Abstract
The tricyclic anti-depressant amitriptyline is widely used in the treatment of chronic tension-type headache. The aim of the present study was to investigate whether the analgesic effect is caused by a reduction of muscle pain or by a general reduction of pain sensitivity. Thirty-three non-depressed patients with chronic tension-type headache were treated with amitriptyline 75 mg/day and with the highly selective serotonin reuptake inhibitor citalopram 20 mg/day in a 32-week, double-blind, placebo-controlled, three-way crossover study. At the end of each treatment period, actual headache intensity and pericranial myofascial tenderness were recorded, pressure pain detection and tolerance thresholds were measured in the finger and in the temporal region and the electrical pain threshold was measured at the labial commissure. Amitriptyline reduced tenderness and headache intensity significantly more than placebo (P = 0.01 and P = 0.04, respectively). The reduction in tenderness could be ascribed solely to the group of patients who responded to amitriptyline treatment by at least 30% reduction in headache while tenderness was unchanged in non-responders. Amitriptyline did not affect pressure or electrical pain thresholds at any of the examined locations. Citalopram had no significant effect on any of the examined parameters. These findings indicate that amitriptyline elicits its analgesic effect in chronic myofascial pain by reducing the transmission of painful stimuli from myofascial tissues rather than by reducing overall pain sensitivity. We suggest that this effect is caused by a segmental reduction of central sensitization in combination with a peripheral anti-nociceptive action.
Introduction
It is well established that the tricyclic anti-depressant amitriptyline possess analgesic activity in various chronic pain conditions including chronic myofascial pain (1, 2) and that the analgesic action of this drug is independent of its anti-depressant effect (3, 4). However, the exact analgesic mode of action is unknown. The blockade of serotonin and noradrenaline reuptake in the central nervous system probably plays an essential role (3, 5), but other mechanisms such as reduction of central sensitization, i.e., increased excitability of neurons in the central nervous system, may also be important (6, 7).
Chronic tension-type headache is one of the most prevalent types of myofascial pain and one of the chronic disorders in which amitriptyline has a positive effect (4, 8–10). The most prominent abnormal finding in chronic tension-type headache is a considerably increased tenderness of pericranial myofascial tissues (11–13). Extensive research for peripheral abnormalities as a cause of the increased tenderness has not yet given conclusive results (14). It has been suggested that segmental central sensitization may contribute significantly to the increased tenderness and to chronification of the headache disorder (15, 16). Furthermore, a generalized hypersensitivity to pain (17–19) probably also contributes to the chronic pain in these patients.
The primary aim of the present study was to examine whether amitriptyline mainly inhibits muscle pain as reflected by a reduction of myofascial tenderness, or whether it reduces pain sensitivity in general as reflected by an increase in electrical or extracranial mechanical pain thresholds. The secondary aim was to clarify the relative importance of serotonin reuptake inhibition for the analgesic effect of amitriptyline by investigating whether the highly selective serotonin reuptake inhibitor citalopram affects the above-mentioned parameters in patients with chronic tension-type headache.
Patients and methods
Patients
Forty patients with chronic tension-type headache diagnosed according to the criteria of the International Headache Society (20) were included (Table 1). Seven patients with coexisting infrequent migraine (less than 1 day/month) were accepted. The patients were recruited from the out-patient headache clinic at Glostrup University Hospital, Copenhagen, Denmark, without respect to the presence or absence of myofascial tenderness. The inclusion criteria were a diagnosis of chronic tension-type headache and age between 18 and 65 years. The exclusion criteria were migraine more than 1 day/month, serious somatic or psychiatric diseases including depression (Hamilton Depression Score (21) ≥17), intake of prophylactic medications for tension-type headache, abuse of simple analgesics (corresponding to more than 2 g of aspirin/day), regular intake of opiates or benzodiazepines and previous treatment with anti-depressants. All patients underwent a general physical and a neurological examination and completed a diagnostic headache diary (22) during a 1-month run-in period. Thereafter, the patients were included in a trial of the prophylactic effect of amitriptyline and of citalopram in chronic tension-type headache. The trial was designed as a 32-week, double-blind, placebo-controlled, three-way crossover study and has previously been described in detail (4). Briefly, each drug was given in random order for 8 weeks, separated by 2-week wash-out periods (Fig. 1). Amitriptyline was administered in a dose-increasing regimen starting with 25 mg per day in the first week followed by 50 mg per day in the second week. In the last 6 weeks of treatment the daily dose of amitriptyline was 75 mg. The daily dose of citalopram was 20 mg during all 8 weeks of treatment. During the trial, the area under the headache curve (AUC) was recorded as headache intensity × headache duration. All patients gave written informed consent to participate in the study, which was approved by the regional ethics committee and conducted in accordance with the Declaration of Helsinki.
Clinical characteristics of tension-type headache patients

Median values with quartiles in parentheses are given.
The 33 patients described completed all recordings of headache intensity, myofascial tenderness and pressure pain thresholds throughout the study. Recordings of electrical pain thresholds were completed in 27 patients.
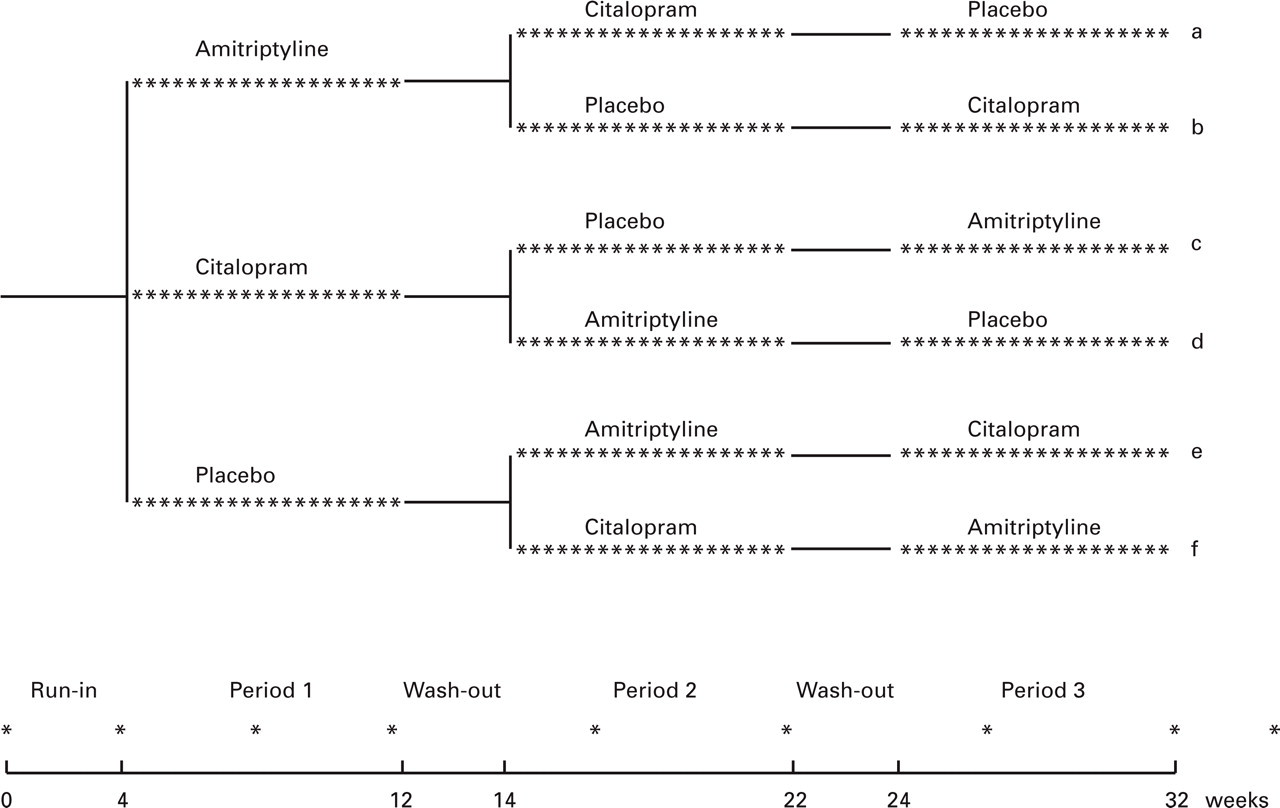
Study design of the 32-week, double-blind, placebo-controlled, three-way crossover trial. Each patient was randomly allocated to one of the six treatment sequences a–f. Clinical visits are indicated by ∗. Reproduced from (4) by permission of BMJ Publishing Group.
Methods
The recordings were performed in a standardized manner by the same investigator, a trained technician (H.A.), throughout the study. The patients were examined sitting in a dental chair with headrest. The examinations were performed in the last week of each of the three 8-week treatment periods, i.e. during treatment with 75 mg amitriptyline daily, during treatment with 20 mg citalopram daily and during placebo. The patients were examined irrespective of their headache state and they were not allowed to take analgesics on the days of examination.
Apparatus
An electronic pressure algometer (Somedic, Stockholm, Sweden) was used to measure pressure pain thresholds. The algometer has been described in detail elsewhere (23). A circular stimulation probe (0.50 cm2) and a pressure loading rate of 22 kPa/s were used.
Headache intensity
The actual headache intensity was recorded on a 100-mm visual analogue scale with endpoints designated ‘no pain’ and ‘unbearable pain’.
Myofascial tenderness
Tenderness of pericranial myofascial tissues was recorded according to the Total Tenderness Scoring system (11), which has previously proved to be reliable (24). Briefly, eight pairs of muscles and tendon insertions (frontal, sternocleidomastoid, masseter, temporal and trapezius muscles, coronoid and mastoid processes, and neck muscle insertions) were palpated. Palpation was performed with small rotating movements of the observer's second and third fingers. Pressure was sustained for 4–5 s. Prior to the study a palpometer (25) was used to train the observer to exert a palpation pressure of moderate intensity. Tenderness was scored on a four-point (0–3) scale and values from left and right sides were summed to a Total Tenderness Score (TTS) (maximum possible score = 48).
Pressure pain thresholds
Pressure pain thresholds were measured at the dorsum of the second finger (middle phalanx) and at a fixed point at the anterior part of the temporal region. Measurements were performed at the non-dominant side. The pressure pain detection threshold (PPDT) was defined as the pressure at which the sensation changed from pressure alone to a combination of pressure and pain. The pressure pain tolerance threshold (PPTO) was defined as the maximal pressure tolerated by the subject. The subjects indicated that the pain threshold was reached by pressing a hand-held button. The algometer display was thereby ‘frozen’, and the pressure was immediately released. Each threshold was calculated as the mean of five consecutive determinations performed with intervals of approximately 30 s.
Electrical pain threshold
Electrical pain thresholds were recorded as previously described (26). Briefly, a bipolar surface stimulation electrode was placed with the stimulation tips on either side of the left labial commissure. Electrical stimuli with a duration of 0.5 ms were delivered by a constant current stimulator (Counterpoint; Dantec, Copenhagen, Denmark). To minimize the variability caused by differences in the placing of the stimulation electrode, both the electrical sensory detection threshold (ESDT) and the electrical pain detection threshold (EPDT) were measured. ESDT was defined as the weakest stimulus detectable by the subject, and EPDT was defined as the weakest stimulus perceived as painful by the subject. The relative electrical pain threshold (REPT) was defined as EPDT −ESDT.
Statistical analysis
Results are presented as median values with quartiles in parentheses. As previously (27), patients were defined as responders if the AUC was decreased by at least 30% during the last 4 weeks of treatment with amitriptyline compared with placebo. Wilcoxon's rank sum test was used for paired observations, i.e. testing results within patients on different days. Mann–Whitney's U-test was used for unpaired observations, i.e. testing results between patients. Five percent was accepted as level of significance.
Results
Drop-outs
Recordings of tenderness and pressure pain thresholds were obtained in 33 of the 40 patients during all three treatments (Table 1). Six patients dropped out of the trial for various reasons (4) unrelated to the present study, while one patient did not want to participate in the present study during the trial. The recordings of electrical pain sensitivity were not completed in another six patients (five women and one man) due to technical problems (n = 2) and unwillingness to be subjected to electrical current (n = 4).
Modulation of area under the headache curve
Amitriptyline reduced AUC by 30% compared with placebo, which was highly significant, while citalopram had only a slight (12%) and insignificant effect (4). Although amitriptyline has clearly identifiable side effects, effective blinding was ensured (4) by the rather complicated three-way, crossover design.
Modulation of headache intensity by amitriptyline
On the day of examination, the actual headache intensity was significantly lower during treatment with amitriptyline 10 (0–22) mm than placebo 16 (0–37) mm, P = 0.04.
Modulation of myofascial tenderness by amitriptyline
The TTS was significantly lower during treatment with amitriptyline 8 (3–12) than placebo 11 (5–16), P = 0.01 (Fig. 2a). In responders to amitriptyline treatment (n = 19), TTS was significantly lower during treatment with amitriptyline 3 (2–11) than placebo 10 (2–13), P = 0.005. In non-responders (n = 14), there was no difference in TTS during treatment with amitriptyline 15 (8–25) and placebo 13.5 (7–25), P = 0.36. During amitriptyline treatment, responders had significantly lower TTS than non-responders, P = 0.003 (Fig. 2b).

(a) Total Tenderness Score (TTS) in 33 patients with chronic tension-type headache during treatment with amitriptyline 75 mg/day, citalopram 20 mg/day or placebo. ∗∗Significant difference between amitriptyline and placebo, P = 0.01. There was no significant difference between citalopram and placebo. (b) TTS during treatment with amitriptyline 75 mg/day in responders, i.e. patients who had at least 30% analgesic effect of amitriptyline (n = 19), and in non-responders (n = 14). ∗∗Significant difference between responders and non-responders, P = 0.003.
Modulation of overall pain sensitivity by amitriptyline
There were no significant differences between amitriptyline and placebo for any of the examined pain thresholds; PPDT in the finger 432 (336–606) kPa, P = 0.80; PPTO in the finger 918 (624–1138) kPa vs. 808 (712–1102) kPa, P = 0.28; PPDT in the temporal region 286 (218–398) kPa vs. 278 (212–398) kPa, P = 0.21; PPTO in the temporal region 544 (406–782) kPa vs. 531 (405–729) kPa, P = 0.31; or REPT 4.0 (2.1–6.0) mA vs. 3.6 (1.9–5.4) mA, P = 0.43 (Fig. 3).

Pain thresholds to mechanical (n = 33) and electrical (n = 27) stimuli in patients with chronic tension-type headache during treatment with amitriptyline 75 mg/day, citalopram 20 mg/day or placebo. There were no significant differences in pain thresholds between the two active drugs and placebo. PPDT and PPTO indicate pressure pain detection and tolerance thresholds. REPT indicates relative electrical pain threshold. Finger indicates index finger. Temporal indicates temporal region. Comm indicates labial commissure.
Effects of citalopram
Compared with placebo, there were no significant effects of citalopram on either actual headache intensity 19 (0–25) mm, P = 0.95; TTS 10 (5–17), P = 0.75 (Fig. 2a); PPDT in the finger 434 (308–548) kPa, P = 0.59; PPTO in the finger 844 (688–1146) kPa, P = 0.12; PPDT in the temporal region 276 (212–406) kPa, P = 0.61; PPTO in the temporal region 571 (438–735) kPa, P = 0.06; or REPT 3.8 (2.0–5.3) mA, P = 0.73 (Fig. 3).
Discussion
Only few previous studies have investigated the effect of tricyclic anti-depressants on myofascial tenderness or pain thresholds in patients with chronic pain conditions. One study found that amitriptyline reduced both pain and tenderness in patients with fibromyalgia (28), while three studies found that amitriptyline reduced pain but not tenderness in these patients (29–31). Göbel et al. (9) found that the pain sensitivity to strong, but not to mild and moderate, mechanical stimuli was reduced during treatment with amitriptyline but not during placebo in patients with chronic tension-type headache. No information about a possible difference in pain sensitivity between amitriptyline and placebo treatments was given. Grace et al. (32) found no effect of amitriptyline on either joint pain or joint tenderness in patients with rheumatoid arthritis, while Sharav et al. (33) found that amitriptyline reduced clinical pain but not experimentally induced heat pain in patients with chronic facial pain. Sindrup et al. (34) demonstrated that treatment with the tricyclic anti-depressants clomipramine and desipramine did not affect heat pain thresholds in patients with diabetic neuropathy, and Chapman & Butler (35) found that chronic dosing with the tricyclic anti-depressant doxepin did not modulate pain sensitivity in healthy controls. Poulsen et al. (36) demonstrated that acute dosing of the tricyclic anti-depressant imipramine influenced some but not other pain modalities in healthy controls during experimental pain. These results seem conflicting, but are in fact incomparable, since the analgesic effect of the tricyclic anti-depressants may differ between the various drugs, between acute and chronic dosing, between patients and healthy controls, and between the various pain conditions (2).
The present study demonstrates that amitriptyline reduces the degree of pericranial myofascial tenderness in patients with chronic tension-type headache. The reduction in tenderness was of the same magnitude (27%) as the reduction in the area under the headache curve (30%) in the last 4 weeks of treatment with amitriptyline compared with placebo (4). Explanatory analyses demonstrated that tenderness was reduced considerably in patients who had the analgesic effect of amitriptyline, while tenderness was unaltered in non-responders, i.e. the reduction in tenderness could be ascribed solely to the group of patients who responded to amitriptyline treatment.
Amitriptyline did not modulate the sensitivity to pressure or electrical pain stimuli applied to various parts of the body, indicating that pain sensitivity in general was unaffected. The lack of significant difference in overall pain sensitivity could be due to lack of power. However, while there is a large inter-individual variability in the measurement of pressure pain thresholds, the intra-individual variability is relatively low (23). Thus, it has been estimated that 10 patients is sufficient to detect a 30% change in pressure pain thresholds in paired studies (23). The 33 patients included in the present study therefore provide sufficient power to conclude that pressure pain thresholds were largely unaffected by treatment with amitriptyline. It may be somewhat surprising that pain detection and tolerance thresholds were unaffected by amitriptyline, contrary to tenderness. One possible explanation could be that amitriptyline mainly modulates low-threshold mechanosensitive afferents, which have been hypothesized to mediate the major part of the increased tenderness in patients with myofascial pain and segmental central sensitization (see below and (16, 49)). Thus, the present data indicate that amitriptyline reduces nociception from myofascial tissues rather than pain sensitivity in general.
The present finding of a differential effect of amitriptyline and citalopram on myofascial tenderness is in agreement with our previous finding of a prophylactic effect of amitriptyline but not of citalopram in chronic tension-type headache (4). This further supports that the tricyclic anti-depressants are more effective than the selective serotonin reuptake inhibitors for the treatment of chronic pain conditions (37, 38), and that the analgesic effect of amitriptyline cannot solely be explained by the inhibition of serotonin reuptake. Thus, while citalopram is an extremely specific serotonin reuptake inhibitor (39), amitriptyline also has several other effects, e.g. effects on reuptake of noradrenaline as well as effects on serotonergic (40), adrenergic (41), cholinergic (40), and histaminergic (34) receptors. Of these effects, especially inhibition of noradrenaline reuptake (42, 43) and the effects on various serotonin receptor subtypes (44, 45) have been considered important. Moreover, amitriptyline potentiates the effect of endogenous opioids (46, 47). Most interesting, it has been demonstrated that amitriptyline may act as a N-methyl-
Clinical studies indicate that central sensitization at the level of the spinal dorsal horn/trigeminal nucleus induced by prolonged nociceptive input from pericranial myofascial tissues is of major importance for the conversion of episodic into chronic tension-type headache and for the maintenance of this chronic pain disorder (15–17). This segmental central sensitization probably contributes significantly to the increased pericranial tenderness in these patients, while the slightly increased general pain sensitivity presumably plays a minor role (49). Therefore, it is most likely that the reduction of tenderness exerted by amitriptyline, at least in part, is caused by a segmental action in the central nervous system, probably at the level of the spinal dorsal horn/trigeminal nucleus. This was supported by a recent study in which the nitric oxide synthase inhibitor L-NG-methyl-arginine hydrochloride (L-NMMA) was administered to patients with chronic tension-type headache with the aim of reducing central sensitization. The study showed that L-NMMA not only significantly reduced headache (50), but also significantly reduced pericranial muscle hardness and tended to reduce pericranial tenderness in these patients (51).
Possible peripheral mechanisms leading to tenderness and myofascial pain include activation or sensitization of nociceptive nerve endings by chemical mediators and dysfunction of afferent nerves. Until recently, there was no evidence of a peripheral anti-nociceptive action of amitriptyline. On the contrary, previous studies of the analgesic effect of tricyclic anti-depressants in animal models of chronic pain (52–54) and in human experimental pain (36) uniformly indicated that this effect was due to an action in the central nervous system and not in the periphery. This has been challenged by recent studies showing a peripheral analgesic effect of amitriptyline in addition to its central effect. An electrophysiological study by Su & Gebhart (55) points towards a direct analgesic effect of tricyclic anti-depressants on afferent nerves, while studies by Esser & Sawynok (56) and Sawynok et al. (57) indicate that interaction with endogenous nociceptive mediators in peripheral tissues plays a role in the anti-nociceptive effect of amitriptyline. Thus, it is possible that a combined action on peripheral and central sites may be of importance for the analgesic effect of amitriptyline in chronic myofascial pain.
The present study does not reveal whether the reduction in myofascial tenderness induced by amitriptyline is a consequence or a cause of the reduction in headache. However, it is most likely that myofascial factors are of primary importance for the development and maintenance of tension-type headache, as previously discussed (15–17). We therefore suggest that the analgesic effect of amitriptyline in chronic tension-type headache can be ascribed primarily to the reduction in myofascial tenderness.
In conclusion, the present study demonstrates that amitriptyline reduces myofascial tenderness and headache in patients with chronic tension-type headache. Furthermore, the results indicate that amitriptyline elicits its analgesic effect in chronic myofascial pain by reducing the transmission of painful stimuli from the myofascial tissues rather than by reducing overall pain sensitivity. On the basis of previous and present results, we suggest that the reduction of myofascial pain exerted by amitriptyline may be caused by reduction of segmental central sensitization in combination with a peripheral anti-nociceptive action.
Footnotes
Acknowledgements
We would like to thank Mrs Hanne Andresen for skilful technical assistance and Professor Jes Olesen and Dr Messoud Ashina for valuable comments during the preparation of the manuscript. The study was supported by the Lundbeck Foundation (145/91).
