Abstract
Introduction
The co-occurrence of headache and epilepsy is well-documented in the adult population. The aim of the prospective study was to analyse in the paediatric population the correlations between the types of peri-ictal headaches and types of seizures. Furthermore, an attempt was made to find trends in characteristic features of peri-ictal headaches.
Results
A total of 913 seizure and 325 peri-ictal headache episodes were noted during the study. Post-ictal headaches were most common, occurring in < 1 h after the seizure, lasting minutes to hours and more likely to occur after generalised seizures, whereas pre-ictal headaches occurred 30–240 min before the seizure. In the analysed group, peri-ictal headaches were most often moderate in intensity. Only 30% of patients took analgesic medication, usually to treat post-ictal headaches.
Conclusion
Peri-ictal headaches are a significant health problem for patients with epilepsy. The most common type are post-ictal headaches, and they are most likely to appear after a generalised seizure.
Introduction
The co-occurrence of headache and epilepsy has been observed for centuries. In patients with epilepsy, headaches are classified as interictal and peri-ictal (1). Peri-ictal headaches were first described by Sir William Gowers in 1881 (2). Peri-ictal headaches are further classified according to their occurrence in relation to the seizure: Pre-ictal, ictal and post-ictal (1–6). It is noteworthy that a patient may experience more than one type of peri-ictal headache, as they are not mutually exclusive (4).
Although the frequency of peri-ictal headaches is high among patients with seizures, none of the International League Against Epilepsy recommendations define the term “peri-ictal headache” (7). The first edition of the International Classification of Headache Disorders was published in 1988 and it does not mention peri-ictal headaches either. Although the later editions (ICHD-2) do describe peri-ictal headaches, they do so using non-uniform terminology that may cause some confusion. In this article, we refer to the latest (third) edition of ICHD, which describes “ictal epileptic headache” and “post-ictal headaches” (3,8–10).
Pre-ictal headache (PreIH) is not included in ICHD-3; however, according to the literature, it occurs among 1.2–35.5% of adults and 3.1–30% of children with epilepsy (11–17). It is suggested by the authors of ICHD-3 that more research is needed to establish the existence of pre-ictal headache as well as to distinguish PreIH “from migraine aura-triggered seizure” (9).
According to the literature, PreIH precedes the seizure. Some authors describe pre-ictal headaches that begin at > 5 min before the seizure, while others define them as headaches with onset < 24 h before it. All authors agree that a PreIH is not part of the seizure episode (11,18,19).
A headache that begins during the seizure episode is described in the majority of the literature as an ictal headache (IH) (1,3–6,19). According to the available data, IH occurs in < 5% of patients with epilepsy, more frequently among children (3,4,14,15,19). Headaches are usually one of the components of a seizure and, rarely, an isolated headache is the only manifestation of a seizure. IHs occur in some of the epilepsy syndromes, particularly those with autonomic symptoms, such as Panayiotopoulos syndrome or frontal lobe epilepsy (20).
In order to meet the ICHD-3 criteria, an IH must begin simultaneously with the focal seizure in an alert patient and should be ipsilateral to the ictal epileptic activities and/or be resolved or significantly reduced upon termination of the seizure (9).
Post-ictal headache (PIH) is the most common type of peri-ictal headaches, occurring in 23.5–64% of adults and 6.4–62% of children diagnosed with epilepsy and is one of the most common peri-ictal symptoms (4,11–13,15,16,21). PIH is defined as a headache with onset < 3 h after the seizure and duration of < 72 h in patients with focal or generalized seizures (3,9,10).
Although the literature about peri-ictal headaches is extensive, the majority of it is focused on the adult population. It is difficult to compare data in the literature due to disparate definitions and criteria applied for peri-ictal headaches and methodological limitations. Furthermore, the majority of the available studies were conducted at specialized epilepsy centres where patients with the most severe, often drug-resistant, cases of epilepsy are treated. Finally, most of the available research about peri-ictal headaches consists of retrospective studies.
Our aim was to conduct a prospective study to determine the frequency of the various peri-ictal headache subtypes and to analyse their characteristics in the paediatric population.
Material and methods
A total of 103 patients with epilepsy, treated in the years 2013–2017 at the Department of Developmental Neurology and its outpatient clinic at the University Clinical Centre were screened. The participants (49 girls and 54 boys) were 6–18 years of age. The exclusion criteria were moderate and severe intellectual disability (or such that prevented reliable communication), speech and communication disorders, nocturnal seizures, non-epileptic episodes (including psychogenic nonepileptic seizures) and neurodegenerative disease as well as lack of peri-ictal headache.
All participants were previously diagnosed with epilepsy according to the ILAE guidelines. Types of seizures were named in accordance with the 2010 ILAE Proposal for Revised Terminology for Organization of Seizures and Epilepsies. For the sake of clarity, in this article we used the past terminology “secondarily generalised seizure” instead of the newly suggested “focal seizure evolving to bilateral tonic-clonic seizure”. Primary generalised seizures were subdivided into generalised convulsive seizures (tonic, clonic, tonic-clonic) and generalised non-convulsive seizures (myoclonic seizures, absence seizures).
We used ICHD-3 terminology for IH and PIH (10), and for PreIH we used the definition based on Wang and Svyvertsen (headache occuring in < 24 h prior to seizure episode and lasting until its onset (11,22). The types of headache (migraine, tension-type or unclassified headaches) were defined in accordance with ICHD-3 (10).
A total of 57 participants (27 girls and 30 boys) with peri-ictal headaches continued in the study. The participants’ guardians were asked to keep a diary of all seizure and peri-ictal headache episodes during a 180-day period. Patients visited their neurologist three times: At the initial visit, 3 months later and 6 months after the initial visit. During the second and third visits to the outpatient clinic, the diaries were reviewed for the following information: Type of peri-ictal headache, headache intensity (VAS scale 0–10), headache onset in relation to seizure onset, additional symptoms (e.g. nausea, vomiting, photophobia, hyperacusis, visual disturbances), analgesics taken for headache relief (Figure 1).
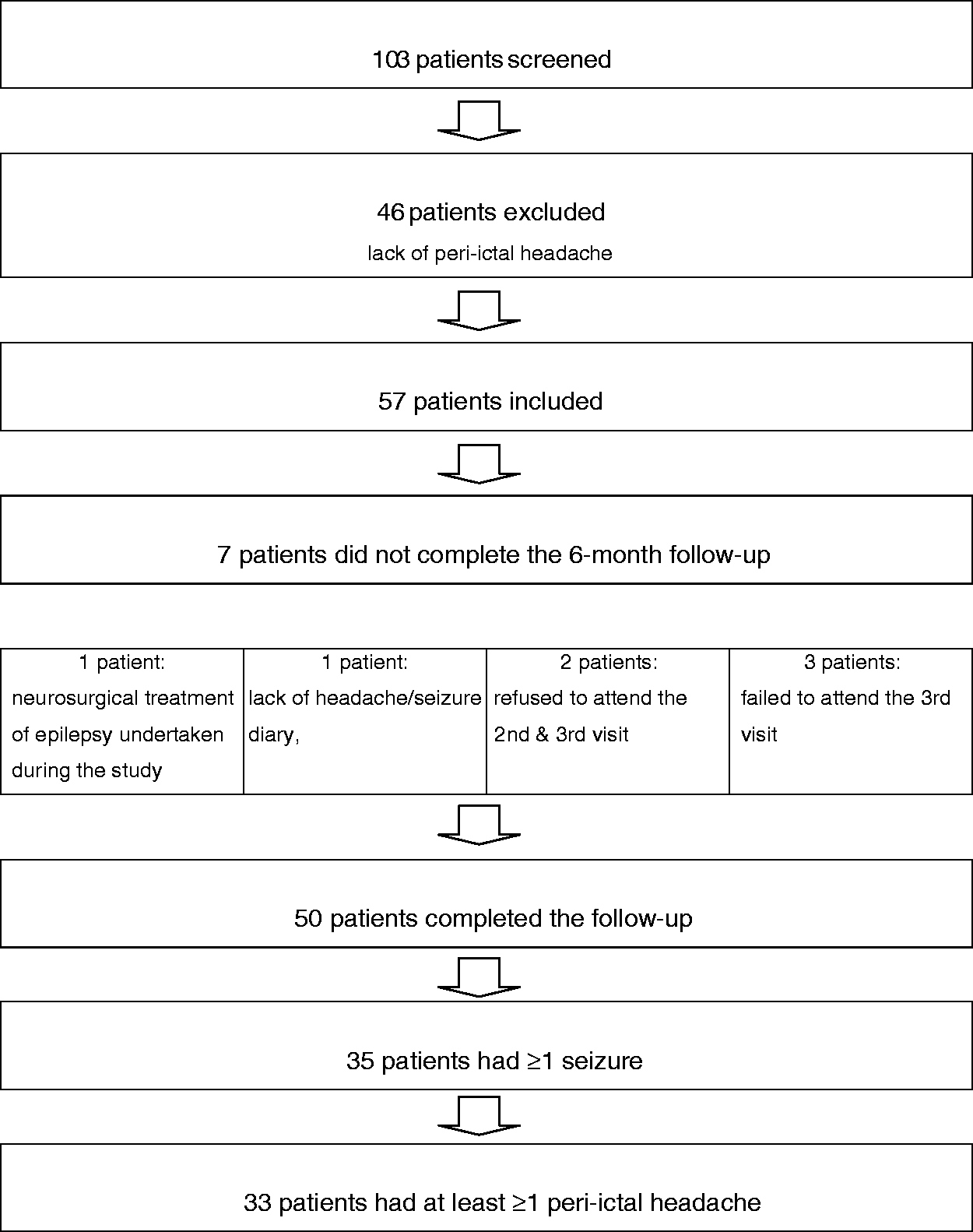
Chart.
The study was performed following the approval of the Ethics Research Committee of the Medical University in Gdańsk. Written consent was obtained from the guardians of all participants < 16 years of age, whereas those who were ≥16 years of age signed written consent forms on their own.
Data were collected in Excel spreadsheets (Microsoft, USA). Statistical analysis was performed using the STATISTICA 10.0 software (StatSoft Inc., USA). All of the quantitative variables were tested using the Kolmogorov-Smirnov test, for meeting the criteria of a normal distribution (Gaussian distribution). Depending on whether the variable met the normality condition, appropriate statistical tests were applied at further stages. In the case of three peri-ictal groups, the parametric analysis of variance (ANOVA) or non-parametric ANOVA test according to Kruskal-Wallis were used. The analysis of qualitative variables in the subgroups was assessed using the Contingency tables. Due to small sample sizes of the subgroups and low expected frequencies, the two tailed Fisher’s exact test (with Freeman-Halton extension) was employed. The statistical significance threshold was set at p < 0.05.
Results
Out of the 57 participants, seven did not complete the 6-month follow-up (one due to neurosurgical treatment of epilepsy undertaken during the study, one did not provide the headache/seizure diary, two refused to attend the second and third visit, while three failed to attend the third visit).
Fifty participants (24 girls and 26 boys) completed the 6-month follow-up and out of that group, 70% (n = 35) had ≥1 seizure episode, whereas 33 of those 35 had at least ≥1 peri-ictal headache.
Type of peri-ictal headache vs. type of seizure
During the 6 months of follow-up, our 50 participants recorded a total of 913 seizures. We also noted 325 peri-ictal headache episodes: 214 (23.44%) were PIH, 75 (8.21%) were PreIH and just 36 (3.94%) IH (Figure 2). Of the 913 seizures, the most common type were focal impaired awareness seizures (n = 566), of which 86 (15%) were associated with PIH and 45 (7.95%) with PreIH. The second most frequently recorded were focal aware seizures (n = 211), of which 64 (30.33%) were associated with PIH, 21 (9.95%) with PreIH and 36 (17%) with IH. Secondarily generalised seizures were less frequent (n = 116) and PIH occurred after 47 (40.5%), while PreIH only occurred before five (4.31%). We also recorded 20 primary generalised convulsive seizures, including 17 (85%) with PIH and 4 (20%) with PreIH. None of the participants experienced a generalised non-convulsive seizure.

Number of patients according to the type of periictal headache.
Peri-ictal headache features
Second visit
No statistically significant differences were found in the severity of the three types of peri-ictal headache. The average reported severity was 6 points on the VAS scale for PreIH, 7 for IH and 6.69 for PIH. Based on the reported data, PIH had onset < 30 min after the seizure ended (average 5.3 min) and lasted from 15 min up to 12 h (average 3.5 h). Participants with PIH were most likely to take analgesics (46.2%). Despite similar headache intensity, patients with PreIH were unlikely to use analgesics (8.33%) (Table 1).
Characteristics of peri-ictal headaches by type (second visit).

Third visit
Once again, we did not find statistically significant differences in the average reported headache intensities: 5.3 (PreIH), 6.8 (IH) and 6.3 (PIH). The headaches began < 60 min after the seizure episode (average 8.62 min) and lasted from 15 min to 12 h (average 2.5 h). Patients with PIH were once again most likely to use analgesics (50%) and none of the participants with PreIH used them (p = 0.03638). Once again, none of the patients took prescription headache medication such as triptans (Table 2).
Characteristics of peri-ictal headaches by type (third visit).

Type of peri-ictal headache and its classification based on ICHD-3
The most frequent characteristics of PIH were localised to the forehead (61.54% on the second visit and 38.46% on the third visit) and pulsating (54% on the second visit and 46% on the third visit) or pressing (31% on the second visit and 38% on the third visit) in quality. Accompanying symptoms were seen in 12 (second visit) and 10 (third visit) patients. Of those symptoms, nausea and/or vomiting were seen in 10 (second visit) and 7 (third visit) children. PIH consistent with the characteristics of migraine headaches according to the International Headache Society criteria (ICHD-3) were observed in 12 (second visit) and 10 (third visit) patients. The most common type of PIH reported were tension-type or not classified (14 patients on second visit and 16 on third visit) (Tables 3,4).
Type of peri-ictal headache and its classification based on ICHD-3 (second visit).

Type of peri-ictal headache and its classification based on ICHD-3 (third visit).
Patients with PreIH described their headache as localised to their forehead (50% on the second visit and 28.57% on the third visit) or the entire head (25% on the second visit and 42.86% on the third visit). Headaches were pulsating in nature in only one patient. Accompanying symptoms were rare (one patient with nausea). The majority of participants with PreIH reported tension-type or unclassified headache, whereas only one met the criteria for migraine.
Five patients suffered from ictal headache. Among patients with ictal headache, 60% experienced vomiting and nausea accompanying the headache. The headaches were localised to the forehead in all patients. The mean intensity of headache assessed on the VAS scale was 7 points. Despite the accompanying symptoms and intensity typical of migraines, these headaches were not classified as migraines due to the short duration of headache.
Discussion
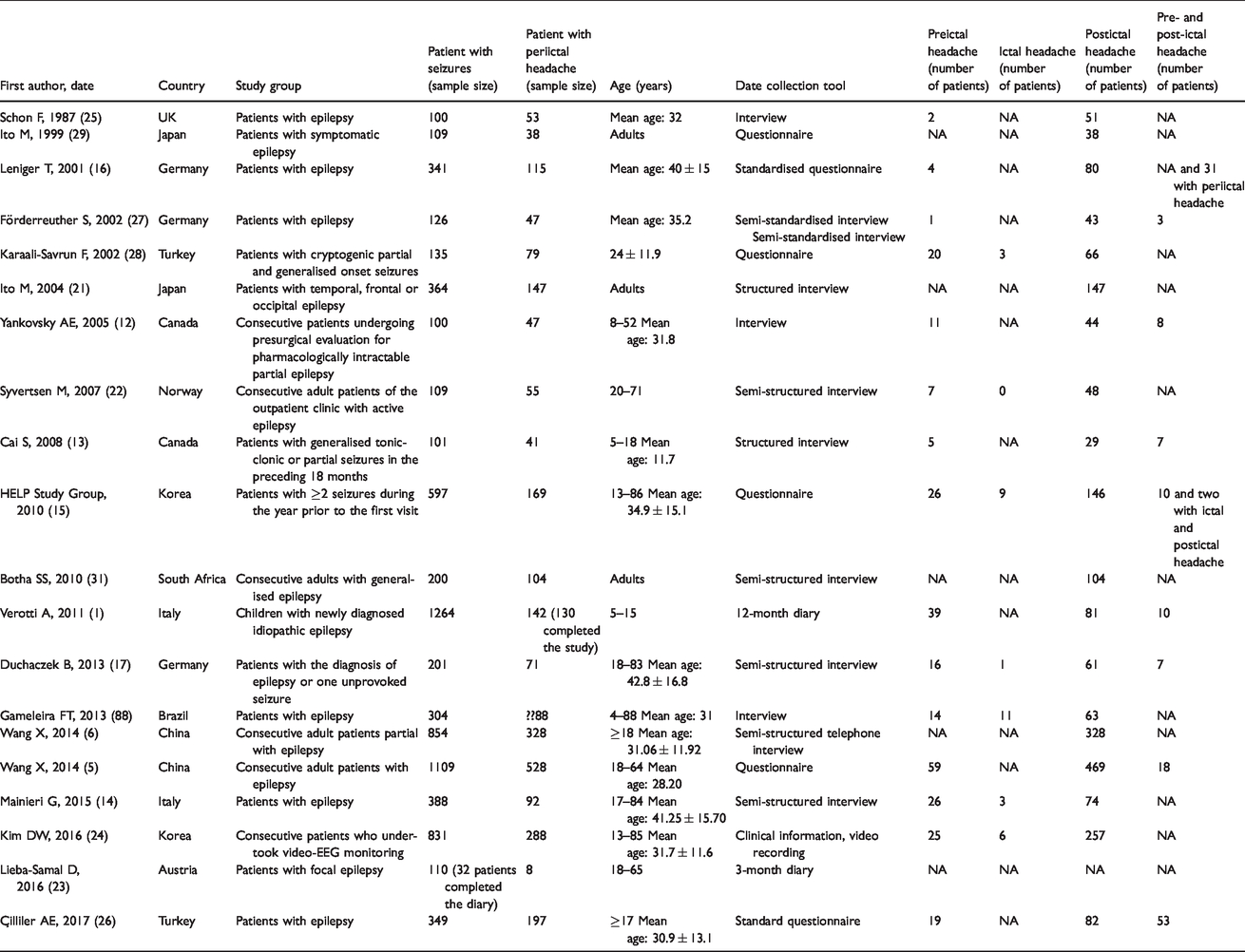
Our study is one of the few prospective study assessing the frequency of the various peri-ictal headache subtypes and analysing their characteristics in the paediatric population. The advantage of this study is the focus on individual seizures and the accompanying headaches using the diary tool. According to our knowledge, this is so far the second analysis of this type, after the Lieba-Samal study (23) (Table 5). Data analysis included 9024 patient days, 913 seizures and 325 episodes of peri-ictal headache. In our study, we confirmed the existence of PreIH, which preceded 8.21% of seizures in the studied population. PIH were most common, occurring < 1 h after the seizure, lasting minutes to hours and more likely to occur after generalised seizures, whereas PreIH occurred 30–240 min before the seizure.
Comparison results of Lieba-Samal and Zawadzka studies using diary tool.

The strength of this study is its prospective design using 6-month, detailed daily diaries. The limitation is provision of a method to avoid a sham effect. It is however difficult to provide a control group in such a setting (epilepsy patients without headaches? healthy children?). Additionally, the study group was small, but it should be noted that the number of analysed days, seizures and peri-ictal headaches was high.
In our study, 36% (325/913) of seizure episodes included peri-ictal headaches. Similar to other reports, PIH were the most frequently reported peri-ictal headache (13,14,23–26]. The majority of PIH in our sample occurred after primary or secondary generalised seizures (47%). The correlation between generalised tonic-clonic seizures and PIH has been reported in numerous studies on the adult population, as these patients complain of PIH more frequently than those with other types of seizures (6,14,17,21,27–29). In studies on the adult population, PIH was confirmed in 93% of patients with primary generalised tonic-clonic seizures and in 42% of patients with secondary generalised tonic-clonic seizures (27,29), whereas in studies with mixed samples (children and adults) PIH was noted in 28.3% of patients with primary generalised tonic-clonic seizures and in 88% of patients with secondary generalised tonic-clonic seizures (15,28). Gameleira et al. report that 55.6% of patients with PIH (n = 63, 4–88 years of age) had generalised tonic-clonic seizures (18). In the adult population PIH after focal seizures occurs less frequently than after generalised tonic-clonic seizures (22–43%) (6,15,27,29). Focal aware seizures coincide with PIH rarely or not at all (14,28,29). However, Çilliler et al. reported contrary observations that in their sample (Turkish adults > 17 years of age) there was no correlation between PIH and the type of seizure (26).
Reports about the correlation between the seizure type and PIH are scant and differ in the paediatric and adult populations and also differ from our results. It is worth underlining how few reports about the paediatric population are available. In a multi-centre prospective study of 142 patients 5–15 years of age with newly diagnosed idiopathic focal or generalised seizures, PIH were the most frequently reported headache type (62.3%) and most frequently after focal seizures. The authors noted that the contrasting results might be explained by the fact that they included only patients with idiopathic epilepsy or by the epidemiological differences between focal seizures and other seizure types (1). On the contrary, Cai et al. did not report any correlation between PIH and seizure type in a sample of children and teenagers (13).
Although PreIH are not included in the ICHD-3, several studies report their existence (9,10). In our study, PreIH most often occurred before primary generalised tonic-clonic seizures. In the literature there is a significant disparity about the correlation between PreIH and particular seizure type, as they were generalised or focal (24,26,28). In a study on the paediatric population, PreIH were reported before focal aware seizures (1). Some authors concluded that the presence of PreIH might indicate the severity of the upcoming seizure episode (27,28). It appears that further studies are needed to verify the correlation between the seizure type and the frequency of PreIH.
In our study, participants reported IH only during focal aware seizures and these were the least frequently reported headache type. The literature about IH is modest. Similar to our study, Kim et al. reported that all patients with IH had focal seizures (24). Fanella described an adult treated for idiopathic generalised seizures, who developed a sudden intense headache during a 24-h electroencephalographic observation. During the headache, authors noted spike and wave complexes, polyspikes and slow wave complexes that lasted 60 min until the termination of headache. This is one of the few published reports of an EEG-documented isolated ictal headache in a patient with an idiopathic generalised seizure (19).
Some authors suggested that peri-ictal headaches are ignored by physicians and the patients’ parents alike. However, the available literature and our results indicate that peri-ictal headaches have a duration and intensity that may interfere with the paediatric patients’ daily activity (16).
The majority of the peri-ictal headaches reported by our participants were either moderate (VAS 4–6) or strong (VAS 7–9). The average intensity of PreIH, IH and PIH were respectively 6.0, 7.0 and 6.7 (second visit) and 5.3, 6.8 and 6.3 during the third visit. The majority of the published studies report similar average peri-ictal headache intensity (VAS 5.9–6.7) described as moderate to strong (6,12,15–17,22,27,31).
Studies that included children and adults report average intensity of PreIH as VAS 6.1–7.1 (12,15), whereas a study of adults only indicates an average VAS of 6.9 (17). Our results are similar: VAS 5.3 (second visit) and 6.0 (third visit).
The intensity of IH was analysed in only one adult study, reporting an average VAS of 5.4 (15). In our study, we noted higher intensities of IH (7.0 on the second visit and 6.8 on the third visit). It is difficult to compare these results as the two studies have different age samples, methodologies, and small sample sizes.
According to ICHD-3, a PIH should appear within ≤3 h after the seizure ended (3,9). The time span between the end of the seizure and the onset of headache was examined in just one paediatric study, which reported that in 92% of the analysed patients PIH began in ≤5 min (13). This is consistent with our results.
The duration of PIH described in the adult population was much longer than in our sample and varied from 5–20 h (15–17,22,28). Paediatric studies do not seem to mention the average headache duration. Cai et al. reported that 61% of their patients had PIH > 2 h, whereas Verrotti et al. indicated that 52% of their patients had PIH lasting > 60 min and 48% lasting 30–60 min. In both studies, the authors did not report the maximum duration of headaches (1,13).
The differences between the presented results and those reported in adult studies might be age related (differences in central nervous system development), methodology related, or due to the influence of analgesics on headache duration. In addition, the studies do not report whether the participants took analgesics for their other chronic diseases, which might have modified the onset and duration of their peri-ictal headaches.
In our study, PreIH began 30–240 min before and lasted until the onset of the seizure episode. Most authors did not describe the timespan between the headache onset and seizure onset. Some used an arbitrary mark, such as headaches that began ≤30 min before the seizure and those that began 24 h to 30 min before (11,12). None of the paediatric studies reported the average duration of PreIH. Cai et al. reported two participants with PreIH lasting < 30 min, eight with > 2 h duration and four who were not sure (13). In Verrotti’s sample, 89.2% of participants reported PreIH lasting 30–60 min (1).
The only studies that described the duration of PreIH are retrospective analyses of a Turkish sample > 10 years of age (5.9 h) and a Korean sample > 13 years of age (12.6 h), as well as a retrospective analysis of German adults (2.75 h) (15,17,28). In our study, the average duration of PreH was shorter: 1.8 h (second visit) and 1.2 h (third visit). The differences in results might be due to the same factors as in the case of PIH, though it appears that age might be the single most important factor because children have much greater difficulty in estimating the duration of symptoms than adults (Table 6).
Comparison results of previous studies.
In our study, about 40% of patients with PIH reported migraine (46% on the second visit, 38% on the third visit) in contrast to PreIH, which was rarely of that type (only one of seven participants on the third visit).
According to the literature, migraine or migraine-like headaches occur in 34–60.98% of adults with PIH (6,11,12,15,22,27,31). Among children with PIH, migraine occurs in 58–81.4% of cases, which is similar to our results (13,32).
Other authors did not report such small number of patients with pre-ictal migraine headache; however, all of the results differ from each other. Wang et al. reported migraine-like headache in 64% (38 of 59) of patients with PreIH, whereas Duchaczek et al. noted migraines in 49% of patients with PreIH (5,17). In another study, four of seven adults reported PreIH that met the criteria for migraine (30). Paediatric studies report migraine-like headaches in 50–93% of patients with PreIH (1,13). As mentioned before, the disparity in results might be due to the fact that it is more difficult to assess headache characteristics among children than adults. Despite regular entries in the headache diary, some of the descriptions were unclear or imprecise; therefore, the headache was automatically deemed as unclassified, which might have had significant impact on the number of migraines we reported.
According to the literature, a relatively small number of patients with peri-ictal headaches take analgesics or received such instructions from their physician (12,13,22,26,27). This might be surprising given the fact that 60–90% of patients with peri-ictal headaches report relief from over-the counter (OTC) analgesics (12,13). In addition, there are also reports of successful treatment of peri-ictal migraines with triptans (33).
The use of analgesics for peri-ictal headaches seems to vary depending on country. In a study of Chinese adults with epilepsy, none of the patients used analgesics for their PIH (6). In a German study from 2002, 30% of patients regularly used analgesics for PIH, whereas in a 2013 German study, only 39% did so (17,27). On the contrary, in a Norwegian study, the majority of the participants reported using analgesics (22).
The use of analgesics for peri-ictal headaches is also not uniform in the paediatric population either. Yankovski et al. report that 36% of Canadian children and teenagers used over-the-counter analgesics for PreIH and as many as 81% for PIH (12), whereas Cai et al. report that 60% of their Canadian participants used analgesics due to PreIH and 50% in the case of PIH. Interestingly, as many as 79% of Cai’s sample reported relief after ibuprofen and 53% after paracetamol (13). All authors seem to agree that the physician’s role in treating peri-ictal headaches is unfortunately small. Syvertsen reported that only 4% of patients with epilepsy received headache treatment recommendations from their physician, whereas Forderreuther and Yamane noted 0% (22,27,30).
In our study, participants with PIH were most likely to use analgesics (46.2% on the second visit, 50% on the third visit). Interestingly, despite similar headache intensity, participants with PreIH were unlikely to use analgesics (8.33% on the second visit, 0% on the third visit). None of our participants used prescription analgesics or triptans.
The reported differences in the amount of anti-convulsant medication used are most likely related to cultural and knowledge differences among patients. Existing data does not support a correlation between the symptom intensity and the amount of medication taken. As noted above, the majority of the literature describes headaches of similar intensity, > 7 points on the VAS. Our results might indirectly reveal a lack of awareness among the guardians and doctors about the burden of headaches in the lives of patients with epilepsy.
Anti-convulsant drugs such as topiramate and valproic acid can be used in prevention of migraines, therefore it is logical to assume that using them in epilepsy treatment might reduce peri-ictal headaches (34). However, the existing literature do not reveal a correlation between the occurrence of peri-ictal headache and the type anticonvulsant medication (22).
Conclusions
Our observations confirm the existing data that peri-ictal headaches most often appear shortly after the seizures (post-ictal headaches). The results of our study on the paediatric population correlate with previous reports on the adult population that post-ictal headaches most frequently occur after generalised convulsive seizures.
Similar to previous reports, we conclude that post-ictal headaches are migraine, moderate to severe in intensity. In addition, we noticed that despite the post-ictal headache frequency and intensity, our patients received a minimal amount of prescription headache medication and the use of over-the-counter medication was also infrequent, though this varied depending on where the patient lived.
Although many published articles report the frequency of pre-ictal headaches, they are not described in the International Classification of Headache Disorders. In our study, we also confirmed the existence of pre-ictal headaches, which preceded 8.21% of seizures in the studied population. However, unlike in existing literature, we classified the pre-ictal headaches as tension-type or not elsewhere classified headaches, rather than as migraine. This discrepancy might be due to differences in methodology and patient enrolment into the study. It appears that more research is needed to describe the frequency of pre-ictal headaches, their clinical features and to formulate their diagnostic criteria.
Clinical implications
The most common type of peri-ictal headache in the paediatric population is post-ictal headaches, and they are most likely to appear after a generalised seizure. Although many published articles report the frequency of pre-ictal headaches, they are not described in the International Classification of Headache Disorders. In our study we confirmed the existence of pre-ictal headaches, which preceded 8.21% of seizures in the studied population. In a paediatric population, most post-ictal headaches occurred in < 1 h after the seizure and lasted minutes to hours, whereas pre-ictal headaches occurred 30–240 min before the seizure. Peri-ictal headaches were most often moderate in intensity. Only 30% of patients took analgesic medication, usually to treat post-ictal headaches.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
