Abstract
Background
CGRP plays a key role in the transmission and modulation of nociceptive signals and is a critical component in the pathogenesis of migraine.
Objective
To assess saliva as a substrate to measure CGRP by comparing interictal levels in patients with episodic migraine and controls; and to evaluate CGRP’s temporal profile during migraine attacks.
Methods
This prospective observational pilot study included young women with episodic migraine and healthy controls. We monitored salivary CGRP-like immunoreactivity (CGRP-LI) during 30 consecutive days and during migraine attacks. We considered six timepoints for the analysis: interictal (72h headache free), preictal (PRE-24h before the attack), ictal (headache onset, after 2h, after 8h), postictal (POST-24h after the attack). CGRP levels were quantified by ELISA.
Results
44 women (22 with episodic migraine, 22 healthy controls) were recruited. Differences in interictal salivary levels of CGRP between patients and controls (Me [IQR]: 98.0 [80.3] (95% CI 56.6, 124.0) vs. 54.3 [44.0] (95% CI 42.2, 70.1) pg/mL, p = 0.034) were found. An increase in CGRP levels during migraine attacks was detected (pre:169.0 [95% CI 104.2–234.0]; headache onset: 247.0 [181.9–312.0]; after 2h: 143.0 [77.6–208.0]; after 8h: 169.0 [103.5–234.0], post: 173.0 [107.8–238.0]). Patients were classified as having CGRP-dependent (79.6%) and non-CGRP dependent migraine attacks (20.4%) according to the magnitude of change between preictal and ictal phase. Accompanying symptoms such as photophobia and phonophobia were significantly associated to the first group.
Conclusions
Salivary CGRP-LI levels, which interictally are elevated in episodic migraine patients, usually increase during a migraine attack in the majority of patients. However, not every attack is CGRP-dependent, which in turn, might explain different underlying pathophysiology and response to treatment.
Introduction
Migraine is a highly prevalent and a disabling neurological disease (1) and, so far, specific biomarkers have yet not been validated, which reflects its complexity and dynamic character (2–4). However, the most studied molecule is the calcitonin gene related peptide (CGRP), which has a clear implication in migraine pathophysiology (5,6), underpinned by the arrival of effective therapies targeting CGRP pathways (7). CGRP is implicated in the development of neurogenic inflammation and it is upregulated in conditions of inflammatory and neuropathic pain (8,9), and it has been hypothesized as a potential diagnostic (10–12) and treatment predictive biomarker in migraine (13–16).
Saliva as a substrate to study biomarkers is a worthwhile approach because its collection is non-invasive and it allows the monitoring of neuropeptides because samples can be repeatedly obtained from subjects (17). The feasibility of CGRP detection in human saliva has been previously demonstrated and used as a marker of trigeminovascular activation in migraine (10,11,14,18–20).
Variability between CGRP levels across the studies is probably due to methodological differences such as type of matrix, collection techniques, type of assay, time of sampling and heterogeneous study population. Standardized procedures in collection and analysis are mandatory in order to be able to use them as a valuable resource to gain scientific information in the migraine field. Therefore, the objectives of this study were: a) to compare interictal salivary CGRP levels between episodic migraine (EM) patients and healthy controls (HC) and, b) to assess the temporal profile of salivary CGRP during migraine attacks.
Methods
Participants and study design
This is a prospective longitudinal pilot study. Patients were recruited from the outpatient headache clinic and carefully interviewed by a headache specialist. Recruitment period was from March 2018 to November 2019.
Patients were women between 18–65 years old fulfilling the criteria for migraine with or without aura, according to the International Classification of Headache Disorders (ICHD-3) (21). Specifically, they had to report between 1–6 migraine days per month (d/mo). Participants with a smoking habit; medical diagnosis of anxiety/depression; medical diagnosis of chronic pain disorders; subjects taking medication affecting central nervous system; subjects taking migraine preventive medication in the past year prior to the study or history of any medical condition that could alter saliva content were excluded. Healthy controls (HC) were age-matched women with no personal or family history of migraine or headache, excluding sporadic tension-type headache, recruited from residency training program. At the screening visit, demographical and clinical data were collected.
All participants were given detailed verbal and written instructions for saliva collection. They were provided with appropriate material for saliva collection at home including pre-labelled tubes; diaries to register sample collection time and menstrual cycle; questionnaires to record migraine attack characteristics such as pain intensity and duration, accompanying symptoms and acute treatment used. During the study, subjects treated their migraine attacks as usual with an approval of the investigator at the initial visit (triptans and non-steroidal anti-inflammatory drugs were allowed). No migraine attack was treated before collecting the first sample of each attack.
Saliva collection
HC and EM patients collected saliva samples consecutively during a 30-day period (baseline samples). EM patients collected three extra saliva samples during the migraine attack (painful condition samples): headache onset, after 2h and after 8h.
Saliva collection was carried out with the resting unstimulated whole saliva method (17,19) using the following step-by-step indications:
To collect saliva at the same time of the day, early in the morning, fasting condition. Not to eat, drink or brush their teeth before collection. To rinse their mouth with water, discarding initial saliva in order to avoid contaminated saliva with debris. To collect the fluid by spitting into a sterile tube of 5 mL of polypropylene material for 5 minutes with a minimum quantity of 3 mL. Each tube was used once only. Not to use citric acid since it could degrade CGRP.
After collection, all baseline saliva samples were kept in the freezer of participants at −18°C degrees. At the end of the study, the samples were carried to the laboratory on ice in order to avoid thawing. All samples were stored in the laboratory freezer at −80°C degrees.
Plasma collection
Plasma samples were also collected from each participant on day 1 in order to make a saliva-plasma correlation. It was noted if it was a free pain period (outside of an attack). Samples were collected from the subject’s antecubital vein between 8–10 am and transferred to EDTA coated 10.8 mg tubes (BD Vacutainer System, K2E). All samples were centrifuged at 3500 rpm at 4°C for 15 min and supernatants were immediately stored at –80°C.
CGRP extraction
Saliva samples were thawed slowly at room temperature and then placed on ice. The samples were centrifuged for 20 minutes at 3500 rpm −4°C, and the supernatant was aliquoted into 1.5mL centrifuge sterile and polypropylene Eppendorf tubes and stored at −80°C or immediately analyzed. Prior to Enzyme-linked Immunosorbent Assay (ELISA), samples were thawed slowly and then centrifuged for 5 minutes at 3500 rpm at room temperature to pellet cellular debris. −80°C storage helped avoid protein degradation and denaturalization. Moreover, to minimize degradation of unstable antigens, samples were kept in ice, and, repeated freeze-thaw cycles were avoided.
Assay protocol
Quantitative determination of plasma and salivary CGRP was measured by using human enzyme linked immunosorbent assay (ELISA) kits (Cusabio, detection range: 1.56–100 pg/ml, minimal detectable dose: 0.39 pg/ml). The assay was performed according to specification of the manufacturer. Intra-assay precision and inter-assay precision is declared with a coefficient of variation (CV) of <8%, respectively <10%. Duplicate measurements were performed for each sample. CGRP concentrations were determined from calibration curves using a 4PL fitting as implemented in Analysis software Gen 5 resulting in a fit with R2 > 0.99 in every case. The final CGRP level of each sample was calculated as the average of the two measurements. An internal validation of the test was performed, ensuring that the ELISA assay used was reliable (quality control was included in the kit). CGRP concentration from immunoassay procedure was corrected by inter and intra-assay coefficients of variability for each ELISA plate.
Regarding saliva samples we found important: to dilute samples 1:10 with sample dilutent; to homogenize the sample with the dilutent (repeated up-down cycles) and to discard blood contaminated samples. In plasma samples, it is important to discard lipemic and hemolyzed samples in order to reduce the effect on the ELISA results. Those samples were not diluted due to the low concentration of CGRP in plasma.
In order to analyze CGRP levels over the different migraine phases, we considered six timepoints: interictal (median value of five consecutive days, for migraine patients when they were headache-free for 72 hours), preictal (pre-24h before the migraine attack), ictal (headache onset, after 2h, after 8h) and postictal (post-24h after the migraine attack).
Statistical analysis
Nominal (categorical) variables were reported as frequencies (percentages) while mean ± standard deviation (age, disease evolution time, Headache Impact Test (HIT-6) and perceived stress scale [PSS]) or median and interquartile range (Me [IQR]) (Migraine Disability Assessment (MIDAS), Hospital Anxiety and Depression Scale [HAD]) and CGRP levels) were reported for continuous variables. For CGRP levels we also report 95% Confidence Interval (CI). Normality assumption of quantitative variables was checked through visual methods (Q-Q plots) and normality tests (Shapiro-Wilk test).
Statistical significance for intergroup variables was assessed by Pearson’s chi-square when comparing categorical variables. In the case of having an expected count less than 5 in more than 20% of cells in the contingency table, Fisher’s exact test was used. Linear trend chi-square was considered for ordinal variables. Independent t-test for continuous variables that followed a normal distribution (age and PSS) was used in order to assess differences between migraine patients and healthy controls and, Mann-Whitney U test was used for the rest variables that did not follow any normality assumption (HAD Scales and CGRP basal levels). The degree of association between interictal CGRP levels and clinical variables was computed by Spearman’s rank correlation and summarized by Spearman’s rho coefficient and related p-values.
We estimated the ictal log2Fold-Change (
Equation 1. Ictal Fold-Change: Measure concept for the evaluation of the change of the CGRP salivary level between premonitory and the attack onset phases.
Finally, to determine the significance of CGRP intraindividual changes according to patient’s migraine phase, we used non-linear mixed effects modeling fit by restricted maximum likelihood. Mixed models are an extension of simple models to allow both fixed and random effects, and are used when there is no independence in the data (e.g. different CGRP measurements from the same patient). Variance inflation factors (VIFs) for all the parameters were computed in order to estimate how much the variance of an estimated regression coefficient is inflated due to correlated variables so that we could avoid an overfitting problem in the model. The non-linear mixed effect models were generated using the lme function in the R package nlme (22), with CGRP levels as a dependent variable, migraine cycle (time), age and stress score (PSS) as a fixed factors and patients as a random factor. To model the non-linear relationship between CGRP over the course of migraine attack, we added a cubic trend component for time. Post-hoc pairwise comparisons of estimated marginal means of CGRP was conducted between each subsequent time point (interictal, preictal, headache onset, after 2h, after 8h and postictal), adjusting for multiple comparisons using the false discovery rate (FDR) correction. Univariate linear mixed models, were used to test the interaction between time and dichotomized basal frequency (<5 d/mo or 6–10 d/mo) on the CGRP levels and treated attacks (None, NSAIDs or Triptans), again covarying for age and PSS. A statistical power calculation was not conducted prior to the study because the sample size was based on the available data for this exploratory analysis. However, effect size for each statistical test is reported. No adjustment for multiple comparisons was made to the statistical clinical inferences, but exact p-values were reported to allow post adjustments. P-values presented are for a two-tailed test and p-values <0.05 were considered statistically significant.
All statistical analysis were conducted in R v3.6.3 (23) and figures were produced using the package ggplot2 (24).
Approval for this study was obtained from the Vall d’Hebron Ethics Committee (PR(IR)292/2017). All participants gave their consent for data collection.
Results
Descriptive
In total, 22 EM and 22 HC with a mean age of 30.8 ± 10.2 years old were included (Table 1). EM patients had statistically significant higher proportion of perceived stress (95.5% vs. 68.2%; p = 0.046). Twelve patients (54.5%) reported headache frequency of ≤5 days/month and ten patients (45.4%) reported headache frequency of 6-10 days/month.
Baseline demographics, clinical characteristics and comorbidities of participants.
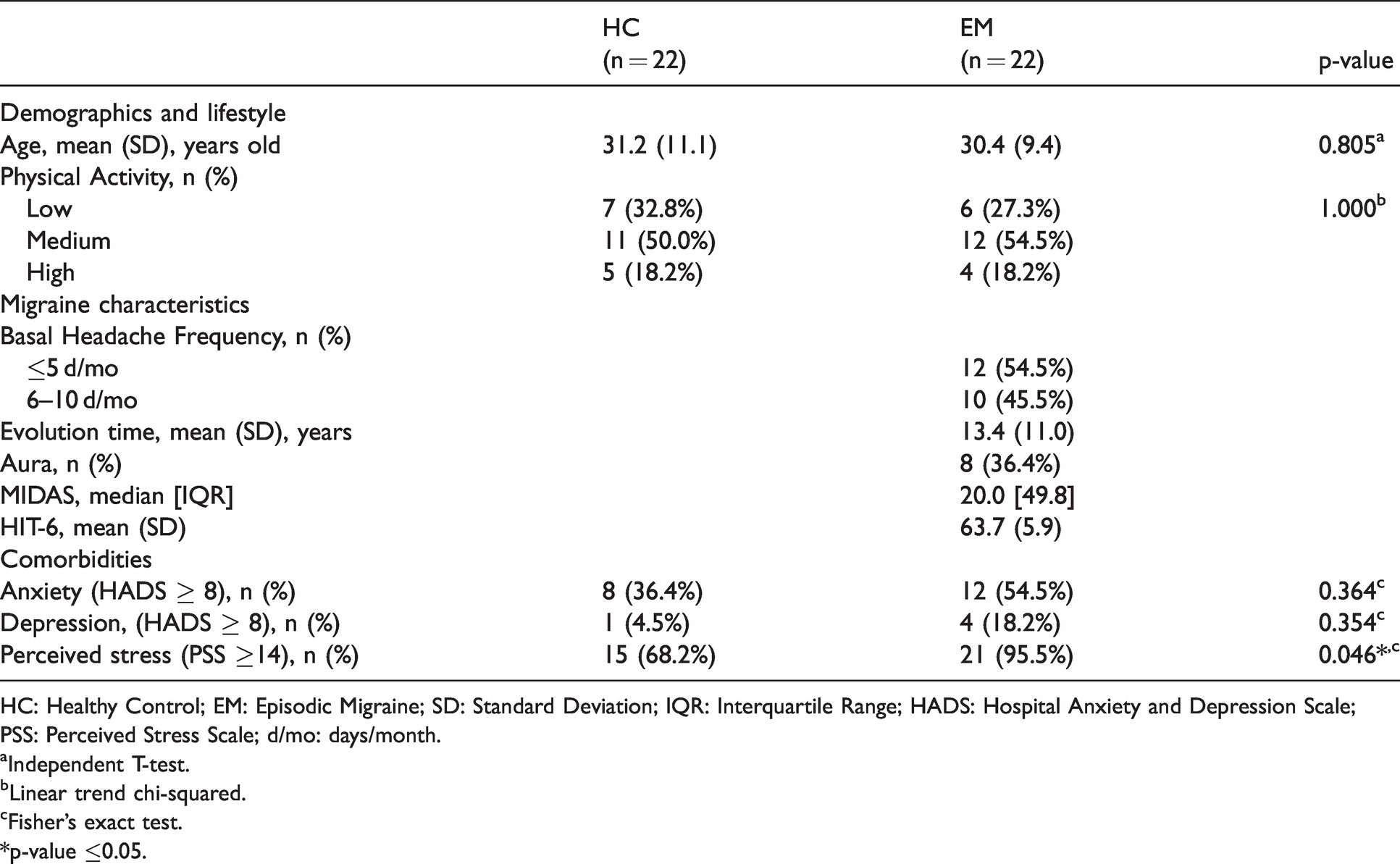
HC: Healthy Control; EM: Episodic Migraine; SD: Standard Deviation; IQR: Interquartile Range; HADS: Hospital Anxiety and Depression Scale; PSS: Perceived Stress Scale; d/mo: days/month.
aIndependent T-test.
bLinear trend chi-squared.
cFisher’s exact test.
*p-value ≤0.05.
Interictal CGRP levels
Following results are expressed as CGRP-like immunoreactivity (CGRP-LI) due to the cross-reactivity. We found statistically significant higher interictal CGRP-LI salivary levels in EM compared to HC: EM, 98.0 [80.3] (95% CI 56.6, 124.0) vs. HC, 54.3 [44.0] (95% CI 42.2, 70.1) pg/mL (p = 0.034, Wilcoxon effect size r = 0.420) (Figure 1A). We did not find any correlation between CGRP-LI levels in regards to: age (ρ = 0.192, p = 0.493), HADS-A (ρ = 0.190, p = 0.217), HADS-D (ρ = 0.259, p = 0.162), or PSS score (ρ = 0.202, p = 0.189) in all participants. In regards to plasma CGRP-LI levels, we did not find statistically significant differences in this substrate between study groups (EM, 6.0[5.2] (95% CI 4.5, 8.4) vs. HC, 5.1[4.2] (95% CI 3.2, 7.1) pg/mL; p = 0.113, Wilcoxon effect size r = 0.241) (Figure 1B). There was not a correlation between salivary and plasma CGRP-LI levels.

Basal salivary levels of CGRP. Box plots and 95% confidence intervals for interictal CGRP levels in saliva (A) and plasma (B).
Longitudinal analysis: salivary CGRP levels through migraine attack
A total of 49 migraine attacks were collected. We analyzed salivary CGRP-LI at each timepoint of the attack. We found that age and cubic trend of time (migraine cycle) drove statistically significantly changes on CGRP-LI concentration (Table 2). Post-hoc analysis showed statistically significantly higher concentration of CGRP-LI during headache onset. Estimated marginal means and 95% CI from the mixed-effect model were: 169.0 [95% CI 104.2–234.0] in the preictal; 247.0 [95% CI 181.9–312.0] during headache onset; 143.0 [95% CI 77.6–208.0] after 2h; 169.0 [95% CI 103.5–234.0] after 8h and 173.0 [95% CI 107.8–238.0] in the postictal (Figure 2).
Results from the linear mixed effects model.

*p-value ≤0.05.
**p-value ≤0.01.

CGRP values changes through the different migraine phases.
Furthermore, we analyzed the possible interaction between time (migraine phase effect) and basal headache frequency, but this interaction did not reach the level of statistical significance (Table 2, p = 0.088). However, patients with higher basal frequency (>6–10 d/mo) presented a statistically significantly higher concentrations of CGRP in all time points (Table 2, Figure 2A). In regards to acute treatment, we also found no statistically significant interaction between migraine cycle and medicated attacks with neither non-steroidal anti-inflammatory drugs (NSAID) nor triptans (Table 2). However, post-hoc analysis revealed that attacks treated with triptans presented a statistically significantly reduction on CGRP after 2 h from headache onset (Figure 3B).

Changes in CGRP levels in the different migraine phases amongst (A) basal headache frequency or (B) acute treatment for migraine attacks.
Subtypes of migraine patients
We found that 79.6% (39/49) of migraine attacks were CGRP dependent (dCGRP) and 20.4% (10/49) of migraine attacks were non-CGRP dependent (nCGRP). CGRP dependent group presented a statistically significant higher levels of CGRP-LI: dCGRP, 171.6 [130.8] (95% CI 160.0, 235.0) vs. nCGRP, 101.7 [153.9] (95% CI 39.8, 155.0); p = 0.009). In regards to accompanying symptoms, a statistically significant association between photophobia (dCGRP: 76.9% vs. nCGRP: 40.0%; p = 0.024) and phonophobia (dCGRP: 69.2% vs. nCGRP: 30.0%; p = 0.036) was found in dCGRP group. Dizziness (dCGRP: 30.8% vs. nCGRP: 70.0%; p = 0.047) was statistically significantly associated with nCGRP group (Table 3). When we analyzed migraine patients, 13 out of 22 patients only showed dCGRP migraine attacks; 3 out of 22 patients only showed nCGRP migraine attacks and 6 patients showed both types of migraine attacks. Regarding time of collection (circadian variations), we did not find statistical differences between two patterns: 69.4% of registered migraine attacks (34/49) occurred during afternoon (1–11PM): GCRP-dependent (30.8% AM and 69.2 PM) vs. non CGRP dependent (30.0% AM and 70.0% PM) (p = 0.962).
Clinical and molecular parameters associated with CGRP dependent and non-CGRP dependent migraine attacks.
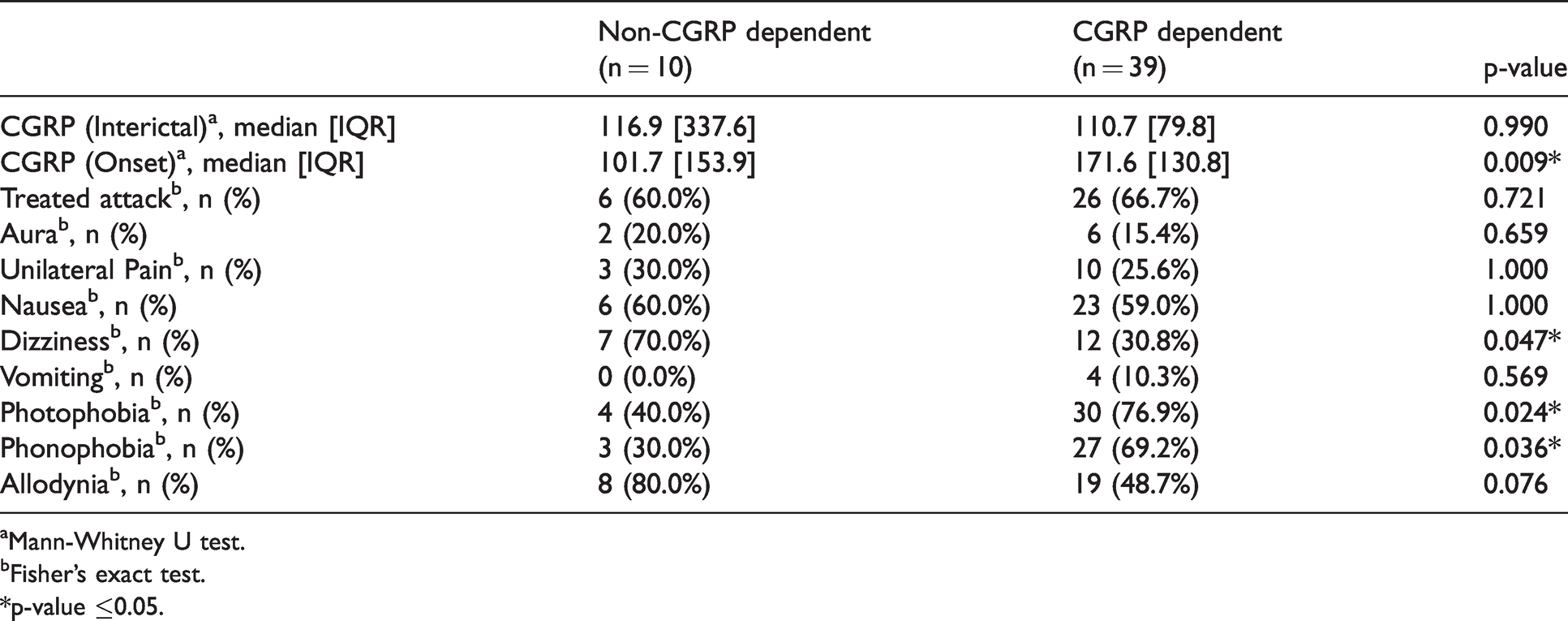
aMann-Whitney U test.
bFisher’s exact test.
*p-value ≤0.05.
Discussion
In this study we performed an ELISA assay to measure and monitor salivary levels of CGRP in episodic and treatment naïve patients over the different migraine phases. With this longitudinal approach over 30 days we wanted to properly identify not only the onset, but also what happened right before and after the attacks, and also confirm that reliable interictal levels could differentiate patients from controls. Then, EM patients showed higher interictal CGRP levels compared to HC. Intraindividual CGRP levels can change over the course of a migraine attack. Moreover, EM patients with CGRP dependent attacks presented with classical migraine clinical symptoms, creating different phenotypes of migraine patients.
Our results show that interictal salivary levels of CGRP are significantly higher in patients with EM, even if they suffer from infrequent attacks, compared to HC. Bellamy et al. also found that patients with EM had elevated salivary levels of CGRP outside the attacks compared to controls (14). There is only one previous study that found higher CGRP levels in HC (10). Different results across the studies maybe lie in methodological aspects, in particular in the sample collection time, since migraine is a cyclic disease. Other than saliva, previous studies using other substrates such as plasma (25,26) or tear fluid (27) also showed higher levels of CGRP outside the attacks in patients with EM. So, our finding endorses the presence of elevated CGRP interictal salivary levels in migraine patients. Moreover, we found an association between larger variations in CGRP concentration and higher headache frequency, which may support a greater activation of the trigeminovascular system in more severe forms of the disease (28,29). In this regard, Cernuda-Morollón et al. found that chronic migraine (CM) patients exhibited the highest levels of CGRP, followed by EM patients and HC (30). In contrast, Lee et al. did not find any significant differences between groups (31). In cerebrospinal fluid (CSF), there are 3 studies that found increased levels of CGRP in CM (32–34). There is no data on CGRP in CSF from EM patients.
As a cycling brain disorder, monitoring migraine over its different phases is important in order to understand the underlying mechanisms and pathogenesis (35) as it has previously been demonstrated in neuroimaging studies (36). At a molecular level, our study reveals that salivary levels of CGRP are dynamic and change over a migraine attack according to the timepoint analyzed: they were shown to increase as headache progressed from preictal to the ictal phase and decreased in the postictal phase at or below interictal levels. Our results are the first ones to see this gradual change of CGRP levels during an attack, and confirm previous studies which also showed an increase in salivary levels of CGRP during the ictal phase, interpreting it as a sign of trigeminovascular activation (10,15). This change reflects that CGRP is a dynamic neuropeptide in a disease that is not static; and, considering intraindividual change and not mean population CGRP levels as a baseline to start working on perhaps developing CGRP as a practical clinical biomarker.
Based on our fold-change (FC) analysis, we observed three different types of patients: those with CGRP dependent migraine attacks, those with non-CGRP dependent migraine attacks and those with two types of migraine attacks. The highest pain intensity peak occurred at the onset of headache in most migraine attacks. Some diagnostic migraine symptoms such as photo- and phonophobia were significantly related to presence of elevated CGRP. This could be interesting both to understand the link between CGRP and migraine symptoms (37); which could also help, if this is confirmed with larger cohorts in the future, to create an algorithm to clinically predict anti-CGRP treatment response according to the percentage of attacks with different symptoms. In this sense, it is worth mentioning the similarity found between the percentage of patients nCGRP dependent and those patients who are non-responders in anti-CGRP monoclonal antibodies clinical trials (38) or the percentage of patients who do not develop a migraine attack after provocation (39). Migraine diagnosis is currently based on clinical criteria according to the ICHD-3, which may result in misdiagnosis due to the recall bias. Then, our results may support the concept of classifying migraine from a pathophysiological point of view. This information might help us to start practicing precision medicine in migraine.
During the past decades, saliva has received growing attention as a substrate to study biomarkers in chronic pain disorders (40). Measuring CGRP closer to the afferents seems to be more effective than in plasma, easier and less-invasive, and is a reliable reflection of trigeminovascular activation (41). We have found that CGRP levels were higher in saliva than in plasma and there were no significant differences in CGRP plasma levels between EM and HC. Hence, serum perhaps is not the ideal matrix to measure CGRP levels since neuropeptides are circulating in low concentrations. Salivary glands are innervated by the third branch of the trigeminal nerve and therefore are closer to the trigeminovascular system. These CGRP-containing trigeminal nerves release this neuropeptide in conditions such as migraine and cluster headache (10,42). As in previous studies, we did not find a correlation between CGRP salivary and plasma levels (10,14,15).
Since the first demonstration of an increase in CGRP levels in the external jugular venous blood during a migraine attack (5), researchers have been seeking the most adequate technique to measure CGRP levels and, controversial results have been published possibly due to methodological differences (41,43). Differences lie in the type of matrix used (plasma, serum, saliva, CRF, tears), different type of collection methods, different immunoassays or brands, different timepoints of sample collection or phenotypically heterogeneous patients. It is worth mentioning that negative studies cannot be taken as evidence for the lack of importance of CGRP in migraine pathophysiology, as CGRP analysis can be challenging. When measuring CGRP there are several factors to take into account: age, gender, menstruation, fasting or circadian variation (44). However, with the approval of new therapies targeting CGRP or its receptor (45–47), the role of CGRP in migraine pathophysiology has been consolidated (48–50).
The study has some limitations. First, the small size of the sample. Some patients only collected one migraine attack so we need to be cautious when classifying patients into different types. However, we really wanted to include episodic migraine patients, in order to clearly differentiate the attack from the interictal period. Secondly, most of the migraine attacks were treated. Thus, the majority of our CGRP levels were measured in medicated migraine attacks, proving that differences can be found even if we treat patients. Finally, if patients had a migraine attack far from home, they placed ictal samples in a fridge without temperature control which in turn could reduce the saliva quality; moreover, commercial freezers can vary slightly their freezer temperature between −18 and −20°C. However, this shows that in these conditions CGRP can be found in saliva.
Our study has a longitudinal approach collecting samples over a 30-day period, which in migraine is less frequent. Moreover, our study participants were strictly and carefully selected, resulting in a very homogeneous sample. Future research should be focused on finding a molecular, anatomical, genetical and physiological way of defining migraine which in turn could help to develop a pathophysiological driven classification.
Conclusions
In conclusion, our data confirm that CGRP levels vary according to the migraine phase; and, finds CGRP dependent and non-CGRP dependent migraine attacks. In the future, the CGRP migraine patient profile might allow clinicians to better phenotype patients, may help predict response to treatment and increases our understanding of migraine pathophysiology.
Article highlights
CGRP varies intraindividually according to the migraine phase Patients with migraine can have CGRP dependent and/or non-CGRP dependent attacks, which is correlated with differences in clinical symptoms
Footnotes
Author contributions
AA, VJG, MTF and PPR made substantial contributions to the conception and design of the study. LA made substantial contribution to the methodology and she performed the ELISA protocol. AA wrote first draft of the paper. VJG performed statistical analysis. All authors participated in acquisition, analysis and/or interpretation of data. All authors have critically revised and finally approved the version to be published.
Acknowledgement
The authors thank the migraine patients who carefully followed all of our instructions for 30 days and did not take any acute treatment until the first sample of each migraine attack was collected.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AA has received honoraria as speaker for Abbie-Allergan and for education for Novartis and Eli Lilly. VJG and LA have nothing to disclose. EC has received honoraria as speaker for Novartis. MTF has received honoraria as a speaker for Abbie-Allergan, Chiesi, Eli Lilly and Novartis. PP-R has received honoraria as a consultant and speaker for Allergan, Almirall, Biohaven, Chiesi, Eli Lilly, Medscape, Neurodiem, Novartis and Teva. Her research group has received research grants from Allergan, AGAUR, la Caixa foundation, Migraine Research Foundation, Instituto Investigación Carlos III, MICINN, PERIS; and has received funding for clinical trials from Alder, Electrocore, Eli Lilly, Novartis and Teva. She is a trustee member of the board of the International Headache Society and a member of the Council of the European Headache Federation. She is in the editorial board of Revista de Neurologia. She is an associate editor for Cephalalgia, Frontiers of Neurology and Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She is the founder of ![]() . PPR does not own stocks from any pharmaceutical company. In relation with this paper the authors have nothing to disclose.
. PPR does not own stocks from any pharmaceutical company. In relation with this paper the authors have nothing to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant from Instituto de Salud Carlos III (ISCIII - PI16/01525) and Alicia Alpuente has been partially financed by a grant from the Headache Study Group of the Spanish Neurological Society 2018.

