Abstract
Introduction
Headache during and/or after coronary intervention is common but has received little attention from cardiologists and neurologists. The purpose of this study was to investigate the incidence, risk factors, and possible mechanism of coronary intervention-related headache.
Methods
Using a prospective observational design, we identified consecutive patients with coronary intervention from May 2020 to August 2020. Patients were followed up with questionnaires immediately after coronary intervention and 24 h, 72 h, 1 week and 2 weeks after the intervention.
Results
In total, 94 patients were enrolled, and 71 patients ultimately completed the 2-week follow-up. Among 71 patients, headache developed during and/or after coronary intervention in 18 (25.4%) patients. Two different types of headache related to coronary intervention were observed: One during and another after coronary intervention. Headache characteristics are described in detail. A history of previous headache was an independent risk factor for coronary intervention-related headache (p < 0.01).
Conclusions
Coronary intervention-related headache has an incidence of 25.4%, and previous headache history was an independent risk factor. Moreover, considering that there are no relevant diagnostic criteria, it is suggested that the definition of coronary intervention-related headache should be established in the International Classification of Headache Disorders.
Keywords
Background
Coronary intervention (CI) is an invasive diagnostic and therapeutic technique that is very useful for heart diseases and usually includes coronary angiogram (CAG) and percutaneous coronary intervention (PCI). The complication rate of cerebral digital subtraction angiography (DSA) has been reported to be 0.5–3.0%. Embolic or haemorrhagic stroke, seizures, puncture site-related problems, contrast agent-related problems and headache have been reported to occur in relation to cerebral DSA (1,2). Headache holds an important place among the complications of cerebral angiography, and its incidence varies between 0.3–51% (3–5). For example, 6.7.1 Headache attributed to an intracranial endarterial procedure and 6.7.2 Angiography headache have been included in the third edition of the International Classification of Headache Disorders (ICHD-3) (6). As with cerebral angiography, CI can lead to some neurological complications such as contrast-induced encephalopathy (7,8). In clinical practice, we found that many patients who underwent CI complained of headache. Through further investigation, we found that CI-related headache was common in cardiovascular medicine but rarely attracted attention from cardiologists and neurologists. To date, there is no definition of this type of headache in ICHD-3. The purpose of this study was to investigate the incidence of CI-related headache as well as its risk factors and possible mechanism.
Methods
This prospective observational study was approved by the Research Ethics Committee of the Chinese PLA General Hospital. All of the patients provided written informed consent.
Endovascular procedures were performed by senior interventional cardiologists with the assistance of a resident. Moreover, operative schemes were designed by three to four interventional cardiologists prior to surgery and were immediately altered according to the findings obtained during the procedures. Clinical records were analysed, and demographic information (age, sex, education level, and past history) and operation information (operation indications, types of interventional operations, types of contrast agent, quantity of contrast agent and time of exposure to X-ray during operations) were collected for each case.
A detailed headache questionnaire was designed by our team based on the ICHD-3 diagnostic criteria and included questions soliciting detailed information about history of headache, headache start time and duration, with predefined time intervals. Other headache-related information, such as headache intensity, quality, location, and associated symptoms, was also investigated in detail. Intensity was assessed with a numeric rating scale (NRS), which is a segmented numeric version of the visual analogue scale (VAS), in which the patients select a whole number from 0 to 10 that best reflects their pain level (9). After completing the informed consent process, patients were asked to report the status of headaches at each time point. Relevant information was collected through a face-to-face questionnaire survey before endovascular treatment. Then, the patients were investigated again immediately after the procedure and 24 h, 72 h, 1 week and 2 weeks after the procedure. If discharged, patients were telephoned to follow up on their headaches. It is well known that contrast injection frequently causes a temporary sense of heat in the head and face called “flushing” (10). In the immediate postoperative investigation, all of the patients were informed about the possibility of “flushing” after the injection of contrast media and were instructed to differentiate this phenomenon from a headache. During follow-up, if a patient with a previous headache reported a headache attack, the characteristics of headache (such as headache duration, quality, location, and associated symptoms) were analyzed by two independent neurologists to determine whether the headache type was similar or different from the previous headache. If the two neurologists have different opinions on the diagnosis, a third neurologist will make the judgment.
Consecutive patients admitted to the Cardiovascular Department of the First Medical Center of PLA General Hospital from May 2020 to August 2020 were selected. The inclusion criteria were: a) being older than 18 years of age and b) having interventional surgery, such as CAG or PCI, under local anaesthesia. The exclusion criteria were as follows: a) having had a headache occurring within 24 h prior to the procedure, b) renal failure requiring dialysis, c) severe organ failure, unconsciousness, or endotracheal intubation; and d) refusal to complete the questionnaire or follow up regarding the questionnaire.
CI included CAG and PCI, as well as routine procedures using a 6 F catheter (three patients used a 7 F catheter); local anaesthesia from the right radial artery (65 patients) or the right femoral artery approach (six patients); intraoperative use of heparin and a small amount of nitroglycerine; contrast agent using the non-ionic contrast agent Ultravist or Iodixanol; and postoperative routine administration of 1000 ml of intravenous hydration.
Statistical analysis
Statistics were analysed with the help of IBM SPSS statistics software, version 25.0. Categorical data are reported as numbers and percentages, while continuous data are reported as the mean ± standard deviation (SD). Relationships between categorical variables were analysed by Fisher’s exact test or Pearson’s chi-square test. Hypotheses on differences in means between groups were tested by the independent samples t test. Using a two-tailed testing, p < 0.05 was considered statistically significant.
Results
In total, coronary intervention was performed in 94 consecutive patients. One had headache before interventional surgery, two were excluded due to renal failure, one refused to participate in the investigation, and one was unable to participate in the investigation after intubation. Fourteen patients were lost to follow-up. Since the vast majority of patients with angina and myocardial infarction require vasodilators to dilate the coronary arteries, which can also dilate the intracranial vasculature and cause headaches, if patients reported that each headache attack followed the vasodilator, and the headache was relieved when the vasodilator was discontinued, we believed that the headache was directly caused by the vasodilators, rather than by the operation. In our study, there were 80.3% (57/71) patients who used nicorandil or nitrates regularly over a long period of time after the procedure. During the 2-week follow-up, four patients developed 8.1.1.1 Immediate NO donor-induced headache (three patients after the use of nicorandil and one patient after the use of nitrates) (6). In these four patients, mild to moderate intensity headaches had developed within 1 h after each use of nicorandil or nitrates, and headaches had resolved within several hours. All four patients complained of medication-related headaches and refused to take these medications, and the headaches did not recur after they stopped taking them. Since other patients who regularly took nicorandil or nitrates for a long period of time did not experience this time and dose-dependent vasodilator-associated headache, we believe that the headache in these four patients was caused by nicorandil or nitrates, while the other patients’ headaches were not. Therefore, these four patients were not included in the analysis. There were 77 procedures performed on 71 patients, including six patients who underwent two procedures. Only the last procedure was included in the analysis. Ultimately, 71 patients completed the 2-week follow-up (Figure 1).

Study enrolment.
Of the 71 patients, 25.4% (18/71) were female, and 74.6% (53/71) were male; their mean age was (63.3 ± 12.3) years. Eighteen of 71 (25.4%) patients developed headache within 2 weeks. Of the 18 patients with CI headache, 72.2% (13/18) had no history of headache, 11.1% (2/18) had different headache characteristics from their previous headaches, and 16.7% (3/18) had headaches similar to their previous headaches. Of the 18 patients with CI headaches, 55.6% (10/18) were male, 44.4% (8/18) were female, and 33.3% (6/18) of patients had more than one headache attack.
During the procedure, headache was seen in five patients (Patients 1–4, 17), and none of them had a history of headache. Within the first 24 h after the procedure, headache was seen in four patients (Patients 5–7, 13). Patient 6 had a history of headache, but his headache was different from the previous headache. Between days 1 and 3 after the procedure, headache was seen in seven patients (Patients 8–10,13, 14, 17 and 18). Patients 8, 14, and 18 had a history of headache. Among them, the headaches of Patients 14 and 18 were similar to their previous headaches, while the headache of Patient 8 was different from the previous headache. Between days 4 and 7 after the procedure, headache was seen in five patients (Patients 14–18). Finally, between days 8 and 14 after the procedure, headache was seen in four patients (Patients 11, 12, 16 and 18). Patients 13–16 had two headache attacks, Patient 17 had three, and Patient 18 had five (Tables 1 and 2).
Characteristics of headache in patients with CI headache and history of primary headache.

aPatients 13–16 had two headache attacks, Patient 17 had three, and Patient 18 had five.
bRefers to the time after the beginning of the procedure (Patients 1–4, Patient 17-1st); in others, refers to the time after the end of the procedure.
cF/T/O/P/G = Frontal/Temporal/Occipital/Parietal/Generalized
dT/P/S/B/O = Throbbing/Pressure-like/Stabbing/Burning/Other
CI: coronary intervention; EH: exercise headache; M: migraine; MA: migraine with aura; TTH: tension-type headache; HA: headache; VAS: visual analogue scale.
Headache in patients with CI headache and the relationship with the history of primary headache.

CI: coronary intervention; HA: headache; N/A: not applicable.
Headache that occurred during the procedure started at a mean time of (15.4 ± 13.6) min after the beginning of the intervention, lasted for an average of (28.0 ± 20.8) min, and the median VAS was 4. The headache occurred on bilateral sides (60%), on unilateral sides (40%), in the temporal region (80%), parietal region (40%), occipital region (20%) and full head (20%) with pain associated with throbbing (60%) or pressing (40%) and accompanied by nausea (20%), photo- (40%) or phonophobia (20%) or visual aura (40%) (Table 3).
Characteristics of CI headache related to coronary intervention.
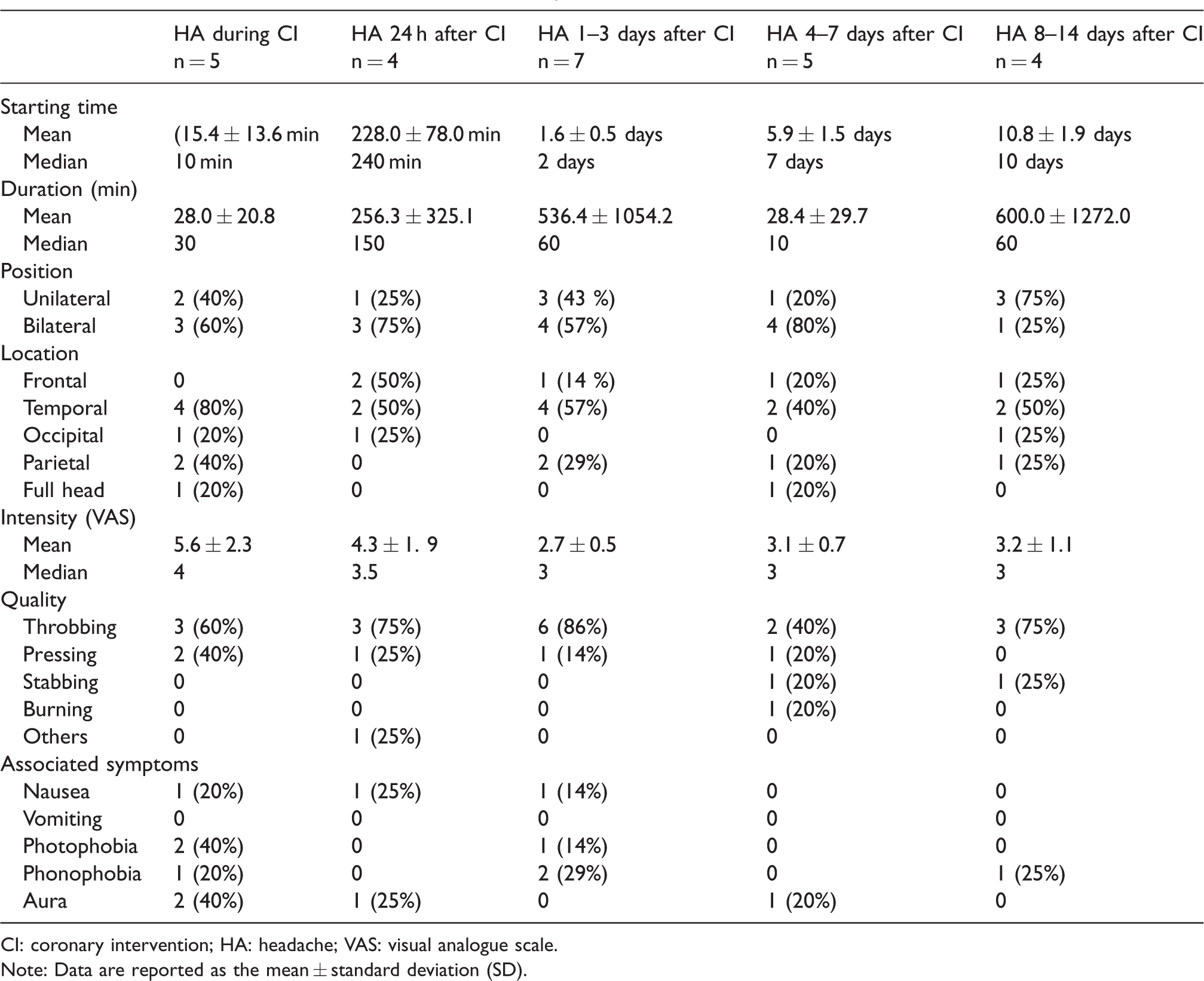
CI: coronary intervention; HA: headache; VAS: visual analogue scale.
Note: Data are reported as the mean ± standard deviation (SD).
Headache that occurred during the first 24 h after the procedure started at a mean time of (228.0 ± 78.0) min after the end of the intervention, the mean duration was (256.3 ± 325.1) min, and the median VAS was 3.5. The headache was 75% on bilateral sides and 25% on unilateral sides, was 50% located in the frontal, 50% in the temporal and 25% in the occipital areas, 75% were with a throbbing sensation, 25% with pressing and could be accompanied by nausea (25%) and visual aura (25%) (Table 3).
Headache that occurred 1–3 days after the procedure began at a mean time of (1.6 ± 0.5) days after the intervention, lasted for an average of (536.4 ± 1054.2) min, and the median VAS was 3. The headache was 57% on bilateral sides and 43% on unilateral sides, was 57% located in the temporal areas, 29% in the parietal areas and 14% in the frontal areas, 86% were with a throbbing sensation, 14% with pressing, and it could be accompanied by nausea (14%) and photo- (14%) or phonophobia (29%) (Table 3).
Headache that occurred 4–7 days after the procedure began at a mean time of (5.9 ± 1.5) days after the intervention, lasted for an average of (28.4 ± 29.7) min, and the median VAS was 3. The headache was 80% bilateral and 20% unilateral, was 40% located in the temporal areas, 20% in the frontal areas, 20% in the parietal areas and 20% in the full head, 40% were with a throbbing sensation, 20% with pressing, 20% with stabbing, 20% with burning and 20% were accompanied by visual aura (Table 3).
Headache that occurred 8–14 days after the procedure began at a mean time of (10.8 ± 1.9) days after the intervention, lasted for an average of (600.0 ± 1272.0) min, and the median VAS was 3. The headache was 25% on bilateral sides and 75% on unilateral sides, was 50% located in the temporal areas, 25% in the frontal areas, 25% in the parietal areas and 25% in the occipital areas, 75% were with a throbbing sensation, 25% with stabbing and 25% were accompanied by phonophobia (Table 3).
During hospitalization, most patients with headaches did not actively complain about their headaches to their doctors and nurses, nor did they request treatment for their headaches. Only one patient complained of headache during hospitalization, which was relieved after taking a compound acetyl-salicylic acid tablet half an hour later. Another patient also took a compound acetyl-salicylic acid tablet when experiencing a headache after discharge, and none of the other patients with headaches sought analgesics for relief during the 2-week follow-up.
Patient demographics (n = 71) are shown in Table 4. A history of primary headaches was present in six patients (8.5%) (two migraine, two tension-type headaches, one exercise headache, one headache to cerebral ischaemic event). Patients who had a history of primary headaches were more likely to have CI headaches (p < 0.01). No statistical differences were found in sex (p = 0.06), age (p = 0.66), education (p = 0.37), tobacco history (p = 0.54) and alcohol use (p = 0.23), hypertension (p = 0.84), diabetes (p = 0.54), cerebral infarction (p = 0.67), hyperlipidaemia (p = 0.51) or other aspects in the headache and no-headache groups. There was no significant difference between the two groups in type of contrast agent used (p = 0.12), type of intervention (p = 0.82), quantity of contrast agent used (p = 0.11), X-ray exposure time during intervention (p = 0.45), previous history of intervention (p = 0.79) or indication for intervention (p = 0.20).
Potential risk factors for related to CI headache.

HA: headache; CAG: coronary angiogram; PCI: percutaneous coronary intervention; AMI: acute myocardial infarction; CI: coronary intervention.
Note: Data are reported as the mean ± standard deviation (SD).
Discussion
In our study, the incidence of headache was 25.4% (18/71) among 71 patients who underwent CIs during the 2 weeks of follow-up. Among the 18 patients with headaches, more than half (88.9%, 16/18) had headaches within 1 week after the procedure; among them, 77.8% (14/18) had headaches within 3 days of the procedure, but 22.2% (4/18) still had recurrent headaches in the second week.
Similar to the study about cerebral angiography headache by Aktan et al. (10), we also observed two different types of CI headaches in our patients: One during and one after the procedure. The headache during the procedure lasted less than 60 min, was of medium intensity, with a pressure-like or throbbing character, and might be accompanied by photo- or phonophobia or visual aura. The second type of headache we observed has developed several hours to 14 days after coronary artery intervention, lasted less than 24 h and was of a mild to moderate intensity, also with a pressure-like or throbbing character, and might accompanied by photo- or phonophobia or visual aura.
According to our survey, coronary interventional headaches are very common in cardiology; however, no cohort studies of coronary interventional related headache have been reported before, and there is no classification of this type of headache in ICHD-3. Cardiologists and neurologists lack diagnostic criteria to follow when facing such type of headache, which brings difficulties to diagnosis. We hope that our research will improve both cardiologists and neurologists’ ability to correctly diagnose and treat this type of headache. So we suggest that the diagnostic criteria for headache associated with coronary intervention in Table 5. At present, since our study was the only study on CI headache, the diagnostic criteria presented by us may contain bias. Further researches are needed to refine and modify the diagnostic criteria.
Possible diagnostic criteria for coronary interventional associated headache.

Obermann reported a case of unilateral cerebral oedema after coronary angiography in a patient with a history of ipsilateral migraine, supporting that a previous history of headache is a risk factor after CI (8). A previous history of headache is a common risk factor for CI-related headache as was demonstrated in our study, as well as in previous studies of post-cerebral DSA headache (3,11). The possible mechanism is that some individuals with a previous history of headache have lower thresholds for trigeminal activation (11). Interestingly, none of the five patients who developed headache during the procedure had a history of headache.
Female sex is regarded as a risk factor for post-cerebral DSA headache, as supported in some studies but denied in others (3,11–13). In our study, the incidence of CI headache in women (44.4%, 8/18) was higher than in men (18.9%, 10/53), though it was not statistically significant. A higher level of education was a risk factor for cerebral angiography-associated headache, which could be due to patients with a higher level of education being more nervous, anxious, and/or stressed about undergoing cerebral DSA than other patients (14). However, in our study, sex and education were not risk factors for CI-associated headache.
In our study, there was no statistically significant difference between headache occurrence and exposure time to X-rays. For cerebrovascular radiography-associated headaches, studies have shown that neurological complications are more common when cerebral angiography is longer than 10 min (2), but in our study, surgeries that required less than 10 min were rare. Of the five patients who had headache during CIs, four underwent stent implantation, and one underwent coronary angiography. Therefore, we imagine that the incidence of headaches after balloon expansion or coronary stenting in patients is higher than that after coronary angiography, but we did not find that the methods of operation had a statistically significant effect on the incidence of headaches, which might be caused by microembolism or endothelial injury from direct vessel manipulation (15).
A study showed that the frequency of headache after carotid internal stenting was higher than that after cerebral angiography (13). We assumed that the high osmotic pressure caused the intravascular contrast agent to have higher osmotic pressure and to dilate intracranial arteries, causing headaches; however, in our study, two different osmotic pressures of contrast agent for the incidence of the headache groups were not significantly different, which might be due to the relatively small amount of contrast agent entering the brain each time, and it was quickly metabolized, rendering it insufficient to alter the plasma osmotic pressure. There was no significant relationship between contrast media volume and headache occurrence in our study, consistent with a study of cerebral angiography by Leffers et al. (1). This result suggested that headache was independent of contrast agent concentration.
The cause of CI-related headache is unknown. We speculate that the mechanism might be as follows. 1. During the procedure, some of the nitroglycerine enters the cerebral blood vessels and causes intracranial blood vessels to dilate, thus causing headache attacks. 2. Studies have shown that iodine contrast agents can induce vasoconstriction in the human body (16,17). The vasoconstriction and vasodilation change may lead to coronary interventional-related headache. 3. Angina attacks can present as headache symptoms, namely cardiac cephalalgia (18–20). 4. During the procedure, it has been observed that electrocardiographic (ECG) changes can be continuously observed, including ST-segment changes, T-wave low levels, and inverted changes, and these changes disappear after 30–45 sec (21). However, in our research, in one of the five patients who developed headaches during the procedure (Patient 17), ECG was checked 1 h after the operation and still showed the T wave change (I, II, III, aVF, aVL, V3–V6) inversion. We believe that the ECG changes were due to contrast agents affecting myocardial microcirculation, rather than contrast agents or catheters blocking coronary blood flow. Similarly, contrast agent enters the brain and leads to microcirculation ischaemia, further causing brain internal environment disorders and then causing headache. 5. In the study of Gil-Gouveia, female patients were more likely to have headaches after cerebral angiography (3). In our study, the incidence of CI headache in women (44.4%,8/18) was higher than in men (18.9%,10/53). We speculated that hormones might be a mechanism leading to CI headache similar to cerebral angiography headache. 6. The incidence of headache that we report is similar to the incidence of cerebral angiography headache reported by Aktan et al. (10). It is conceivable that the catheter or the contrast material activated platelets or damaged the vascular endothelium, thereby resulting in the release of vasoactive substances such as nitric oxide (NO) or serotonin (5-HT) (5).
Strengths and limitations of the study
To our knowledge, this study is the first study on the association of headache incidence during and/or after CI, with important clinical implications. It was a prospective study, and the patients were followed up several times until 14 days after the procedure. Additionally, it was an observational study, in which participants were not informed of the possibility of headache before the procedure to avoid causing psychological stress to patients and thus affecting the incidence of headache. In our study, relevant procedure information was collected in detail, including the type of contrast agent used, the quantity of contrast agent used, and the time of exposure to X-ray. One limitation was that, among 71 patients, the ratio of women to men was almost 1:3, which might have caused bias in the results. A relatively small number of patients and CI-related headache based on only 18 patients would also be a major limitation of this study. More research is needed to further study CI-associated headache.
Conclusions
Headache during and/or after CI is common in patients undergoing CI in the Department of Cardiology. This study reported for the first time that the incidence of CI-related headache was 25.4% and that 77.8% of headaches occurred within 3 days after CI. This finding might have important clinical guiding significance for cardiologists and neurologists. In the ICHD-3, there is no definition of this type of headache. Thus, we hope to improve the classification of headache in the future.
Clinical implications
There are two different types of headache in relation to coronary intervention: one during and another after coronary intervention. Both headaches are more frequent in patients with a history of prior primary headache, Headache characteristics can be similar to or different from the premorbid headache. There are no relevant diagnostic criteria, and doctors lack diagnostic criteria to follow when facing such headache, which brings difficulties to diagnosis. We hope that our research will improve doctors’ ability to correctly diagnose and treat this type of headache. It is suggested that the definition of coronary intervention-related headache should be established in ICHD.
List of abbreviations
CI: coronary intervention; ICHD-3: International Classification of Headache Disorders, 3rd edition; CAG: coronary angiogram; PCI: percutaneous coronary intervention; DSA: digital subtraction angiography; VAS: visual analog scale; PLA: People’s Liberation Army; SD: standard deviation; HA: headache; TTH: tension-type headache; N/A: not applicable; EH: exercise headache; M: migraine; MA: migraine with aura; AMI: acute myocardial infarction; NSTEMI: non-ST-segment elevation myocardial infarction; STEMI: ST-segment elevation myocardial infarction.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (grant 2018YFC0117003), Chinese PLA General Hospital 2019 Military Medicine Transformation Project (grant ZH19002), National Natural Science Foundation of China (grants 81771180, 81671077, 81771200, 81901134 and 81901145, 82071226), and Beijing Natural Science Foundation Essential Research Project Z170002.
