Abstract
Objective
EMPOwER, a double-blind, randomised, phase 3 study, evaluated the efficacy and safety of erenumab in adults with episodic migraine from Asia, the Middle East, and Latin America.
Methods
Randomised patients (N = 900) received monthly subcutaneous injections of placebo, erenumab 70 mg, or 140 mg (3:3:2) for 3 months. Primary endpoint was change from baseline in monthly migraine days at Month 3. Other endpoints included achievement of ≥50%, ≥75%, and 100% reduction in monthly migraine days, change in monthly acute migraine-specific medication treatment days, patient-reported outcomes, and safety assessment.
Results
At baseline, mean (standard deviation) age was 37.5 (9.9) years, 81.9% were women, and monthly migraine days was 8.2 (2.8). At Month 3, change from baseline in monthly migraine days (primary endpoint) was −3.1, −4.2, and −4.8 days for placebo, erenumab 70 mg, and erenumab 140 mg, respectively, with a statistically significant difference for erenumab versus placebo (P = 0.002 [70 mg], P < 0.001 [140 mg]). Both erenumab doses were also significantly superior to placebo on all secondary endpoints, including the proportion of patients achieving ≥50% reduction from baseline in monthly migraine days, change from baseline in monthly acute migraine-specific medication treatment days and change from baseline in the Headache Impact Test-6™ scores. The safety profile of erenumab was comparable with placebo; no new safety signals were observed.
Conclusions
This study of erenumab in patients with episodic migraine from Asia, the Middle East, and Latin America met all primary and secondary endpoints. A consistent numerical benefit was observed with erenumab 140 mg versus erenumab 70 mg across all efficacy endpoints. These findings extend evidence of erenumab’s efficacy and safety to patients under-represented in previous trials.
Keywords
Introduction
Migraine is a complex neurological disease, affecting nearly 12% of the global population (1), and is the second leading cause of years lived with disability (2).
Migraine is associated with substantial economic and societal burden (3). Data on migraine burden worldwide are derived from epidemiological surveys conducted primarily in the United States and Europe (4,5). In addition to patients from Western Europe and North America, the International Burden of Migraine Study included patients from Taiwan (667/8726 [7.6%]) and demonstrated the high disease burden of migraine across ethnic groups (4). Wang et al. demonstrated a more region-specific assessment of the high disability and disease burden associated with migraine in patients from Taiwan (6). Moreover, the prevalence of migraine is only slightly lower (9.1%) in the Asia-Pacific region (7) than in Europe and North America (10.6%) (1). Therefore, there remains an unmet need for the appropriate management of migraine in Asia, which represents nearly one-third of the world’s population.
Monoclonal antibodies targeting the calcitonin gene-related peptide pathway, involved in the pathogenesis of migraine, represent a new platform of preventive migraine therapies (8). Erenumab (erenumab-aooe in the United States) is a fully human monoclonal antibody that selectively blocks the canonical calcitonin gene-related peptide receptor, involved in the pathogenesis of migraine (8). Previous clinical studies of erenumab were conducted in migraine patients from the United States, Japan, and Europe (9–13).
EMPOwER (ClinicalTrials.gov identifier: NCT03333109) evaluated the efficacy and safety of erenumab in patients with episodic migraine from Asia, the Middle East, and Latin America. This study aimed to strengthen the evidence in a population not adequately represented in previous clinical trials of erenumab.
Methods
Study design
This multicentre, randomised, double-blind, placebo-controlled, parallel-group, phase 3 study was conducted at 83 sites across 11 countries in Asia, the Middle East, and Latin America (Figure 1) from 8 February 2018 to 13 January 2020. The study comprised an eligibility-screening period of 2 weeks, followed by a 4-week baseline period. Patients were randomised 3:3:2, using an Interactive Response Technology (IRT) system, to receive monthly subcutaneous injections of placebo, erenumab 70 mg, or 140 mg during a 3-month double-blind treatment period followed by a 12-week (3-month) safety follow-up period. Randomisation was stratified by prior prophylactic migraine treatment failure. A patient randomisation list was produced by the IRT provider using a validated system that automated the random assignment of patient numbers to randomisation numbers. The randomisation scheme was reviewed and approved by a member of the Novartis randomisation group.

Study design.
Patients, investigator staff, persons performing the assessments, and data analysts were unaware of the group assignments. Identity of the treatments was concealed by use of identical packages, labelling, schedules of administration, appearance, taste, and odour.
This study was conducted in accordance with the International Council for Harmonisation E6 Guideline for Good Clinical Practice with applicable local regulations and guidelines and with the ethical principles laid down in the Declaration of Helsinki. All patients provided written informed consent before participation. The final study protocol, informed patient consent forms, and accompanying materials were reviewed and approved by the relevant Ethics Committee/Institutional Review Boards at participating sites (
Study participants
Adults aged 18 to 65 years, with a diagnosis of migraine according to the International Classification of Headache Disorders (ICHD-3 beta) (14) based on medical records and/or patient self-report, were eligible for study participation.
Eligibility criteria included an average of ≥4 and <15 migraine days per month and <15 headache days per month (migraine and non-migraine) across the 3 months before screening and during baseline (based on electronic diary calculations).
Key exclusion criteria were age of >50 years at migraine onset; no therapeutic response to >2 of the seven categories of migraine-preventive treatments after an adequate therapeutic trial; use of a prohibited medication, device, or procedure prior to the start of the study; use of botulinum toxin within 4 months, ergotamines or triptans on ≥10 days per month, simple analgesics on ≥15 days per month, or opioid- or butalbital-containing analgesics on ≥4 days per month. Detailed inclusion/exclusion criteria are presented in
Assessments and endpoints
Patients recorded all headaches and information about headache medication using a daily electronic diary throughout the baseline and subsequent treatment periods. Patients were encouraged to complete this information at the same time each day. Data entry beyond two days after activity completion was considered missing.
The primary study endpoint was change from baseline in monthly migraine days. A migraine day was defined as any calendar day on which the patient experienced a qualified migraine headache (onset, continuation, or recurrence of the migraine headache) as recorded in the electronic diary. A qualified migraine headache was defined as a migraine with or without aura, lasting for ≥30 minutes, and meeting at least one of the following criteria: 1) two or more of the pain features (unilateral, throbbing, moderate to severe, or exacerbated with physical activity), and 2) one or more of the associated symptoms (nausea and/or vomiting or photophobia and phonophobia).
A qualified headache was defined as a qualified migraine headache (including an aura-only event that is treated with acute migraine-specific medication), or a qualified non-migraine headache, which was a headache that lasted ≥30 minutes and was not a qualified migraine headache, or a headache of any duration for which acute headache treatment was administered.
Secondary endpoints included achievement of ≥50% reduction from baseline in monthly migraine days, change from baseline in monthly acute migraine-specific medication treatment days and change from baseline in the Headache Impact Test-6™ scores (measured monthly). Exploratory endpoints included achievement of ≥75% and 100% reduction from baseline in monthly migraine days, and patient-reported outcomes (change from baseline in the Migraine Physical Function Impact Diary [measured daily]; modified Migraine Disability Assessment [measured monthly]; and EuroQoL, 5-dimension, 5-level scale [EQ-5D-5L; measured monthly]). Specific details of the patient reported outcome tools used in this study (HIT-6, modified MIDAS, MPFID, EuroQOL, or EQ-5D-5L) are presented in
Safety evaluations included the reporting of adverse events, coded using the Medical Dictionary for Regulatory Activities, version 22.0. Safety assessment included evaluation of laboratory values, vital signs, and electrocardiograms. Additionally, serum samples were collected for the measurement of anti-erenumab-binding antibodies. Samples testing positive for binding antibodies were further tested for neutralising antibodies and if found positive, the sites were not notified until the patient’s final study visit. These patients were dosed throughout the study.
Statistical analysis
Considering a 3:3:2 randomisation ratio among placebo, erenumab 70 mg and 140 mg, a total of 880 patients (including a 10% dropout rate) would enable both dose arms to achieve >90% power under overall 0.05 full alpha level to detext the treatment difference with respect to the primary and secondary efficacy endpoints.
Patients were analysed according to their randomised treatment. Demographic characteristics and study disposition were reported using the intention-to-treat (ITT) analysis set, which included all randomised patients in the study.
Efficacy evaluations used the modified ITT (mITT) analysis set, which comprised all patients who started study treatment and completed at least one post-baseline efficacy measurement during the double-blind phase.
The safety analysis set comprised all randomised patients who received at least one dose of treatment and were analysed based on the actual treatment received.
Demographic characteristics were summarised using descriptive statistics. The primary and secondary efficacy outcomes were analysed using a linear mixed-effects repeated measures model based on observed monthly data during the double-blind treatment phase and pairwise comparisons versus placebo were conducted. The model was fitted with a linear mixed effects repeated measures with treatment group, baseline value, stratification factor, scheduled visit, and the interaction of treatment group with scheduled visit. In repeated statement, an unstructured covariance matrix was used, and no random effect was assumed. The dependent variable is change from baseline scores. The estimation method was restricted maximum likelihood (REML). No missing data were imputed for the mixed effects model. Dichotomous endpoints were analysed by Cochran-Mantel-Haenszel test with patients missing data on monthly migraine days at Month 3 of the double-blind treatment phase counted as non-responders. The model was fitted for each time-point for the Odds Ratio (OR). This was adjusted by the stratification factor. Treatment difference and OR compared with placebo were generated. Nominal 95% confidence intervals (CI) and P values were reported.
For safety analyses, patient incidence of treatment-emergent adverse events were reported using the system organ class and preferred term by treatment group. Separate summaries were provided for fatal adverse events, serious adverse events, adverse events leading to discontinuation of treatment, treatment-related adverse events, and treatment-related serious adverse events. Summary statistics were provided for laboratory data, electrocardiogram, vital signs, and immunogenicity assessments.
Results
Demographics and disease characteristics
Of 1423 screened patients, 900 were randomised to either placebo (N=338), erenumab 70 mg (N=338), or 140 mg (N=224) (Figure 2). Overall, 840 (93.3%) patients completed the study and 865 (96.1%) completed the double-blind treatment phase. A total of 29 (3.2%) patients discontinued the double-blind treatment phase, with patient or guardian decision being the most frequent reason (Figure 2). Most patients were enrolled by participating sites in Asia (
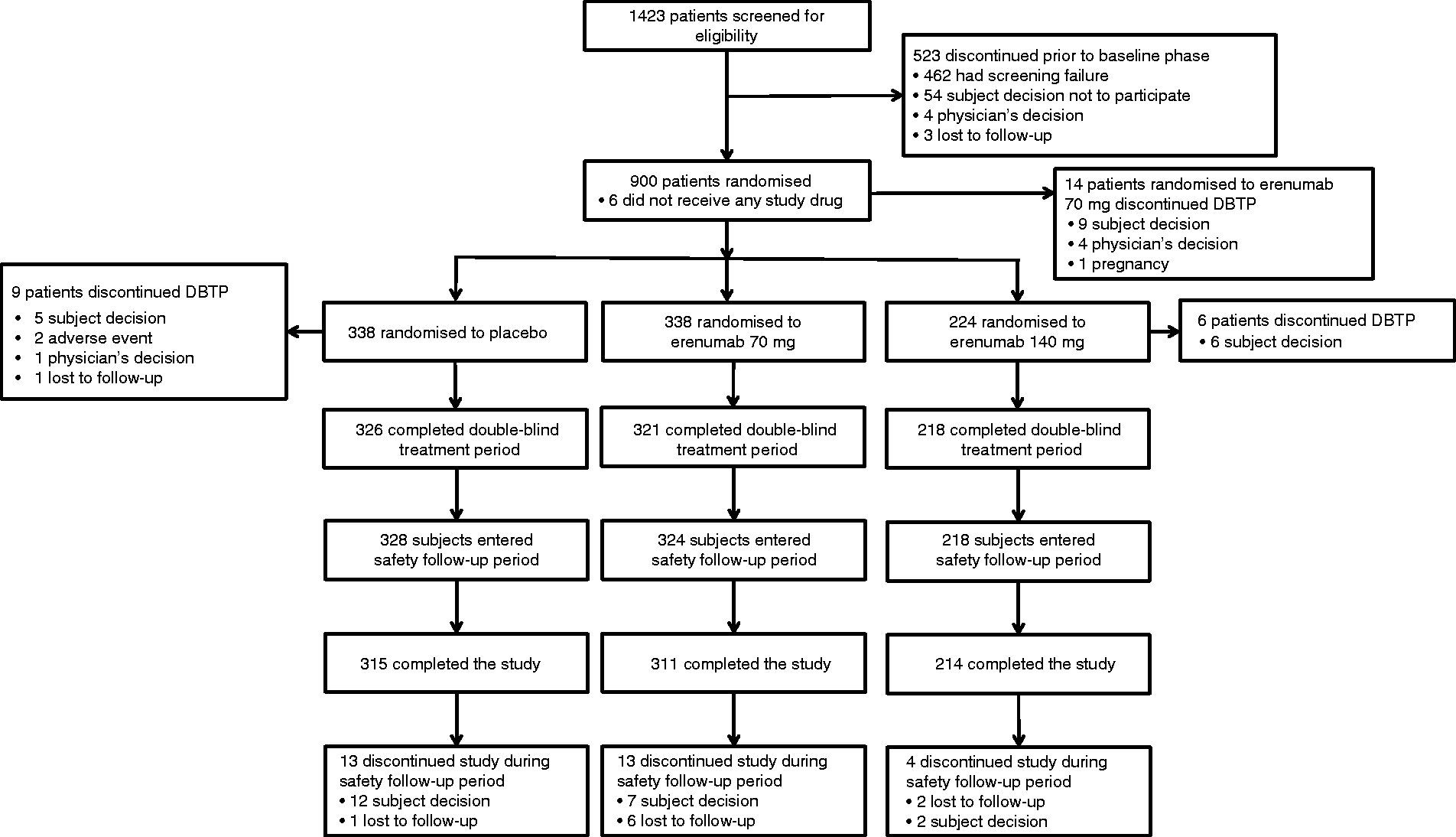
Trial profile (randomised analysis set).
Baseline demographic and disease characteristics were balanced across treatment groups (
Baseline demographic and clinical characteristics.

Data are mean (standard deviation) unless specified. EQ-5D-5L, EuroQoL, 5-dimension, 5-level scale.
The mean (standard deviation, SD) age of patients was 37.5 (9.9) years. At baseline, the mean (SD) monthly migraine days were 8.4 (2.8) in the placebo group, 8.1 (2.6) in the erenumab 70 mg group, and 8.3 (3.1) in the erenumab 140 mg group. Overall, 53.2% patients had prior prophylactic medication treatment, whereas 46.8% were treatment naive. Most patients (68.0%) had not previously taken preventive migraine treatment or experienced preventive migraine treatment failure (due to lack of efficacy or poor tolerability). Details on prior migraine preventive medication categories are provided in
Efficacy
Change from baseline in mean monthly migraine days at month 3 was −3.1 days for placebo, −4.2 days for erenumab 70 mg, and −4.8 days for erenumab 140 mg. The mean differences versus placebo (95% CI) were −1.1 (−1.8, −0.4), P=0.002 (70 mg); −1.7 (−2.5, −0.9), P<0.001 (140 mg) (Figure 3). There was a statistically significant greater reduction in monthly migraine days observed with both doses of erenumab compared with placebo at all time-points of assessment, starting from Month 1. Details on change from baseline in monthly migraine days are provided in

Change from baseline in monthly migraine days by treatment and visit (modified intention-to-treat analysis set).
At Month 3, 148/330 (44.8%) patients receiving placebo, 182/329 (55.3%) receiving erenumab 70 mg, and 140/219 (63.9%) receiving erenumab 140 mg showed ≥50% reduction from baseline in monthly migraine days. The resulting OR (95% CI) were 1.5 (1.1, 2.1), P = 0.007 (70 mg vs placebo) and 2.2 (1.6, 3.2), P<0.001 (140 mg versus placebo) (Figure 4a). Additionally, a statistically significant greater proportion of patients in both erenumab dose groups demonstrated ≥50% reduction in monthly migraine days during Months 1 and 2 than in the placebo group.

Proportion of patients with: (a) ≥50% reduction from baseline in monthly migraine days, (b) ≥75% reduction from baseline in monthly migraine days, and (c) 100% reduction from baseline in monthly migraine days (modified intention-to-treat analysis set).
At Month 3, 86/330 (26.1%) patients receiving placebo, 124/329 (37.7%) receiving 70 mg erenumab, and 94/219 (42.9%) receiving 140 mg erenumab showed a ≥75% reduction in monthly migraine days (OR 1.7 [95% CI 1.2–2.4]; P=0.001 for 70 mg vs placebo and OR 2.2 [95% CI 1.5–3.1]; P<0.001 for 140 mg vs placebo). Similarly, 47/330 (14.2%) patients receiving placebo, 73/329 (22.2%) receiving 70 mg erenumab, and 50/219 (22.8%) receiving 140 mg erenumab had a 100% reduction in monthly migraine days (OR 1.7 [95% CI 1.2–2.6]; P = 0.008 for 70 mg vs placebo and OR 1.8 [95% CI 1.2–2.8]; P = 0.009 for 140 mg vs placebo). The proportion of patients with ≥75% and 100% reduction from baseline in monthly migraine days at each time-point of assessment was higher in the erenumab groups compared with the placebo group, except for erenumab 70 mg on the 100% threshold at Month 1 (OR 0.8 [95% CI 0.5–1.4]; P = 0.467 for 70 mg vs placebo) (Figure 4b and c).
Statistically significant improvements were observed for both erenumab doses versus placebo for all other secondary (with multiplicity adjustment through hierarchical testing) and exploratory endpoints (without adjusting for multiplicity) across all assessment time-points (
Change from baseline in secondary outcomes at Month 3 (modified intention-to-treat analysis set).

*Monthly acute migraine-specific medication treatment days reported in patients who received migraine-specific medication at baseline. N, the number of patients included in the analysis of particular endpoint. Intention-to-treat analysis set included all patients who were randomised in the study. P<0.05 was considered as statistically significant.
CI, confidence interval; EQ-5D-5L, EuroQoL, 5-dimension, 5-level scale; SE, standard error.
Safety
The proportion of patients receiving all three scheduled doses of study drug were 96.4% in the placebo and erenumab 70 mg group, and 97.3% in the erenumab 140 mg group. Overall, erenumab was well tolerated and the proportions of patients reporting at least one adverse event and serious adverse events were similar between groups. Treatment-emergent adverse events were reported in 123/335 (36.7%) patients receiving placebo, 117/335 (34.9%) patients receiving erenumab 70 mg, and 77/224 (34.4%) patients receiving erenumab 140 mg (Table 3).
Summary of treatment-emergent adverse events, during the double-blind treatment phase (safety analysis set).

A patient with multiple occurrences of an adverse event under one treatment is counted only once in this adverse event category for that treatment; a patient with multiple severity grades for an adverse event is only counted under the maximum grade; MedDRA Version 22.0 has been used for the reporting of adverse events. Grading categories determined using CTCAE version 4.03.
e, the total time at risk during the DBTP, summed across all the patients in that class; time at risk during the DBTP is the time from first dose of DBTP investigational product (IP) through to onset of first event in the DBTP or the minimum (end of study date, last IP dose + 27 days); N, number of patients in the analysis set; n, number of patients reporting at least one occurrence of an adverse event in that class; r, exposure-adjusted patient rate per 100 patient-years (n/e*100).
Serious adverse events were reported in 2/335 (0.6%) patients receiving placebo, 3/335 (0.9%) receiving erenumab 70 mg, and none receiving erenumab 140 mg. One patient in the placebo group reported two serious adverse events. None of the serious adverse events were considered by the investigators to be treatment-related, except for asthenia in the erenumab 70 mg group. None of the erenumab-treated patients had adverse events leading to treatment discontinuation. No deaths occurred during the study.
In the study, 32/335 (9.6%) patients receiving placebo, 38/335 (11.3%) receiving erenumab 70 mg, and 24/224 (10.7%) receiving erenumab 140 mg experienced treatment-related adverse events. The most frequent treatment-emergent adverse events (≥2% in any treatment group) were constipation, nasopharyngitis, dizziness, and pyrexia (Table 3). Most observed adverse events were mild or moderate in severity.
There were no significant differences observed between the erenumab and placebo groups regarding hepatic function testing, creatinine concentrations, vital signs, or electrocardiogram findings (data not shown). Overall, 15/549 (2.7%) patients, 12/329 (3.6%) receiving erenumab 70 mg, and 3/220 (1.4%) receiving erenumab 140 mg, with an on-study result developed anti-erenumab-binding antibodies during the double-blind treatment phase. Of these, neutralising antibodies against erenumab developed in one patient receiving erenumab 70 mg. Immune disorder-related adverse events were not reported at any time during the study.
Discussion
EMPOwER is the first study of erenumab conducted in patients with episodic migraine from Asia (81.2%), the Middle East, and Latin America (18.8%). In this phase 3 study, erenumab 70 mg and 140 mg demonstrated a significant benefit over placebo in reducing the mean number of migraine days per month (difference versus placebo, −1.1 days per month [70 mg] and −1.7 days per month [140 mg]) at Month 3. Statistically significant superiority of erenumab compared with placebo on all secondary and exploratory endpoints was observed. Treatment effects were apparent within Month 1 after initiation of erenumab.
The results from EMPOwER are consistent with two previous studies of erenumab versus placebo in patients with episodic migraine (11,13). The pivotal phase 3 STRIVE study of erenumab in episodic migraine, which mainly recruited patients in North America and Europe, compared erenumab 70 mg and 140 mg doses versus placebo and showed a relative change of −1.4 and −1.9 migraine days per month over the final 3 months of the 6-month double-blind treatment phase, respectively (11). As with EMPOwER, an early onset of efficacy was apparent and secondary endpoints were met.
A phase 2, double-blind, placebo-controlled study of erenumab in Japanese patients compared erenumab 28 mg, 70 mg, or 140 mg versus placebo (13). Treatment with erenumab 70 mg and 140 mg led to statistically significant improvements versus placebo for the primary endpoint (−2.3 [70 mg] and −1.9 [140 mg] migraine days per month versus placebo [P<0.001], respectively) over Months 4–6 of the double-blind treatment phase and for secondary endpoints. In all three studies above, both erenumab 70 mg and 140 mg were significantly better than placebo. In both STRIVE and EMPOwER, erenumab 140 mg had numerically superior efficacy than the 70 mg dose (11), which was not observed in the phase 2 Japanese study (13). The exact reasons for these differences are not known. Potential explanations might include the mean body mass index of the Japanese patients being lower than that observed in the EMPOwER and STRIVE studies (22.0, 24.6 and 27.2 kg/m2, respectively), which might have led to differences in erenumab exposure between the three patient populations.
Over 70% of patients in the EMPOwER study reported migraine with aura at baseline which differs from the predominance of migraine without aura observed in the Asian population (6). It is noteworthy that in the EMPOwER study, the baseline aura status was self-reported using the electronic diary rather than being diagnosed by a physician. Patients may confuse prodromal symptoms with aura symptoms in the checklist accompanying the electronic diary. Therefore, it is possible that this self-reported prevalence of migraine with aura does not reflect the prevalence of diagnosed migraine with aura.
Another observation is the slightly lower age of patients (37.5 years) enrolled in this study as compared to STRIVE (40.9 years), the phase 2 Japanese study (44.3 years), and the 5-year open-label extension study (41.3 years) (11,13,15).
The proportions of acute migraine-specific medication use at baseline and mean medication days at baseline were 36.8% and 1.9 days in the EMPOwER study, 58.8% and >3.2 days in the STRIVE study (11) and >90%, >5.4 days in the Japanese phase 2 study (13). A higher use of acute/preventive migraine medications is likely to be associated with lower expectations for improvement in migraine and also may reflect a higher burden of disease. This may explain a lower placebo response in the STRIVE (reduction of 1.8 MMD in the placebo group over months 4-6) and the Japanese phase 2 study (increase of 0.06 MMD in the placebo group over months 4-6) compared with EMPOwER (reduction of 3.1 MMD at Month 3). Although the difference in placebo response rates between different studies may be attributable to the intervention, study population characteristics, ethnic and regional differences, and variations in study design, the higher placebo response in the EMPOwER study may reflect a higher expectation of improvement in disease management with an agent proven to have a beneficial effect in other studies.
As in other placebo-controlled studies, both doses of erenumab significantly improved all measures of patient-reported outcomes (Migraine Physical Function Impact Diary [domain scores for Physical Impairment and Everyday Activities], modified Migraine Disability Assessment, EQ-5D-5L, and Headache Impact Test-6™), providing supportive evidence of meaningful benefit.
As with previous trials of erenumab in migraine prevention, the rate of adverse events reported in the EMPOwER study for erenumab was generally low and similar to those reported with placebo, and discontinuation of the study due to adverse events was infrequent (11–13). Also, in line with other studies, the most frequent treatment-emergent adverse events (≥2% in any treatment group) were nasopharyngitis and constipation with a higher incidence of constipation in the erenumab groups (11–13). Although, the prescribing information for erenumab in the European Union (16) and the United States (17) include warnings about constipation with serious complications (16,17) hypertension (17), and hypersensitivity reactions including anaphylaxis (16,17); the current study reported constipation (Table 3) and a single case of hypersensitivity (erenumab 140 mg), drug-induced hypersensitivity (placebo), and hypertension (erenumab 70 mg) each. The safety profile for erenumab in the EMPOwER study did not differ from that established in other studies (11–13). In line with other studies (11,12), the immunogenicity of erenumab observed in the EMPOwER patient population was low. Treatment emergent serious adverse events were low in patients receiving placebo (one case of viral infection and two cases of abortion) and erenumab 70 mg (one case each of asthenia, gastroenteritis and labyrinthitis), while none were reported in patients receiving erenumab 140 mg. None of these except asthenia were deemed to be related to erenumab treatment. Overall, the safety profile of erenumab appears to be similar for the different ethnicities enrolled across erenumab studies.
As EMPOwER measured efficacy and safety over a 3-month double-blind treatment phase, the longer-term efficacy and safety in this population remains to be established. However, the long-term efficacy and safety of erenumab in patients with migraine has been demonstrated in open-label extension studies of up to five years duration (15,18). To date, these studies have revealed no new safety signals (15,18).
Study limitations
The study was limited to patients with episodic migraine and, therefore, results should not be generalised to include those with chronic migraine.
Conclusions
The EMPOwER study conducted in patients from Asia, the Middle East and Latin America met the primary and secondary endpoints. Treatment with erenumab was associated with a statistically significant reduction in monthly migraine days and in the use of acute migraine-specific medication, significantly increased achievement of ≥50%, ≥75% and 100% reduction in monthly migraine days, and improved patient-reported outcomes over a period of 3 months compared with placebo. Compared with the erenumab 70 mg dose, erenumab 140 mg was associated with a consistent numerical superiority across all efficacy endpoints, a finding consistent with previous pivotal studies of erenumab.
Combined with the efficacy results, the low proportion of patients who discontinued the study due to adverse events and the lack of any new safety findings, extend the evidence for erenumab as a safe preventive treatment for episodic migraine to ethnicities previously under-represented in prior erenumab trials.
Clinical implications
EMPOwER is the first study of erenumab conducted in patients with episodic migraine from Asia, the Middle East, and Latin America This phase 3 study demonstrated a significant benefit of erenumab 70 mg and 140 mg over placebo in reducing the mean number of migraine days per month at Month 3 This study adds evidence to existing data from prior global erenumab studies indicating that erenumab at doses of 140 mg and 70 mg is a safe and well-tolerated treatment for ethnicities previously under-represented in prior erenumab trials
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211024160 - Supplemental material for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study
Supplemental material, sj-pdf-1-cep-10.1177_03331024211024160 for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study by Shuu-Jiun Wang, Artemio A Roxas Jr Bibiana Saravia, Byung-Kun Kim, Debashish Chowdhury, Naji Riachi, Mei-Ling Sharon Tai, Surat Tanprawate, Tai Tran Ngoc, Zhao Yi Jing, Daniel D Mikol, Shaloo Pandhi, Shihua Wen, Subhayan Mondal, Nadia Tenenbaum and Peggy Hours-Zesiger in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024211024160 - Supplemental material for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study
Supplemental material, sj-pdf-2-cep-10.1177_03331024211024160 for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study by Shuu-Jiun Wang, Artemio A Roxas JrBibiana Saravia, Byung-Kun Kim, Debashish Chowdhury, Naji Riachi, Mei-Ling Sharon Tai, Surat Tanprawate, Tai Tran Ngoc, Zhao Yi Jing, Daniel D Mikol, Shaloo Pandhi, Shihua Wen, Subhayan Mondal, Nadia Tenenbaum and Peggy Hours-Zesiger in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_03331024211024160 - Supplemental material for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study
Supplemental material, sj-pdf-3-cep-10.1177_03331024211024160 for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study by Shuu-Jiun Wang, Artemio A Roxas JrBibiana Saravia, Byung-Kun Kim, Debashish Chowdhury, Naji Riachi, Mei-Ling Sharon Tai, Surat Tanprawate, Tai Tran Ngoc, Zhao Yi Jing, Daniel D Mikol, Shaloo Pandhi, Shihua Wen, Subhayan Mondal, Nadia Tenenbaum and Peggy Hours-Zesiger in Cephalalgia
Supplemental Material
sj-pdf-4-cep-10.1177_03331024211024160 - Supplemental material for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study
Supplemental material, sj-pdf-4-cep-10.1177_03331024211024160 for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study by Shuu-Jiun Wang, Artemio A Roxas JrBibiana Saravia, Byung-Kun Kim, Debashish Chowdhury, Naji Riachi, Mei-Ling Sharon Tai, Surat Tanprawate, Tai Tran Ngoc, Zhao Yi Jing, Daniel D Mikol, Shaloo Pandhi, Shihua Wen, Subhayan Mondal, Nadia Tenenbaum and Peggy Hours-Zesiger in Cephalalgia
Supplemental Material
sj-pdf-5-cep-10.1177_03331024211024160 - Supplemental material for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study
Supplemental material, sj-pdf-5-cep-10.1177_03331024211024160 for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study by Shuu-Jiun Wang, Artemio A Roxas JrBibiana Saravia, Byung-Kun Kim, Debashish Chowdhury, Naji Riachi, Mei-Ling Sharon Tai, Surat Tanprawate, Tai Tran Ngoc, Zhao Yi Jing, Daniel D Mikol, Shaloo Pandhi, Shihua Wen, Subhayan Mondal, Nadia Tenenbaum and Peggy Hours-Zesiger in Cephalalgia
Supplemental Material
sj-pdf-6-cep-10.1177_03331024211024160 - Supplemental material for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study
Supplemental material, sj-pdf-6-cep-10.1177_03331024211024160 for Randomised, controlled trial of erenumab for the prevention of episodic migraine in patients from Asia, the Middle East, and Latin America: The EMPOwER study by Shuu-Jiun Wang, Artemio A Roxas JrBibiana Saravia, Byung-Kun Kim, Debashish Chowdhury, Naji Riachi, Mei-Ling Sharon Tai, Surat Tanprawate, Tai Tran Ngoc, Zhao Yi Jing, Daniel D Mikol, Shaloo Pandhi, Shihua Wen, Subhayan Mondal, Nadia Tenenbaum and Peggy Hours-Zesiger in Cephalalgia
Footnotes
Acknowledgements
The authors thank the EMPOwER study participants and investigators for their commitment to this study. The authors thank Aditi Kataria and Fatima Hasan (both of Novartis Healthcare Pvt. Ltd., India) for medical writing support in accordance with the Good Publication Practice guidelines. Fatima Hasan confirms that she has received authorization from all listed authors to submit this manuscript on their behalf.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Novartis Pharma AG, Basel, Switzerland. Employees of the funding source were involved in the trial design, data collection, analysis, interpretation of results, and writing of the manuscript. All authors were provided full access to the study data and had the final responsibility for the decision to submit the publication.
Contributors
SJW participated in the conceptualization of the study. SP, NT, PHZ participated in the development or design of methodology and creation of models. The chief investigators were SJW, AAR, BS, BKK, DC, NR, MLST, ST, ZYJ, TTN, PHZ. SJW and DC were involved in provision of study materials, reagents, materials, patients, laboratory samples, animals, instrumentation, computing resources, or other analysis tools. AAR was involved in preparation, creation and/or presentation of the published work, specifically visualization/data presentation. SW and SM participated in patient data collection and were the study biostatisticians responsible for the statistical analyses. PHZ was the medical lead for the study. SJW, DM and PHZ were responsible for oversight and leadership responsibility for the research activity planning and execution, including mentorship external to the core team. PHZ was responsible for the management and coordination responsibility for the research activity planning and execution. All authors were involved in the writing, review and interpretation of the data. All authors agreed on the content of the manuscript, reviewed drafts and approved the final version.
Data availability statement
The data for the analyses described in this report are available by request from the authors or Novartis Pharma AG, sponsor of this clinical research.
Statistical analysis
Statistical analysis was conducted by Shihua Wen, Novartis Pharmaceutical Corporation, East Hanover, NJ, USA and Subhayan Mondal, Novartis Healthcare Pvt. Ltd., Hyderabad, India.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
