Abstract
Background:
Headache (HA) following traumatic brain injury (TBI) is common, but predictors and time course are not well established, particularly after moderate to severe TBI.
Methods:
A prospective, longitudinal cohort study of HA severity post-TBI was conducted on 450 participants at seven participating rehabilitation centers. Generalized linear mixed-effects models (GLMMs) were used to model repeated measures (months 3, 6, and 12 post-TBI) of two outcomes: HA density (a composite of frequency, duration, and intensity) and HA disruptions to activities of daily living (ADL).
Results:
Although HA density and ADL disruptions were nominally highest during the first three months post-TBI, neither showed significant changes over time. At all time points, history of pre-injury migraine was by far the strongest predictor of both HA density and ADL disruptions (odds ratio (OR) = 8.0 and OR = 7.2, averaged across time points, respectively). Furthermore, pre-injury non-migraine HA (at three and six months post-TBI), penetrating-type TBI (at six months post-TBI), and female sex (at six and 12 months post-TBI) were each associated with an increase in the odds of a more severe HA density. Severity of TBI (post-traumatic amnesia (PTA) duration) was not associated with either outcome.
Conclusion:
Individuals with HA at three months after moderate-severe TBI do not improve over the ensuing nine months with respect to HA density or ADL disruptions. Those with pre-injury HA, particularly of migraine type, are at greatest risk for HA post-TBI. Other independent risk factors are penetrating-type TBI and, to a lesser degree and post-acutely only, female sex. Individuals with these risk factors should be monitored and considered for aggressive early intervention.
Background
Headache (HA) is the most frequent chronic pain problem in persons with traumatic brain injury (TBI) (1,2). Published prevalence rates of HA among patients with TBI vary widely, ranging from 18% to 93% depending on sample makeup and identification methods (3–6). The diagnosis of post-traumatic headache (PTHA) is applied when there is onset of new HA or worsening of pre-morbid HA severity temporally related to TBI. In the current classification of headache disorders, the ICHD-2 (International Classification of Headache Disorders, revised), PTHA is considered a secondary rather than a primary HA disorder (7). Despite its high prevalence, the predictors for PTHA are far from established, so it is not clear why some individuals develop PTHA and why others do not.
One notable gap in the literature on PTHA is that cohort studies have overwhelmingly consisted of persons with mild TBI and the associated post-concussion disorder (8–13). In mild TBI samples, there is compelling evidence that female gender and history of mood disorders are predictive of PTHA. Furthermore, one study identified an association between scalp lacerations and PTHA (14). Several prior investigations have studied severe or mixed severity TBI cohorts but have significant methodological limitations regarding subject selection and/or lack of prospective or well-designed HA outcome measures (15–18). More recently, Hoffman et al. (19) and Walker et al. (20) prospectively studied large moderate-severe TBI cohorts using well-defined quantitative HA measures for the first one to two years after injury. Hoffman and colleagues showed an association between HA and female gender in a civilian sample (19), while Walker and colleagues showed an association between HA and mood disorders (20) in a military and veterans sample that was overwhelmingly male.
One intriguing potential predictor of PTHA, the severity level of TBI, has received considerable attention in past investigations. Several investigators have reported more HA in mild versus severe TBI, suggesting an inverse relationship between TBI severity and PTHA. However, these samples were largely biased by inclusion of only patients presenting to a tertiary-care outpatient clinic. Moreover, De Benedittis and De Santis (21) reported the opposite, showing worse HA symptoms in patients with longer periods of lost consciousness and post-traumatic amnesia (PTA), respectively. Neither Hoffman et al. (19) nor Walker et al. (20) found a relationship between injury severity and HA after TBI in their samples. Surprisingly, the type of TBI, namely penetrating versus closed, has been largely ignored in PTHA predictor studies, probably because previous studies have been limited by sample sizes of participants who have sustained a penetrating TBI.
In summary, little can be generalized from the existing literature and much is to be learned about predictors of PTHA, especially for persons with moderate and severe TBI. Accordingly, the objectives of the current study were to determine predictors and assess for change over time in PTHA density and PTHA functional impairment within a large cohort of patients with moderate-severe TBI. These objectives are an extension of the recently published study by Hoffman et al. that analyzed potential associations with HA (presence versus absence) within the same cohort of patients. It adds to this previous study by using more responsive and clinically meaningful outcome measures: 1) HA density as a summary score of HA activity and levels over time, and 2) the impact of HA on activities of daily living (ADL). HA density is a composite of frequency, duration, and intensity of all HA episodes occurring during a time period and represents the dimension of HA impact that is typically assessed in clinical trials (22,23). Given the commonality of HA within the general population, HA density represents a more discriminating and clinically relevant outcome compared to the presence or absence of any HA. Although best obtained by prospective daily HA recordings (e.g. HA diary), it can also be measured by obtaining self- (or surrogate-) reported typical HA episode frequency, duration, and intensity over a specified retrospective time frame.
This study’s hypotheses are that the predictive factors found in the literature, including history of HA/migraine HA and being female, are associated with increased HA density and negative impacts on ADL, including their change over time. We also examine whether other demographic and injury-related factors are associated with HA density and impact on ADL. A relatively understudied potential predictor, type of TBI, is of particular interest given our experiential evidence that PTHA is more common and severe after penetrating versus closed TBI.
Methods
Participants
The Natural History of HA study was developed as a module of the larger overarching TBI Model Systems (TBIMS) cohort study and was conducted at seven participating centers: Virginia Commonwealth University (Richmond, VA), University of Washington (Seattle, WA), Craig Hospital (Englewood, CO), Mayo Clinic (Rochester, MN), University of Alabama at Birmingham (Birmingham, AL), University of Texas Southwestern Medical Center (Dallas, TX), and Moss Rehabilitation Research Institute (Philadelphia, PA). Recruitment for the HA study occurred in tandem with the TBIMS study and enrolled subjects during acute inpatient rehabilitation hospitalization following TBI. There were 450 participants with TBI who consented and were consecutively enrolled into the HA study between February 2008 and June 2009. Participants completed follow-up assessments by July 2010.
HA study inclusion criteria were the same as for the TBIMS whereby participants: 1) presented to the designated TBIMS acute-care hospital within 72 hours of injury; 2) received both acute medical and acute rehabilitation care within the same system; 3) had sustained a TBI with at least one of the following characteristics: Glasgow Coma Scale (GCS) score <13 on emergency admission (not due to intubation, sedation or intoxication); loss of consciousness >30 minutes (not due to sedation or intoxication); PTA > 24 hours; or trauma-related intracranial abnormality on neuroimaging; and 4) were aged 16 years or older. Participants were excluded if they were incarcerated or did not have telephone access. Informed consent was obtained as approved by the institutional review boards of all seven institutions. If a subject was unable to consent because of persistent confusion, proxy consent was obtained from the legally authorized representative.
Procedures
Longitudinal data were collected on HA symptoms and HA treatment by trained research assistants using fully structured interviews. Patient (self-reported) information was the primary source of data. Proxy respondents completed more limited interviews when participants were either cognitively unable to respond validly or were unavailable during the interview windows. Initial interviews were conducted prior to or within one week after discharge from inpatient rehabilitation. Follow-up interviews were conducted over the telephone at three, six, and 12 months after injury. The 12-month interview occurred in conjunction with the planned TBIMS follow-up. Of the 450 participants enrolled, follow-up rates were 84% at three months, 90% at six months, and 89% at one year post-injury. Questionnaires completed by proxy occurred with 80 patients at initial evaluation, 61 at three months, 71 at six months, and 51 at 12 months post-injury. A comparison of proxy to participant responses found no significant difference in report of HA (vs. no HA) pooled over time, but under-reporting of HA at three and six months post-injury by proxies compared to participants with TBI.
HA density outcome variable
Headache density definitions.
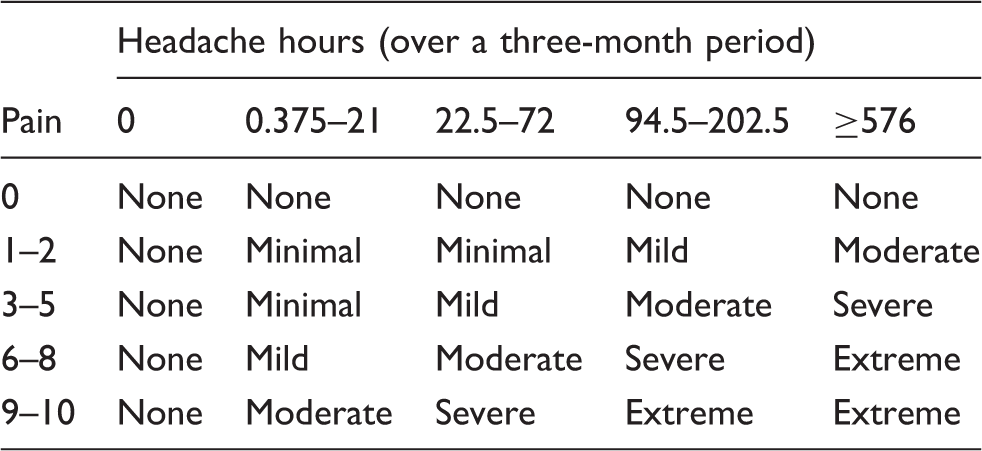
The following are three actual patient examples:
Patient A: Has HA several times a week lasting four to <24 hours with a pain rating of 8 out of 10 → Extreme Patient B: Has HA once a week lasting 30 minutes to <four hours with a pain rating of 5 out of 10 → Mild Patient C: Has HA every day lasting 30 minutes to <four hours with a pain rating of 3 out of 10 → Moderate
Lastly, an expert physician reviewed a random sample of individual subject data to ensure the density category they were assigned by this procedure had a high level of face validity.
HA functional limitations outcome variable
Functional limitations were measured on a five-level ordinal scale representing the degree to which HA interfered with ADL, “How often do HAs limit your ability to do usual daily activities including household work, work, school, and social activities?” Response options were Never, Rarely, Sometimes, Very Often, and Always.
Predictor variables
The primary demographic predictor of interest was sex (male or female) and the primary pre-injury characteristic was pre-injury history of HA. The pre-injury history of HA variable asked the participants if they had a “problem with HAs” prior to injury. For those responding yes, pre-injury HA problem was categorized as migraine versus non-migraine type based on the subjects’ response to “Have you ever (before injury) been given the diagnosis of migraine by a qualified health care provider?” In addition, we also examined presence of any skull, spine, and/or facial fractures found on computed tomography (CT) scans due to injury (yes or no), type of TBI (closed or penetrating), and severity of TBI (ordinal categories). The category of penetrating TBI was not a defined variable in the TBIMS database so it was extrapolated from having either a gunshot wound injury mechanism and/or skull fragments on imaging. The ordinal categories for severity of TBI were empirically guided ranges of PTA: moderate (one to seven days), severe (one to four weeks), or very severe (>four weeks) (24,25). There were 43 participants whose duration of PTA could not be categorized (still in PTA at inpatient discharge and under four weeks post-injury) so their severity scores were coded as missing. Of note, any subjects enrolled having mild TBI by PTA criteria (<24 hours) were assumed to have a “complicated” mild TBI and therefore grouped together with moderate TBI. The rationale is that because of the inclusion criteria for the overarching TBIMS study, these cases are not representative of mild TBI patients at large, who are rarely admitted for hospitalization at all, let alone inpatient rehabilitation following acute hospitalization.
Statistical methods
Generalized linear mixed-effects models (GLMMs) were used to model the repeated measures (three, six, and 12 months post-TBI) over time, separately for each outcome measure. GLMMs were chosen to model these data because they can (a) incorporate within-subject correlations due to the repeated measures over time, (b) appropriately model ordinal response data, and (c) model both time-varying and time-independent covariates or factors. These models assumed a cumulative logit link and a multinomial distribution for the outcome variables due to the ordinal nature of the outcomes. The models each included fixed-effects for post-injury time period (time-varying factor), the predictors of interest (time-independent covariates and factors), and the predictor by follow-up period interactions (time-varying covariates and factors), as well as random-subject effects to account for within-subject covariability in repeated measures. Estimate statements were used to test for the associations between the factor of interest and the severity of HA density and HA functional impact limitations at each follow-up month as well as to capture the pattern of significant modifiers of the changes in severity and limitations over time when necessary. Odds ratios (ORs) for the 95% confidence limit (CLs) were used to describe the effect sizes. The odds were modeled to represent the odds of having a more severe HA than a less severe HA for the HA density outcome and to represent the odds of having more limitations than fewer limitations for the function limitation outcome. Odds ratios then represent the odds of a more severe HA (or more severe limitations) for one time point (or sub-group), as compared to another time point (or sub-group). The
Results
Average age for the sample was 43.0 years (standard deviation (SD) = 19.2, range = 16–88) with the majority of participants being male (71%), white (74%), not married (65%), and having at least a high school level of education (59%). Etiology of injury was primarily due to motor vehicle accidents (53%), with falls comprising 27% and violence another 10%. Additional etiologies (10%) were sports related, hit by a falling object, or other causes. Median time in acute care was 17 days (interquartile range (IQR) = 9–28), median time to follow commands was two days (IQR = 0.5–7), and median time of PTA was 17 days (IQR = 6–31). Penetrating injuries were present in 6% of the sample, 62% exhibited a skull, spine, and/or facial fracture, and 3% reported difficulty with pre-injury ADL due to HA. Most had no history of HAs pre-injury (82%), with 13% and 5% reporting pre-injury non-migraine and migraine HAs, respectively.
Density model
The final HA density model with all covariates had a sample size of 344 (out of 450) participants. The reduction in sample size was due to (1) missing or incalculable outcome variable (reduction to
Distribution (percentage) of headache density severity categories by time (over a three-month period).

After adding the set of predictor variables and their interactions with time into the model (
Odds ratios from adjusted headache density model, irrespective of time.

OR: odds ratio; CL: confidence limits; HA: headache; TBI: traumatic brain injury; PTA: post-traumatic amnesia. aIndicates statistical significance at α = 0.05.
Odds ratios from adjusted headache density model by time (

OR: odds ratio; CL: confidence limits; HA: headache; TBI: traumatic brain injury; PTA: post-traumatic amnesia. aIndicates statistical significance at α = 0.05; CLs with 1 on the boundaries may or may not be statistically significant depending on rounding (i.e. <1.00 vs. >1.00).
Functional limitation model
The final functional limitation model had a sample size of 366 (out of 450 participants). The reduction in sample size was due to (1) missing outcomes at all follow-up occasions (reduction to
Distribution (percentage) of functional limitation scores by time.
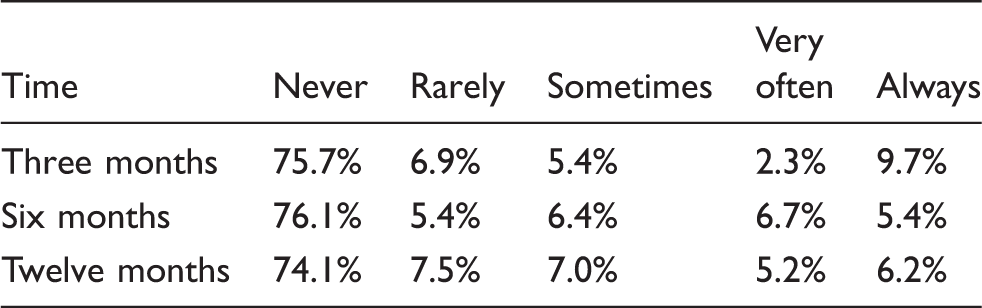
After adding the set of predictor variables and their interactions with time to the model (
Odds ratios from adjusted ADL model, irrespective of time.

ADL: activities of daily living; OR: odds ratio; CL: confidence limits; HA: headache; TBI: traumatic brain injury; PTA: post-traumatic amnesia. aIndicates statistical significance at α = 0.05.
Odds ratios from adjusted ADL model by time (

ADL: activities of daily living; OR: odds ratio; CL: confidence limits; HA: headache; TBI: traumatic brain injury; PTA: post-traumatic amnesia. aIndicates statistical significance at α = 0.05; CLs with 1 on the boundaries may or may not be statistically significant depending on rounding (i.e. <1.00 vs. > 1.00).
Discussion
It is widely believed that earlier targeted treatment can reduce the rate and severity of chronic HA after TBI (26). Establishing predictors of persisting HA should help guide the clinical care of individuals with TBI. If clinically significant HA can be more effectively identified and treated after TBI, long-term disability and morbidity may be avoided or reduced. Several conclusions can be drawn from this study that refine knowledge on predicting HA after moderate-severe TBI.
Firstly, we demonstrated that pre-injury history of HA is by far the most significant predictor of HA severity (both density and functional limitation) following moderate and severe TBI. The association was particularly robust when the pre-injury HA was categorized as migraine-type HA. These finding corroborate those of Hoffman et al. (19) that the presence of pre-injury HA is a predictor for HA following TBI, and of Lucas et al. (27) that pre-injury migraine-type HA is a predictor for migraine-subtype HA after TBI; but provide additional information on the strength of their correlation. The implications from this are that a significant proportion of HA reported after TBI may be a continuance of a chronic HA condition (i.e. not meeting diagnostic criteria for PTHA). It should be pointed out that those reporting pre-injury HA may still meet diagnostic criteria for PTHA if their post-injury HA is significantly worse than pre-injury (e.g. an exacerbation of premorbid HA condition). Unfortunately, quantitative pre-injury measures were not available in the data in order to make this comparison. Regardless of the diagnostic label, these findings nonetheless emphasize the strong prognostic significance that pre-injury history of HA has on HA problems post-TBI.
Sex was also found to be a significant predictor of HA severity after TBI, where females are more susceptible to worsened outcomes. In the earlier study from the same sample by Hoffman and colleagues, an association with HA prevalence was found, whereas in the current study the correlation was found with higher density of HA and for worse functional limitation at certain time points. Although in both cases the ORs for females were small in comparison to those for pre-injury history of HA, our findings clearly indicate that HA after TBI is more problematic for females compared to males. Interestingly, sex was also a modifier of changes in functional limitations from HA over time. At early time points females and males are not different with respect to functional limitations. However, females become more susceptible to having HA that limits function as the first post-injury year progresses. Specifically, the OR comparing females to males increased from 0.88 at three months where males were nominally worse, to 1.50 at six months and 2.64 at 12 months post-injury, where females had greater odds of limitations. This indicates that being female may have greater influence on the persistence of PTHA compared to the onset of PTHA. Either way, this study corroborates the mounting evidence that female gender is associated with PTHA, not only after mild TBI but also moderate-severe TBI.
Within the same cohort as the current study, Hoffman (19) previously showed that the period prevalence of HA is static (41%–44%) over the first year after TBI. In the current study we hypothesized that even though the rate of patients reporting HA does not change, the severity of HA among them diminishes over time. Disappointingly, neither HA density nor limitations from HA changed over time during the first year after TBI. In the unadjusted model, there was only a trend (
In a novel finding, this study demonstrated that a penetrating type of TBI, as categorized by gunshot mechanism and/or skull fragments on head CT, is associated with worse HA after TBI. The large sample size and the use of a responsive quantitative outcome measure of HA allowed us to examine the effect of this previously underexplored predictor. The odds of greater HA density was in the range of two to four times greater for those with penetrating vs. closed TBI. Additionally, significantly greater functional limitation from HA was found for those with penetrating type TBI irrespective of time, and particularly at six months post-injury.
Another finding in this study was a questionable early association of skull/face/cervical fractures with early HA after TBI. The main effects and the interactions with time for skull/facial/cervical fractures were not significant in either model. However, it was suggestive that significantly greater ADL limitations were present at three months post-injury in those with skull, facial, or cervical fracture(s). There was not a corresponding significant (or even trend toward) increase in HA density for this variable. This raises the possibility that poorer functional limitation was present but was perhaps mischaracterized by participants as being due to HA.
Lastly, we clearly demonstrated that severity of TBI as measured by PTA duration is not significantly associated with post-injury HA in our sample. Severity was the only independent variable that was neither a significant nor a marginally significant predictor in either outcome model. This provides further corroboration to the mounting evidence that there is not likely an inverse relationship between TBI severity and PTHA that some have claimed.
The strengths of this study were its 1) large sample size, 2) prospective longitudinal design, 3) use of multivariable ordinal logistic regression models, and 4) use of responsive and clinically meaningful outcome measures. The chief limitation was that pre-injury HA was determined by self-reported retrospective recall, and that although post-injury HA data were collected in prospective fashion, recall over up to a three-month period rather than a daily HA journal was used to record HA information. These types of recalled data may be influenced by bias and/or deficits in memory. A second limitation was selection bias incurred by missing or incalculable data that necessitated excluding 24% of the sample (106 out of 450) for the final HA density model and 19% (84 out of 450) for the final functional limitation model. Compared to the final sample, patients excluded from the density model were more severely injured. This was an expected finding since more severely injured patients would more likely need surrogate responders and more likely have incalculable PTA (still in PTA at discharge under four weeks) categories. Those excluded from the functional limitation model also had a higher proportion of penetrating injuries compared to its final sample. Other limitations were that 1) pre-injury HA phenotype was based on participants’ report of physician or provider-based diagnosis, and 2) penetrating-type HA was inferred from injury mechanism and imaging findings.
In summary, this study showed HA symptoms do not improve during the first year after TBI and provides additional data on HA risk factors in persons with moderate-severe TBI. Previous research has usually shown female sex to be a risk factor for PTHA, whereas evidence is mixed on whether severity of TBI is a risk factor. We found that female participants became more susceptible to worse HA (higher density and more ADL limitations) as the first year after injury progressed, with a significant gender effect appearing around six months post-TBI. Severity of TBI (PTA duration) was not associated with either outcome. In evaluating additional predictors less assessed in past studies, there were several noteworthy findings. The effect of pre-injury history of HA (vs. no pre-injury history of HA) on HA density and functional limitations from HA was much stronger than the effect of female gender. Pre-injury history HA having been diagnosed as migraine-type HA was an especially robust predictor of higher HA density and HA-related functional limitations after TBI. Additionally, penetrating- (vs. closed) type TBI emerged as a significant predictor for HA density. Individuals with these risk factors should be closely monitored for PTHA and receive aggressive early intervention when discovered.
Clinical implications
Individuals with pre-injury headache (HA), particularly of migraine type, are at greatest risk for HA after traumatic brain injury (TBI). Other independent risk factors are penetrating-type TBI and, to a lesser degree and post-acutely only, female sex. Individuals with these risk factors should be monitored and considered for aggressive early intervention.
Footnotes
Acknowledgements
This study was conducted as part of the Department of Education, National Institute on Disability and Rehabilitation Research, TBI Model Systems module projects. Data were contributed by seven centers: University of Washington Traumatic Brain Injury Model System H133A070032; Rocky Mountain Regional Brain Injury System H133A070022; Mayo Clinic Traumatic Brain Injury Model System H133A070013; UAB TBI Model System H133A070039; North Texas Traumatic Brain Injury Model System H133A0252604; The Moss Traumatic Brain Injury Model System H133A070040; and the Virginia Commonwealth Traumatic Brain Injury Model System H133A070036.
Funding
This work was supported by the National Institute on Disability and Rehabilitation Research (NIDRR), Office of Special Education and Rehabilitative Services (OSERS), Dept. of Education, Washington, D.C. (grant numbers H133A070013, H133A070032, and H133A070036).
Conflict of Interest statement
Dr Walker, Ms Marwitz, Dr Ketchum, Dr Hoffman, and Dr Brown receive research funding from NIDRR. Dr Lucas received funding from NIDRR and Wadsworth Foundation for this study. Other research funding for her has been from Merck, GSK, Map, Nupathe, St. Jude Medical (formerly AGA), BiogenIdec, Sanofi-Aventis, John L. Locke Foundation and the National Headache Foundation. Honoraria, advisory or consulting fees have been received from Merck, GSK, BiogenIdec, EMD Serono-Pfizer, Zogenix, Map, Novartis and Allozyne.
