Abstract
Chronic myofascial pain is very common in the general population. The pain is most frequently located in the shoulder and neck regions, and nociceptive input from these regions may play an important role for tension-type headache. The mechanisms leading to the frequent occurrence of muscle pain in the shoulder and neck regions are largely unknown. It is possible that the pain is caused by increased sensitivity of muscle nociceptors or by central sensitization induced by nociceptive input from muscle. The primary aim of the present study was to compare muscle pain sensitivity in the trapezius and anterior tibial muscles. The secondary aim was to investigate whether temporal summation, a clinical correlate of wind-up, is more pronounced in muscle than in skin and, if so, whether such a difference is more pronounced in the trapezius than in the anterior tibial region. Sixteen healthy subjects were included. Pressure-pain thresholds and electrical cutaneous and intramuscular pain thresholds were measured at standard anatomical points in the trapezius and anterior tibial regions. Temporal summation was assessed by repetitive electrical stimulation. Pressure-pain thresholds (P = 0.005) and intramuscular electrical pain thresholds (P = 0.006) were significantly lower in trapezius than in anterior tibial muscle. Temporal summation was present in skin and muscle of both regions (P < 0.001). The degree of temporal summation was significantly higher in muscle than in skin in the trapezius region (P = 0.02), but not in the anterior tibial region (P = 0.47). In conclusion, we found that muscle pain sensitivity was higher in the trapezius than in the anterior tibial muscle. We also demonstrated that temporal summation could be induced in both muscle and skin and, importantly, that temporal summation was significantly more pronounced in muscle than in skin in the trapezius but not in the anterior tibial region. These data may help to explain why chronic muscle pain most frequently is located in the shoulder and neck regions.
Keywords
Introduction
Chronic muscle pain is a very prevalent disorder, which affects some regions of the body more often than others. Andersson et al. (1) found that the most common location of chronic muscle pain was the shoulder and neck regions, in which chronic muscle pain was reported by 30% of the general population, followed by low back pain affecting 23% of the population. Muscle pain from the shoulder and neck probably plays an important role in the most common primary headache disorder, tension-type headache (2, 3).
The pathophysiological mechanisms responsible for muscle pain are not fully clarified and it is unknown why muscle pain is more prevalent in some regions than in others. In particular, it has never been tested whether this could be due to differences in muscle pain sensitivity. Moreover, experimental evidence suggests that continuous nociceptive input from myofascial tissues may lead to increased excitability of neurones in the CNS, i.e. central sensitization (4), and thereby chronification of muscular pain conditions (2). In addition, it has been shown that nociceptive input from muscle sensitizes the CNS to a higher extent than input from cutaneous tissues (5). The experimental models used to investigate central sensitization include the study of wind-up in animals (6–8) and temporal summation of pain in humans (9–14). In rats, extracellular recordings in dorsal horn neurones have demonstrated that wind-up produces some characteristics of central sensitization (7). In humans, temporal summation has been described as a gradual increase in pain sensitivity to repetitive stimuli (9). These studies suggested that temporal summation might be a clinical correlate of wind-up in humans and one of several initiators of central sensitization (15, 16).
The primary aim of the present study was to compare pain sensitivity in the trapezius and anterior tibial muscles by intramuscular electrical stimulation. Furthermore, we aimed to examine whether temporal summation is more pronounced in muscle than in skin and, if so, whether such a difference is more pronounced in the trapezius region than in the anterior tibial region.
Subjects and methods
Subjects
Sixteen healthy volunteers were included (Table 1). Exclusion criteria were: more than 12 headache days per year; headache on the day of examination; neurological, systemic and psychiatric disorders; any form of drug abuse or dependency; moderate to severe tenderness of tibialis anterior and trapezius muscles (local tenderness score > 1 on a 4-point (0–3) scale) (17); pregnancy or nursing. The study was approved by the local ethics committee, and was undertaken in accordance with the Helsinki Declaration of 1975, as revised in 1983. All subjects gave informed consent to participate in the study.
Clinical data on healthy subjects

Values are medians (quartiles).
Apparatus
Electrical pain and sensory thresholds
Intramuscular electrical stimulation was induced by two needle electrodes with uninsulated tips (3 mm) (Dantec Measurement Technology A/S, Copenhagen, Denmark). Cutaneous electrical stimulation was induced by a bipolar saline-soaked surface stimulation electrode (Dantec Measurement Technology A/S). Electrical stimuli were generated by a constant current stimulator (Counterpoint; Dantec Measurement Technology A/S). A single stimulus lasted 1 ms. Repetitive stimuli consisted of single stimuli delivered for 5 s at 100 Hz, i.e. a series of 500 stimuli.
Pressure–pain thresholds
An electronic pressure algometer (Somedic AB, Hörby, Sweden) was used to measure pressure–pain thresholds. A circular stimulation probe (0.50 cm2) and a pressure loading rate of 22 kPa/s were used. The algometer has been described in detail elsewhere (18).
Electromyography
The electromyographic (EMG) signals from the tibialis anterior and trapezius muscles were recorded by an electromyograph (Counterpoint; Dantec Measurement Technology A/S).
Procedures
The subjects were examined sitting in a dental chair with headrest. All measurements were performed by the same observer (S.A.) throughout the study. The subjects were assigned to muscular and cutaneous stimulations and to examination at the trapezius and anterior tibial muscles in a randomized and balanced design. Thresholds to single stimuli were recorded first, followed by repetitive stimuli. Measurements of pressure–pain, electrical sensory and pain thresholds were performed at standard anatomical points on the tibialis anterior and trapezius muscles, at the non-dominant side. On anterior tibial muscle the standard point was located 15.5 cm distal to the lower tip of knee cap (apex patellae), and on trapezius muscle the point was located on the centre of the descending part of the trapezius muscle midway between the processus spinosus of the 7th cervical vertebra and the acromion.
Pericranial tenderness recording
Pericranial tenderness was recorded according to the total tenderness score (TTS) (total score of eight pairs of pericranial muscles and tendon insertions scored on a 4-point (0–3) scale at each location with maximum possible score = 48), which has proved to be reliable (17).
Pressure–pain thresholds
The pressure–pain threshold (PPT) was defined as the pressure at which the sensation changed from pressure alone to a combination of pressure and pain. The subject indicated that the pain threshold was reached by pressing a handheld button. The algometer display was thereby ‘frozen’ and the pressure was immediately released.
Electrical sensory and pain thresholds
The surface and the needle electrodes were placed with the cathode 5 mm proximally and with the anode 5 mm distally to the standard points. The needle electrodes were inserted 15 mm into the muscle. The correct positioning of electrodes in muscle was verified by demonstration of EMG activity in response to voluntary muscle contraction.
The sensory threshold was defined as the weakest stimulus detectable by the subject, and the pain detection threshold was defined as the weakest stimulus perceived as painful by the subject. The sensory and pain thresholds were measured by the method of limits (19). The stimulus strength was gradually increased up to the first report of sensation with notation of value in mA. The procedure was performed three times and the threshold was calculated as the mean of the three values. A minimum interval of 1 min between successive determinations was used to avoid hyperalgesia in the area of stimulation.
Data analysis and statistics
To investigate whether the ratio between skin and muscle pain sensitivity was higher in the trapezius than in the tibialis region, we calculated the ratio of cutaneous to muscle pain thresholds, R CM, by dividing cutaneous electrical pain thresholds by muscle electrical pain thresholds. A high ratio will indicate that muscle is relatively more sensitive than skin and vice versa.
Temporal summation was considered to be present if the threshold to repetitive stimuli was significantly lower than to single stimuli (10). The degree of temporal summation was evaluated by calculation of the summation ratio defined as the difference between pain thresholds to single and repetitive stimuli divided by pain thresholds to single stimuli (11). A high summation ratio will indicate a high degree of temporal summation and vice versa.
We used the Shapiro–Wilk test to determine whether the data were normally distributed. Normally distributed results (PPTs) were presented as mean and 95% confidence interval (CI) and t-test was used to test paired observations. Results that were not normally distributed were presented as median with quartiles and the Wilcoxon signed ranks test was used to test recordings for paired observations. Spearman's test was used for calculation of correlation coefficients, R. These analyses were done using SPSS®, version 10.0.5 software (SPSS Inc., Chicago, IL, USA). Five percent was accepted as level of significance.
Results
All subjects completed the study and their clinical characteristics are presented in Table 1.
Muscular electrical and mechanical pain thresholds
The muscular electrical pain thresholds to single stimuli were significantly lower in trapezius muscle than in anterior tibial muscle (P = 0.006, Table 2, Fig. 1). Muscular pain thresholds to repetitive stimuli tended to be lower in trapezius muscle compared with anterior tibial muscle (P = 0.05, Table 2).
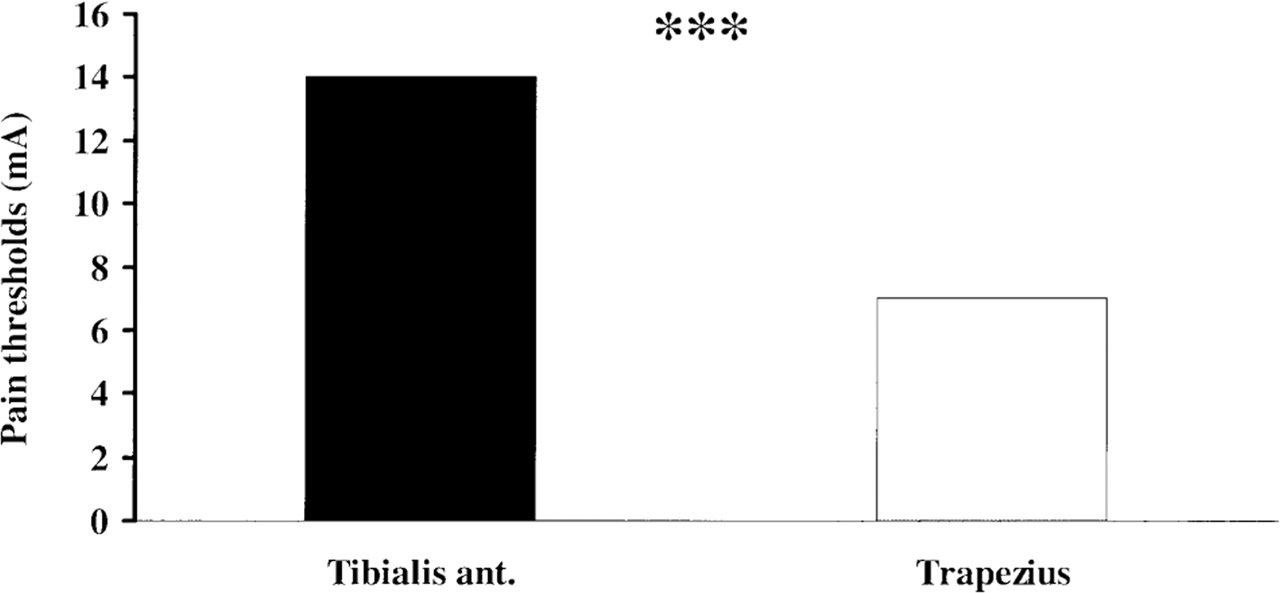
Pain thresholds to single electrical stimuli were significantly lower in trapezius muscle than in anterior tibial muscle. Values are mA given as medians. ∗∗∗P = 0.006.
Cutaneous and muscular electrical pain thresholds (mA) in the trapezius and anterior tibial regions
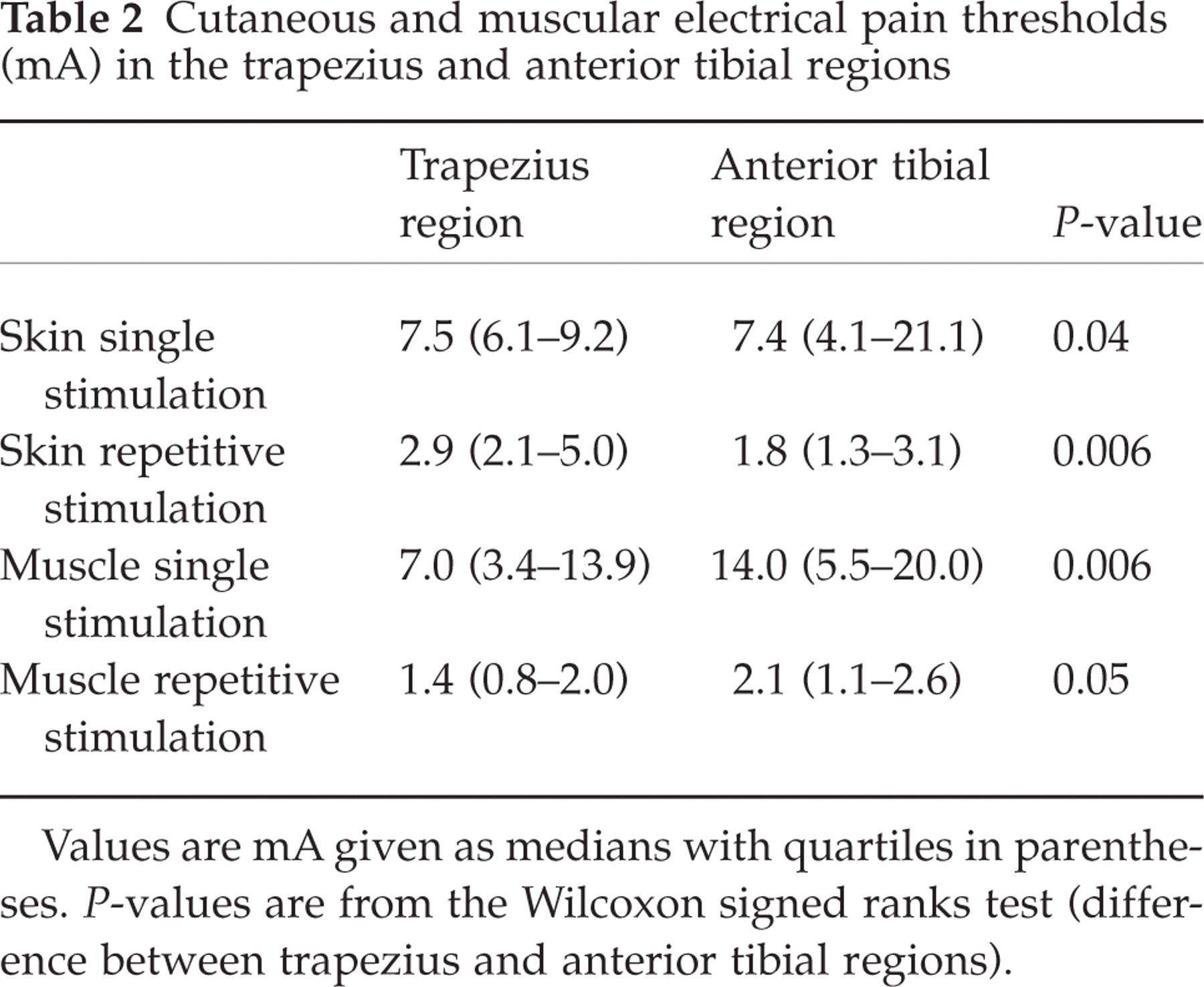
Values are mA given as medians with quartiles in parentheses. P-values are from the Wilcoxon signed ranks test (difference between trapezius and anterior tibial regions).
The PPT in trapezius muscle, 453 ± 123 kPa, was significantly lower than in anterior tibial muscle, 563 ± 135 kPa (P = 0.005).
Cutaneous electrical pain thresholds
The cutaneous electrical pain thresholds to single stimulation were significantly lower in the trapezius region than in the tibialis region (P = 0.04, Table 2). The apparent discrepancy between the significantly decreased thresholds and the slightly higher median value for the trapezius region can be explained by the fact that 10 subjects had the lowest pain thresholds at this region, four subjects had the lowest values at the tibialis region, while two subjects had the same values in the two regions. The cutaneous pain thresholds to repetitive stimuli were significantly lower in the tibialis region than in the trapezius region (P = 0.006, Table 2).
Cutaneous and muscular electrical sensory thresholds
In muscle, we found no difference in sensory thresholds to single electrical stimuli between trapezius, 1.0 (0.5–1.5) mA and tibialis anterior, 1.3 (0.3–2.5) mA, regions (P = 0.61). Also, there was no difference in muscular sensory thresholds to repetitive electrical stimuli between trapezius, 0.6 (0.3–0.8) mA, and tibialis anterior, 0.7 (0.3–1.2) mA, regions (P = 0.36).
In skin, sensory thresholds to single electrical stimuli in the trapezius region, 1.2 (1.0–1.6) mA, did not differ from the anterior tibial region, 1.2 (0.8–1.7) mA (P = 0.95). In addition, no difference was found between sensory thresholds to repetitive electrical stimuli in skin in the tibialis anterior, 0.6 (0.4–0.7) mA, and the trapezius, 0.6 (0.4–0.7) mA, regions (P = 0.65).
Temporal summation
Temporal summation was found to be present both in skin and muscle in both regions, since pain thresholds to repetitive stimuli were significantly lower than to single stimuli both in the anterior tibial region (P < 0.001) and in the trapezius region (P < 0.001) (Fig. 2).
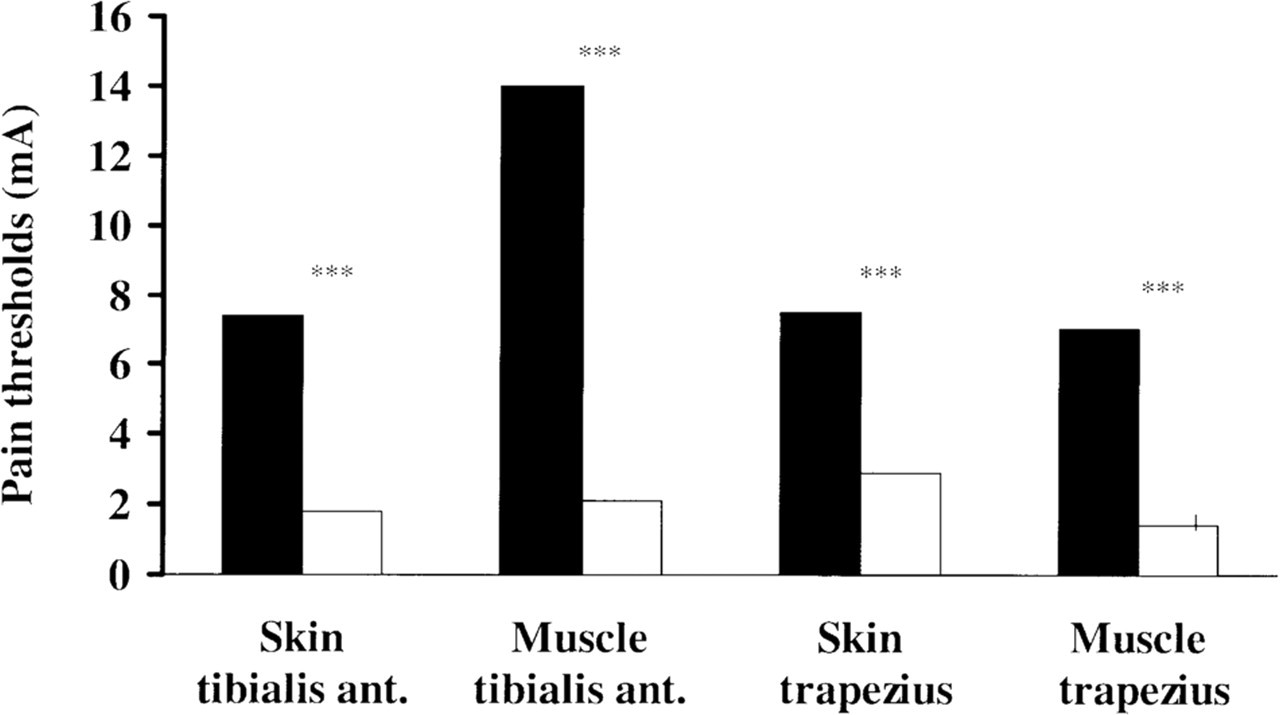
Cutaneous and muscular pain thresholds to repetitive stimuli (□) were lower than to single stimuli (▪) in both the trapezius region and in the anterior tibial region, demonstrating the presence of temporal summation at all sites tested. Values are mA given as medians. ∗∗∗P < 0.001.
The summation ratio was significantly higher in muscle than in skin (P = 0.02, Table 3) in the trapezius region. In the anterior tibial region no difference in the summation ratio between muscle and skin was found (P = 0.47, Table 3).
Summation ratios of electrical pain thresholds
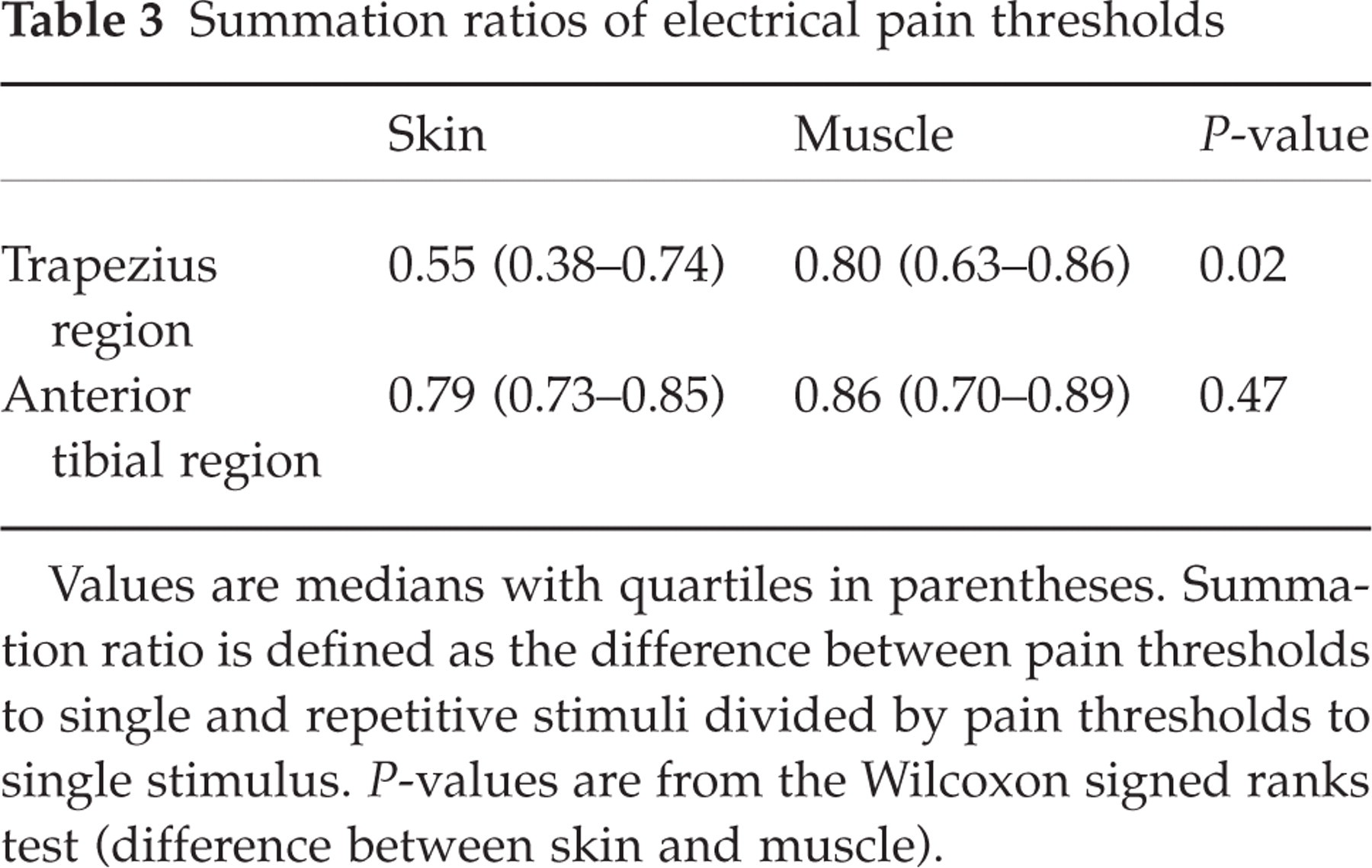
Values are medians with quartiles in parentheses. Summation ratio is defined as the difference between pain thresholds to single and repetitive stimuli divided by pain thresholds to single stimulus. P-values are from the Wilcoxon signed ranks test (difference between skin and muscle).
Ratio between cutaneous and muscular thresholds
The ratio between skin and muscle pain thresholds, R CM, for repetitive stimuli was significantly higher in the trapezius region, 2.5 (1.0–5.5), than in the tibialis region, 1.0 (0.5–1.3) (P = 0.008). For single stimuli there was no difference between R CM in the trapezius region, 1.0 (0.7–2.3), and in the tibialis region, 0.7 (0.4–1.3) (P = 0.28).
Correlations between pressure–pain thresholds and muscular electrical pain thresholds
In the trapezius muscle there was a significantly positive correlation between PPTs and muscular pain thresholds to single stimuli (r = 0.51, P = 0.04), while no significant correlation was found in anterior tibial muscle (r = 0.23, P = 0.40).
Discussion
Muscle pain sensitivity
The major finding of the present study was a higher pain sensitivity in trapezius muscle than in anterior tibial muscle. To our knowledge, muscle pain sensitivity in the shoulder and neck regions has not previously been compared with muscle pain sensitivity in the extremities. We found that both pain thresholds to single electrical stimuli and pressure–pain thresholds were lower in the trapezius muscle than in the anterior tibial muscle. We also showed that pain thresholds to single stimuli and pressure–pain thresholds were positively correlated in trapezius muscle but not in anterior tibial muscle. It has previously been speculated whether pressure–pain thresholds mainly reflect muscle or skin pain sensitivity (20). Our findings of considerably lower electrical muscular pain thresholds and practically equal cutaneous thresholds in the trapezius region compared with the anterior tibial region indicate that the increased pressure–pain sensitivity in the trapezius region can be ascribed mainly to increased muscle sensitivity.
Several factors may be responsible for the higher pain sensitivity in the trapezius muscle than in the anterior tibial muscle. Since the sensory detection thresholds did not differ in the two muscles, it seems unlikely that the sensory innervation in general is more dense in the trapezius muscle. However, it is possible that the density of nociceptors is higher or that the nociceptors in the trapezius muscle are more sensitive than in the anterior tibial muscle. Electrophysiological experiments in rabbits have shown that shoulder muscles contain a high density of nociceptors (21). Another possible explanation could be an increased strain on shoulder and neck muscles, since these muscles have to balance the head.
It has been shown that in rats projection territories of cutaneous primary afferent inputs from the forelimb and shoulder occupy more segments of spinal cord than do inputs from hindlimb (22). This could also be possible for muscular primary afferent inputs. Furthermore, several studies (23–25) have shown that nociceptive activity in human S1 cortex is somatotopically organized. Thus, a possible different somatotopic representation of the anterior tibial and trapezius regions in the somatosensory cerebral cortex could also explain the higher cutaneous and muscular electrical and pressure–pain thresholds in the latter region.
Temporal summation
Temporal summation in the present study was found both in skin and in muscles in both regions, which is in accordance with previous studies with electrical stimulation (9, 10, 26). Another important finding in the present study was the demonstration of significantly more pronounced temporal summation in muscle than in skin in the trapezius region, but not in the anterior tibial region. However, the absolute temporal summation values were quite similar in the two muscles. The findings from the trapezius region are in agreement with basic animal studies showing that nociceptive input from muscle sensitizes the central nervous system to a higher extent than input from cutaneous tissues (5). To our knowledge this has not previously been tested in humans. Arendt-Nielsen et al. (10) suggested that in humans, summation of input from deep structures might be more important than summation of cutaneous input. We also found a significantly higher ratio between cutaneous and muscular pain thresholds to repetitive stimuli in the trapezius region than in the anterior tibial region. Interestingly, we found lower cutaneous pain thresholds to repetitive electrical stimulation and, subsequently, high absolute values of summation ratio in skin in the anterior tibial than in the trapezius region. The significance of this finding is unknown.
Vecchiet et al. (27) previously reported that pain thresholds to repetitive electrical stimuli were lower in overlying skin, subcutis and muscle in deltoid, trapezius and quadriceps regions of patients with chronic fatigue syndrome than in healthy controls. However, the authors did not compare pain thresholds between these regions.
Tension-type headache
The present data may have implications for chronic myofascial pain disorders such as tension-type headache. Myofascial nociception from the pericranial muscles may play a major role in the pathophysiology of this disorder (2, 3). Thus, increased pericranial myofascial tenderness and hardness are the most prominent findings in patients with tension-type headache (28–31). The painful impulses from the pericranial muscles are probably referred to the head and perceived as headache. It is assumed that prolonged nociceptive input from the pericranial myofascial tissues is responsible for the central sensitization found in patients with chronic tension-type headache and thereby for chronification of the muscle pain and headache (2, 3). The present results might indicate that increased pain sensitivity in pericranial muscles could be a possible mechanism leading to myofascial pain and tenderness in patients with tension-type headache (3), and that continuous nociceptive input from pericranial myofascial tissues such as trapezius muscle may induce and maintain central sensitization in these patients (2). However, further studies are needed to support the importance of above-mentioned mechanism of central sensitization in patients with tension-type headache.
Methodological considerations
It should be kept in mind that temporal summation reflects only part of the process of central sensitization (15, 32), and the present results should therefore be supported by other studies before firm conclusions can be drawn. Temporal summation is induced by stimuli that are sufficiently intense to activate C-fibres and delivered with frequencies above 0.3 Hz (15, 16). Li et al. (7) suggested that nociceptors are likely to discharge increasingly at higher frequencies, and that central sensitization may depend on the frequency and duration of initial C nociceptor discharge. A stimulation frequency of 100 Hz has previously been used to induce long-term potentiation in animal studies (33, 34), and we therefore chose this frequency for repetitive stimulation. Further studies are needed to demonstrate possible differences between a stimulation frequency of 100 Hz and lower stimulation frequencies, such as 0.5 and 2 Hz.
It must be added that localization of regions, their proximity to sensitive areas and their threatening potential could affect responses of the subjects to painful stimuli. The trigeminal system has a large central representation and a presumed high degree of somatotopy (23), and facial stimuli generally are more threatening to the patients than stimulation of the limbs. Together, the neuroanatomical and the psychological fear factors can influence patients’ need to stop the stimulus faster.
The pathophysiological mechanisms responsible for the higher degree of temporal summation from muscle than from skin should be examined in future studies. At present we do not know whether this difference might be due to peripheral factors, e.g. increased sensitivity or firing pattern of nociceptors, or due to central factors, e.g. increased sensitivity to incoming stimuli at the level of the spinal dorsal horn or decreased level of descending inhibition from supraspinal structures. It should be noted that our measurements were made on healthy younger subjects, and other studies with patients are needed for further conclusions.
Footnotes
Acknowledgment
We thank Dr Messoud Ashina for his excellent technical assistance, and thoughtful and critical comments on this manuscript.
