Abstract
Abstract
Background
Clinical presentation is the key to the diagnosis of patients with migraine and tension-type headache, but features may overlap when both become chronic. Psychophysical parameters may distinguish both conditions. We aimed to compare psychophysical aspects of patients with chronic migraine, chronic tension-type headache and headache-free controls, and to determine whether these can predict headache frequency.
Methods
An examiner blinded to the diagnosis assessed 100 participants (chronic migraine (n = 38), chronic tension-type headache (n = 31) and controls (n = 31)). Assessed variables included painful area, pressure pain thresholds, temporal summation, cervical range of motion, neck posture, headache and neck impact, quality of life, and kinesiophobia. Comparison between groups was performed with one-way ANOVA and multiple linear regression was used to assess the headache frequency predictors.
Results
We found differences of both headache groups compared to controls (p < 0.01), but not between headache groups. Neck disability was a significant predictor of headache frequency for chronic tension-type headache (adjusted R2 = 0.14; β = 0.43; p = 0.03) and chronic migraine (adjusted R2 = 0.18; β = 0.51; p < 0.01).
Conclusions
Chronic tension-type headache and chronic migraine showed similar psychophysical results, but were significantly worse when compared to controls. The psychophysical examination did not discriminate between headache types. The variable best explaining headache frequency for both headache types was neck disability.
Introduction
Migraine and tension-type headache are exclusively diagnosed according to the quality and duration of the symptoms and associated features such as nausea and phonophobia/photophobia (1). However, when headaches become chronic, these features may overlap (2). According to Lipton et al. migraine is frequently mistaken for tension-type headache and this is considered a common diagnostic error in primary care settings (3). Although migraine and tension-type headache have been extensively studied, the pathophysiology underlying the chronification process is not fully understood and both headache types remain a major problem from socio-economic and quality of life perspectives (4,5).
Among all symptoms, neck disability is one of the most common in patients with tension-type headache and migraine (6,7). Neck pain is present in 76.2% and 88.4% of patients with pure migraine and tension-type headache, respectively (8). However, the cervical musculoskeletal findings of both episodic forms are not the same. Episodic migraine is more associated with cervical active range of motion reduction (9), while episodic tension-type headache is more associated with postural alterations and increased extratrigeminal muscle tenderness (greater number of cervical trigger points) (10). Direct comparison of both conditions showed a significant reduction of pressure pain thresholds (PPTs) in episodic tension-type headache compared to episodic migraine (11).
Decreased pressure pain thresholds (PPTs), increased pain area, referred pain and temporal summation (TS) have been associated with several chronic pain conditions and seem to contribute to the development and/or maintenance of chronic disorders (12–16).
In this regard, PPTs and TS have been reported in patients with chronic primary headaches when compared to episodic primary headaches or to asymptomatic controls (11,17–21). Furthermore, Uthaikhup et al. observed larger painful areas in chronic migraine patients compared to episodic migraine (22). Hence, all these psychophysical variables have been widely investigated in patients with chronic migraine (CM) and chronic tension type headache (CTTH), however there is a lack of evidence attempting to compare the two chronic conditions.
Thus, it is well documented that clinical phenotypes, cervical musculoskeletal features and pathophysiology are different between episodic forms of migraine and tension-type headache (1,9–11). However, since patients with CM and CTTH are often not distinguished by pain phenotypes, (3), studies are needed to identify if chronic forms of migraine and TTH present differences in clinical psychophysical variables (e.g., neck range of motion, head posture, and psychosocial variables) in an attempt to open perspectives for distinguishing both conditions. Additionally, whether Quantitative Sensory Tests (PPTs, TS and the extension of pain) can help to better explain the underlying mechanisms. The distinction between the two chronic conditions is crucial in the clinical setting and needed to design future tailored non-pharmacological strategies, such as physiotherapy and psychology to help in the management of both headache types. Therefore, the aim of the present study is to compare psychophysical outcomes, as well as PPTs and TS in the trigeminal area, the neck and distal regions between patients with CM, CTTH and controls. Finally, based on a previous Delphi study to determine which outcome variables are the most relevant in clinical practice (23), our secondary aim is to determine which psychophysical variables best explain headache frequency, quality of life and cervical disability.
Methods
A single blinded observational cross-sectional study was conducted at a specialized headache clinic in La Paz University Hospital in Madrid. Consecutive patients diagnosed with CM or CTTH were recruited through the specialized headache clinic between December 2019 and October 2020. Asymptomatic participants were recruited through advertisements.
This study was developed according to the Strengthening the Report of Observational Studies in Epidemiology statement (STROBE) to increase the methodological quality of the reports of observational studies (24). It was approved by the Ethics Committee for Clinical Research of a public hospital in Madrid (HULP, PI-1241). Furthermore, written informed consent was obtained from all the participants before enrolment in the study.
Participants
Patients who met the following criteria were included: adults diagnosed with CM or CTTH by an experienced headache neurologist according to the 3rd edition of the International Classification of Headache Disorders (ICHD-III) (1). Patients with another concomitant headache diagnosis, a history of trauma or surgery in the cervical-cranio-mandibular region, drug or analgesic medications abuse, meningitis, fibromyalgia or other chronic pain diagnosis different than CM or CTTH were excluded. Additionally, diagnosed peripheral neuropathies, rheumatic diseases, or other diseases with potential involvement of the sensory pathways were also excluded. Due to ethical aspects, subjects with CM and CTTH were allowed to continue their drug treatment. However, they had to be headache free at the moment of the assessment. Patients were instructed to reschedule the examination appointment in case of headache on the same day. Controls were considered eligible if they reported two or fewer episodes of headache in the previous year and did not meet any of the exclusion criteria listed above.
Procedures
On the day of the assessment, participants were informed verbally and in writing about study procedures. After signing the informed consent form, they were asked to complete sociodemographic and psychosocial questionnaires. The Spanish validated questionnaires of headache impact (HIT-6) (25), quality of life (SF-12) (26), neck disability (NDI) (27), Beck depression inventory (BDI) (28) and Tampa scale of kinesiophobia (TSK-17) (29) were assessed.
Size of painful area (pain drawings)
Headache patients were asked to colour in the area of pain using an electronic three-dimensional body chart [anterior, lateral (left, right) and posterior views] on a 10.1” tablet (Samsung Galaxy Note 10.1, Navigate Pain application, Aglance Solutions, Aalborg, Denmark) with a tablet pen (1.5 mm pen tip).
The tablets, with four different views of the body chart, were given to each participant who were further instructed to highlight the entire area of pain instead of indicating it by a cross or drawing a circle around the area. Digitally collected spatial characteristics of pain have been shown to be valid, replicable and comparable to that collected on paper (30).
For the data analysis of the size of the painful area, each participant was asked to paint the strictly “cephalic” in a different colour from the “extracephalic”. In this sense, the nuchal line (posterior view) the mastoid process (lateral views) and the mandible (anterior view) were established as reference cut-off areas to differentiate “cephalic pain” (above these areas) from “extracephalic pain” (below these areas). Spatial characteristics of the “cephalic” and “extracephalic” pain were analysed separately combining all views (frontal, posterior and lateral) and were automatically extracted and expressed in (%) of the body by the app.
The above information was collected by examiner 1, who was not blinded to the diagnosis. After these assessments, examiner 1 left the room and examiners 2 and 3 (blinded to the diagnosis) continued the examination described below (Figure 1).
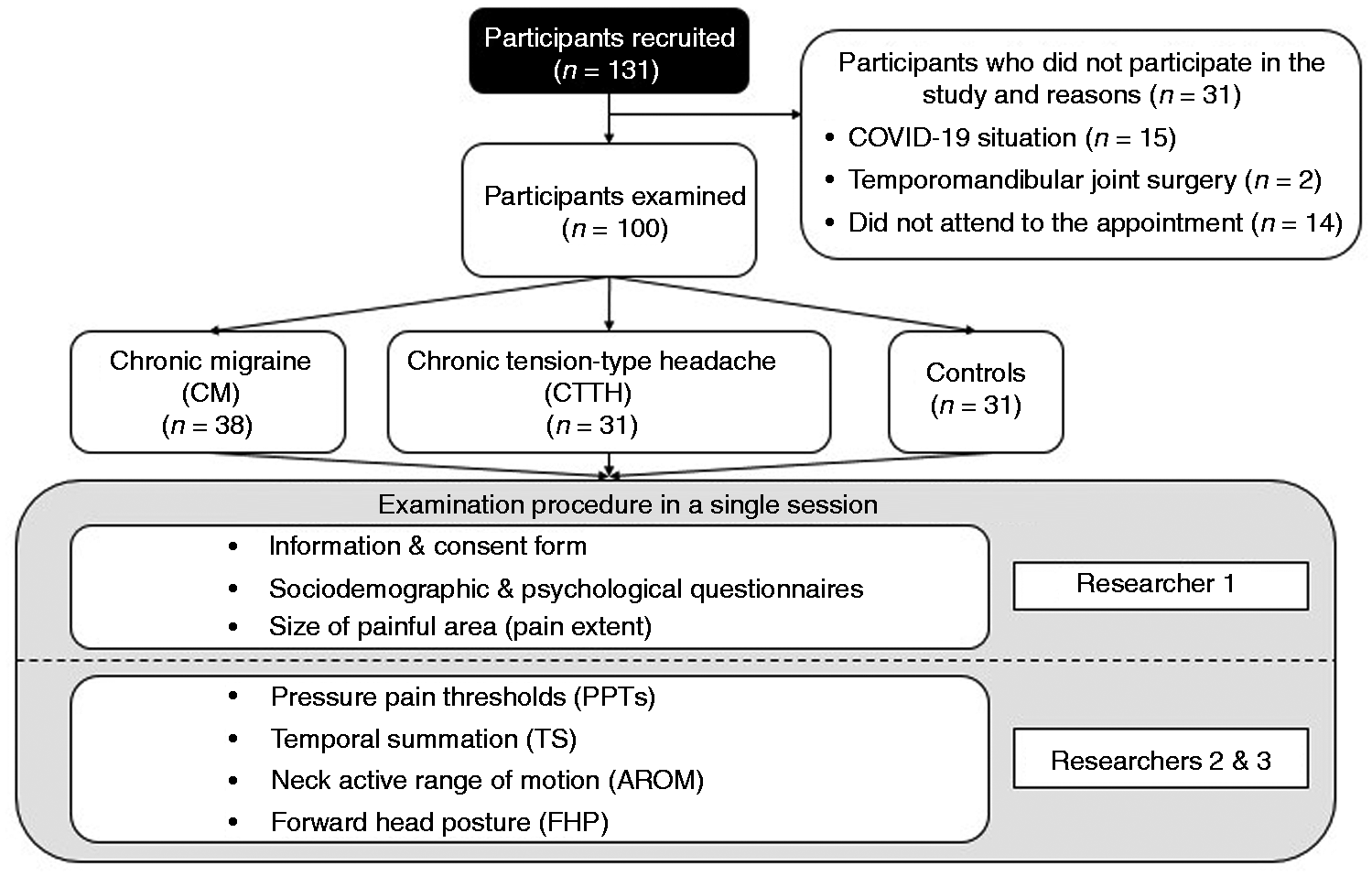
Study flow chart.
Pressure pain thresholds (PPTs)
An algometer (model Fx. 25 Force Gage, Wagner Instruments, Greenwich, CT, USA) was used to measure the minimal amount of pressure which would elicit the first sensation of pain. It had a 1 cm2 rubber tip attached to a pressure meter (31). A gradually increasing pressure was applied with the algometer at a rate of 49 kpa/s or 98 kpa/s over different trigeminal or extra-trigeminal locations, respectively (32,33). All participants were instructed to respond as soon as the sensation of pressure became painful. PPTs were measured in three different regions. 1) “The trigeminal region” was measured bilaterally over the anterior and posterior fibers of the temporalis muscle and over the upper and lower part of masseter muscle (eight locations). 2) “The neck region” was assessed over the spinal process of C2 and bilaterally over the upper trapezius and suboccipital muscles (five locations). 3) “A distal region” was assessed bilaterally over the tibialis anterior muscle (two locations) (Figure 2). Three consecutive measurements, at each location, were assessed with an interval of 30 seconds between each measurement. The mean of the three measurements was calculated for each location and side. The data were recorded in kpa.

Locations of pressure pain thresholds (PPTs). (a) Bilaterally over the anterior and posterior parts of temporalis muscle, bilaterally over the upper and lower part of masseter muscles. (b) Spinous process of C2, bilaterally over suboccipitalis muscles and upper trapezius. (c) Tibialis anterior muscle.
Temporal summation (TS)
To assess temporal summation (TS), Von Frey filaments (Pressure Aesthesiometer®, Stoelting, Chicago, IL) were used. Participants were positioned in supine and the measurement was performed on the dominant headache side over the anterior part of temporalis muscle and over the tibialis anterior muscle. The dominant headache side was defined as the side were >60% of headache occurs. If there was no dominant headache side, the assessment side was randomized. Initially, a single stimulus was applied to each location. Subsequently, the patient rated the perceived pain intensity using a visual analogue scale. Then, ten rhythmic stimuli were applied to the same location guided by a metronome at 60 bpm. The pain intensity was once again rated for the perceived intensity of the final stimulus (34). To calculate the definitive value the following formula was used: Wind-up Ratio = Last measurement/1st measurement (35).
Neck Active Range of Motion (AROM)
To quantify the cervical active range of motion (AROM) the validated cervical inclinometer (CROM; Cervical Range of Motion Instrument, Performance Attainment Associates, 1988, University of Minnesota) was used (36). Participants were positioned sitting on a chair and the CROM was placed on the head. Participants were asked to perform three consecutive repetitions maximum flexion and extension, lateral bending (left to right) and rotation (left to right). Data were recorded in degrees of movement. The total AROM was determined by adding all measured values.
Forward head posture (FHP)
The CROM with forward head arm and a vertebral locator (with a bubble leveller) was used to measure the position of the head in a relaxed position. Participants were asked to sit in a comfortable upright position with the thoracic spine in contact with the back of the chair. The CROM instrument were positioned in the same way as for the AROM measurement. Additionally, the forward head arm was placed in the frame of the CROM. The base of the vertebral locator was placed on the spinous process C7, while keeping the bubble leveller centred. The second examiner was standing to the left of the participant keeping a neutral flexion-extension position and measuring the posture using as a reference the cut of the two CROMS arms, in centimetres. Excellent reliability has been showed for the measurement of FHP using the CROM instrument (37).
Sample size calculation
The sample size was calculated based on data from a pilot study (n = 15) using G*Power 3.1 (University of Düsseldorf, Germany). The required sample size to detect differences between groups in the PPT of the trigeminal region was 99 subjects in total, with an effect size of 0.32, power of 80% and α of 5%. Considering the neck region, the sample size required to detect differences among groups is 51, with an effect size of 0.44, power of 80% and α of 5%. For the distal region it required a total of 66 participants, with an effect size of 0.40, power of 80% and α of 5%. Due to the planned regression analysis (10 subjects per variable), the number of cases was increased to n = 100 in order to obtain stable estimates for the regression coefficients (38).
Data analysis
The data analysis was performed using IBM SPSS (v.25.0; SPSS, Inc, Chicago, IL) statistical software. Shapiro-Wilk test (N < 50 per group) was carried out to test for normal distribution of the data. Descriptive data were presented as mean and standard deviation for quantitative variables, and frequency and percentage for categorical variables.
Comparisons between groups
Variables related to headache symptoms (e.g., duration, size of painful area or frequency of headache) were compared between both headache groups using a Student’s t-test for independent samples. To assess the effect size of the mean differences, Cohen’s d was calculated [Cohen´s d between (0.21–0.50), (0.51–0.80) or >0.81 are considered as small, medium or large effects, respectively] (39).
Psychophysical variables were analysed using a one-way analysis of the variance (ANOVA) with Bonferroni-corrected post hoc test. The effect size was calculated using partial eta-squared (η p 2) from the ANOVA analysis [ηp2 between (0.01-0.039), (0.06-0.11), and >0.14 was considered as small, medium or large, respectively] (39).
A repeated-measures ANOVA was carried out to analyse differences in PPTs and TS, considering “area” as the within factor and “group” as between factors. PPT results were pooled for three areas (“trigeminal region”, “neck region” and “distal region”). Thus, variables were created by calculating the PPT average for each area (“trigeminal region”: both masseter and both temporalis locations; “neck region” upper trapezius, suboccipital muscles and spinous process; “distal region” tibialis anterior muscle). There were no differences between sides. Consequently, the mean of each pair of muscles (left and right) was calculated and considered for further analysis. For TS measurements, data were kept separate for “temporalis muscle” and “tibialis anterior muscle”. In case of significant interaction effects, Bonferroni corrected post-hoc t-tests were performed. All p-values are presented with Bonferroni correction. The level of the significance was set at p < 0.05.
Multiple linear regression
Considering that not all the variables showed a normal distribution, Spearman’s rank correlation coefficient was used to analyse correlation between dependent and independent variables. Scores range from −1 to 1, with values less than 0.3 meaning weak correlation; 0.3 to 0.7, moderate correlation; and more than, 0.7 strong correlation (40) According to correlation test results, only the three independent variables with highest values of Spearman’s Rho were entered into a predictive model (41,42).
Multiple backward linear regression models were used to examine the capacity of physical and psychosocial variables to predict the variability of the frequency of headache, quality of life and neck disability. The strength of the association was determined using coefficient (B), R2, adjusted R2 and p-values. Also, standardized beta coefficients (β) were included for all predictors in the final model to allow direct comparisons between different predictor variables. The level of the significance was set at p < 0.10.
Results
A total of 131 participants were recruited. 31 participants were excluded because they did not meet the inclusion criteria. Finally, a total of 100 participants (31 CTTH, 38 CM and 31 healthy controls) were assessed (Figure 1).
No differences between groups regarding demographic characteristics and also, no differences between patient groups regarding headache characteristics were found (Table 1). The ANOVA and T-Test analyses showed differences between groups for all variables except for the FHP [F(2,97) = 1.61, p = 0,21, η2 = 0.03]. Post-hoc analyses found differences for these variables comparing both patient groups with controls, with large effect sizes (d > 0.8). T-Test resulted in no differences between headache groups regarding pain location (cephalic [t(67) = 0.55, p = 0.55; d = 0.14] and extracephalic [t(67) = 0.75, p = 0.46; d = 0.18]) (Table 2). Figure 3 shows the pain frequency maps of both patient groups.
Sample characteristics and comparison of headache features between groups.

CTTH: Chronic tension-type headache; CM: Chronic Migraine; SD: Standard deviation.
Comparison between groups of psychosocial and physical outcomes.

F†: F obtained from statistical Welch’s test; CTTH: Chronic tension-type headache; CM: Chronic Migraine; SD: Standard deviation; HIT-6: Headache Impact Test; NDI: Neck disability index; BDI: Beck´s depression inventory; SF-12: Quality of life; TSK-17: Tampa scale kinesiophobia; AROM: Active range of motion; FHP: Forward head posture; PPT: Pressure pain threshold; *p < 0.05; **p < 0.001

Distribution of pain frequencies, generated by overlapping body charts of each headache groups.
The repeated-measures ANOVA of PPTs showed significant differences for the area*group interaction (F[2,97] = 14.80; p < 0.001; ηp2 = 0.24), “Area” (F[2,97] = 206.28; p < 0.001; ηp2 = 0.70) and “group” (F[2,97] = 30.26; p < 0.001; ηp2 = 0.39). The Bonferroni corrected post-hoc tests showed differences between both headache groups compared to the controls in the “trigeminal”, “neck” and “distal” regions (p < 0.001; d > 0.8) but not between both patient groups. Additionally, Bonferroni corrected post-hoc tests showed differences within the three groups for “distal region” compared to “trigeminal” and “neck” regions (p < 0.001; d > 0.8). Also, post-hoc tests revealed differences within the CM group for “trigeminal” vs “neck” regions (p < 0.001; d = -0.29) (Figure 4). The analysis of TS showed no significant area*group interaction (F[2,97] = 1.71 P = 0.19; ηp2 = 0.04) but “area” (F[2,97] = 8.35 P = 0.005; ηp2 = 0.082) and “group” (F[2,97] = 12.36 P < 0.001; ηp2 = 0.20). The Bonferroni corrected post-hoc analysis showed between-group differences when both headache groups were compared to the controls in the “trigeminal”, “neck” and the “distal” regions (p < 0.001; d > 0.8) (Figure 5 and Appendix, Repeated-measures ANOVA for PPTs and TS).

Repeated-Measures ANOVA for pressure pain thresholds. All differences are presented with effect size (Cohen’s d); (**) p < 0.001. (CTTH) Chronic Tension-type headache; (CM) Chronic migraine.

Repeated-Measures ANOVA for temporal summation. All differences are presented with effect size (Cohen’s d); (**) p < 0.001. (CTTH) Chronic Tension-type headache; (CM) Chronic migraine.
The regression model for criterion variables (frequency of headaches, SF-12 and NDI) is presented in Table 3. NDI was a significant covariate for the frequency of headaches for CTTH (adjusted R2 = 0.14; β = 0.43; p = 0.03) and CM (adjusted R2 = 0.18; β = 0.51; p < 0.01). For quality of life (SF-12) the regression model showed that a combination of BDI and HIT-6 were significant predictors for CTTH (adjusted R2 = 0.50). A combination of BDI and location of pain was able to explain a 66% of the variance of quality of life scores within the CM group (adjusted R2 = 0.66). Finally, a combination of size of body pain and HIT-6 was able to predict 41% of the NDI scores within the CTTH group (adjusted R2 = 0.41) and a combination of BDI and TSK-17 explained 53% of the variance of the NDI scores in the CM group (adjusted R2 = 0.53).
Multiple backward linear regression analysis for frequency of headache episodes, SF-12, and NDI.

CTTH: Chronic tension-type headache; CM: Chronic migraine; HIT-6: Headache Impact Test; NDI: Neck disability index; BDI: Beck´s depression inventory; SF-12: Quality of life; SF-12 PH subscore: Quality of life, physical health subscore; SF-12 MH subscore: Quality of life, mental health subscore; TSK-17: Tampa scale kinesiophobia; FHP: Forward head posture; PPT: Pressure pain threshold; VIF: Variance inflation factor; D: Highest value of Cook’s Distances; †: Table shows p-values of Shapiro-Wilk test; *p < 0.05.
Discussion
The aim of this study was to compare psychophysical parameters between patients with CM and CTTH. Our results showed that patients with CM and CTTH presented similar levels of PPTs, TS, and size of the painful area. Furthermore, neither neck range of motion or psychosocial variables (depression, kinesiophobia, neck disability and quality of life) discriminated the two groups of patients, yet all patients differed from controls regarding all assessed variables. Neck disability was considered a significant predictor of greater headache frequency for both conditions. On the other hand, a combination of depression symptoms with headache impact or with location of pain were able to explain the quality of life scores for CTTH and CM, respectively. The variables that best explained neck disability were the extent of pain area and the headache impact in the CTTH group and depressive symptoms together with kinesiophobia in the CM group.
It can be suggested that alterations in somatosensory outcomes (PPTs, TS and the size of painful area of pain) are manifestations of alterations within the central nervous system such as central sensitization and thus can contribute to headache chronification (43,44). Musculoskeletal and psychosocial disorders have been reported to be associated with a higher frequency headache in patients with CM and CTTH, which are the most prevalent headaches (45–47). According to Arendt-Nielsen et al. central sensitization is present if PPTs and TS are measured at different body regions, including distal pain-free areas, and compared with those of a matched healthy sample (48).
In this study, the size of the painful area during headache episodes, PPTs and TS were measured in local (trigeminal) and remote regions (lower limbs). In addition, PPTs were measured in the trigeminal area and just outside of the area innervated by the trigeminal nerve. The present data indicate that the results were not dependent on the regions evaluated, i.e., alterations in TS and PPTs were observed in headache-affected regions as well as in remote body locations. These results differ from those found in subjects with episodic migraine (49). Szikszay et al. found altered pain modulation in the trigeminal system but not in the somatic system (49).
Furthermore, according to the literature the clinical phenotype and neck musculoskeletal examination, including pain location AROM and FHP allow to differentiate between migraine and episodic tension-type headaches (1,9,10,22). However, neither our pain maps, nor AROM nor FHP allowed us to differentiate both when they become chronic.
Moreover, previous studies investigated several quantitative sensory tests parameters in patients with headache (2,11,18,22,44–46,50–56). From these cited articles, only episodic tension-type and episodic migraine were compared regarding PPTs (11).
The absence of differences between the two chronic headache types are in line with a recent study based on magnetoencephalography that compared the cortex excitability in chronic and episodic TTH and migraine during electrical stimulation of the index finger (57). Chen et al. reported altered inhibition of the primary somatosensory cortex in both episodic and chronic TTH (57). In migraine patients, just patients with the chronic form showed deficits in inhibition. Authors suggested that this disinhibition is inherent to tension-type headache diagnosis but an acquired maladaptation occurring with migraine chronification. Moreover, a recent meta-analysis carried out on data from migraine patients, reported decreased PPTs just before and during episodes while data tended to return to normal between episodes (20). Olesen et al. indicated that although peripheral and central sensitization changes were reported in episodic migraine, peripheral trigeminal sensitization prevails interictally (58).
Thus, considering the shared pathophysiological mechanisms reported across chronic headache types, it might be suggested that central sensitization is a key factor in the chronification of the two disorders (59). However, the difference in clinical phenotypes, confirmed in this current study by mechanical hyperalgesia in the “trigeminal” vs “neck region” in CM but not found in CTTH, may suggest that CM and CTTH are not similar in all aspects.
While there is current consensus that migraine and tension type headache present different pathophysiological characteristics in their episodic forms, the results of this research suggest that when both diseases become chronic, central sensitization become a prominent feature in both headache types. Therefore, we found characteristics overlapping not only in somatosensory features and pain phenotypes, but also in psychological characteristics.
Interestingly, these current results are also in line with previous evidence showing that CM and CTTH present higher levels of depressive symptoms than healthy subjects, with no differences between both groups of patients (60,61). In addition, Buse et al. reported that higher depressive symptoms is associated to lower levels of quality of life in patients with migraine and tension-type headache (62), and Ashina et al. reported that depressive symptoms is an important covariate to explain lower level in the mental health subscore of the quality of life in CTTH (63). In line with Ashina et al. we also evidenced that depressive symptoms were a covariate to predict quality of life of both headache groups (63). These findings may explain why we were unable to differentiate between headache groups in relation to quality of life with the mental health subscale. However, our results as Ashina et al. 2021 also reported, showed that pericranial PPTs were not a covariate of quality of life, neither in the physical nor in the mental sphere in CTTH (63).
Ours results have therefore implications for clinical practice and future studies. Since both chronic headache types exhibited similar psychophysical presentation, we suggest that no tailored approach may be needed for the non-pharmacological, such as physiotherapy and psychology treatment addressing the same aspects assessed in the present study. However, future studies should continue to investigate physical function and psychological factors of both conditions to allow more precise diagnosis and therefore treatment.
Some limitations of this study must be acknowledged. First, the study is cross-sectional in nature thus causal inference is not possible; future studies should employ longitudinal designs. Second, external validity is reduced given the fact that our sample represented a specific geographical area and a specialised headache clinic. Third, there might be a gender bias in the current study, although our data reflect the usual male-to-female ratio (1:3) found in previous studies (64). Fourth, the quality features of pain were not recorded in the present study, so we recommend its inclusion in future studies. Fifth, although we ensured that patients were headache-free on the day of the examination, we did not record presence of pain in the 72 hours before and after the examination, therefore patients could have been assessed during their preictal or post-ictal phase. Finally, since patients were allowed to continue their usual pharmacological treatment, this limitation should be taken into consideration and monitored in future studies.
In conclusion, we can point out that patients with CM and CTTH showed no differences in all psychophysical measures. Nevertheless, trigeminal sensitization is higher than extra-trigeminal in the CM group. All outcomes were significantly worse in patient groups compared to controls. The variable best explaining headache frequency in both patients groups was NDI. Furthermore, the predictors in both groups for quality of life and neck disability included depression symptoms, headache impact, size of painful area and kinesiophobia.
Clinical Implications:
Patients with chronic tension-type headache and chronic migraine showed similar psychophysical results, but were significantly worse when compared to controls. The sensitization of the trigeminal system was more pronounced in the chronic migraine group compared to the chronic tension type headache group and to controls. Neck disability was the variable best explaining headache frequency in both patient groups.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211060315 - Supplemental material for Psychophysical testing in chronic migraine and chronic tension type headache: An observational study
Supplemental material, sj-pdf-1-cep-10.1177_03331024211060315 for Psychophysical testing in chronic migraine and chronic tension type headache: An observational study by Ignacio Elizagaray-García, Gabriela F Carvalho, Tibor M Szikszay, Waclaw M Adamczyk, Gonzalo Navarro-Fernández, Paula Alvarez-Testillano, Javier Díaz-de-Terán, Kerstin Luedtke and Alfonso Gil-Martínez in Cephalalgia
Footnotes
Acknowledgements
We thank Shellie A Boudreau for their support in making pain frequency maps. Waclaw M Adamczyk is suppoted by the START program of Polish Foundation for Science.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
A single blinded observational cross-sectional study was approved by the Ethics Committee for Clinical Research of a public hospital in Madrid (HULP, PI-1241). Furthermore, written informed consent was obtained from all the participants before enrolment in the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
