Abstract
Background
Allodynia in adults with migraine is related to disease duration. In pediatric patients with migraine, the same proportion reported allodynia in the first six months of migraine presentation as in prolonged disease. This study examined a possible association between migraine pediatric allodynia and maternal allodynia.
Methods
We interviewed children with migraine first, and then their mothers, regarding allodynia and headache symptoms. We reviewed hospital charts on pediatric medical background and headache symptoms. Mothers and children older than 11 years filled the Strengths and Difficulties Questionnaire.
Results
Ninety-eight children with migraine, mean age 13.49 ± 3.1 years, and their mothers, mean age 43.5 ± 6.2 years were recruited to the study. Pediatric allodynia was associated with maternal allodynia; the latter was reported in 82.8% of children with allodynia versus 35.3% of children without allodynia (p < 0.001). Maternal migraine was reported in 44 (68.7%) of children with allodynia versus 16.3% without allodynia, p < 0.001. No difference was found in Strengths and Difficulties Questionnaire scores, between children with and without allodynia.
Conclusions
Pediatric allodynia is associated with maternal migraine. Genetic and environmental factors such as maternal behavior may contribute to reduced pain threshold.
Introduction
Cutaneous allodynia is defined as pain provoked by stimulation of the skin that would ordinarily not produce pain (1). Studies regarding pediatric allodynia are sparse. The prevalence of cephalic cutaneous allodynia in children with migraines has been reported in the range of 31–37% (2–4). In adults with migraines, the proportion of allodynia was found to be 60–80%, and related to chronic migraine and disease duration; the prevalence reportedly decreases after age 65 years (5–8).
The underlying mechanism of cutaneous allodynia is sensitization of the nociceptive neurons in the trigeminal nucleus caudalis, which receives convergent afferent input from the dura mater and periorbital skin (9). Allodynia is known to reflect central sensitization (10), and to result from a common mechanism that appears more frequently in migraine with aura.
Central sensitization has been found to be related to other pain syndromes, including: fibromyalgia, osteoarthritis, rheumatoid arthritis, Ehlers-Danlos syndrome, upper extremity tendinopathies, rheumatoid arthritis, osteoarthritis, musculoskeletal disorders, complex regional pain syndrome, post-surgical pain, non-cardiac chest pain, irritable bowel syndrome and esophageal hypersensitivity (11,12).
Retrospective studies of data from pediatric clinics reported the presentation of cutaneous allodynia already in the first six months of migraine onset (2), and in children under the age of 7 years (4). In contrast to reports in adults, those studies found no associations of allodynia with migraine frequency or prolonged disease duration.
Tommaso et al. found that allodynia was prevalent among pediatric patients who had had migraines for up to one year. This is another indication that in pediatric patients, allodynia is not necessarily related to prolonged pediatric migraine (13). Notably statistically significant genetic components of pain sensitivity have been shown to differ between dizygotic and monozygotic adult twins (14).
We hypothesized that children and adolescents with migraine and cutaneous allodynia are more likely to have mothers with allodynia, than are children without allodynia
We further hypothesized that this may be due to a genetic predisposition, environmental or psychologic cause related to the mothers. The first aim of our study was to evaluate a possible association of migraine allodynia in children and adolescents, whose mothers had allodynia. The second aim was to identify clinical parameters that may be associated with pediatric and adolescent allodynia.
Methods
Participants and Setting
The participants were children and adolescents with migraine, aged 7–18 years, and their mothers. The setting was a pediatric and adolescent headache clinic of a tertiary university affiliated pediatric medical center, during March–September 2019. Comorbidities were based on review of the medical records from the hospital's ambulatory clinic and hospitalization departments, and review of data from the clinics of primary physicians. Younger patients were not included due to possible difficulty in understanding the allodynia questionnaire. Patients were excluded from the analysis as follows: were not accompanied by their mothers, not understand the allodynia questionnaire and had developmental delay. The diagnosis of children and their mothers was evaluated for this study according to the International Classification of Headache Disorders (ICHD)-3 (15).
Headache clinic protocol
Children and adolescents were interviewed at the beginning of their visits to the headache clinic. Study inclusion criteria were a diagnosis of children with migraine according to ICHD-3 classification (15) and aged 7 years or older. Children not accompanied by their mothers, not able to understand the questionnaire or with developmental delay were not included in the study. All the children and adolescents were first interviewed by the senior physician (TEM) who heads the pediatric headache clinic. A routine questionnaire used in the headache clinic was based on the headache society criteria ICHD-3 (15) to diagnose headache. Children were also interviewed routinely as part of our headache clinic protocol, which includes clinical questionnaires to diagnose allodynia. Mothers and also patients older than 11 years filled the Strengths and Difficulties Questionnaire as part of their routine first visit at the pediatric headache clinic. This questionnaire is used to screen for behavioral, psychological and social problems in children and adolescents aged 4–18 years (16).
Headache diagnosis
As part of the routine headache clinic visit, pediatric and adolescent patients were interviewed regarding their specific headache characteristics and symptoms including the frequency, duration and the nature of the headache attacks, the time from the start of the headaches to the visit at the clinic, phonophobia, photophobia, etc. For young children, headache frequency and symptoms were determined by their complaints and their mothers' impressions of their behavior (according to the ICHD-3 criteria [15]).
Allodynia diagnosis
To diagnose allodynia in the pediatric patients in our headache clinic, we routinely administered allodynia questionnaires that were found validated in previous studies (2–4,7,17).
Similar questions were used to diagnose ictal and interictal allodynia in children and adolescents, as was done in previous studies (2–4). Ictal allodynia was diagnosed when two positive answers were recorded for the relevant questions during a migraine attack (2–4). Pediatric interictal allodynia was diagnosed when two positive answers were recorded for the questions relating to interictal attacks (18). Pediatric allodynia was also diagnosed when only one and the same positive answer was given for both ictal and interictal allodynia.
The following questions were asked for the diagnosis of ictal cephalic allodynia. The possible responses were yes/no/not applicable.
During a headache attack, have you ever had an unpleasant sensation when combing your hair? Do you try to avoid combing your hair when you have an attack? During a headache attack, have you ever had an unpleasant sensation when touching your head? Do you try to avoid touching your head when you have an attack? During a headache attack, does it bother you to wear an object on your head or neck, such as jewelry (earrings, necklace), glasses or a hat? During a headache attack, does it bother you to have a ponytail? During a headache attack, do you avoid putting your head or face on a pillow?
For interictal symptoms the same questions were asked but with reference to the period of time between migraine attacks.
Maternal allodynia diagnosis
After a child was diagnosed with migraine, with or without aura, his/her mother was offered to participate in the study and to sign informed consent. The mothers were interviewed by TEM according to the ICHD-3 criteria (15), to diagnose headache type. Maternal questionnaires were based on previously published allodynia questionnaires (1,7,8,17,18). As with the pediatric patients, for the mothers with headaches (any type, and not only migraine), ictal allodynia was diagnosed when mothers cited cited discomfort and /or pain during at least two activities.
Interictal allodynia was diagnosed when two or more activities between headache attacks were cited as causing pain or discomfort; or alternatively allodynia was also diagnosed when the same item was cited both interictally and during an attack (13).
To diagnose ictal allodynia, mothers who reported headaches were asked if during headache attacks they felt pain or discomfort while doing any of the following, or if they avoided these activities:
Combing hair. Pulling hair back (ponytail) Wearing eyeglasses Wearing contact lenses Wearing earrings Wearing a necklace Wearing tight clothing Taking a shower (when shower water hits face) Resting face or head on a pillow Exposure to heat (e.g., cooking, washing face with hot water) Exposure to cold (e.g., using an ice pack, washing face with cold water)
To diagnose interictal allodynia, mothers who reported headaches, were asked about pain or discomfort, or avoidance of the same activities listed above, in regard to their experience between headache attacks.
Mothers without headache were asked if, during daily life, they felt pain or discomfort while doing the following activities, or if they avoided such activities due to expected pain or discomfort. Allodynia was diagnosed as an affirmative response regarding two or more activities:
Combing hair. Pulling hair back (ponytail) Wearing eyeglasses Wearing contact lenses Wearing earrings Wearing a necklace Wearing tight clothing Taking a shower (when shower water hits face) Resting face or head on a pillow Exposure to heat (e.g., cooking, washing face with hot water) Exposure to cold (e.g., using an ice pack, washing face with cold water)
Ethics
Mothers gave their informed consent for their participation and their children's participation in the study. Demographic and clinical parameters of the pediatric participants were collected from the patients’ files.
The study was approved by the Research Ethics Committee of Rabin Medical Center (approval no. RMC-0294-18RMC).
Statistical analysis
With the proposed sample size of 35 children without allodynia and 65 children with allodynia, respectively, the study would have power of 93.0% to yield a statistically significant result. Data were analyzed using BMPD software (19).
Continuous variables were calculated as means and standard deviations, and compared between groups using analysis of variance. Parameters that did not have a Gaussian distribution were compared between groups using the nonparametric Mann-Whitney U-test. Discrete variables were calculated as numbers and percentages, and compared between groups using Pearson’s chi-square. A p value of 0.05 was considered significant. Odds ratios (OR) and 95% confidence intervals (CI) were determined. The effect size for continuous variables was calculated with Cohen’s d. For the stepwise logistic regression, we included the variables found to be significant at p ≤ 0.1 on univariate analysis. We also included age.
Results
During the study period, 108 pediatric patients with migraine were admitted to our headache clinic. Ten patients were excluded: 2 had developmental delay, 5 were not accompanied by their mothers and 3 did not understand the allodynia questionnaire, (Figure 1). Ninety-eight children with migraine and their mothers were recruited to the study. The mean age of the pediatric participants was 13.5 ± 3.1 years (range 7.0–18.2, median 14.3). Fifty-six (57.1%) were females. Forty-nine (50.0%) of the children had migraine with aura. Forty-four (44.9%) children were diagnosed with a psychiatric comorbidity and 58 children (59.2%) with an organic comorbidity, during the study period or in the past.

A flow chart of the pediatric patients with migraine who were included in the study.
For the children and adolescents, the mean duration of migraine before participation in the study was 49.8 ± 39.9 months (range 3–166, median 36). The mean migraine frequency per month was 11.4 ± 11.2 (range 0.3–30, median 8). Migraine duration in hours was 16.6 ± 29.9 (range 2–164, median 5). The mean age of onset of children's migraines was 9.3 ± 3.7 years (range 2.9–16.6, median 10).
Sixty-four (65.3%) children with migraine reported allodynia (64 during the migraine attack and 6 between migraine episodes in addition to during an episode. None of the children reported interictal allodynia alone. The mean number of allodynic symptoms of child during migraine was 2.3 ± 1.67 (median 2).
Compared to the pediatric participants without allodynia, those with allodynia were characterized by higher proportions of females (65.6% vs. 41.2%, p = 0.03 CI 95% 1.07–7.03 OR 2.73); awakening headache 58.7% vs. 23.5%, p = 0.001 95%CI 1.67–13.56 OR 4.63), organic comorbidities (68.7% vs. 41.2%, p = 0.01, 95%CI 0.99–2.17 OR 1.63) and migraine with aura (57.8% vs. 35.3%, p = 0.055 95% CI 0.92–6.01 OR 2.37). Among the children with allodynia compared to those without, higher proportions of their mothers had migraine (68.7% vs. 16.3%, p < 0.001), maternal allodynia (82.8% vs. 35.3 (Table 1), p < 0.001 95% CI 3.08–25.7 OR 8.83), and maternal headache of any type 76.6% vs. 47.1%, p = 0.012 95% CI 1.23–8.7 OR 3.27). The mean number of symptoms of ictal allodynia was higher among mothers of children with allodynia than without (4.7 ± 2.4 vs 3.1 ± 3.0, p = 0.002, Cohen's d 0.4) (Table 1).
Comparison of demographic and clinical parameters of pediatric patients with migraine, between those with and without allodynia, and between their mothers.

The data are presented as mean ± standard deviation, or as number (percentage).
*Mothers without headache were included regarding allodynia symptoms not related to headache.
Statistically significant differences were not found between the pediatric participants with and without allodynia, in the following migraine characteristics: unilateral pain, nausea, vomiting, photophobia, phonophobia and osmophobia. Statistically significant differences were not found between children with and without allodynia in scores on the following maternal and pediatric scales of the Strengths and Difficulties Questionnaire: emotional problems, behavioural problems, hyperactivity, peer problems, prosocial problems and total difficulties.
Tables 2 and 3 detail pediatric and psychiatric comorbidities in patients with and without allodynia.
Organic comorbidities in pediatric patients with and without allodynia.
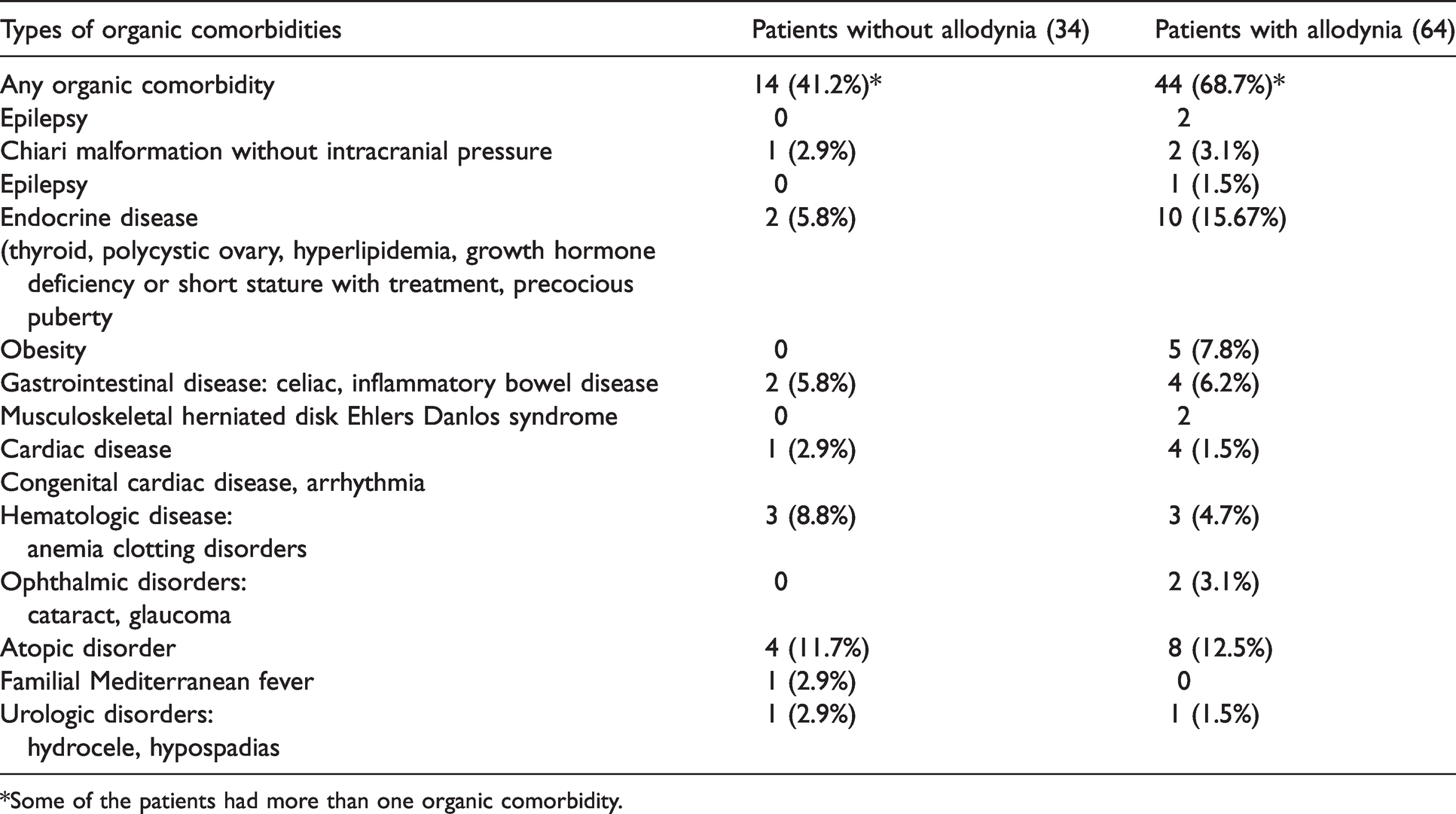
*Some of the patients had more than one organic comorbidity.
Psychiatric comorbidities in pediatric patients with and without allodynia.

*some of the patients had more than one psychiatric diagnosis.
Maternal allodynia
Ninety-eight mothers, mean age 43.5 ± 6.2 years (range 30-57, median 43) participated in the study. Their mean age at onset of maternal migraine was 22.9 ± 11.0 years (range 5-55, median 20). Sixty-six mothers (67.3%) reported headache; the diagnoses were as follows: 60 (90.9%) migraine, 35 migraine without aura, 25 migraine with aura, 5 (7.6%) tension type headache and 1 (1.5%) headache not otherwise classified. Sixty-five (66.3%) mothers were diagnosed with allodynia according to study criteria; of them, 53 (81.3%) were diagnosed with migraine and four with tension type headache.
Sixty-five of the 98 (66.3%) mothers reports allodynia; of them, 57 had allodynia only during headache episodes (ictal allodynia). Nine of these mothers had interictal allodynia in additional to ictal allodynia. Organic comorbidities were reported for 21 (32.3%) mothers with allodynia and 4 (12.1%) mothers without allodynia (p < 0.001) (p < 0.001) (Table 4). Eight of the 65 (12.3%) mothers with allodynia me the criteria with allodynia without having headache. Their mean number of increased pain sensitivity and perception (allodynia) during daily activities were 5.75 ± 21.
Comparison of pediatric and maternal parameters between mothers with and without allodynia.

The data are presented as mean ± standard deviation, or as number (percentage).
*Calculated from 60 mothers with migraine.
Five of these mothers had conditions that may be related to pain hypersensitivity due to central sensitization (11): 1 irritable bowel syndrome, 2 non-cardiac pain, 1 tendinopathy and 1 low back pain and neck pain. Six of the 8 mothers with allodynia without headache had children who had migraine with allodynia. The other 13 mothers with allodynia and organic comorbidities had the following diseases: 1 melanoma, 1 remission after breast cancer, 4 asthma, 2 cerebrovascular accident, 1 fibromyalgia, 2 clotting disorder and 2 hypothyroidism.
The four mothers without allodynia and organic comorbidities had the following organic diseases: 1 hypertension, 1 clotting disorder and 2 hypothyroidism. Fifty-three (81.5%) of 65 mothers with allodynia had migraine vs. 7 (21.2%) of the mothers without allodynia, p < 0.001 (Table 4). Fifty-three (81.5%) of the mothers with allodynia had a child with allodynia vs. 11 (33.3%) of the mothers without allodynia, p < 0.001 95% CI 3.08-25.77 OR 8.83.
Stepwise logistic regression analysis
Table 5 shows the results of the stepwise logistic regression analysis: five variables were found to be significantly associated with allodynia in children and adolescents with migraine: maternal allodynia, organic comorbidity, awakening pain, migraine with aura and female gender.
Stepwise logistic regression: factors associated with allodynia in pediatric and adolescent patients with migraine.

Area under the curve = 0.873.
No statistically significant difference was found between children with and without allodynia in the following maternal and pediatric scores of the Strengths and Difficulties Questionnaire: total score, emotional score, conduct score, hyperactivity score, peer score and prosocial score.
Excluded patients and missing data
Five patients who were not accompanied by their mothers were excluded from the analysis:, 4 of them were accompanied by their fathers and 1 by a grandmother . In addition, 3 who did not understand the allodynia questionnaire and 2 who had developmental delay were excluded.
Data of some parameters were missing for some patients. No significant differences were observed in missing data among pediatric patients, between those with and without allodynia: awakening pain of child, 1 vs. 0, p = 1.0; dizziness, 3 vs. 0, p = 0.548; child headache frequency per month, 3 vs 0, p = 0.548; duration of migraine attack, 1 vs. 0, p = 1.0
Discussion
The main finding of this study is the demonstration of significant associations of pediatric allodynia with maternal allodynia, a higher mean number of ictal allodynia symptoms among mothers, maternal headache of any type and maternal migraine,
Pediatric and adolescent allodynia was also associated with female gender, awakening headaches at night and organic comorbidity. The diagnosis of migraine with aura was more prevalent among pediatric patients with than without allodynia; however, this difference had borderline statistical significance.
Our finding of higher prevalence of allodynia among females concurs with the results of a population study (20). In an experimental model of chemically induced (capsaicin) trigeminal sensitization in women of childbearing age, the area of brush-evoked allodynia was larger in the menstrual phase of the cycle than in the luteal phase (21,22). Our finding of an association of pediatric allodynia with awakening headache corroborates our earlier findings (3). Lovati et al. suggested allodynia is strongly related to sleep quality, in a bi-directional way: sleep disturbances may favour central sensitization, and allodynia may in turn impair sleep (23).
Since our patients are treated in tertiary centers, a high proportion had organic comorbidities. Some of the organic diseases have been reported as related to central sensitization and pain syndrome. For example, four patients had inflammatory bowel disease, one of whom did not have allodynia. However, the small numbers with each comorbidity preclude reaching a conclusion regarding the influence of organic comorbidity in our patients (11,12).
Our rate of 66% allodynia compares to 31-37% previously reported in studies conducted in the headache clinic of the same tertiary pediatric hospital (2,3).
The higher rate may be due to the different study methodology, in which we interviewed only children and their mothers, in contrast to studies in which mothers and/or fathers were interviewed regarding children's allodynia. Moreover, in the current study, we identified headache type with ICHD-3 criteria (15). In previous studies conducted in the same headache center, migraine was diagnosed according to ICHD-2 criteria (3) and ICHD-3 beta version (2).
We found that pediatric allodynia was associated with maternal allodynia and maternal allodynia was associated with pediatric allodynia. Six children who were diagnosed with allodynia had mothers who had allodynia but not headache Five of these mothers with allodynia but without headache had organic diseases that have been reported to be associated with central sensitization (11,12) and pain syndrome. This may explain the appearance of allodynia in some mothers who did not have headache.
The relation between pediatric allodynia and maternal allodynia may explain the presentation of allodynia already in children under the age of 7 years with migraines (4).
According to our study hypotheses, the association of pediatric allodynia and maternal allodynia may be related to genetic, environmental or psychological factors. The observation that pain thresholds are often also reduced in individuals with allodynia during pain-free periods, and in episodic as well as chronic migraine, suggests possible mechanisms other than central sensitization as the cause of sensory hypersensitivity. These may act independently or synergistically with temporal summation in migraine (24).
Sand et al. demonstrated a reduced threshold for thermal pain sensitivity in the minutes preceding a migraine attack. This apparently cannot be attributed to pain-induced central sensitization (24), thus raising the possibility of other mechanisms as the cause of sensory hypersensitivity. Such mechanisms may act independently or synergistically with temporal summation in migraine.
Our finding of cutaneous allodynia in four mothers with tension headache concurs with other reports of cutaneous allodynia in primary forms of headache other than migraine (25). These reported findings and our study findings of the association between pediatric allodynia and maternal allodynia may be related to genetic, environmental or psychological factors.
Genetic/epigenetic factors
Eight mothers of our study reported allodynia without having headache. Six of their children had allodynia. The backgrounds of five mothers without headache included a pain syndromes associated with central sensitization.
This suggests an internal cause as a genetic and or epigenetic factor in maternal and child allodynia
Norbury et al. (14) performed a classical twin study to evaluate the relative contributions of genetic and environmental factors in responses to painful stimuli in women. Ninety-eight pairs of twins, 51 monozygotic and 47 dizygotic, were recruited. Statistically significant associations of genetic components (with frequencies in the range of 22-55%) were observed for the responses to the majority of painful stimuli. These included: heat pain threshold, the pain rating during induction of a thermal burn, the secondary areas of punctate hyperalgesia and brush evoked allodynia following the induction of a 45°C thermal burn, and pain ratings during the iontophoresis of adenosine triphosphate.
A number of genes have been suggested to have critical roles in determining pain sensitivity or susceptibility to chronic pain. One of these genes, catechol-o-methyltransferase, is known to be involved in the inactivation of dopamine, epinephrine and norepinephrine neurotransmission. Other genetic factors are polymorphisms within GCH1, the gene encoding GTP cyclohydrolase and polymorphisms of the serotonin transporter gene (SLC6A4) (26).
Valder et al. (27) identified genetic variants of potential target genes that may contribute to the development of neuropathic tactile allodynia in rats. In humans, carriers of TRPM8 rs10166942T nucleotide polymorphism alleles reported more allodynic symptoms and allodynia chronification than did non-T allele carriers, but this was not associated to allodynia severity (28). Kayama et al. revealed that the activation of TRPM8 reversed the heat allodynia in a mouse meningeal inflammation model, thus suggesting that the activation of TRPM8 may be protective against allodynia in migraine (29).
An association of maternal with pediatric allodynia may be explained by epigenetic changes in allodynia. Epigenetics involves several mechanisms such as DNA methylation, histone acetylation and methylation, and miRNA expression. All of these are involved in changes at various sites of the central nervous system under chronic pain states; and influence chronic pain-related behaviors. Epigenetic changes of the nervous system are involved in persistent pain and pain perception. Modifications in supraspinal sites related to pain perception, brain circuits and neural activity regions have been demonstrated under chronic pain conditions (30).
Environment
Chambers et al. (31) investigated the impact of maternal behavior on pain experiences of 121 children aged 8-12 years, using self-reports of intensity and affect, coding of facial activity, tolerance, and heart rate responsiveness. Mothers were randomly assigned and trained to interact with their children in one of three ways while the children were exposed to lab-induced cold pressure pain: 1) a pain-promoting interaction, 2) a pain-reducing interaction, and 3) a no-training control group. Girls whose mothers interacted with them in the pain-promoting manner reported more pain than daughters of mothers in the control group, who in turn reported more pain than girls whose mothers interacted with them in the pain-reducing manner. This effect of the intervention was not found in boys (31).
In our cohort, the association between allodynia in mothers and their children may be related to a low level of pain perception among the mothers, which influences their children’s pain perception. Contrary to the finding of Chambers (31), our study found no associations of maternal allodynia with children's gender and allodynia However, pediatric allodynia was associated with a higher number of ictal maternal allodynia symptoms, and this suggests that more allodynic symptoms in mothers may have impacted allodynia in their children.
Psychological factors
We report no statistically significant differences in responses to the Strengths and Difficulties Questionnaire, nor in the rate of psychiatric comorbidity, between our patients with and without allodynia. This corroborates the findings of a study by Lovati et al. (32) of 213 children with headache. The psychological profile was assessed in each patient by the SCL90R (Symptom Checklist-90-Revised), a 90-item self-report of broad psychological symptoms questionnaire. No statistically significant difference was found in the scores between those with and without allodynia, neither in analysis of the whole cohort nor in analyses that compared subgroups according to headache type and episodic/chronic pattern.
Study limitations
The study was performed in a tertiary care pediatric medical center that serves as a referral center for patients with more severe condition. This may have created a selection bias due to the inclusion of patients with more severe headache. Another limitation is that the study was conducted in a single center, thus limiting the generalizability of the findings. Allodynia symptoms in children and their mothers were diagnosed by a questionnaire that has been used commonly for research and clinical purposes and not by objective measurements. Notably, allodynia questionnaires have been shown to be reliable for assessing both adults and children (3,4,17,18).
In an attempt to mitigate bias, patients were interviewed before their mothers regarding allodynia symptoms
It is possible that the eight mothers without headache and with allodynia gave unreliable responses to our questionnaire. However, the high number of daily activities that the mothers cited in regard to increased pain perception (allodynia) may preclude this. Additionally five of the mothers had medical conditions that may be related to central sensitization and a pain syndrome; and this may explain their responses regarding allodynia.
Conclusions
Pediatric allodynia is associated with maternal allodynia and with maternal migraine. Genetic epigenetic and environmental factors may contribute to the reduced pain threshold. The lack of association of pediatric allodynia with scores on the Strengths and Difficulties Questionnaire and with psychiatric comorbidity does not support the involvement of psychological factors.
Clinical Implications
Pediatric and adolescent allodynia was associated with maternal allodynia in a tertiary pediatric headache center. The association of pediatric and maternal allodynia suggests the involvement of genetic, epigenetic and environmental factors. The lack of difference in scores on the Strengths and Difficulties Questionnaire between children with and without allodynia suggests that psychologic factors may not have a central role in the pathogenesis of pediatric allodynia.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
