Abstract
Background
Women show increased prevalence and severity of migraine compared to men. Whether small molecule calcitonin gene-related peptide receptor (CGRP-R) antagonists (i.e., gepants) and monoclonal antibodies targeting either the CGRP-R or the CGRP peptide might show sexually dimorphic outcomes for acute and preventive therapy has not been established.
Methods
We conducted a subpopulation analysis of available published data from FDA reviews to evaluate potential sex differences in the response rates of ubrogepant, rimegepant and zavegepant for acute migraine therapy. Available data from FDA reviews of erenumab, fremanezumab, galcanezumab and eptinezumab, approved CGRP-R and CGRP monoclonal antibodies and of atogepant were examined for prevention outcomes based on patient sex. Preventive outcomes were analyzed separately for patients with episodic migraine and chronic migraine.
Results
In women, the three approved gepants produced statistically significant drug effects regardless of dose tested on the FDA mandated co-primary endpoints, the proportion of patients achieving two-hour pain-freedom and the proportion of patients free of their most bothersome symptom at two hours post-dose. In women, the average placebo-subtracted two-hour pain-freedom proportion was 9.5% (CI: 7.4 to 11.6) and the average numbers needed to treat was 11. The free from most bothersome symptom at two hours outcomes were also significant in women. The gepant drugs did not reach statistically significant effects on the two-hour pain-freedom endpoint in the men, with an average drug effect of 2.8% (CI: −2.5 to 8.2) and an average number needed to treat of 36. For freedom from most bothersome symptom at two hours post-dose endpoint, differences were not significant in male patients. The treatment effect in each of the gepant studies was always numerically greater in women than in men. In evaluation of prevention outcomes with the antibodies or atogepant using the change from the specified primary endpoint (e.g., monthly migraine days), the observed treatment effect for episodic migraine patients almost always favored drug over placebo in both women and men. For chronic migraine patients the treatment effects of antibodies were similar in men and women and always favored the drug treated group.
Keywords
Background
Migraine is a multi-symptom, multiphasic and sometimes progressive neurological disorder that impairs the quality of life of patients around the world (1). Options for treatment of migraine now include small molecule drugs and biologics that interfere with calcitonin gene-related peptide (CGRP) signaling (2). The gepant class of molecules are CGRP receptor (CGRP-R) antagonists. The gepants include ubrogepant, rimegepant and zavegepant that are used for acute migraine therapy and atogepant and rimegepant that are used for preventive therapy. Additionally, erenumab is a CGRP-R and fremanezumab, galcanezumab and eptinezumab are CGRP peptide targeting monoclonal antibodies used for migraine prevention. Multiple lines of evidence, including the clinical effectiveness of these drugs, suggest a causal role of CGRP in migraine pathophysiology. However, CGRP based therapies are not effective in all patients (3,4). Understanding which patient groups preferentially respond to CGRP-based therapies and which are less likely to respond could reduce trial and error and facilitate the selection of effective treatment for patients.
Female sex hormones have been recognized as factors in promoting migraine (5,6). While CGRP neurotransmission promotes migraine in some patients, whether this mechanism applies equally in both sexes is unknown. Women show increased severity, persistence and co-morbidity profiles as well as increased prevalence of migraine relative to men (7,8). These observations raise the possibility that treatment effects targeting CGRP or the CGRP-R may not be uniform in men and women. We therefore analyzed the totality of results of the approved gepant drugs for acute migraine therapy stratified by sex across all approved dose levels using publicly available data from FDA reviews. We also reviewed results for CGRP targeted monoclonal antibodies and atogepant by sex in the preventive treatment of patients with episodic (EM) and chronic (CM) migraine from the FDA reviews. We did not include analysis of rimegepant for migraine prevention as the Center for Drug Evaluation and Research (CDER) data separated by sex were not publicly available at the time of submission.
Methods
Clinical and statistical reviews by the FDA Center for Drug Evaluation and Research (CDER) for the New Drug Applications (NDA) of ubrogepant (9), Rimegepant (10), and zavegepant (11) in the acute treatment of migraine are publicly available. We used these data to conduct a subpopulation analysis to evaluate potential sex differences in the response rates for acute migraine therapy of these three gepants. Results from each confirmatory study used to support a labeled dose level were used. Therapeutic effect (sometimes called therapeutic gain) was determined by subtracting the proportion of patients responding to placebo from the proportion responding to active treatment on the two co-primary endpoints (a) the two-hour pain freedom (PF) (Table 1) and (b) the two-hour freedom from most bothersome symptom (MBS) (Table 2). Additionally, we further analyzed pooled data from all studies to estimate overall treatment effects in men and women (Tables 1 and 2). The pooled estimates used Mantel-Haenszel weighting (12,13).
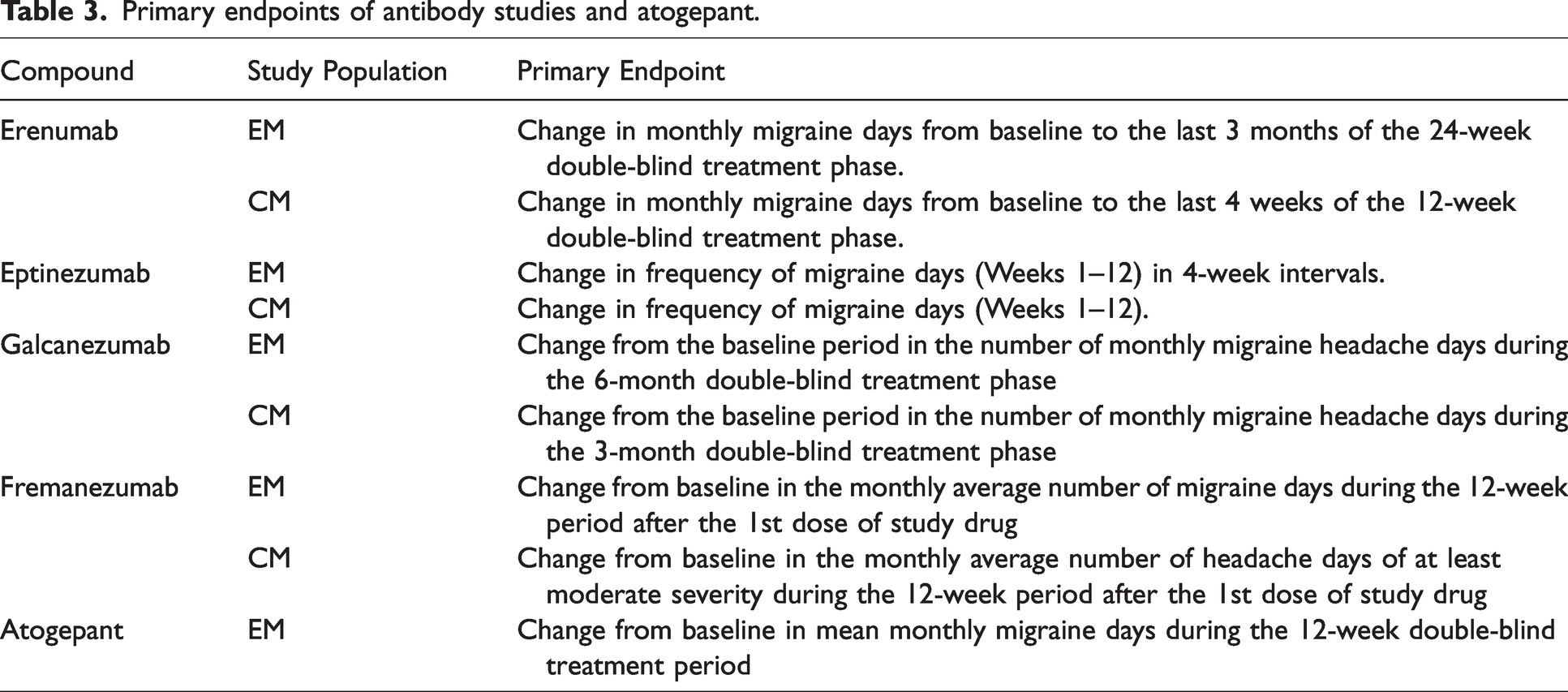
Placebo and gepant drugs response rates, drug effect, and number needed to treat (NNTs) for 2-hour pain freedom (PF) in women and men.

Number of responders/total number of participants (% of responders); Mantel-Haenszel weighting was used.
Placebo and gepants drugs response rates, drug effect, and number needed to treat (NNTs) for 2-hour most bothersome symptom (MBS) freedom in women and men.
Number of responders/total number of participants (% of responders).
Data were also extracted from published CDER reports for studies of anti-CGRP-R or anti-CGRP peptide antibodies in both EM or CM patients (14–17) and for atogepant (18) in prevention in EM patients. The patient numbers, Change from Baseline (CFB) of a specific primary endpoint for a study, the Treatment Effect (TE, defined as “active CFB – placebo CFB”) and the associated 95% Confidence Interval (CI) were tabulated for each antibody or atogepant. The CI for fremanezumab was not available within the FDA review and was therefore calculated from the standard error (SE) that was provided. We used the concept of test inversion to identify findings with P-values <0.05, where confidence intervals for the treatment effect that exclude zero would, if a test had been conducted, have resulted in a P-value <0.05. The studies used similar, but not identical, primary endpoints of change in migraine or headache days over a specified period of time, complicating comparisons between studies (see Table 3). However, there were no limitations in comparing results for male and female patients within studies (Tables 4–6). The ratio of the treatment effect (males:females) within a study was calculated in order to normalize comparisons between studies. A male:female (M:F) ratio < 1 indicates a smaller TE for men than women while a M:F ratio >1 indicates a TE that is larger for men.
Primary endpoints of antibody studies and atogepant.

Preventive treatment results of antibodies by sex from EM studies.

Two EM studies combined;
Each of the two active treatment arms included three doses: either 675 mg drug/placebo/placebo or three doses of 225 mg;
Weighted M/F ratio was calculated from pooled data with the weights proportional to the study sample size.
N = sample size; CFB = change from baseline for the primary endpoint; TE = treatment effect for the primary endpoint and analysis method; CI = 95% confidence intervals; Plb = placebo.
Preventive treatment results by sex from atogepant studies in episodic migraine (EM) patients.

N = sample size; TE = treatment effect for the primary endpoint and analysis method calculated as the LSMD (least squares mean difference); CI = 95% confidence intervals.
Preventive treatment results of antibodies by sex from chronic migraine (CM) studies.
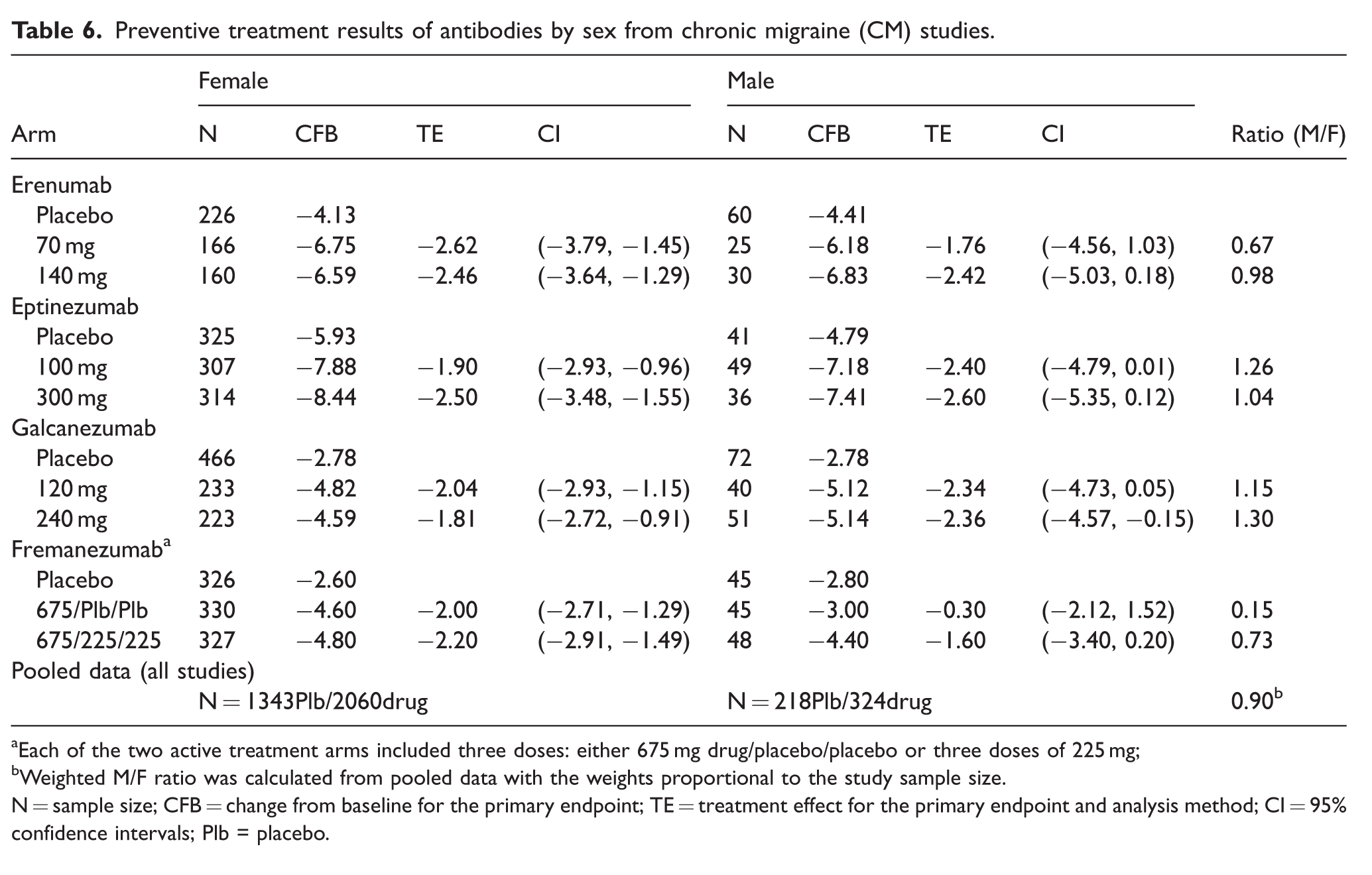
Each of the two active treatment arms included three doses: either 675 mg drug/placebo/placebo or three doses of 225 mg;
Weighted M/F ratio was calculated from pooled data with the weights proportional to the study sample size.
N = sample size; CFB = change from baseline for the primary endpoint; TE = treatment effect for the primary endpoint and analysis method; CI = 95% confidence intervals; Plb = placebo.
Results
CGRP-R antagonists for acute treatment of migraine in men and women
In women, the three approved gepants for the acute treatment of migraine (ubrogepant, rimegepant, zavegepant) (9–11) produced a statistically significant drug effect on the 2h-PF endpoint ranging from 7.4% to 11.8% which corresponded to an average (Mantel-Haenszel weighting) drug effect of 9.5% (CI: 7.4 to 11.6) and an average Number Needed to Treat (NNT) of 11 (9 to 14) (Table 1). These three gepants had a lower observed effect on the 2h-PF endpoint in the men, with drug effects ranging from −6.5% to 6.5% with confidence intervals that always included zero (i.e., no effect). The average drug effect of the gepants in males was 2.8% (CI: −2.5 to 8.2) with an average NNT of 36 (16 to ∞) (Table 1).
In the 2h-MBS endpoint in the women, the effects of these three gepants ranged from 8.5% to 12.4% which corresponded to an average drug effect of 10.2% (CI: 7.6 to 12.9) and an average NNT of 10 (9 to 12) (Table 2). In men, these drugs produced observed treatment effects on 2h-MBS ranging from −7.4% to 7.4% with confidence intervals always overlapping zero. The average drug effect of the gepants for 2h-MBS in men was 3.2% (CI: −3.3 to 9.7) and the average NNT was 32 (14 to ∞) (Table 2).
The directionality of point estimates provided in the FDA reviews allow statistical exploration of the results. Under the assumption of no effect between treatment and placebo or no difference between the effects for females and males, there would be a 50% probability that the point estimates would favor either group. Thus, it is possible to compare how often the point estimates favor treatment over placebo or favor females over males to 50%. In all treatment-dose comparisons for both co-primary endpoints (2h-PF and 2h-MBS freedom, i.e., 12 total comparisons) the effect was larger in women for each contrast (Figure 1c, g). Under the assumption of no sex difference, a result this extreme would arise in only 1 of 4096 times (P = 0.00024); this finding is consistent with a preferential effect of CGRP-R antagonists in women for the acute treatment of migraine. The CDER data for analysis of the outcomes by sex of gepants showed that in each comparison, the female treatment effect was positive, and the resulting P-value is <0.02 in 100% of cases. In contrast, the P-values for the male treatment effect are >0.20 in all cases, a finding that is not surprising given the relatively small sample sizes for male subjects. Notably, however, in men five out of 12 estimates favor placebo over active treatment (two of six for the 2h-PF and three of six for the 2h-MBS) (Tables 1 and 2 and Figure 1b, f). While this outcome does not prove a lack of effect, it would be the expected outcome for a treatment with little to no effect. As noted above, the pooled treatment effect is positive but not significantly different from zero for men (2.8% for PF and 3.2% for MBS) (Figure 1d, h). The similarly pooled treatment effects for women are three times as large at 9.2% and 10.2%, respectively (Figure 1d, h). In fact, for the pooled data the upper limit of the confidence interval for the men excludes (is less than) the observed average for the women and the lower limit of the confidence interval for women excludes (is greater than) the observed average for the male subjects. It should be noted, however, that the upper CI for men overlaps with the lower CI of women. Direct comparisons of effects show that the TE always favors women (Figure 1c, g). Note however that the placebo response rate for the two of three ubrogepant studies and one of two zavegepant studies were higher in men than in women (Table 1). Other studies with ubrogepant, rimegepant and zavegepant had similar placebo response rates in women and men. The pooled placebo responses were 12.9% and 15.7% in women and men, respectively (Table 1).

Drug effect of ubrogepant, rimegepant and zavegepant for the acute treatment of migraine in female and male patients. (a, b) 2h-PF in six different treatment regiments investigated in the indicated clinical reviews for women (a) and men (b). (c) 2h-PF combined for all gepant treatments. (d, e) 2h-MBS co-primary endpoint in six different treatment regiments plotted separately for women (d) and men (e). (f) 2h-MBS combined for all gepant treatments. Graphs and analyses were generated using data from the corresponding Center for Drug Evaluation and Research (CDER) clinical reviews for the New Drug Applications (NDA) (9–11). Data are presented as the mean drug effect size (%) and 95% CI.
CGRP-R and anti-CGRP antibodies and atogepant for migraine prevention in men and women with episodic migraine
All analyzed studies (14–17) used slightly different outcome measures as shown in Table 3. The TE was found to always numerically favor active treatment for both women and men with EM. (Table 4, Figure 2). For women with EM, with one exception (atogepant 60 mg, QD), the CI always excluded zero indicating that the P-value is <0.05, suggesting a significant effect for all individual studies. For men, the CIs are very wide and generally cover zero; exceptions were for galcanezumab, when two EM studies were combined providing a larger sample size (Table 4) and for some atogepant comparisons (Table 5). In these contrasts, the CI do not include zero, indicating a P-value of <0.05 for men with EM. Additionally, for men with EM, the TE was always numerically smaller than for women (Figure 2c) with the male:female TE ratio ranging from 0.01 to 0.79 (Table 4) except for atogepant where this pattern was reversed (Table 5). The TE ratio of pooled data from all antibody studies of EM patients was 0.55 (Table 4) indicating a treatment effect about half as large in males as females.

Drug effects of erenumab, eptinezumab, galcanezumab, fremanezumab and atogepant for the prevention of episodic and chronic migraine in female and male patients. Treatment effects of monoclonal antibodies targeting CGRP for the primary endpoint and analysis method in patients with episodic migraine are plotted separately for women (a) and men (b) and for both sexes for direct comparison (c). Treatment effects of atogepant for the primary endpoint and analysis method in patients with episodic migraine are plotted for women (d) and men (e) and for both sexes (f). Treatment effects of monoclonal antibodies for the primary endpoint and analysis method in patients with chronic migraine are plotted for women (g) and men (h) and for both sexes for direct comparison (i). Graphs and analyses were generated using data from the corresponding Center for Drug Evaluation and Research (CDER) clinical reviews for the New Drug Applications (NDA) (14–18). Data are presented as the mean treatment effect and 95% CI.
CGRP-R and anti-CGRP antibodies for migraine prevention in men and women with chronic migraine
In CM patients, the treatment effect always favored active treatment over placebo for both women and men (Table 5, Figure 2). For female patients, the CI excluded zero for all treatments indicating that the P-value would be <0.05 (Figure 2d). The TE for male CM patients was larger than for females for eptinezumab and galcanezumab and smaller for erenumab and fremanezumab (Table 5, Figure 2f). The CIs are very wide for males due to the small sample sizes and generally cover zero (Figure 2e). Note, however, for galcanezumab (240 mg) the CI for male CM patients does not cover zero indicating the P-value <0.05 and demonstrating efficacy at the 0.05 significance level for one of the treatments. The TE male:female ratio was >1 in four of eight comparisons in CM patients and <1 in the other four. The pooled TE male:female ratio in CM patients was close to 1 (0.9).
Conclusion/Interpretation
Published data evaluating the acute treatment effects of gepant drugs from more than 2595 women and 422 men show significant effects on PF and MBS outcomes in women but the available data do not show significant effects in men. The magnitude of the TE observed was always larger in women than in men. Additionally, the calculated NNTs were higher for men than for women for all gepant studies. Evaluation of 2140 women and 382 men with EM showed that all antibody prevention treatments were effective but whether there was a sex difference in outcomes remains unclear. Analysis of 2060 women and 324 men with CM showed that all antibody prevention treatments favored drug treatment. The TE for antibody prevention in EM studies was larger in women than in men in all studies. In contrast, the TE for antibody prevention was similar in men and women with CM. Analysis of 972 women and 133 men with EM showed that atogepant was effective for migraine prevention in women and men. As the placebo response in atogepant prevention studies was not reported separately for men and women, the differences in TE across sexes is difficult to interpret.
Direct comparisons of the efficacy of CGRP-based therapies in acute migraine treatment and in prevention between sexes are complicated by the differences in outcome measures used in these studies. Evidence of efficacy for acute migraine is based on the FDA required co-primary endpoints of pain freedom at two hours and freedom from most bothersome symptom at two hours with dosing at moderate to severe pain. In contrast, for migraine prevention, the primary endpoint is a reduction in monthly migraine days or monthly headache days (see Table 3). In addition, other factors may be considered that complicate direct comparisons of efficacy of acute and preventive therapy across sexes including the likelihood of different patient populations being evaluated (e.g. more low-frequency EM patients in acute treatment trials) and potential differences in the role of CGRP in initiating a migraine attack versus maintaining an already established migraine headache phase of the attack.
Despite difficulties in comparing efficacy of CGRP targeting therapies for use as acute or preventive treatments, some insights into potential sexual dimorphism in each of these approaches can be ascertained from the data. While individual studies for acute therapy were not powered to determine sex differences in outcomes, the analysis of pooled data failed to reveal evidence of effectiveness in men. However, the male sample size was 17.3% that of females and even the pooled data are under-powered for a modest population effect. For this reason, the lack of statistically significant results within individual studies for males is not dispositive. It should also be noted that the placebo response was higher in men than in women in some studies possibly contributing to the smaller drug effect observed in men. However, placebo response in men was not uniformly higher and small TEs were observed in men even in studies where the placebo effect was similar in men and women. Based on the low effect levels observed in these studies, properly powered studies to detect treatment effects in men would require very large sample sizes.
Comparison of point estimates between men and women across studies, however, does not rely on power within each study but rather on the consistency of results among studies. For acute therapy, gepant drugs were always more effective in women than in men raising the possibility of sex differences in the role of CGRP in maintaining an ongoing migraine headache. These outcomes suggest the need for further exploration and understanding of the underlying biology of CGRP across sexes in sustaining an established migraine headache. Differences in target engagement and pharmacokinetics of these drugs might contribute to the reported treatment effects for acute migraine in men and women. Dose-response curves for the gepants however are shallow suggesting that little therapeutic gain is obtained with higher doses of the same drug (i.e., ubrogepant; 50 mg, 100 mg); it is not clear that higher doses would have yielded better therapeutic results in men (Table 1). It should be noted, however, that dose-ranging studies with gepants for acute migraine may have been carried out predominately in women and it is possible that higher doses might be needed in men to produce appropriate receptor occupation. If true, this possibility would be consistent with sexual dimorphism for acute migraine therapy. Pharmacodynamic effects, such as the high protein binding and CYP3A4 metabolism of gepants are also possible explanations for the differences observed (19). An additional possibility is that associated symptoms of migraine are less frequent in men than in women (20). This would result in men having less most bothersome symptoms which would also decrease power of the analyses. It should also be noted that possible sexually dimorphic effects of CGRP-R antagonists in acute therapy cannot be compared to other drug classes such as triptans where differential effects are not observed between sexes (21). CGRP-R antagonists act by preventing CGRP signaling without influencing the actions of other neurotransmitters that may promote migraine pain and other symptoms. In contrast, triptans are 5HT1B/1D receptor agonists that can influence the release and actions of multiple neurotransmitters.
Analysis of outcomes from prevention studies reported in the CDER reviews for possible sex differences in EM patients with CGRP/CGRP-R targeting monoclonal antibodies and atogepant do not allow firm conclusions. In contrast, the data for CGRP/CGRP-R monoclonal antibodies were very similar in prevention in patients of both sexes with CM. A recent European collaborative study of 1154 women and 256 men did not observe a difference in response for erenumab at the 12-week time point (22). It should be noted that 75.1% of women and 66.0% of men in this evaluation had CM with an average of 17.1 ± 8.0 migraine days per month. The lack of sex difference in this study would be consistent with the conclusions from CDER analysis of antibodies for prevention in female and male CM patients. Additionally, recent real world registry data on approximately 5000 patients with high-frequency episodic migraine (HFEM) or CM treated with CGRP monoclonal antibodies also did not identify differences in responses at six months between men and women (23,24). Again, these conclusions are largely consistent with analysis from the CDER reports that support similar efficacy of preventive therapies in both men and women with CM. It should also be noted that the majority of participants in CM clinical trials may also have concomitant medication overuse. Preclinical studies have suggested that CGRP expression increases in the trigeminal ganglion in a model of medication overuse headache (25) supporting the possibility that the influence of CGRP may be similar in males and females with CM.
There are limitations in this analysis. This is a post hoc investigation. The limited data and power to detect an acute treatment effect of CGRP-R antagonists in males is also a limitation, which reflects a general limitation as well in the diversity of the participant populations in migraine clinical trial. An additional consideration is whether antibodies targeting the CGRP-R and those targeting the CGRP peptide might show differential outcomes when analyzed by patient sex.
In conclusion, the analysis of publicly available data suggests that (a) gepants are preferentially effective in women for the acute treatment of migraine; (b) based on data available at this time, there is no definitive evidence that supports an effect of gepants for acute migraine in men and (c) it is unlikely that there is a difference in the efficacy of CGRP targeting therapies for migraine prevention particularly in patients with CM. Collectively, these observations suggest that the contributions of CGRP to initiating a migraine attack and maintaining a migraine headache may be different and that the role of CGRP may evolve as migraine transitions from episodic to chronic states, especially as the coexistence of medication overuse increases in the CM population. These possibilities require additional studies to specifically address the question of sex differences for CGRP targeting medications both with regard to migraine subtypes as well as the target (peptide versus receptor) of the CGRP therapeutic. More broadly, these results highlight the importance of considering sexual dimorphisms in the evaluation of migraine therapeutics.
Key findings
For acute migraine treatment, small molecule CGRP-receptor antagonists (i.e., gepants) are effective in women; data available at this time do not demonstrate effectiveness in men. For migraine prevention, CGRP targeting antibodies or atogepant are effective in both men and women with chronic migraine; possible sex differences for patients with episodic migraine are inconclusive at this time. Future studies should be designed to determine efficacy of small molecule CGRP-receptor antagonists for acute migraine treatment in men.
Footnotes
Declaration of conflicting interests
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
