Abstract
Background
Potential fremanezumab doses for pediatric patients were evaluated using pharmacokinetic modeling and simulation. An open-label phase 1 pharmacokinetic and safety study was conducted in pediatric patients with migraine. This study’s results together with refinement of the adult population pharmacokinetic model were used to determine fremanezumab dose recommendations for phase 3 pediatric studies.
Methods
Initial application of the adult model suggested that a 75 mg dose in pediatric patients would match exposures determined safe and efficacious in adults; thus, in the phase 1 study, 15 patients, aged 6–11 years and weighing 17–45 kg received a single subcutaneous 75 mg fremanezumab dose. The sparse pharmacokinetic data collected were used to refine the adult model and simulate concentration-time profiles for monthly subcutaneous doses (60 to 225 mg) in a virtual pediatric population.
Results
In the phase 1 pediatric study, the safety profile was similar to that of adults. A two-compartment model with first-order absorption and elimination and body weight effects on clearance and central volume was found to adequately describe the pediatric pharmacokinetic data.
Conclusions
Using exposure matching to the effective adult fremanezumab dose (225 mg subcutaneous monthly), modeling and simulations predict recommended dose of 120 mg in pediatric patients weighing < 45 kg.
Keywords
Introduction
Fremanezumab is a fully humanized IgG2Δa/kappa monoclonal antibody that selectively targets the calcitonin-gene-related peptide (CGRP) and prevents it from binding to the CGRP receptor. It is approved for the preventive treatment of migraine in adults in the US, in Europe, and in other countries worldwide. In phase 3 studies of adult patients with episodic and chronic migraine, fremanezumab significantly reduced the number of migraine days and headache days compared to placebo treatment (1,2). In pharmacokinetic (PK) analyses of adult patients and healthy volunteers, fremanezumab has a median time to maximum concentrations (tmax) of 5–7 days, a median half-life of 30 days and an absolute bioavailability of 55% to 66%. Steady-state systemic exposure is expected by approximately 6 months in adult patients following the 225 mg monthly and 675 mg quarterly dosing regimens (3).
Preventive therapy is indicated for all individuals with chronic migraine and for those with episodic migraine with frequent attacks (4). It is recommended that children who experience a minimum of one headache per week or 3–4 headaches per month, or those reporting intensive or prolonged headaches (lasting more than 48 h) even if infrequent, receive preventive medication (5). Topiramate is approved as a migraine preventive medication in the US, but its approved use in pediatric patients is limited to adolescents 12 through 17 years of age (6,7). To address this unmet medical need, including in children under 12 years, phase 3 clinical studies were planned to assess the safety and efficacy of fremanezumab as a preventive therapy.
The FDA has developed guidelines on ways to adequately study the effects of products in pediatric patients, while at the same time minimizing the number of studies involving pediatric patients. Part of these recommendations involves partial or full extrapolation of efficacy data from adults to pediatric populations (8). Two assumptions for the use of an extrapolation approach include determining that pediatric and adult patients have similar disease progression and a similar treatment response, but a third assumption involves ensuring similarity in the exposure-response relationship between adults and children. Given the extent to which similarity between adult and pediatric patients can be claimed, a full or partial extrapolation approach may be considered, in both cases involving the collection of PK data in pediatric patients and confirmation of the similarity in exposure between pediatrics and adults at the efficacious dose.
Based on clinical experiences and theoretical considerations, there are a variety of different drug disposition mechanisms that may be affected by age and thus can result in differences in the PK exposure of monoclonal antibodies in children compared to adults (9). While these age-based differences can be addressed to a large degree by body size-based dosing, especially by adequately considering the non-linear relationship between clearance and body size according to allometric principles, additional age-related dose adjustments may be necessary in younger pediatric populations (10). To this end, the present report describes an open-label phase 1 pediatric study conducted to assess safety and tolerability of fremanezumab administration in pediatric patients and the population pharmacokinetic (PopPK) modeling and model-based simulations performed to support the fremanezumab dose recommendation for pediatric patients weighing < 45 kg in the phase 3 program.
Methods
Phase 1 study of pediatric patients
Study design: Eligible participants of this open label phase 1 study were diagnosed with migraine (ICHD-3), aged 6–11 years, and weighing between 17–45 kg; more information on the study design and patient characteristics is provided in Table S1. As part of the inclusion criteria, patients needed to be in good health as determined by a medical and psychiatric history, physical examination, 12-lead ECG, and clinical laboratory tests including serum chemistry, hematology, coagulation, and urinalysis. Written informed consent was obtained from each patient’s parent(s)/legal representative(s), and each pediatric patient (if the patient was able) before any study procedures or assessments were done. The study registration number is EudraCT number 2018-000734-35.
Patients who met eligibility criteria during the 2-week screening period were enrolled into the study, which consisted of a clinic visit with a single dose administration on day 1 followed by 16 weeks of follow-up visits. Blood samples for measurement of plasma concentrations of fremanezumab were obtained before study drug administration and on days 2, 11, 29, 85 and 113 during the 16-week follow-up. The samples were analyzed using a validated ELISA method with a concentration detection range from 250–3500 ng/mL as described in detail in Cherniakov et al. (11). Safety was assessed throughout the study by monitoring the occurrence of adverse events, including injection site reactions, clinical laboratory test results (chemistry, hematology, coagulation, and urinalysis), vital sign measurements, 12-lead ECG findings, physical examination findings, and use of concomitant medications.
Statistics: The sample size (n = 12 evaluable patients) was calculated based on the PopPK model for fremanezumab in adults available at that time and was expected to achieve a 95% confidence interval within 60% and 140% of the geometric mean estimates of apparent clearance and apparent central volume of distribution with at least 80% power. In anticipation of dropouts, a sample size of 15 enrolled patients was expected to result in a total of 12 evaluable patients.
Plasma concentrations were summarized using descriptive statistics and mean plasma concentrations for the body weight groups (17 to < 30 kg and ≥ 30–45 kg) and overall were depicted in linear plots. Fremanezumab PK parameters were calculated by non-compartmental methods using appropriate validated software (Phoenix® WinNonlin Version 6.3 in conjunction with Pharsight® Knowledgebase Server™ version 4.0.3; Certara, Princeton, NJ, USA).
Dose selection for the pediatric PK phase 1 study: To determine the dose for the phase 1 pediatric study, a modeling and simulation-based approach was conducted wherein the adult population PK model for fremanezumab was used to simulate exposures in virtual pediatric patients following a range of possible doses. For this analysis, a virtual population of pediatric patients aged 6–11 years was simulated with 100 patients per year of age per gender. The dose level in virtual pediatric patients was selected such that levels of exposure parameters were similar to those of adults administered 225 mg once monthly (data on file, Teva Pharmaceutical Industries, Ltd). A single dose of 75 mg was determined to provide a similar distribution of fremanezumab exposure for patients weighing < 45 kg to those following 225 mg once monthly in adults (online supplement Figure S1).
Dose selection processes for pediatric patients weighing <45 kg
The processes for the modeling and simulation analyses performed for the dose selection for pediatric patients weighing below 45 kg are outlined in Figure 1.

Dose selection processes for pediatric patients weighing < 45 kg
Re-estimating the PopPK model with the phase 1 pediatric data
The fremanezumab adult PopPK model (12) was used as prior information for fitting the observed pediatric PK data from the phase 1 study (PK samples n = 75; Table S1) to establish a PopPK model for pediatric patients < 18 years of age and weighing < 45 kg. The base model was a 2-compartment model with first-order absorption and elimination, log-normally distributed inter-individual variability for clearance (CL), central volume of distribution (Vc), and absorption rate constant (ka). Covariates already included in the previously developed model (allometric weight scaling on CL and Vc) were re-evaluated for precision and plausibility of parameter estimates based on the pediatric data. Initial residual variability was described with an additive-plus-constant coefficient of variation model structure. Model evaluation was performed throughout the modeling steps using prediction-corrected visual predictive checks (pcVPC), which provided a graphical assessment of the agreement between the time course of observed and model-predicted concentrations (13,14). Following refinement of the PopPK model based on the phase 1 pediatric data, a stepwise forward selection (α = 0.01 plus at least a 5% reduction in inter-individual variability in the parameter of interest) followed by backward elimination (α = 0.001) methodology was used for re-assessment of body weight, age, and sex as covariate effects on PK model parameters.
Generation of a virtual population and simulations to support phase 3 dose selection
A virtual population of 2400 pediatric patients (6–17 years of age) was generated (200 patients per year of age) and used along with the final pediatric PopPK model estimates to simulate concentration-time data for monthly subcutaneous (sc) doses ranging from 60 to 225 mg. In this setting, weight was assigned based on the Centers for Disease Control and Prevention (CDC) growth chart using a random number to determine the Z score (percentile of weight) for each simulated age (15). The median (range) of baseline weight was 27.2 kg (17.0–60.5 kg) and 51.50 kg (28.8–96.3 kg) for patients 6–11 and 12–17 years of age, respectively. Frequency distributions of weight, by gender, for pediatric virtual patients are provided in Figure 2. Among the 2400 simulated virtual pediatric patients, 1453 had a body weight that was < 45 kg and were included for simulations.
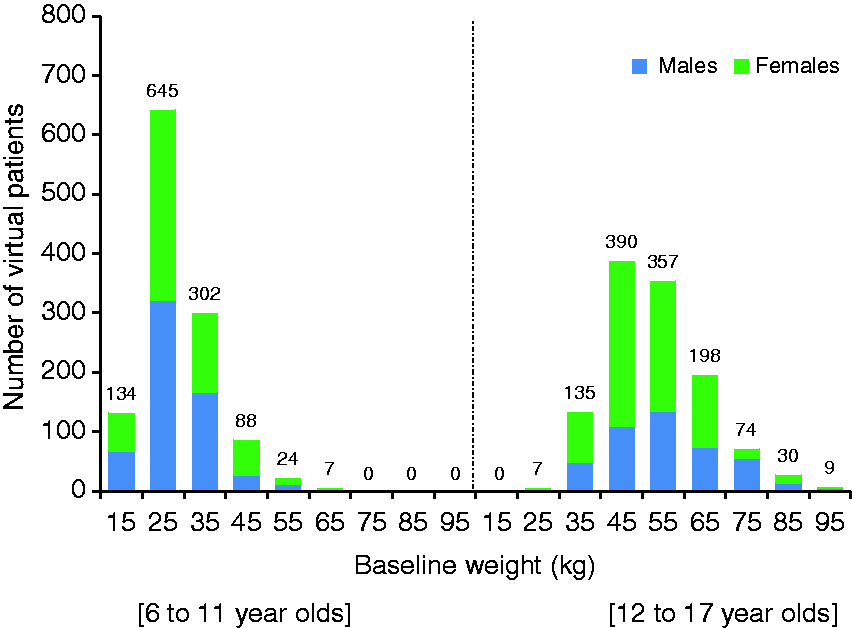
Frequency distributions of weight by gender for virtual pediatric patients. The number under each bar represents the median of the range of weight values for that bar. The number above each bar represents the number of virtual patients in each bar.
For the virtual pediatric patients weighing < 45 kg, simulated exposure measures (i.e. average concentration [Cav], cumulative exposure represented by the area under the concentration-time curve from time 0 to 28 days [AUC28d], minimum drug concentration [Cmin], and maximum drug concentration [Cmax]) were calculated at steady-state and compared to exposure parameters measured at steady-state in the adult population receiving fremanezumab 225 mg sc monthly.
Results
Open-label phase 1 study
Seventeen pediatric patients with migraine were screened, and of these, 15 were enrolled (six in the 17 to < 30 kg weight group and nine in the ≥30–45 kg weight group). The average age was 9.3 years (range 6–11 years). The majority of patients (67%) were black or African American and the remaining were white. The gender distribution was balanced, eight males and seven females. All 15 patients met the inclusion/exclusion criteria. Twelve of the 15 enrolled patients had at least one medical history finding; the most frequently reported medical history abnormalities (other than migraine diagnosis) were nervous system disorders involving headaches (eight patients [53%]). Others included attention deficit/hyperactivity disorder (two patients), insomnia (one patient), asthma (three patients), and psoriasis (one patient). All 15 participants received one dose of study drug, were evaluable for pharmacokinetics, safety and tolerability and completed the study. Study period was from June 2018 to June 2019.
Safety and tolerability: There were no deaths, serious adverse events, treatment-emergent adverse events of special interest (ophthalmic adverse events of at least moderate severity, liver events, Hy’s Law events, or events of anaphylaxis and severe hypersensitivity reactions). Seven (47%) patients reported at least one adverse event, the majority of which were of mild severity (6 [40%] patients). One adverse event, laceration, was considered moderate, and none were severe. The most common treatment-emergent adverse event, injection site erythema, was reported in five patients (33%). The remaining treatment-emergent adverse events: Upper abdominal pain, injection site induration, neutrophil count decreased, increased prothrombin international normalized ratio (INR) > 1.5, prothrombin time prolonged, and laceration were each reported in one patient (7%). There were no clinically meaningful trends in mean changes from baseline for any serum chemistry, hematology, coagulation, and urinalysis variables.
Pharmacokinetics: Pre-dose fremanezumab concentration values for all patients were below the lower limit of quantification (LLOQ) and post-dose fremanezumab concentrations were quantifiable in all patients up to the final time point (i.e. 2688 hours [day 113]). Up to day 28, in general, differences were found in the mean serum concentrations of fremanezumab between the two weight groups; fremanezumab concentrations were lower in patients weighing ≥30–45 kg compared with patients weighing 17 to < 30 kg, and there also was no detectable absorption phase for patients in the lower weight group, although the limited sampling during the absorption phase should be noted. Plasma concentrations are shown in Figure 3.
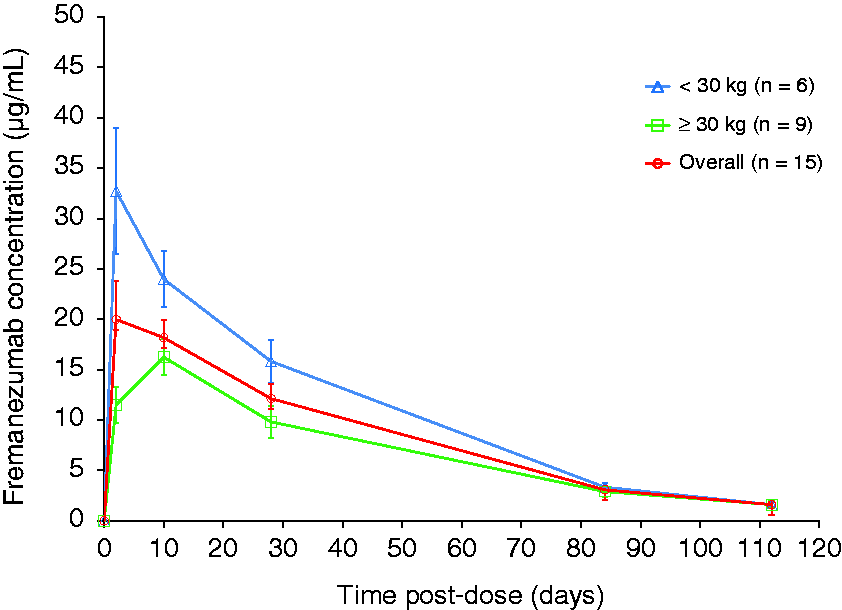
Mean (± SD) plasma concentration over time of fremanezumab by weight group.
For the PK parameters, in the overall group (n = 15), the geometric mean Cmax of fremanezumab was 20.74 μg/mL and the median tmax was 7.99 days. The geometric mean AUC∞, Vz/F, and CL/F values were 23259.9 μg*h/mL, 3.119 L, and 0.05377 mL/min, respectively, and the mean t½ was 28.4 days (Table 1). The exposure parameters (Cmax and AUCs) of fremanezumab were moderately variable with CVs ranging from 47.9 to 54.8%. These findings suggest that the systemic exposure following a 75 mg sc dose was lower in the pediatric study compared to adult patients exposed to 225 mg (11), therefore 75 mg was deemed to be a suboptimal dose. For the pediatric patients, as in adult patients, differences due to body weight were observed; the geometric mean Cmax of the 17 to < 30 kg weight group was approximately two-fold higher than that of the ≥30–45 kg weight group and the AUC values were about 1.5-fold higher in the 17 to < 30 kg weight group than in the ≥30–45 kg weight group. The mean t½ values of the two weight groups of pediatric patients were similar; however, the median tmax values were 1.00 day and 9.98 days for the 17 to < 30 kg and ≥30–45 kg weight groups, respectively.
Pharmacokinetic parameters of fremanezumab phase 1 pediatric study.

aMedian, min-max; bgeometric mean (geometric % CV); carithmetic mean (arithmetic % CV); d95% CI of the geometric mean values of the overall CL/F and Vz/F were within the boundaries of 60–140% of the geometric mean.
% AUCext: percentage extrapolated area under the plasma concentration-time curve; AUC28d: AUC from time 0 to 28 days after study drug administration; AUC∞: AUC from time 0 to infinity; CL/F: apparent clearance; Cmax: maximum observed plasma drug concentration; % CV: percent coefficient of variation; max: maximum; min: minimum; n: number of patients with evaluable data; t½: elimination half-life; Tmax: time to Cmax; Vz/F: apparent volume of distribution.
Refining the PopPk model with pediatric data
Initial base model development in the pediatric population began with the previously established adult PopPK model utilizing informative prior information on all population mean PK parameters and the variance thereof, re-estimating parameters based on the pediatric patient data. Fixed parameters in the adult model were held fixed in the pediatric model while covariates already included in the model (allometric weight scaling on CL and Vc) were re-evaluated for precision and plausibility of parameter estimates based on the pediatric data. Inter-individual variability on ka was not identifiable based on the limited available data in pediatrics, likely due to the limitations of available PK data during the absorption phase. In addition, no absorption lag time was observed in the pediatric patient data, so removal of the absorption lag time parameter was necessary to stabilize the model estimation. The estimated bioavailability from the adult model was not considered as prior information in the pediatric PopPK model; hence, bioavailability in the pediatric model was treated as apparent bioavailability (F1 = 1). Refinement of the residual variability model to a simple proportional error model structure by removing the additive component was required to properly fit the pediatric data. Removing the prior information on the allometric weight scaling components on CL and Vc was necessary due to a general trend for over-prediction across the range of fremanezumab concentrations. Following covariate analysis, only an additional effect of age on central volume Vc was statistically significant, but, due to the high correlation between age and body weight (|r| = 0.89) in this pediatric population and the inclusion of the effect of weight on Vc, the age effect was deemed not clinically relevant and was not retained in the model. Thus, in the re-estimation of the adult PopPK model using the pediatric data, the final PK parameter estimates, with the exception of ka and the allometric exponents for CL and Vc, were comparable between adult and pediatric patients (Table 2).
Parameter estimates for fremanezumab pediatric PopPK model.

aParameters are apparent; bTypical value for CL from pediatric model = 0.0905 x (Weight/71)^0.245; cCentral volume of distribution, subcutaneous from adult model; dTypical value for Vc from pediatric model = 1.89 x (weight/71)^1.20.% CV: coefficient of variation expressed as a percent; NE = not estimated; % RSE = relative standard error expressed as a percent.
The pcVPC plots for the final pediatric model indicate that a two-compartment model with first-order absorption and elimination including covariate effects of body weight on CL and Vc adequately predicts both the central tendency of the concentration data over time, as well as the extent of variability in the observed pediatric data (Figure 4).
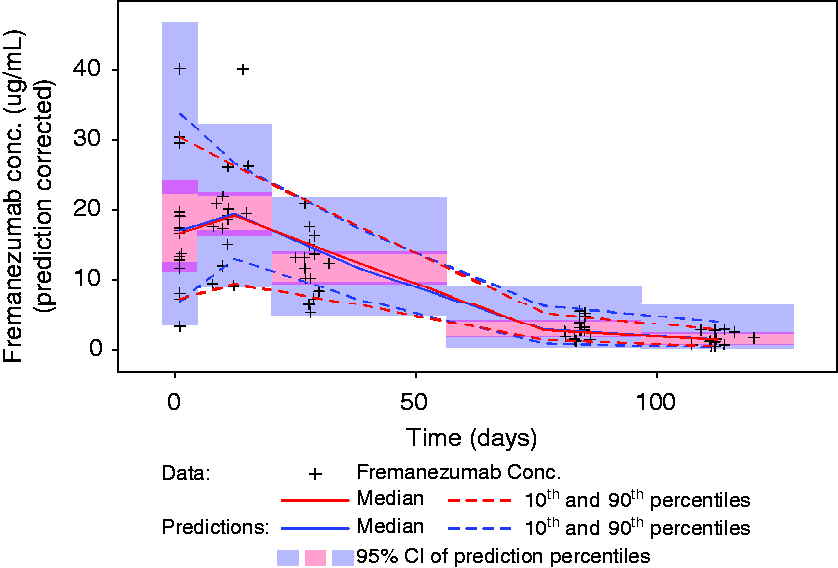
Prediction-corrected visual predictive check for the final fremanezumab pediatric PopPK model. Fremanezumab concentrations are plotted (black plus symbols) versus time since dose administration. The red solid line represents the median of the observed data, with the dashed lines representing the 10th and 90th percentiles of the observed data. The blue solid and dashed lines represent the corresponding percentiles of 1000 simulations of the data based on the model, with the shaded regions about each blue line illustrating the 95% confidence interval about the predicted percentiles.
Simulations
Dose selection for pediatric patients weighing <45 kg
Simulated exposure measures AUC28d and Cmax were calculated at steady-state for the virtual pediatric patients and compared to simulated exposure measures at steady-state in the adult population receiving fremanezumab 225 mg sc monthly (11). Boxplots of the simulated steady-state exposure parameters following fremanezumab doses of 60–225 mg sc monthly are shown in Figure 5, panels (a)–(c) for virtual pediatric patients (6–17 years with body weight < 45 kg). For virtual pediatric patients administered 120 mg sc monthly, the simulated AUC28d distribution is nearly identical to the adult patient distribution following 225 mg sc monthly (Figure 5(a)), with similar patterns observed for Cav and Cmin. The simulated Cmax distribution following 120 mg sc monthly in the pediatric population suggests slightly higher Cmax than that achieved in the adult population with 225 mg; however, the overall upper exposure range extends only slightly above the upper range of the adult distribution (Figure 5(b)). Weight is a significant covariate affecting exposure, hence when comparing the simulated Cmax distribution following 120 mg sc monthly in the pediatric population with the adult Cmax distribution by weight quartile (Figure 5(c)), the Cmax distribution for the pediatric population is comparable to that of the two lower body weight quartiles of the adult population.
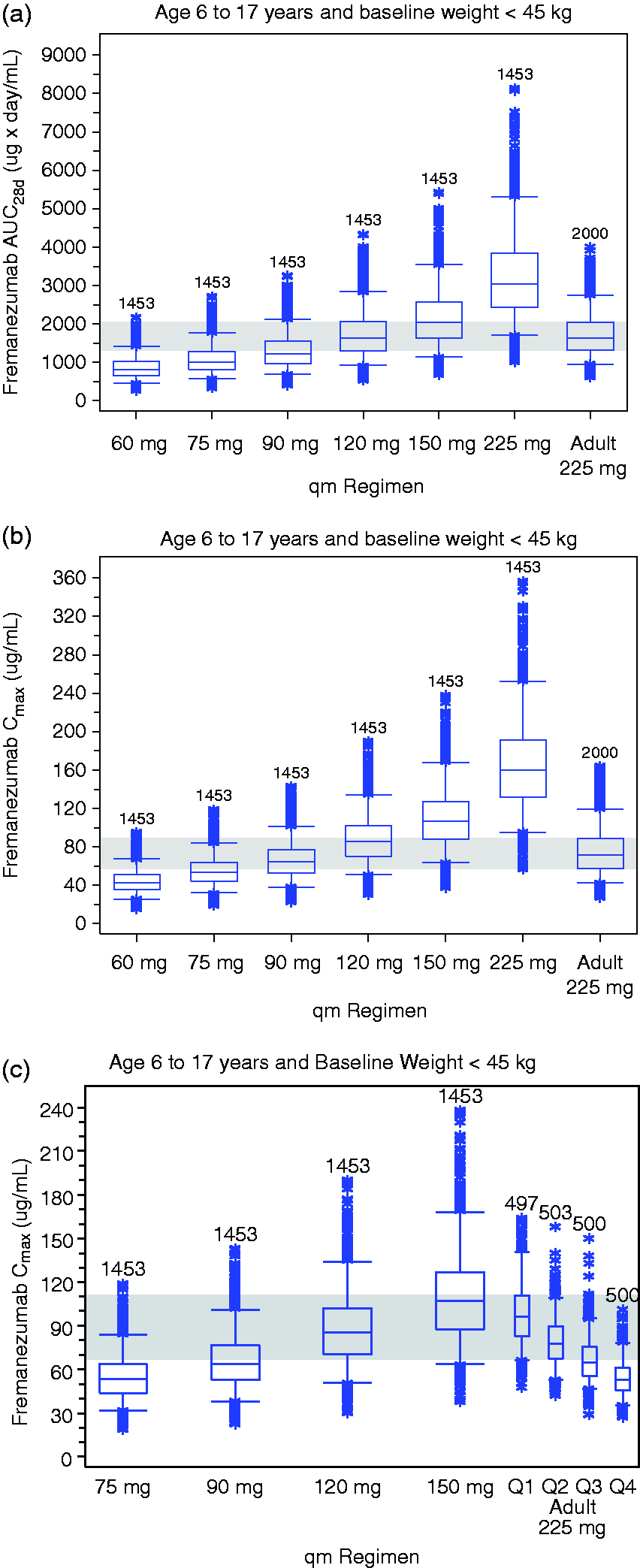
Simulated steady-state exposure parameters for pediatric patients, ages 6–17 years and with body weights <45 kg, compared to adults following fremanezumab 225 mg sc administration and stratified by body weight quartile. (a) Simulated AUC28d by dose for pediatric patients compared to adults following fremanezumab 225 mg sc administration. (b) Simulated Cmax by dose for pediatric patients compared to adults following fremanezumab 225 mg sc administration. (c) Simulated Cmax by dose for pediatric patients compared to adults following fremanezumab 225 mg sc administration and stratified by body weight quartile.
The simulated steady-state cumulative exposure (i.e. AUC28d) following fremanezumab 120 mg sc monthly dosing and body weight in a virtual pediatric population are presented in Figure S2. As expected, based on the significant effect of body weight on fremanezumab PK, the simulated data predict slightly higher exposures with lower body weight. Despite this relationship, it is notable that the range and central tendency of the simulated exposures are similar between 17 and 45 kg. To further explore the relationship of fremanezumab exposure and pediatric body weights, the pediatric body weight range for patients 6–17 years of age weighing < 45 kg was further subdivided into two body weight categories: 17 to < 30 kg and 30 to < 45 kg. Boxplots of simulated steady-state fremanezumab AUC28d by dose for pediatric patients 6–17 years of age divided into these two body weight categories compared to the adult distribution following 225 mg sc monthly doses are shown in Figure 6. The distribution of exposures following a 120 mg sc dose is nearly identical in both body weight categories (17 to < 30 kg and 30 to < 45 kg) and matches the exposure in adults at the effective dose of 225 mg sc monthly. Similar results were observed for the other PK exposure parameters (Cav, Cmin, and Cmax). Based on the similarity in exposures over this body weight range, no additional body weight cutoff value < 45 kg was deemed necessary.
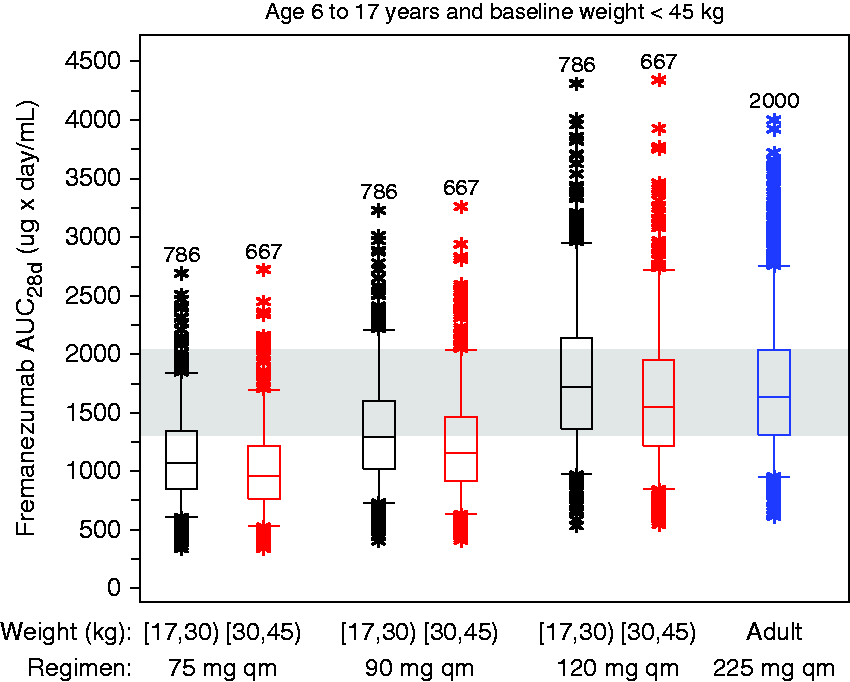
Predicted exposures by two body weight categories <45 kg (17 to <30 kg and 30 to <45 kg) compared to an adult exposure distribution following 225 mg sc administration.
Discussion
Phase 3 studies assessing fremanezumab as a migraine treatment for pediatric patients are under way. The present report describes the open-label pediatric phase 1 study and population PK modeling and simulation conducted to recommend doses for these pediatric phase 3 fremanezumab studies. The recommended pediatric doses result in simulated matching exposures known to be associated with efficacy in adults treated with the approved fremanezumab dose of 225 mg sc monthly.
In the open-label phase 1 study, fremanezumab 75 mg as a sc injection was well tolerated when administered to pediatric patients with migraine, and the safety profile was similar to that observed in previous studies with adult patients. Injection site reactions were the most common adverse event reported during the study and all were mild in severity. Abnormal clinical laboratory tests, ECGs, and vital signs occurred infrequently. It must be mentioned that two-thirds of the study participants were African-American or Black, with the remaining 30% being White. However, race was evaluated in the adult population PK model to not statistically significantly influence exposure of fremanezumab (12). This lack of effect on exposure was also demonstrated in the phase 1 study of Japanese and Caucasian healthy subjects, which deduced no differences in fremanezumab pharmacokinetics due to racial diversity (16). In the study of Japanese and Caucasian subjects (16), fremanezumab exposures were generally higher with lower body weight and tended to decrease as body weight increased. Moreover, body weight was also found to be a significant predictor of fremanezumab exposure in the adult PopPK model. Therefore, the potential effect of body weight was closely examined in the present study by dividing the participants into two body weight subgroups (17 to 30 kg and 30 to < 45 kg). For the pediatric patients in the lower weight group (17 to < 30 kg), a single fremanezumab sc injection of 75 mg produced higher exposures, in terms of Cmax and AUC, when compared to the higher weight group (30 to < 45 mg). The time to reach maximum concentration (median Tmax) also varied greatly between the two body weight groups, suggesting that the rate of absorption was more rapid in the 17 to <30 kg weight group. However, because the Tmax ranges of the two groups were similar and the PK sampling was limited during the absorption phase (one sample at day 2 followed by another at day 11), any conclusions about the absorption phase should be interpreted with caution. PK measures (CL/F and Vz/F) increased with increasing body weight and mean half-lives were similar between the two weight groups.
In the present study, a dose of 75 mg was selected using a simulation-based approach based on a previously developed adult population PK model. Given the anticipated similarity between adult and pediatric patients with migraine and the effect of body weight on fremanezumab PK in adults, exposure measures were simulated in a virtual pediatric population by extrapolating to appropriate pediatric body weight values. Upon completion of the pediatric study, the adult PopPK model was then updated to generate a pediatric PopPK model using the observed data collected from pediatric patients with migraine in the PK phase 1 study.
An apparent difference in the absorption was observed in the pediatric patients from the pediatric phase 1 PK study when compared to the adult fremanezumab concentration time profiles. Multiple attempts were made in the PopPK model to describe the variability seen in fremanezumab absorption in the pediatric population but, likely due to the limitations of available PK concentrations during the absorption phase, these were not successful. A small pediatric study reporting the variability in absorption kinetics after administration of an anti-TNFα monoclonal antibody, also indicated higher ka for pediatric patients aged less than 16 years (17), which is consistent with the observations in the current pediatric study. As generally observed with therapeutic monoclonal antibodies, body weight was a significant predictor of variability in the fremanezumab PK with respect to both CL and Vc, and higher body weight was associated with increased CL and Vc. A statistical evaluation of other covariates, such as sex and age, demonstrated that the covariate either had no effect on the PK of fremanezumab (sex) or had a high correlation with body weight (age), such that the effect on the PK parameters was deemed not to be clinically relevant. VPC and diagnostics indicated that the final pediatric population PK model provided an adequate fit to the data.
A range of possible pediatric doses of 60–225 mg monthly were evaluated in this analysis, and the proposed dose of 120 mg sc monthly in pediatric patients aged 6–17 years and weighing <45 kg manifested the greatest overlap in simulated exposures to those achieved in adults after administration of 225 mg sc monthly dose, the approved adult dose associated with efficacious response. Furthermore, the distribution of simulated exposures following a 120 mg dose is nearly identical in both 17–30 kg and >30–45 kg weight categories, while matching the exposures in adults at the effective dose of 225 mg, thus there is no need to have an additional weight cut off below 45 kg.
While the proposed dose of 120 mg sc monthly for pediatric patients differs from the initial proposed dose of 75 mg sc monthly for pediatric patients aged 6–17 years and weighing <45 kg, this change is justified by the present inclusion of pediatric data into the population PK model. The results from the current pediatric population PK model indicate that the PK model provided an adequate fit to the pediatric data and both the central tendency of the concentration data over time, as well as the extent of variability in the observed pediatric data, was adequately predicted, supporting the use of the pediatric population PK model for simulations and dose selection decision making.
For patients weighing ≥45 kg, dose selection for the phase 3 pediatric study was determined to be the approved adult dose of 225 mg monthly. A 2013 investigation by Momper et al. (18) from the Food and Drug Administration analyzed 92 products approved between 2007 and 2012 with similar adult and pediatric indications across different therapeutic areas, and 87 (94.5%) had equivalent dosing for adults and adolescent patients. Similarly in 2019, Yang et al. (19) conducted an analysis to identify body weight thresholds for therapeutic monoclonal antibodies above which pediatric patients can receive adult dosages, and found that a 40 kg body weight threshold is generally recommended if the reference median adult population is ≤80 kg and the product has a wide therapeutic window. The clinical studies for fremanezumab in adult patients had a median body weight of 71 kg and a large therapeutic window of up to 900 mg sc monthly (1,2). The 40-kg threshold is also consistent with the FDA’s recent draft guidance for industry for including adolescents into adult oncology trials (20). The FDA guidance recommends that adolescents weighing above 40 kg can receive the same fixed adult dose without risks to clinically relevant drug exposure and safety. For the phase 3 program, the fremanezumab weight cutoff was slightly higher (45 kg) to further reduce the likelihood of exceeding a pediatric exposure above the adult exposure.
In conclusion, administration of fremanezumab was found to be safe and well-tolerated in pediatric patients with a safety profile similar to that seen in previous adult studies. The population PK model, including the PK data of the phase 1 pediatric study, was found to provide an adequate fit to the pediatric data, supporting its use for simulations and dose selection. To match exposure to the efficacious adult dose of 225 mg sc monthly, a dose of fremanezumab 120 mg sc monthly is recommended for pediatric patients weighing less than 45 kg. For pediatric patients weighing equal or more than 45 kg, the adult dose of 225 mg monthly is recommended.
Clinical implications
There is an unmet medical need for preventive migraine treatments in pediatric patients. The present report describes an open-label phase 1 pediatric study conducted to assess the safety and tolerability of fremanezumab administration in pediatric patients and a refinement of the adult fremanezumab population PK model using pediatric PK data from this phase 1 study. Fremanezumab 120 mg sc monthly in pediatric patients weighing < 45 kg is the recommended dose based on simulated data and exposure matching with the efficacious adult dose of 225 mg sc monthly. No further stratification (additional body weight cutoff) below 45 kg is warranted.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211007789 - Supplemental material for Dose selection for fremanezumab (AJOVY) phase 3 pediatric migraine studies using pharmacokinetic data from a pediatric phase 1 study and a population pharmacokinetic modeling and simulation approach
Supplemental material, sj-pdf-1-cep-10.1177_03331024211007789 for Dose selection for fremanezumab (AJOVY) phase 3 pediatric migraine studies using pharmacokinetic data from a pediatric phase 1 study and a population pharmacokinetic modeling and simulation approach by Orit Cohen-Barak, Andrijana Radivojevic, Aksana Jones, Jill Fiedler-Kelly, Michael Gillespie, Michael Brennan, Dikla Gutman, Michele Rasamoelisolo, Hussein Hallak, Pippa Loupe, Yoel Kessler, Xiaoping Ning, Micha Levi, Andrew H Ahn and Laura Rabinovich-Guilatt in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: OC-B, MG, MB, DG, MR, HH, PL, YK, XN, ML, AHA, and LR-G are employees of Global Research and Development Teva Pharmaceutical Industries, Ltd. AR is an employee of IntiGrowth LLC, New York, NY, and was paid as a consultant by Teva Pharmaceutical Industries. AJ and JF-K are employees of Cognigen Corporation, a Simulations Plus company, Buffalo, NY, and were paid by Teva Pharmaceutical Industries to perform the PopPK modeling and simulations.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Global Research and Development Teva Pharmaceutical Industries Ltd, Netanya, Israel.
Ethics or Institutional Review Board approval
The open-label phase 1 pediatric pharmacokinetic study (Study TV48125-CNS-10141) was conducted in full accordance with the International Council for Harmonization (ICH) Good Clinical Practice (GCP) Consolidated Guideline (E6) and any applicable national and local laws and regulations. The study was conducted at five sites in the United States. For each site, all study documents were reviewed and approved by their Institutional Review Boards: Premiere Research Institute Palm Beach FL, Associate Institutional Review Board, IRB 00000971; Atlanta Center for Medical Research, Associate Institutional Review, IRB 00000971; Texas Children’s Hospital/Baylor College of Medicine, Western Institutional Review Board, IRB 00000533; Children’s Medical Center Dallas, Associate Institutional Review Board IRB 00000971; PANDA Neurology Atlanta GA, Associate Institutional Review, IRB 00000971. Written informed consent was obtained from each patient’s parent(s)/legal representative(s) and each pediatric patient (if the patient was able) before any study procedures or assessments were done.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
