset, especially after head trauma, infection or exposure to new medications or hallucinogens, and clinical findings such as monocular or hemifield static, accompanying visual loss, other neurological symptoms or optic–nerve/retinal abnormalities on examination (68).
In VSS, the static is persistent, with black-and-white dots (58%), transparent dots (53%), flashing dots (47%) and coloured dots (44%) being reported as the most common subtypes of static, often occurring in combination (7). In MwA, conversely, VS can represent a rare subtype of visual aura, transient in nature, occurring in approximately 7% of patients and usually accompanying other aura phenomena such as zigzag or jagged lights, flickering lights or scotoma (69).
The pathophysiology of VS has yet to be fully elucidated. However, imaging findings from patients experiencing the VS phenomenon during stroke, without additional visual or perceptual symptoms typical of VSS, indicate that lingual and fusiform gyri, occipital pole and cerebellum may form its neuroanatomical substrate (63). Dysregulation within these visual-processing regions therefore appears to be a major pathophysiological mechanism underlying this symptom.
Palinopsia
Palinopsia is a positive visual phenomenon (70) characterized by visual perseveration (i.e. an increased duration, continuation or repetition) of an object/stimulus that is no longer present in the visual point of fixation (71). Different subtypes of palinoptic symptoms have been described, including scene perseveration, categorical incorporation, afterimages and visual trailing (72).
Several conditions can be responsible for palinopsia, including VSS. Depending on the characteristics of palinoptic images, a valuable clinical classification distinguishes hallucinatory and illusionary palinopsia (73). Hallucinatory palinopsia is characterized by long-lasting, isochromatic, high-resolution afterimages, which are generally not affected by environmental conditions of light or motion. It is typically caused by posterior cortical lesions or seizures and is considered to represent a visual memory dysfunction. By contrast, the afterimages in illusionary palinopsia are unformed, low-resolution, in complementary colours and are affected by ambient light and motion. This type of palinopsia could be caused by VSS, migraine, head trauma, prescription drugs or illicit drugs, and it is considered to represent a dysfunction in visual perception (73). However, it is important to note that afterimages can also be a physiological phenomenon, either when perceived after viewing a bright stimulus and shifting visual focus, representing the bleaching of photochemical pigments in the retina (74) or as the result of cortical central mechanisms generating the persistence of visual images (75).
Palinopsia is common in VSS (7) and is frequently described as an “afterimage” or “trailing” of an object, with these two manifestations occurring in approximately 80% and 60% of VSS patients, respectively (7). A phenotypic description of 31 patients confirmed that palinoptic images in VSS are of indistinct and unformed nature, without the vivid detailed clarity of hallucinatory palinopsia, thus fitting the category of illusory palinopsia (76). Afterimages in VSS are mostly similar to physiologic afterimages perceived by those with normal vision. However, it has been suggested that afterimages in VSS patients are induced more readily, and sometimes even by everyday objects that are not expected to elicit physiologic afterimages in a normal visual system (76).
Palinopsia is present in approximately 10% of migraine patients (77,78), particularly in patients with aura (77) who have a longer duration of afterimages compared to those without (79). However, recent evidence suggests that it could be an underdiagnosed phenomenon in this population, being present in more than 50% of migraine patients (78). Interestingly, the afterimages reported by migraine patients are qualitatively similar to those reported by VSS patients (76). Afterimages last longer during headache attacks (78), but their relationship with the frequency of attacks is unclear because contrasting results emerged from different studies (77,78).
The neural basis and the pathophysiology of palinopsia are still unresolved. It was hypothesized that palinopsia could be due to a pathological overactivity of the cerebral visual system, with a lack of inhibitory control (70) and impaired GABAergic transmission (80). This has led to the hypothesis that localized and transient cortical hyperactivity in the form of cortical spreading depression (CSD) can be one of the pathophysiological mechanisms responsible for the perception of palinoptic images and other visual symptoms in MwA (73). In particular, when CSD reaches the parietal projections of the dorsal visual pathway, it may give rise to the generation of visual perseveration (77). This is further supported by the evidence that lesions in the parieto-occipital area have been classically linked to palinopsia (81,82). Subcortical and/or cortical structures, such as the thalamus (79), play an additional role. The hypometabolism of the inferior parietal lobule has been suggested to represent a biological correlate of palinopsia in VSS (21).
However, because of a lack of dedicated studies, the underlying pathophysiological mechanisms of palinopsia and whether they differ between MwA and VSS remain unknown.
Enhanced entoptic phenomena
Entoptic phenomena are defined as visual experiences arising from the eye's own structures (83,84). They include, but are not limited to, floaters, phosphenes, self-light of the eye and blue field entoptic phenomenon (85). While, to some extent, these manifestations can be occasionally and transiently noticed by nearly everyone (86–88) including patients with migraine (89) or other medical conditions (90), in VSS, these phenomena are more frequently perceived and become a prominent symptom (2). Indeed, in VSS, excessive entoptic phenomena are reported at notably high rates: floaters occur in up to 86% of patients, self-light of the eye in 71%, the blue field entoptic phenomenon in 67% and flashes in 63% (7). By contrast, not all of them enter into the typical clinical description of MwA, with only flashes reported as a common symptom during the aura phase. However, in this scenario, they tend to be episodic and of cortical origin (25).
Interestingly, each subtype of entoptic phenomena has a distinct mechanism. The blue field entoptic phenomenon results from the gaps in the columns of red cells created by leukocytes traversing the retinal microvasculature within the macula and is most apparent when viewing a bright, uniform background, such as a clear sky (91,92). Floaters correspond to vitreous opacities, often collagen clumps or debris, that cast shadows on the retina, creating small, moving specks in the field of vision (93). Flashes or phosphenes reflect the perception of light in the absence of external photons, typically arising from mechanical stimulation of the retina, such as vitreoretinal traction (93). Finally, self-light of the eye refers to coloured shapes or waves seen in darkness, likely generated by spontaneous activity in retinal and early visual pathways (7).
While flashes may represent a transient phenomenon of the aura phase (19) likely of cortical origin (25), the entoptic phenomena in VSS are persistent and supposed to be derived from these normally subtle or momentary signals perceived as amplified and long-lasting (94).
Nyctalopia
Nyctalopia, or impaired vision in dim light conditions, is a visual symptom characterized by difficulty seeing in low light or at night. Frequently described as a symptom in retinal disturbances such as retinitis pigmentosa (95), as well as in several metabolic diseases (96,97), it commonly also enters the clinical picture of VSS, being reported by over 75% of the patients (7,98). By contrast, its prevalence in migraine is unknown since it has not been investigated.
The rod system is implicated in mesopic and scotopic vision in conditions of low light intensity (99). Indeed, nyctalopia is generally encountered in conditions that damage this system, such as genetic ophthalmologic diseases (100) or disorders with low vitamin A (101). However, patients with VSS experiencing nyctalopia have normal electroretinography and ophthalmological tests (47), making the dysfunction of the photoreceptors unlikely in VSS (2).
Although the pathophysiology of nyctalopia in VSS is unknown, it may be a consequence of the other symptoms rather than having a unique and stand-alone pathophysiology. In particular, it can be hypothesized that an increased background noise could functionally mask the weaker visual signals arising from dark environments. Indeed, some VSS patients describe an increased combination of palinopsia and entoptic phenomena in low-light conditions or notice more static. This suggests a dysregulation in cortical processing of incoming sensory stimulation as a potential pathophysiological mechanism responsible for this symptom (4).
Photophobia
Photophobia is defined as an abnormal hypersensitivity or intolerance to light that causes discomfort or pain (102). It is characterized by different clinical manifestations, including photic sensitivity, photic allodynia and photo-oculodynia. Photic sensitivity refers to a general discomfort or aversive reaction to light exposure that does not necessarily worsen existing pain (103). Photic allodynia describes an increased intensity of headache pain provoked specifically by exposure to light, whereas photo-oculodynia is characterized by ocular pain triggered directly by light exposure (103).
Several neurological conditions can cause photophobia, including migraine and VSS (104). Specifically, MwA and VSS are particularly associated with high prevalence rates of photophobia, affecting up to 94% (105) and 81% (7) of patients, respectively. Importantly, the burden of photophobia appears to be greater in VSS than in migraine. Indeed, patients with VSS nearly reach very elevated scores on the Leiden Visual Sensitivity Scale (L-VISS), comparable to those seen in individuals with chronic MwA during attacks (106). While patients with migraine typically experience light sensitivity predominantly during headache episodes, patients with VSS endure a similar level of photophobia continuously (106).
From a neurophysiological perspective, photophobia arises from a dynamic interplay among retinal photoreceptors, subcortical relay stations and cortical networks responsible for processing pain and vision (107). Most of the evidence on the pathophysiology of photophobia derives from studies of patients with migraine. However, some of these neuroanatomical considerations can also be extended to VSS because both conditions share overlapping patterns in some crucial areas (9,12).
Crucially, melanopsin-expressing intrinsically photosensitive retinal ganglion cells (ipRGCs) provide direct light input to trigeminovascular dura-responsive neurons in the posterior thalamus, particularly the lateral posterior/pulvinar area involved in meningeal nociception (108). These neurons project to somatosensory (S1/S2), visual (V1/V2) cortices and the insula, directly linking photic input to headache intensification during migraine attacks (109). This is further supported by the clinical observation that blue and, to some extent, reddish and short-wavelength lights tend to exacerbate photophobia (110,111), whereas green light exposure significantly alleviates discomfort (112). Indeed, this differential response reflects varying ipRGC activation across wavelengths (113,114), with green light potentially mediating its analgesic effects via opioid pathways (115).
In parallel, retinal signals also reach the hypothalamus, projecting to the superior salivatory nucleus and the intermediolateral cell column through retinohypothalamic-parasympathetic and retinohypothalamic-sympathetic pathways (116). These pathways underlie various hypothalamic-mediated autonomic symptoms associated with photophobia, including changes in heart rate, dizziness, nausea, lacrimation, feelings of thirst or hunger, and drowsiness (117).
Additionally, cone/rod-ipRGC pathways relay signals to the midbrain pretectal area, communicating with the Edinger–Westphal nucleus and intermediolateral nuclei to control pupil constriction and dilation (118). Abnormal pupillary reflexes observed in migraine patients correlate with greater disease severity and lower tolerance thresholds for light, indicative of central autonomic dysregulation (119). It has been suggested, on the basis of animal studies, that photo-oculodynia occurs when retinal input activates the olivary pretectal nucleus in the midbrain, stimulating the superior salivatory nucleus and causing choroidal vasodilation (103,120). According to this hypothesis, this vasodilation would subsequently activate trigeminal nerve endings enriched in calcitonin gene-related peptide, integral to migraine pathophysiology and central sensitization processes (121).
Finally, reciprocal connections between posterior thalamic neurons and sensory cortices form feedback loops where cortical hyperexcitability heightens the perception of photic stimuli and may sustain persistent photosensitivity (122–124). Enhanced cortical activation reinforces the central role of cortical over-responsiveness in photophobia, which is crucial in both MwA and VSS.
Pathophysiological perspective
Structural and functional neuroimaging evidence
The pathophysiology of VSS is not yet fully understood but has been increasingly elucidated through neuroimaging studies, highlighting similarities and key differences with MwA.
Given the predominance of visual symptoms, it is not surprising that visual areas play a significant role in the VSS. Evidence from a voxel-based morphometry (VBM) study revealed increased grey matter (GM) volume in primary and secondary visual cortices in VSS, including the motion-sensitive V5. Notably, these changes did not correlate with symptom duration or severity, suggesting that they may represent a constitutional trait rather than a consequence (22). By contrast, studies on volumetric brain changes of V5 in MwA yielded conflicting results, with some studies reporting decreased (23) and others revealing increased GM volume (24). However, the V3A area seems to play major role in MwA, with evidence of CSD initiation from this area (25). Additionally, patients with MwA showed increased cortical thickness (24) and indirect evidence of changes said to represent neuroinflammation in the V3A area (32). These findings were further confirmed by resting-state connectivity analyses, where a reduced coupling between the dorsal V3A and the anterior insula emerged as a signature of MwA (26).
Visual associative areas, such as the lingual gyrus, comprising an area involved in face and shape recognition (125,126), are crucial in the pathophysiology of both VSS and MwA. Using a positron emission tomography (PET) scan, Schankin et al.(9) revealed a hypermetabolism of the lingual gyrus in patients with VSS. This result was subsequently confirmed by a multimodal PET/VBM study that showed a tight anatomical–metabolic relationship in the lingual gyrus, with hypermetabolism occurring concurrently with increased cortical volume (21). Interestingly, an altered anaerobic metabolism stands behind these structural and functional alterations. Indeed, a combined functional magnetic resonance imaging (fMRI) and magnetic resonance spectroscopy (MRS) study found increased lactate concentrations in the lingual gyrus, which were negatively correlated with the blood-oxygenation-level-dependent
Although other cerebral areas showed increased GM volume in VSS (middle-temporal, parahippocampal, anterior-cingulate), none displayed a metabolic correlate, leaving their significance uncertain (21). It is also important to note that most of the results discussed for the visual areas in VSS were also found in the lateral cerebellar areas, with hypermetabolism of the left cerebellar anterior lobe (9) and increased GM volume in the left cerebellar crus I/lobule VI (22). Because they are involved in spatial processing functions, somatosensory integration and visual working memory (127–129), it is plausible to hypothesize that they may contribute to the pathophysiology of VSS alongside the visual areas.
Besides these subtle alterations in circumscribed areas, recent multimodal imaging studies have suggested that VSS is a network disorder in which the structural, metabolic, functional and neurochemical fingerprints extend beyond the visual cortex. Indeed, differently from MwA, where a reduced neurovascular coupling was found in the primary visual cortex parcel (VIS-4) of the large-scale visual network (33), studies with arterial-spin-labelling on VSS revealed widespread increases in bilateral regional cerebral blood flow in several areas involved in processing internal experiences over external attention, suggesting that brain energy is over-allocated to internally driven processing in VSS (34).
Studies using fMRI revealed reduced functional connectivity between different nodes of the visual, attentional and salience networks in VSS. These include reduced connectivity between the right anterior insula and anterior/mid-cingulate cortex (ACC/MCC), between V5 and the right precuneus, and between the pulvinar and dorsal caudate (29). Another study confirmed these alterations within the salience network, showing an attenuated activity in the insula in response to a visual stimulation mimicking VS (27). By contrast to VSS, MwA is characterized by less widespread functional connectivity changes than VSS, mainly centred around the visual network (28). Additionally, one study reported evidence of increased functional connectivity between the insula and cerebellar vermis VI, a pain and autonomic-processing hub (30). However, Hougaard et al.(31) failed to show differences in functional connectivity between patients and controls, underscoring heterogeneity across studies.
Neurotransmitter imbalances, particularly glutamate and serotonin, are likely to stand behind the alterations in brain connectivity among the areas of the visual, salience and limbic systems in VSS. One study that used a receptor-enriched analysis of functional connectivity by targets (REACT) analysis revealed a reduced functional connectivity in glutamatergic networks localized in the ACC and serotoninergic networks among occipito-temporo-parietal association areas (35). While the glutamatergic alterations were specific to patients with VSS, serotonergic abnormalities were found in both patients with VSS and MwA, suggesting a molecular overlap between them (35).
Taken together, these results suggest that, although some anatomical and neurochemical substrates overlap, such as serotonergic alterations and involvement of the lingual gyrus and visual cortex, the underlying patterns of functional connectivity fundamentally differ. VSS involves persistent visual, attentional and salience network dysfunction (27), with glutamatergic dysregulation on the background (35), while MwA distinctively shows fMRI signatures of transient cortical spreading depolarization, with an initial focal BOLD rise in V3A that slowly propagates retinotopically across the occipital cortex (25). This suggests that, in VSS, attentional and salience-monitoring circuits are unable to gate or suppress the surrounding continuous visual noise, which allows unfiltered visual activity to remain in conscious awareness as continuous “static” (29). Conversely, MwA is characterized by transient cortical disruptions prominently involving the CSD-associated area V3A, with interictal functional connectivity changes mainly centred around the lingual gyrus (28) and reduced neuro-vascular coupling within the visual network (33) that can predispose to CSD in case of increased energy demand.
Neurophysiological evidence (electroencephalography, visual evoked potentials and transcranial magnetic stimulation)
Neurophysiological studies mostly focused on the visual system and how it processes visual stimuli, providing insights into cortical excitability and excitation-inhibition mechanisms underlying the pathophysiology of VSS and MwA.
Electroencephalography (EEG) and magnetoencephalography (MEG)
Studies performing EEG and MEG on VSS patients have consistently observed altered alpha and gamma oscillations in visual cortices, suggesting excitation–inhibition imbalance in these regions. Comparing resting state alpha rhythm and power spectral density over the individual alpha peak, Klein et al.(36) found decreased alpha power spectral density over parietal and temporal regions corresponding to areas of the secondary visual cortex in patients with VSS. Given the recognised role of alpha oscillations in top-down sensory gating, this focal reduction suggests an electrophysiological correlate of the weakened inhibitory control that VSS patients describe as an inability to “filter out” background visual noise (130,131). This hypothesis was further confirmed by one MEG study using high-contrast grating patterns, which showed that VSS patients exhibit an up-regulation of gamma-band activity (40–70 Hz) in primary visual areas and a breakdown of alpha-gamma phase-amplitude coupling (37). Indeed, because gamma frequency is a key feed-forward carrier of visual information (132,133), an impaired “noise-cancellation” mechanism was suggested. A subsequent MEG study did not replicate resting gamma differences but found that patients showed a higher repetition-related increase in gamma power, indicating an exaggerated activity-dependent plasticity in VSS (38).
By contrast to VSS, MwA presents a markedly different electrophysiological profile when examined under the same paradigms. Interictal quantitative EEG revealed prominent posterior alpha asymmetry, along with a widespread increase in alpha, theta and delta power (39,40). Additionally, a unilateral reduction of alpha–theta activity contralateral to the symptomatic hemifield was described in patients who experience pure visual aura (41). MEG studies extended these observations by showing increased theta-band connectivity within occipital networks and widespread slow-wave (0.1–1 Hz) coherence in frontal regions (42), together with reduced stimulus-driven synchronization and heightened causality and transfer entropy in beta bands, which are changes that differentiate patients without aura from patients with MwA (43,44). Interestingly, EEG microstate analysis also supported altered auditory and visual processing in VSS, with evidence of shorter microstates and abnormal transition probabilities (45). Although with different evidence from VSS, altered microstate dynamics were also found in MwA (46).
Visual-evoked potentials (VEP)
VEPs are a non-invasive and valuable tool for studying the visual pathway. Their application helped to study cortical excitability and understand how the migraine brain responds to visual repetitive stimulation, giving insights into the habituation mechanisms. This has led to the evidence of the “habituation deficit” during the interictal period, which reverts during the ictal phase (48). The lack of habituation has been shown with almost all the sensory modalities, including VEPs (49). It is a common electrophysiological feature of migraine patients, and no differences were found in VEP habituation between migraine with and without aura (50). Different hypotheses are proposed to explain these abnormalities, including thalamo-cortical “dysrhythmia”(134) and the contribution of genetically determined dysfunctions of the synaptic learning mechanisms (49). A deficit in VEP habituation was also observed in several case reports of VSS patients (51,52). Additionally, a small prospective and observational study confirmed the lack of habituation in VSS patients, regardless of migraine comorbidity (53). However, a different study did not confirm this result (54). Although canonical VEP responses showed no abnormalities in the VSS patients (47), one study showed reduced N75–P100 amplitudes and increased N145 latency, which was not seen in participants with migraine, suggesting a dysfunction of extrastriate cortical areas (54).
Transcranial magnetic stimulation (TMS)
TMS has been used in both disorders to assess their cortical excitability, but there is limited evidence regarding the VSS. In one study, Yildiz et al.(53) reported decreased phosphene thresholds only in the subgroup of VSS patients with comorbid migraine, indicating an occipital hyper-excitability in VSS amplified when migraine co-exists. By contrast, MwA has been examined repeatedly and has shown a broader excitability profile: most series report reduced phosphene thresholds and shortened cortical silent period (55–57), while other studies found no changes (58,59). Additionally, extrastriate regions such as V5 have been found hyper-responsive (60) and TMS-EEG mapping revealed an attenuated N100 component over frontal and occipital cortex (61), consistent with deficient GABA mediated inhibition (135).
Taken together, these results suggest that, although both diseases present with cortical hyper-excitability, VSS is characterized by high-frequency alterations, excessive feed-forward drive and impaired top-down suppression. By contrast, MwA predominantly shows slow rhythm alterations and hemispheric asymmetries. This evidence suggests that, although there are some overlapping mechanisms, the neurophysiological mechanisms underlying the pathophysiology of VSS cannot be entirely explained by those of MwA, likely reflecting distinct entities. However, further research with standardized protocols and a larger sample size is needed to elucidate the neurophysiological alterations in VSS and the shared mechanisms with MwA.
Conclusions
VSS and MwA have long been linked, largely due to the common disturbance in visual perception and frequent co-occurrence; however, the collective body of evidence gathered across symptoms, imaging and electrophysiology shows that they are two distinct and separate entities.
Taken together, the data favour a model in which VSS represents a chronic disorder of salience assignment and sensory suppression: retinal and thalamo-cortical inputs that are normally filtered as physiological “noise” are mis-categorised as salient, generating a persistent visual backdrop and exaggerating entoptic, palinoptic and photophobic experiences. Conversely, MwA can be conceptualised as an intermittently unstable network in which cortical excitability periodically exceeds a critical threshold, producing the slow depolarizing wave that underpins clinical symptoms. Shared serotonergic alterations, overlapping involvement of visual cortical areas and the frequent clinical co-occurrence of migraine and VSS do point to some convergence, yet these parallels can be more plausibly explained by a general tendency towards increased cortical excitability rather than a single unified disorder.
In conclusion, recognizing the overlapping but distinct features of VSS and MwA is crucial for advancing our understanding and improving management of these complex conditions. An illustrative example is the newly identified selective glutamatergic role in VSS, which already potentially hints at tailored therapeutic avenues–much needed given the current scarcity of disease-specific treatments. Therefore, future research should aim to further elucidate specific network abnormalities, guiding the development of targeted therapeutic approaches. Ultimately, integrating detailed clinical characterization with advanced neuroimaging and neurophysiological methodologies will enhance our ability to tailor individualized interventions and improve quality of life for patients suffering from these distinct yet interconnected disorders.
Article highlights
VSS presents as a persistent visual disturbance that shares pathophysiological and clinical features with MwA.
Despite the overlapping traits and similarities with MwA, such as serotonergic involvement and cortical hyperexcitability, VSS is a distinct neurological disorder.
Neuroimaging and electrophysiological studies in VSS consistently show abnormalities in visual, salience and attentional networks, with evidence of glutamatergic dysregulation and impaired top-down control.
The review emphasizes the need for future research into VSS-specific network dysfunctions to guide targeted therapeutic development.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
