Abstract
Objective
To assess photophobia and allodynia in subjects with post-traumatic headache and examine how these sensory hypersensitivities associate with clinical measures of disease burden.
Background
Post-traumatic headache is the most frequent and disabling long-term consequence of mild traumatic brain injury. There is evidence of sensory dysfunction in acute post-traumatic headache, and it is known from other headache conditions that sensory amplifications correlate with more severe disease. However, systematic studies in post-traumatic headache are surprisingly scarce.
Methods
We tested light and tactile sensitivity, along with measures of disease burden, in 30 persistent post-traumatic headache subjects and 35 controls.
Results
In all, 79% of post-traumatic headache subjects exhibited sensory hypersensitivity based on psychophysical assessment. Of those exhibiting hypersensitivity, 54% exhibited both light and tactile sensitivity. Finally, sensory thresholds were correlated across modalities, as well as with headache attack frequency.
Conclusions
In this study, post-traumatic headache subjects with both light and tactile sensitivity had significantly higher headache frequencies and lower sensitivity thresholds to both modalities, compared to those with single or no sensory hypersensitivity. This pattern suggests that hypersensitivity across multiple modalities may be functionally synergistic, reflect a higher disease burden, and may serve as candidate markers of disease.
Introduction
Post-traumatic headache (PTH) is the most common and disabling consequence of mild traumatic brain injury (TBI) (1,2). Current International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria define PTH as new or worsening head pain attributed to head injury, beginning within 7 days; whereas continued head pain beyond 3 months is referred to as persistent PTH (3). Interestingly, while ICHD-3 stipulates temporal criteria for onset and persistence, it does not dictate criteria for frequency or clinical characteristics. Thus, PTH subgroups are typically classified using primary headache criteria for clinical and research purposes (4). This speaks to a lack of fundamental knowledge about PTH as a distinct entity, and the sensory features of patients with persistent PTH remain incompletely characterized.
Symptoms of increased light and tactile sensitivity (photophobia and allodynia) and signs of altered sensory function have been reported in PTH (5,6) and in fact, “migraine-like” PTH-related symptoms (including unilateral, throbbing headache with light and/or sound sensitivity) have been associated with a higher likelihood of persistent post-injury symptoms following acute injury (7). These observations have led to the hypothesis that PTH is generated by a combination of cranial, peripheral and central sensitization (5,8). These proposed mechanisms are corroborated by preclinical TBI models implicating sensitization of trigeminal and somatosensory pathways as pain generators (9,10). Importantly, sensory dysfunction may also herald more severe disease, and could serve as treatment targets in those with the greatest disease burdens.
The objective of this study was to examine quantitative sensory symptoms and psychophysical discomfort thresholds to light and mechanical touch within PTH subjects, compared to non-headache healthy control subjects. We present data showing significantly reduced sensory thresholds in PTH subjects, which correlate across multiple sensory modalities and with headache frequency.
Methods
Participants
In total, 65 subjects (40 female, 25 male) aged 15–67 years were recruited into two groups: Persistent PTH and non-headache healthy controls. Recruitment occurred between 2013–2019; subjects were recruited from the local community and University of Utah Headache and General Neurology Clinics, as well as via word of mouth, internet, and flyers. Institutional Review Board approval was obtained from the University of Utah Human Studies Committee (IRB_00085309 and IRB_00064447). All participants completed informed consent; for those < 18 years old, participant assent was paired with legal guardian permission.
Diagnosis of persistent PTH was based on ICHD-3 criteria, inclusive of those meeting criteria for persistent headache attributed to mild, moderate, or severe traumatic injury to the head (ICHD-3, 5.2.1 and 5.2.2 classifications) (3). Exclusion criteria for the persistent PTH group included: < 3 months since head injury; medication overuse headache; recurrent, disabling headaches other than mild, short-lived headaches related to muscle tension or caffeine withdrawal, that were present prior to the subjects’ head injury. Exclusion criteria for all subjects were: History of neurological or systemic disease affecting sensory function (e.g. peripheral neuropathy, multiple sclerosis, diabetes) and use of medication that might affect sensory function (e.g. tricyclic antidepressants, anti-epileptic agents, opioids).
We also subdivided the PTH group into “migraine-like” and “non-migraine-like” subgroups, based on their questionnaire-reported headache features (see Measurements, Questionnaire section below), which included: Intensity (mild, moderate, severe), location, sidedness (unilateral/bilateral), quality, temporal attributes, and aggravating/alleviating factors. In order to adhere to the highest standard definitions, we used ICHD-3 based criteria to classify each headache phenotype, though with the caveat that all PTH subjects did not meet the full criteria for any given primary headache phenotype, based on the key factor that they could be better accounted for by another ICHD-3 diagnosis (i.e. PTH under ICHD-3, 5.2). PTH subjects whose headaches included all features of definite migraine with or without aura were designated as “migraine-like” PTH, whereas those whose headache features were similar to ICHD-3 based “probable migraine” (ICHD 3, 1.5; migraine-like attacks missing one of the required features required to fulfil criteria for migraine with or without aura), cervicogenic and tension headache-like features were designated as “non-migraine-like” PTH.
Post-traumatic headache participants were generally tested after being acute headache attack-free for at least 48 h, though testing during non-acute, chronic daily headaches was permitted (i.e. any headache defined as “mild and non-throbbing” as a surrogate lay-terminology definition to avoid testing during acute, disabling headaches). Subjects had not used opiate medication or migraine-specific abortive medications during the 48 h prior to testing. The group of control subjects reported no history of recurrent, disabling headache, apart from mild, short-lived headaches related to muscle tension or caffeine withdrawal, and were studied in their usual state of health.
Measurements
Questionnaire: Subjects completed a written structured headache interview (12), modified to correspond with current ICHD-3 criteria (3), along with additional questions to report a first degree relative with history of migraine and characterize head injury history and attributes (adapted from Couch and Bearss (13)). Headache-associated sensory symptoms were assessed using the Short Form version of the Choi Photophobia Questionnaire (SF-PhotoQ) (14) and 12-item Allodynia Symptom Checklist (ASC-12) (15). Headache impact and affective symptoms were ascertained using the Headache Impact Test (HIT-6) (16), the 9-item Patient Health Questionnaire (PHQ-9; to measure severity of symptoms related to depression) (17), and the 7-item Generalized Anxiety Disorder tool (GAD-7; to measure severity of symptoms related to anxiety) (18).
Sensory testing: While some definitions use the term “photophobia” to refer to triggering and/or exacerbation of headache by light, we aim to measure light sensitivity that might also be present in healthy, non-headache controls, and in headache subjects, between acute headache attacks. Thus, for the purposes of this report, we use the term “photophobia” to refer to discomfort or pain induced by light (light sensitivity), but do not specifically limit this definition to the context of exacerbation of ocular or head pain. For this study, psychophysical assessment of light sensitivity thresholds was performed using our previously published methods (19). In brief, subjects were seated facing two white-light 500W halogen lamps (SL-1002; Bayco, Wylie, TX), shielded by a double-pane sheet of infrared-filtering glass (Solarban 60, ¼ in Clear; Oldcastle, Atlanta, GA) to mitigate heat exposure. Subjects underwent dark-adaptation for 3 min prior to testing. Using a rheostat calibrated by a luxometer (LX1010B Luxmeter), light intensity was increased in a stepwise fashion at 2-sec intervals from 0.1 lux to the patient-reported discomfort threshold, or a maximum of 16,450 lux. Subjects were asked to look at a fixed point between the light sources and to say “stop” when the intensity of light went from being simply “bright”, to “unpleasant, annoying or disagreeable” (e.g. the level of stimulus that made the subject want to blink or turn away from the light source, and physiologically confirmed by grimace) (20). Each subject completed three trials, with dark adaptation between each trial. Luxometer and rheostat levels were recorded from each of the three trials and averaged to determine participant light sensitivity threshold.
Pain thresholds to cutaneous mechanical (tactile) stimuli were tested using methods adapted from Burstein et al. (21) and Cooke et al. (22) Calibrated von Frey hair (VFH) filaments (Stoelting Co., Wood Dale, IL, USA) were applied sequentially in ascending order for 2 sec each over the subject’s periorbital and forearm skin regions. Standardized anatomic locations were used to guide testing as follows: Forearm testing was performed at midline of the volar side of the forearm, 8–10 cm above the wrist crease; supraorbital testing was performed 2 cm above the exit of the supraorbital branch of the trigeminal nerve (approximately the medial 1/3 of the eyebrow) on each side. The smallest VFH number capable of inducing pain at two of three trials was considered the threshold. Pain thresholds were recorded and expressed in units ranging from 11 to 20, representing the respective ascending forces of each numbered filament (4, 6, 8, 10, 15, 26, 60, 100, 180, and 300 g respectively). Due to the linear relationship between the log force and ranked number of each filament, tactile thresholds were analyzed and reported as their corresponding VFH numbers, rather than their forces (21,22).
Analysis and statistical methods
A priori group size power calculations were based on previously published data examining light (19) and tactile sensitivity (22) thresholds in chronic migraine and healthy non-headache control groups; we determined that sample sizes of 23 and 12 respectively would be needed to have 80% power to detect a significant difference between groups (alpha 0.05); thus, we selected a conservative minimum n of 30 for the primary between-group comparisons planned for this study.
In this study, in addition to psychophysical pain threshold identification, we also used a pre-defined quantitative definition of allodynia (21). Here, allodynia in the PTH group was defined as a pain threshold one standard deviation below that of the control group’s mean threshold for the testing site; all were down to the nearest VFH number unit to provide a conservative cut-off. Thus, individual patients in this study were identified as having allodynia to tactile stimuli overall if the average threshold of all four testing sites was 18 VFH or less (≤ 100 g); facial allodynia if the average threshold of the two peri-orbital sites was less than 17 VFH (≤ 60 g), and forearm allodynia when the average of the two forearm sites was less than 18 VFH (≤ 100 g).
Photophobia is defined as a perceived painful response to light (19). In this study, we used similar methods to those described for allodynia above: Using a pre-defined quantitative definition in which photophobia was defined as a discomfort threshold one standard deviation below that of the control group’s mean threshold. Thus, with a mean light sensitivity threshold in the control group of 2.6 and a SD of 0.8 log lux, photophobia was identified within individuals if their mean light sensitivity threshold was below 1.8 log lux.
Data distributions were visually inspected and tested for normality using the Shapiro-Wilk test; overall, the light sensitivity thresholds, SF-PhotoQ scores, and ASC-12 scores were found to be normally distributed, whereas the remaining parameters, including VFH thresholds and clinical data, were not. Normally distributed data was analyzed using Independent Sample t-tests; nonparametric data was analyzed using the Mann–Whitney U test. Results were considered significant for two-sided p-values ≤0.05, except where multiple comparisons occurred, in which Bonferroni correction was applied as appropriate. Multiple regression analysis, using the standard least squares method, was performed to evaluate the effect of the following covariates: Light sensitivity and VFH thresholds with anxiety (GAD-7) + depression (PHQ-9). Statistical analyses were performed with R for Windows (Version 3.5.2; R Core Team, Vienna, Austria) and JMP Pro® (Version 15.0.0, Windows; SAS Institute Inc., Cary, NC 2020).
Results
Subjects and clinical characteristics
There were no significant differences in age, sex, or reported family history of migraine between persistent PTH and healthy non-headache controls, though PTH subjects did report a greater affective symptom burden (see Table 1 for the clinical data summary). Of the 30 PTH subjects (all without history of recurrent disabling headaches or migraine prior to their head injury, per inclusion/exclusion criteria), 16 (53%) had a migraine-like PTH phenotype, and 14 (47%) had recurrent headache not meeting criteria for definite migraine-like headaches (including seven with probable migraine-like headaches, one with cervicogenic-like headaches, four with tension-like headaches, and two with non-specific headaches that could not be categorized) (3). Persistent PTH subjects with migraine-like headache phenotype had significantly higher depression symptom severity (PHQ) scores than those with non-migraine-like phenotype, though they otherwise did not differ in sex, age, or anxiety-related symptom scores.
Clinical characteristics.
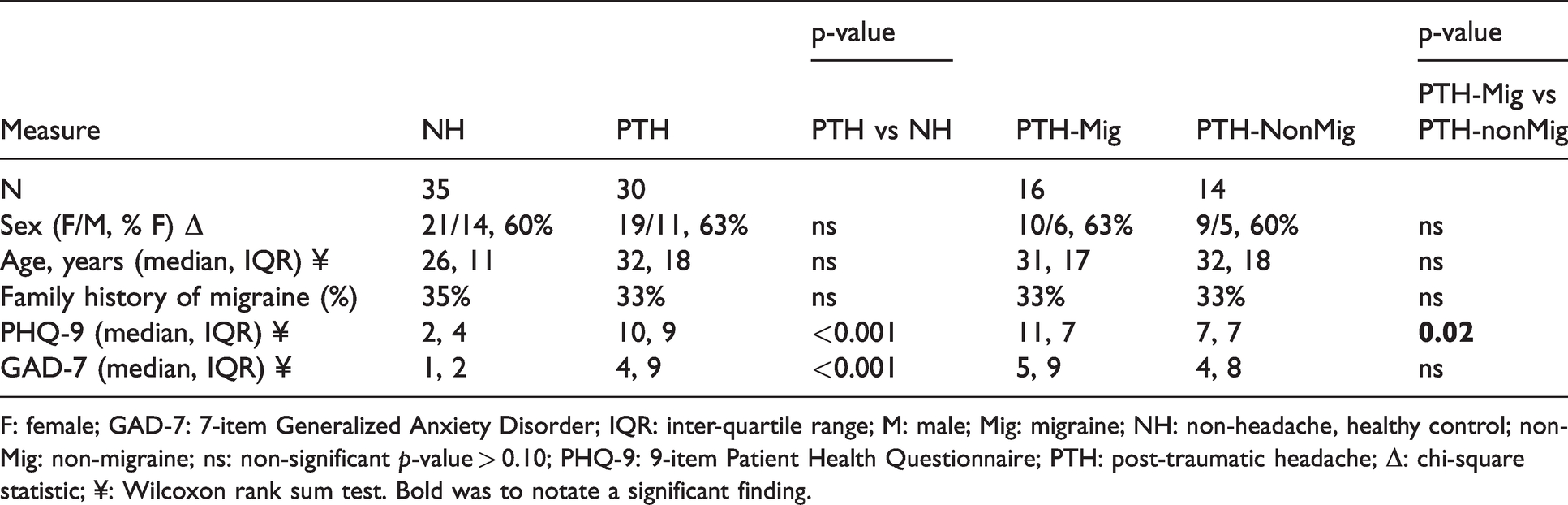
F: female; GAD-7: 7-item Generalized Anxiety Disorder; IQR: inter-quartile range; M: male; Mig: migraine; NH: non-headache, healthy control; non-Mig: non-migraine; ns: non-significant p-value > 0.10; PHQ-9: 9-item Patient Health Questionnaire; PTH: post-traumatic headache; Δ: chi-square statistic; ¥: Wilcoxon rank sum test. Bold was to notate a significant finding.
Headache duration since injury (at time of study evaluation) ranged from 3 months to 15 years, and the majority (83%) experienced mild TBI based on criteria adapted from Couch and Bearss (13). Types and severity of injuries are summarized in Table 2; five individuals reported a history of more than one head injury. Headache impact (HIT-6) scores were significantly higher in persistent PTH subjects with migraine-like headaches, compared to those with non-migraine-likeheadache (p = 0.02). Headache-associated photophobia (SF-PhotoQ) and allodynia (ASC-12) scores did not differ between PTH subgroups (p = 0.63 and 0.22 respectively).
Headache-related measures.
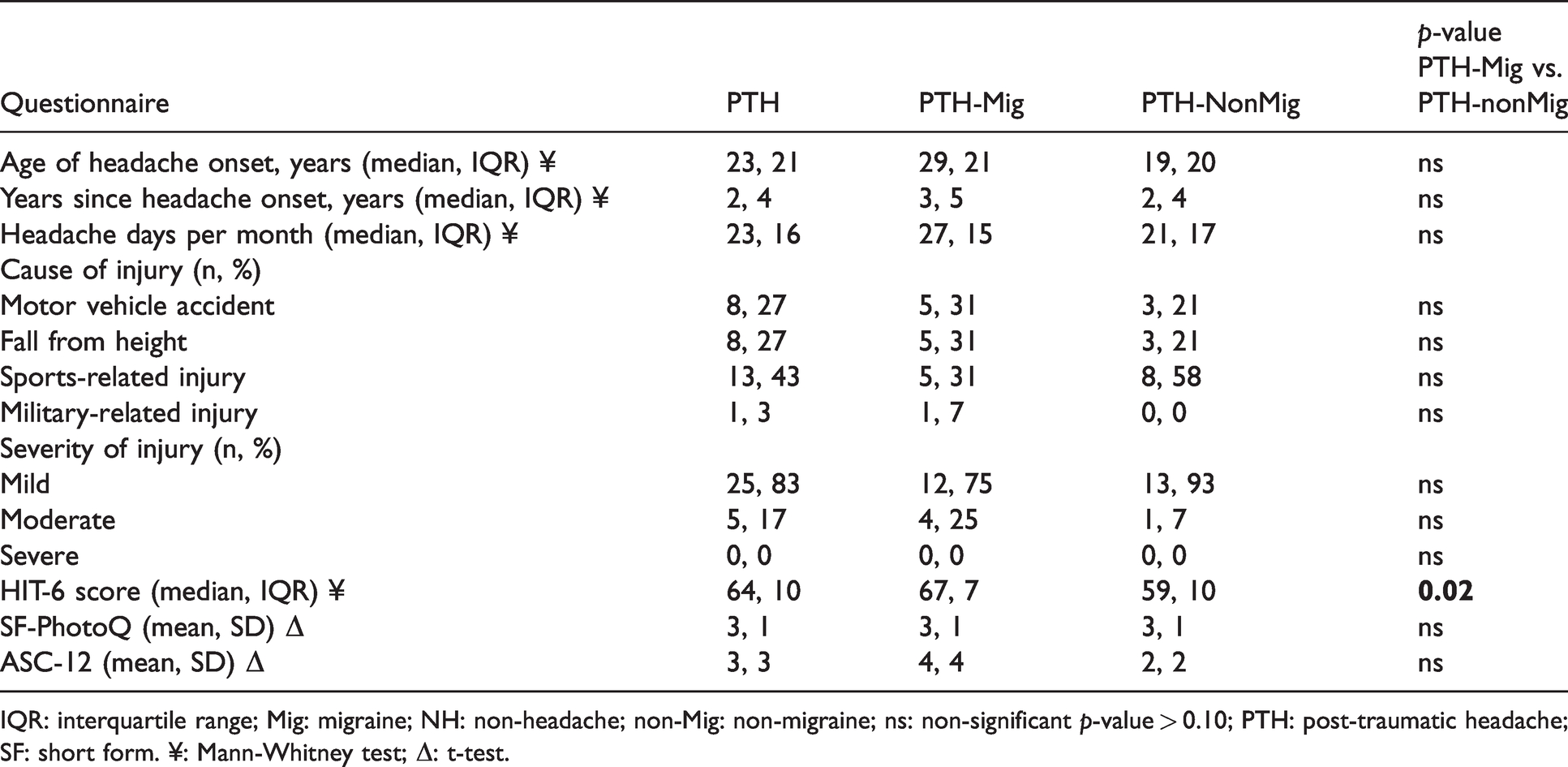
IQR: interquartile range; Mig: migraine; NH: non-headache; non-Mig: non-migraine; ns: non-significant p-value > 0.10; PTH: post-traumatic headache; SF: short form. ¥: Mann-Whitney test; Δ: t-test.
Both visual and tactile sensory thresholds were reduced in PTH compared to controls
Sixty-one percent of PTH subjects were photophobic (based on a mean light sensitivity threshold ≤ 1.8 log lux). Median light sensitivity thresholds were significantly lower in persistent PTH subjects, compared to controls (mean 1.5 log lux vs. 2.6 respectively; p = 0.0006). The significant difference in light sensitivity thresholds between the PTH and control groups remained significant after regression analysis controlling for GAD and PHQ scores (p = 0.0003); comparisons by headache subgroup maintained significance or trended toward significance (control vs. PTH-migraine-like, mean 2.6 vs. 1.7 log lux, p = 0.0002; control vs. PTH-non-migraine type, mean 2.6 vs. 1.9 log lux, p = 0.15; Figure 1(a)).

Light and mechanical touch thresholds were lower in PTH compared to controls. (a) Light sensitivity thresholds were lowest in persistent PTH subjects with migraine- headache phenotype (control vs. PTH-migraine type, mean 2.6 vs. 1.7 log lux, p = 0.0002; control vs. PTH-non-migraine type, mean 2.6 vs. 1.9 log lux, p = 0.15). (b)–(c) Whereas persistent PTH subjects with non-migraine type headache phenotype have the lowest touch sensitivity thresholds ((b) peri-orbital site, control vs. PTH-migraine type, median 19.3 vs. 17.3 VFH, p = 0.08; control vs. PTH-non-migraine type, peri-orbital median 19.0 vs. 19.0, p = 0.0005. (c) forearm site, control vs. PTH-migraine type, median 20.0 vs. 17.8, p = 0.14; control vs. PTH-non-migraine type, median 20.0 vs. 16.0, p = 0.002).
Sixty percent of persistent PTH subjects had allodynia overall (based on an average VFH threshold of ≤18; corresponding with a tactile sensitivity threshold of ≤100 g); 52% had facial allodynia (based on an average peri-orbital VFH threshold of ≤17; threshold of ≤60 g) and 60% had forearm allodynia (based on an average forearm VFH threshold of ≤18; threshold of ≤100 g). Periorbital and forearm median tactile pain thresholds were significantly lower in persistent PTH compared to controls (periorbital median 17.5 vs. 19.3 VFH; forearm median 17.5 vs. 20.0; p = 0.01 and 0.003 respectively). Again, differences in VFH thresholds between the PTH and control groups remained significant after regression analysis controlling for GAD and PHQ (peri-orbital site, p = 0.0008; forearm, p = 0.02). Subgroup comparisons also maintained significance or trended toward significance (control vs. PTH-migraine-like, peri-orbital median 19.3 vs. 17.3 VFH, p = 0.08; control vs. PTH-non-migraine type, peri-orbital median 19.0 vs. 19.0, p = 0.0005. Control vs. PTH-migraine-like, forearm median 20.0 vs. 17.8, p = 0.14; control vs. PTH-non-migraine type, forearm median 20.0 vs. 16.0, p = 0.002; Figure 1(b)–(c)).
Low visual and tactile sensory thresholds correlated with one another and were associated with higher headache frequencies
Low light sensitivity thresholds were positively correlated with low VFH thresholds in persistent PTH subjects, which was statistically significant at the forearm site, an extracephalic site (forearm, rho 0.52, p = 0.004; periorbital, rho 0.28, p = 0.09; Figure 2(b)–(c)). Low sensory thresholds inversely correlated with headache frequency, though this was only statistically significant for light sensitivity (light, Spearman’s rho −0.37, p = 0.03, Figure 2(a); VFH at forearm, rho −0.35, p = 0.08, and periorbital site, rho −0.16, p = 0.26; not shown). Finally, low light sensitivity thresholds were associated with ictal sensory symptom scores, though only photophobia-related scores reached statistical significance (SF-PhotoQ scores, rho −0.34. p = 0.04; ASC-12 scores, rho −0.30, p = 0.07; not shown). There were no significant relationships between light or VFH thresholds and other clinical parameters, including sex, age, GAD score, PHQ score, age at headache onset (injury), or HIT-6 (rho < 0.20 and/or p-value ≥ 0.20 for all associations).
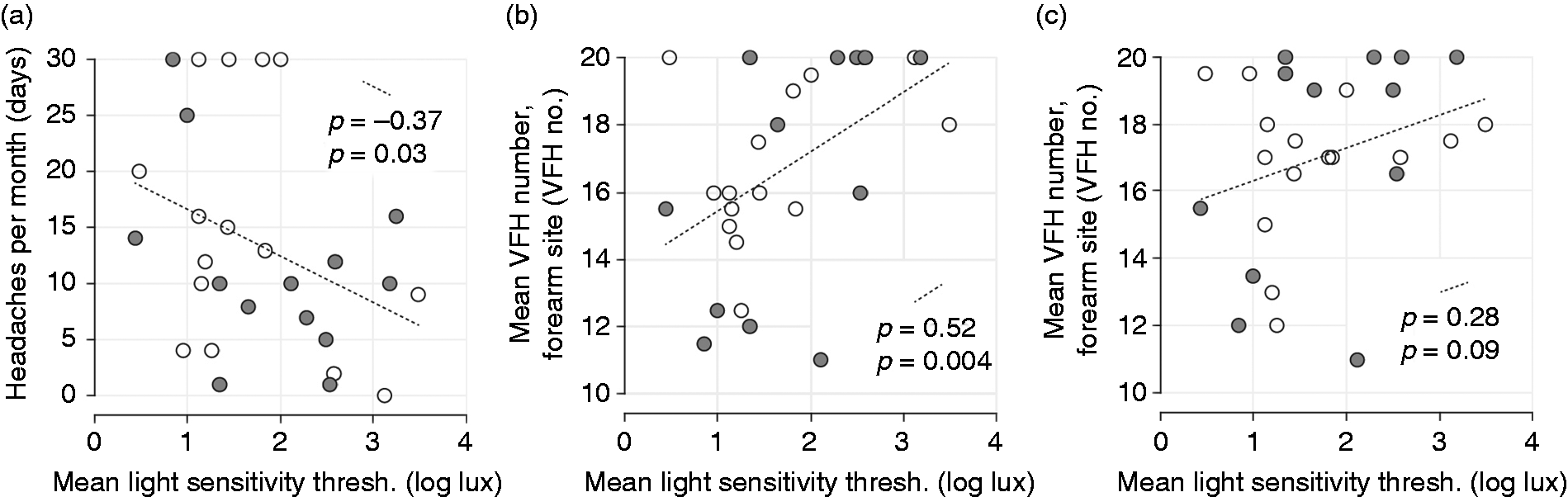
Sensory thresholds correlated between visual and tactile stimuli and were associated with headache attack frequency. Light and tactile sensitivity thresholds significantly correlate at the forearm (b) (rho 0.52, p = 0.004), though not as robustly at the periorbital site (c) (rho 0.28, p = 0.09). Light sensitivity thresholds were inversely correlated with number of headache days per month (a). Tactile sensitivity showed a similar relationship (not shown), though the correlations were not statistically significant (forearm rho −0.35, p = 0.08; periorbital rho −0.16, p = 0.26).
Visual and tactile sensory thresholds were lowest in PTH subjects with both sensory hypersensitivities
Seventy-nine percent of persistent PTH subjects exhibited sensory hypersensitivity to at least one of the two modalities based on psychophysical assessment, with 54% of PTH subjects exhibiting both photophobia and allodynia (25% exhibited hypersensitivity to a single modality and 21% exhibited no hypersensitivities). Individuals with both photophobia and allodynia showed significantly lower sensory sensitivity thresholds during both light and tactile testing (Wilcoxon rank sum test, light thresholds p = 0.004 and VFH threshold p = 0.001), as well as higher headache frequencies (median 30 days/month ± interquartile range of 7 days vs. 15.5 ± 19 days/month; p = 0.02; not shown), compared to those with single or no sensory hypersensitivity as shown in Figure 3(a)–(b). Further, on correlation analysis stratified by subgroup, we see that the positive correlation between sensory modalities noted across the entire PTH group (Figure 2(a)–(b)) appears to be primarily driven by those with both light and tactile hypersensitivities (Spearman’s rho 0.47, p = 0.05; single or no hypersensitivity, rho −0.09, p = 0.60; Figure 3(c)). Finally, SF-PhotoQ (photophobia) and ASC-12 (allodynia) scores, which predominantly reflect ictal sensory symptoms, were also higher in those with multiple sensory sensitivities, compared to persistent PTH subjects with single or no sensory sensitivity (light p = 0.03 and VFH p = 0.07; not shown). Sex, age, age of headache onset, headache type (migraine-like vs. non-migraine type) and HIT-6 did not differ significantly in those with multiple sensory sensitivities (both photophobia and allodynia) versus those with one or no sensory modalities affected (p > 0.20).
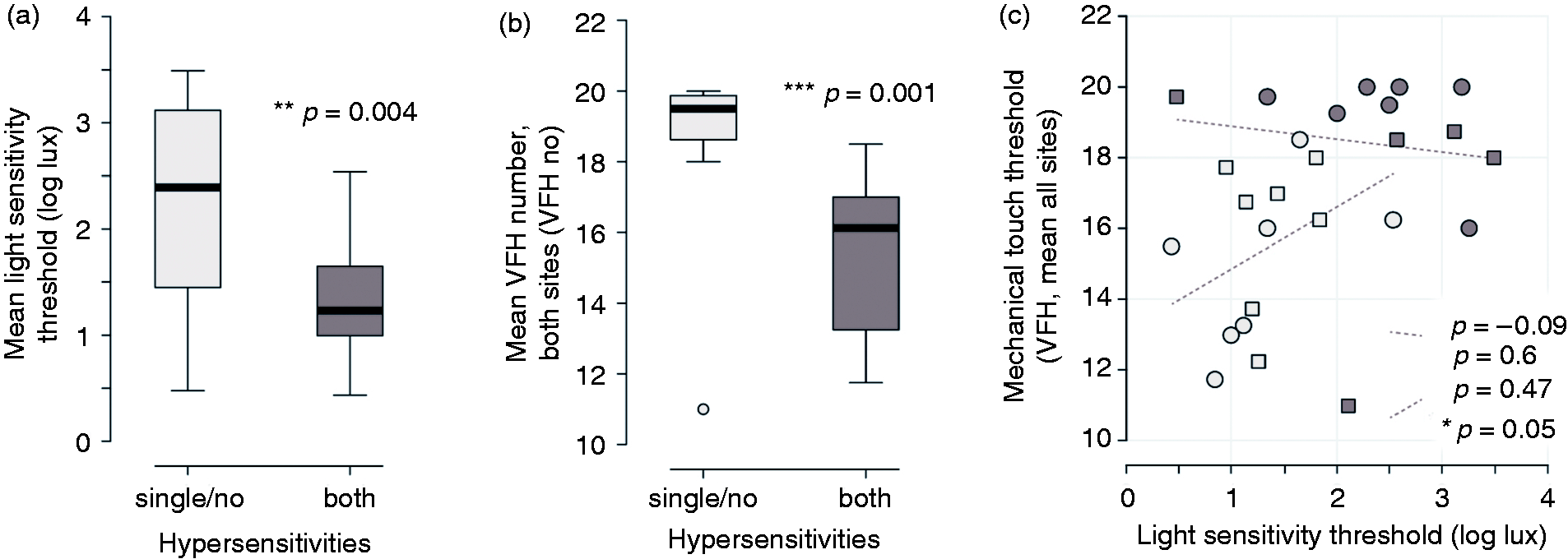
Light and tactile hypersensitivity appeared to be synergistic in post-traumatic headache subjects. Light (a) and tactile (b) sensitivity thresholds were significantly lower in PTH subjects with both sensory hypersensitivities. Further, those with both light and tactile hypersensitivities showed a significant positive correlation between sensory modalities, whereas this relationship was not apparent in those with single or no hypersensitivity (c).
Discussion
We examined sensory symptoms and psychophysical sensory thresholds to assess photophobia and allodynia in persistent PTH subjects compared to healthy non-headache controls (Figure 1). We found evidence of sensory sensitivity in 79% of persistent PTH subjects, as well as a correlation between low sensory thresholds and headache frequency (Figure 2(a)). These findings support both the clinical relevance, as well as the biomarker potential of sensory dysfunction in persistent PTH. We also found that sensory sensitivity involved both light and cutaneous mechanical touch in 54% of PTH subjects, where we saw significantly lower sensory thresholds to both modalities (Figure 3). Further, subjects with both sensory hypersensitivities also reported higher headache frequencies, with more severe headache-associated (ictal) symptoms of allodynia and photophobia. These latter associations raise the hypothesis that sensory hypersensitivity across multiple modalities may be functionally synergistic in persistent PTH, perhaps sharing a common central mechanism, rather than separate distinct afferent origins. To our knowledge, these data represent the first report of multisensory dysfunction in PTH as measured by both psychophysics and patient-reported measures of disease burden, providing evidence for multisensory sensitivity in persistent PTH subjects.
Visual and tactile sensory thresholds as indicators of disease burden
Photophobia and allodynia have been reported following head injury (23,24), though prior studies primarily focus on single sensory modalities and/or the ictal phase. Our findings also demonstrate that light and/or tactile hypersensitivity persist into the interictal phase (i.e. the period in which the subject is free of an acute headache attack) in a majority of persistent PTH subjects, and provide systematic confirmation of the anecdotally observed multisensory sensitivity in this patient population. Further, those with multisensory hypersensitivity reported the lowest sensory thresholds overall (Figure 3), as well as higher headache attack frequencies, and more severe headache-associated (ictal) symptoms of allodynia and photophobia (per ASC-12 and SF-PhotoQ scores respectively). These latter findings raise the question as to whether multiple sensory hypersensitivity in PTH may heighten attack-based sensory dysfunction, perhaps through mechanisms of interictal sensitization of respective nociceptive pathways (25). Finally, given the high frequency of headaches in the group with the lowest sensory thresholds (Figure 3(a)–(b)), one must also consider the possibility of continuous, chronic generalized sensitization wrought by near-daily headaches in the most severely impacted PTH subjects in our study (8,26).
While it is well established that PTH may present clinically as a variety of primary-like headache phenotypes (4,27), several clinical characteristics of PTH-related pain have led to wide use of the term “post-traumatic migraine” (typically defined as post-traumatic onset, unilateral throbbing headache, associated with light and/or sound sensitivity). Indeed, many studies (including this one) rely on shared clinical characteristics in order to classify PTH subjects based on their clinical headache subtype, though definitions of “migraine”-like headache differ notably across studies (5,27–29). Using this approach, prior investigators have reported a migraine-like phenotype in a majority of persistent PTH subjects, ranging from 60–97%, depending on the study population and methodology, with tension-type headache and migraine-like ± combined tension-type headache comprising the next most common phenotypes (27,29). These estimates are comparable to the present study, where 53–77% exhibited migraine-like headaches (depending on whether strict criteria for episodic and chronic migraine-like headaches were applied, or “probable migraine”-like PTH was included). Interestingly, “migraine-like” headache-related symptoms have been associated with a higher likelihood of persistent post-injury symptoms following acute injury (7). With this in mind, we chose to study de novo PTH subjects, without known history of pre-injury, recurrent headache or migraine. Notably, despite the exclusion of those with pre-existing headache, and inclusion of a wide range of headache frequencies (1–30 per month; Figure 2(a)), sensory thresholds in our PTH group were reduced to a similar degree to those published in chronic migraine subjects (22). For comparison, prior studies in chronic migraine (≥ 15 headache days/month) report mean light sensitivity thresholds of 2.2 log lux and average tactile sensitivity thresholds of 16 VFH (≤26 g) (19,22).
Finally, of additional interest, we see that PTH subjects across both study groups exhibited low sensory thresholds, in the range reported in chronic migraineurs (19,22). Specifically, Figures 2 and 3(c) show PTH subjects with both migraine-like and non-migraine type headaches with sensitivity thresholds below 2.2 log lux and 16 VFH, and that low thresholds are even present in PTH subjects with fewer than 15 headache days/month. Here, we hypothesize that sensory dysfunction in PTH may be a result of persistent post-injury sensitization (9,30,31), rather than emerging as a migraine-specific phenomenon. These data highlight the potential utility of sensory dysfunction as both a clinically relevant, and perhaps temporally differentiating, marker of disease in PTH, which could be used to understand PTH across the clinical disease spectrum (e.g. acute and persistent, migraine-like and not).
Hypersensitivity to light and touch appear to be synergistic in PTH
Both interictal photophobia and extra-cephalic allodynia are widely considered to be clinical signs of central sensitization – a change in centrally mediated nociceptive processing that occurs with pain chronification in both headache and extracephalic pain conditions (32). Their co-occurrence, as well as apparent synergistic relationship (Figure 2(b)–(c) and Figure 3), is consistent with centrally mediated sensory circuit dysfunction in persistent PTH (6). Though independent peripheral sensitization of both visual and extracephalic tactile circuits can certainly not be ruled out, work in animal models suggests that even for single sensory modalities (e.g. trigeminal) changes in central nociceptive response occur (9,33), whereas when multisensory phenotypes are involved, the role of central network dysfunction is arguably much more likely (26).
Sensory hypersensitivity as a clinical indicator
In the clinical setting, sensory features in PTH are both prominent and clinically actionable (34) aspects of headache-related disease burden. Importantly, our data highlights the presence of multimodal sensory hypersensitivity in non-migraine type, as well as in migraine-like, PTH subjects (Figures 2 and 3(c)). In other words, the multisensory sensitivity of PTH appears generalizable, and thus could be considered as a disease indicator across multiple clinical subtypes, which may not be strictly linked to a migraine-like phenotype in which ictal sensory sensitivity is a presumed hallmark feature. Two potential limitations of the current study is our focus on persistent PTH (headache beyond 3 months post-injury) and our inclusion of moderate TBI subjects, which prevented us from exploring differences in multisensory hypersensitivity based on PTH acuity and trauma severity. Given that the presence of headache itself (7,35) and sensory hypersensitivity (7,35,36) have been shown to be predictive of prolonged recovery from acute TBI, we suggest that future studies might explore early identification of sensory dysfunction in acute TBI (inclusive of mild, moderate and severe TBI injuries), with the potential that it may serve as a marker of risk for headache persistence – perhaps leading to targeted mitigation strategies that reduce progression to chronic post-traumatic sensory dysfunction and headache (37).
Limitations
The cross-sectional design of the current study restricts our ability to determine whether the signs/symptoms of sensory dysfunction (i.e. mechanical or light hypersensitivity) were present prior to a given subjects’ head injury, or rather developed as a consequence. However, we chose to exclude patients with pre-existing migraine in an effort to minimize confounding from premorbid primary headache. Although our recruitment focused on persistent PTH subjects who suffered mild TBI > 3 months prior, our cohort included a very wide range of chronicities, and thus we are not able to directly examine whether sensory dysfunction differs as a function of time. As a surrogate, we demonstrated that sensory measures do not significantly correlate with duration since injury (Spearman’s rho −0.3, p = 0.22).
In addition, a byproduct of our attempt to include a broad range of PTH subjects, was the lack of a true “interictal” PTH sample. While we did require headache subjects to be “attack-free of their acute headaches for at least 48 hours”, sensory testing during non-disabling, chronic daily headaches was permitted. In our sample, 10 PTH subjects reported some type of daily headache (seven PTH-Mig and three PTH-NonMig). In this case, all subjects were tested on a day > 48 h since their last “migraine-like” or disabling, acute headache, and where their current daily headache was considered “mild and non-throbbing.” Future studies might provide further phenotypic clarification by testing both true interictal and ictal phases, as well as comparing those with truly episodic versus chronic daily headaches.
Affective and post-traumatic distress related symptoms are a prominent and widely associated, clinical consideration in post-TBI and chronic pain care (38). For this study, we used the GAD-7 to measure anxiety-related symptoms, which has been validated in chronic headache disorders (18) and is reasonably sensitive and specific for PTSD-related anxiety (39). Our study supports prior reports with higher depression and anxiety scores in PTH subjects compared to controls, and while we are unable to examine possible temporal interactions of affective symptoms and sensory sensitivity, our regression analysis suggests that affective symptoms are unlikely to fully account for the differences in sensory thresholds between PTH and controls shown here.
Summary and conclusions
We show evidence of multisensory dysfunction in greater than 50% of persistent PTH subjects. We also show that light and tactile sensitivity not only correlate with each other, but appear to be functionally synergistic, in that having both sensory amplifications was associated with the highest headache attack frequencies, greatest ictal sensory symptom burden, and lowest sensory thresholds overall. The correlated, apparently additive nature of sensory amplifications, and their scaling with measures of disease severity, make it appealing to hypothesize that a central process is involved and prompt further pathophysiological investigation into the underpinnings of this multisensory gain in PTH. On a clinical basis, the correlation of multisensory dysfunction with attack/disease severity also suggests the use of sensory amplifications as potential markers of disease, risk of chronification, and treatment response.
Article highlights
Greater than 50% of persistent post-traumatic headache subjects exhibit sensory hypersensitivity to both light and tactile stimuli. Post-traumatic headache subjects with both light and tactile sensitivity have significantly higher headache frequencies compared to those with single or no sensory hypersensitivity.
Footnotes
Acknowledgements
The authors would like to thank Jeremy Theriot for figure preparation.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MMC reports consulting activities unrelated to this paper or topic of research from Eli Lilly. LM, CS, and NR report nothing to disclose. KCB reports consulting activities unrelated to this paper or topic of research from Eli Lilly and Allergan.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH NIMHD LRP and the American Academy of Neurology Clinical Research Scholarship (MMC); American Academy of Neurology Medical Student Summer Research Program (NAR); Fairclough Endowment for Headache Research (LM); NIH NINDS R01 NS 102978, 104742 (KCB).
