Abstract
Little is known about factors associated with migraine pharmacotherapy during pregnancy. Of 60 435 pregnant women in a population-based cohort, 3480 (5.8%) reported having migraine during the first 5 months of pregnancy. Of these, 2525 (72.6%) reported using migraine pharmacotherapy, mostly non-narcotic analgesics (54.1%) and triptans (25.4%). After adjustment for sociodemographic factors and comorbidities in logistic regression analysis, high pregestational body mass index [odds ratio (OR) 1.3, 95% confidence interval (CI) 1.2, 1.4], sleep < 5 h (OR 1.6, 95% CI 1.3, 1.9), being on sick-leave (OR 1.3, 95% CI 1.2, 1.5) and acute back/shoulder/neck pain (OR 0.6, 95% CI 0.6, 0.7) were associated with migraine pharmacotherapy during pregnancy. Many women need drug treatment for migraine during pregnancy, and the choice of pharmacotherapy during this period may be influenced by maternal sociodemographic factors and comorbidities.
Introduction
Between 20% and 80% of pregnant women are estimated to have self-reported migraine (1–3). Many migraineurs may experience an exacerbation of their symptoms at the beginning of the first trimester (3–6). Consequently, these women may require pharmacotherapy during this period, which also corresponds to the most vulnerable period of fetal development. Consumption of both prescription and non-prescription drugs has been shown to decrease during pregnancy, and a higher percentage of women on pharmacotherapy has been shown to use over-the-counter drugs rather than prescription-only medications during this period (7–10). One previous study showed that pregnant migraineurs followed the same pattern, using paracetamol (22.5%), non-steroidal anti-inflammatory drugs (NSAIDs) (7.1%) and antihistamines (5.3%) most frequently (11). Despite their non-prescription status, many of these drugs have limited safety profiles during pregnancy, a fact of which pregnant patients may not be fully aware.
Information on maternal characteristics associated with migraine pharmacotherapy during pregnancy is sparse. One study including 905 pregnant women found an association between parity, maternal age, and migraine drug therapy during pregnancy (11). Migraineurs who mainly used sumatriptan during pregnancy were older and more frequently of first parity when compared with pregnant women without migraine. More detailed information about such factors is warranted in order to enable the identification of women at risk of receiving suboptimal migraine treatment during pregnancy and possibly women at risk of developing pregnancy complications associated with untreated migraine.
The main objectives of this study were to identify maternal characteristics associated with migraine pharmacotherapy during the first months of pregnancy when the fetus is at its most vulnerable.
Materials and methods
Data for this study were retrieved from the newest dataset (version 3) released by the Norwegian Mother and Child Cohort Study, which covers pregnant women recruited between 1999 and 2006. The Norwegian Mother and Child Cohort Study is an observational, prospective cohort study of pregnancy conducted by the Norwegian Institute of Public Health (12). The main objective of The Norwegian Mother and Child Cohort Study is the evaluation of the effect of an extensive number of exposures on pregnancy outcome, the health status of the mother during pregnancy, and the health status of the child during pregnancy and after birth. The present study was approved by the Regional Committee for Ethics in Medical Research region South and the Norwegian Data Inspectorate.
Data collection
All pregnant women living in Norway and attending hospitals or maternity units (about 60 in total) with > 100 births per year (in total 52 units, of which 50 had participated in the study by 2006) are invited to participate in the Norwegian Mother and Child Cohort Study prior to their first routine ultrasound examination. Approximately 55 000 women living in Norway give birth every year, and > 95% of them deliver at maternity units with > 100 births per year. Overall, 42% of the invited mothers agreed to participate in this study during the period 1999–2006. Together with an appointment for the first ultrasound scan between gestational weeks 17 and 18, the pregnant women also receive a postal invitation including an informed consent form, the first questionnaire, an information leaflet, and a questionnaire and consent form for the father of the child. In total, five questionnaires are to be filled out by the mother, the first three during pregnancy and the fourth and fifth 6 and 18 months after delivery. The first questionnaire, to be answered between weeks 17 and 18 of gestation, covers sociodemographic data, maternal medical history and exposures during the 6 months prior to pregnancy and approximately during the first 5 months of the current pregnancy. The questionnaires and a more detailed description of the Norwegian Mother and Child Cohort Study are available at its website (12). Every birth after gestational week 12 is recorded in the Medical Birth Registry of Norway. As each pregnant woman has a unique personal identification number, all data from the Norwegian Mother and Child Cohort Study can be linked to the pregnancy and the birth record forms in the Medical Birth Registry of Norway.
Study population
The study population consisted of 60 435 pregnant women who were recruited for the Norwegian Mother and Child Cohort Study between 1999 and 2006, who had answered questionnaire 1, and who had records in the Medical Birth Registry of Norway.
Dependent variables
The criteria for having migraine included answering ‘yes’ to questions about migraine and/or migraine pharmacotherapy in questionnaire 1. The translated version of the questions asked in the first questionnaire (covering the first trimester of pregnancy) reads as follows: ‘39. Do you have or have you had (migraine)? If you have taken medication (tablets, mixtures, suppositories, inhalers, creams, etc.) in conjunction with the illness or health problem, give the name(s) of the medication(s) and state when you took them’.
The study participants were then allowed to tick any or all of the following choices: ‘Migraine: Before pregnancy/During pregnancy. Use of medication: Last 6 months before pregnancy/During pregnancy: weeks 0–4/weeks 5–8/weeks 9–12/weeks 13+’.
The dependent variables were pregnant women on the following subsets of migraine agents: ‘no migraine agents during pregnancy’ (yes/no), ‘any migraine agent during pregnancy’ (yes/no), ‘triptans during pregnancy’ (yes/no), and ‘other migraine agents during pregnancy’ (yes/no). These subsets were created using answers to questions regarding both migraine agent use and migraine in questionnaire 1 as long as the name of the migraine agent and timing of either medication use or migraine was stated.
Drug therapy was classified according to the Anatomical Therapeutic Chemical Classification System developed by the World Health Organization (13). Migraine agent use was defined as (No), (Yes, during the 6 months prior to pregnancy but not during pregnancy), (Yes, both prior to pregnancy and during pregnancy), and (Yes, during pregnancy).
Independent variables
The selection of the specific maternal characteristics listed below was mainly based on current knowledge of medication use during pregnancy, which, among other factors, may depend on maternal age, parity, economic and educational status, and maternal health and comorbidities occurring simultaneously with migraine and migraine therapy. In addition, based on the fact that the association between serotonin levels and migraine has been shown to play a major role in migraine pathophysiology (14–20), studying the effect of a possible sudden drop in serotonin activity was considered worth pursuing. An independent variable ‘selective serotonin reuptake inhibitor (SSRI) discontinuation’ was therefore created, by taking into account and identifying all pregnant migraineurs with depression who reported the use of SSRIs prior to but not during pregnancy. Also, since β-blockers are used in the prevention of migraine attacks and β2-receptor agonist use is often associated with headache, it was considered to be of interest to analyse whether β2-receptor agonist use would have a possible provoking effect on migraine. The variable ‘β2-receptor agonist therapy during pregnancy’ was created by including all records of β2-receptor agonist use in subjects concomitantly suffering from asthma.
Characteristics selected for initial analysis included maternal age (≤ 19 years/20–29 years/30–39 years/≥ 40 years), marital status (married to or cohabiting with child's father/not), parity (defined as all previous pregnancies lasting ≥ 12 weeks) (primiparous/multiparous), body mass index (BMI) prior to pregnancy (≤ 18.5 kg/m2/18.6–25.0 kg/m2/> 25.0 kg/m2), sleep duration (< 5 h/5–10 h/> 10 h), physical activity (never/yes, but ≤ once a week/> once a week), education (primary/secondary/tertiary), employment (employed/unemployed), annual household income calculated in Euros (< 90 000/90 000–145 000/> 145 000), smoking during pregnancy (yes/no), chronic medical conditions (diabetes, thyroid disorders, epilepsy, asthma, chronic musculoskeletal disorders, psychiatric disorders) (yes/no), high blood pressure (defined as systolic blood pressure ≥ 140 mmHg) (yes/no), and acute back/neck/shoulder pain (yes/no). Sick-leave during pregnancy was coded for as (on sick-leave/not on sick-leave).
Statistical analysis
The Pearson's χ2 test and logistic regression were used to identify associations between maternal characteristics and migraine pharmacotherapy. A P-value of < 0.05 was considered statistically significant. The χ2 tests were used during preliminary analyses to assess which maternal characteristics were eligible for logistic regression analysis individually. The following independent variables were used in the final logistic regression analyses: maternal age, parity, pregestational BMI, sleep duration, sick-leave, high blood pressure, discontinuation of SSRIs during pregnancy, β2-receptor agonist therapy during pregnancy, and acute back, neck and/or shoulder pain. The model was restricted to include statistically significant variables only. The threshold for retaining variables in the model was P < 0.001.
Possible multicollinearity among the independent variables was identified using multiple regression analysis. The tolerance values for multicollinearity were set at > 0.5. Hosmer and Lemeshow goodness-of-fit tests > 0.5 indicated robust models and were considered valid in the logistic regression analyses. All statistical analyses were performed with the Statistical Package for Social Sciences
Results
Of the total cohort of 60 435 pregnant women, 6580 (10.9%) reported having migraine at any time before and/or during pregnancy. The study participants displayed comparable sociodemographic characteristics to the general population except for educational level and household income, both of which were higher among women who reported having migraine in the study (21). The total number of women who reported having migraine during the 6 months before pregnancy was 6099 (of these, 6086 answered ‘yes’ to having had migraine prior to pregnancy and 2268 answered ‘yes’ to having used migraine agents prior to pregnancy). Of the 6099 reporting having migraine during the 6 months prior to pregnancy, 2999 (49.2%) also reported having migraine during pregnancy, whereas 3100 women (50.8%) reported that their migraine symptoms ceased. In addition, 481 of all the women in the cohort (0.8%) reported having had migraine during pregnancy without reporting migraine during the 6 months prior to pregnancy. Thus, the total number of women reporting migraine during pregnancy was 3840 (5.7%), of whom 3334 answered ‘yes’ in questionnaire 1 to having migraine during pregnancy and 2525 answered ‘yes’ to having used migraine agents during pregnancy.
Maternal characteristics associated with migraine
Table 1 shows the characteristics of the study population compared with women who had no migraine. Logistic regression performed on these characteristics shows that after adjustment for possible confounders, pregestational BMI > 25.0 kg/m2, sleep duration < 5 h, unemployment, current or previous depression or asthma, sick-leave during pregnancy, high blood pressure during pregnancy and SSRI discontinuation during pregnancy were all significantly associated with migraine (Table 1). Acute musculoskeletal pain of the back, neck and/or shoulder was the only characteristic found to be negatively associated with migraine.
Maternal characteristics of the study population and their associations with migraine shown as crude and adjusted odds ratios (significant associations are shown in bold)
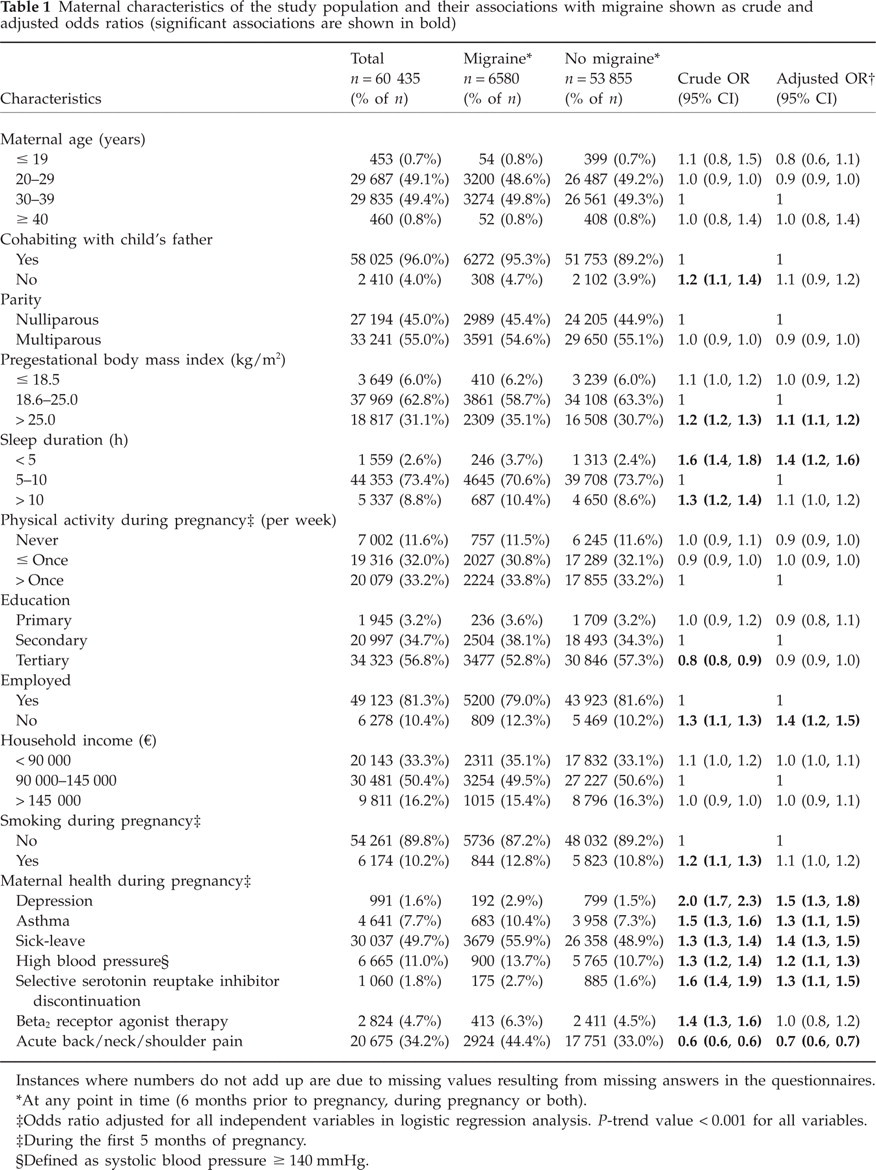
Instances where numbers do not add up are due to missing values resulting from missing answers in the questionnaires.
At any point in time (6 months prior to pregnancy, during pregnancy or both).
Odds ratio adjusted for all independent variables in logistic regression analysis. P-trend value < 0.001 for all variables.
During the first 5 months of pregnancy.
Defined as systolic blood pressure ≥ 140 mmHg.
Migraine pharmacotherapy
In total, 2525 women (72.6 %) reported using a migraine agent during pregnancy (Table 2). The proportion of women using migrainous agents was higher among women who reported migraine both prior to and during pregnancy (75.9%) when compared with women who reported having migraine during pregnancy only (51.8%).
Migraine agent use during the 6 months prior to pregnancy and during pregnancy
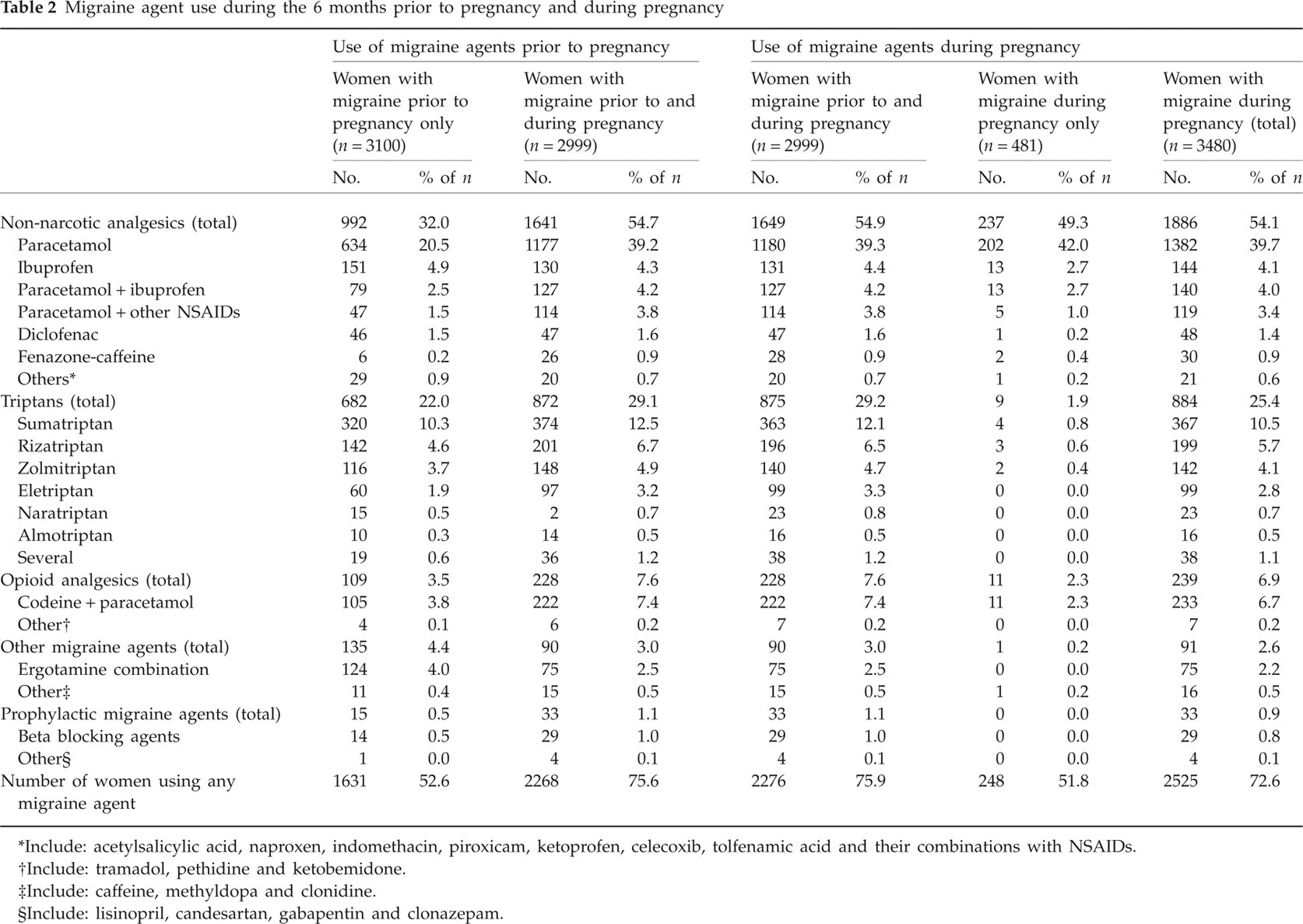
Include: acetylsalicylic acid, naproxen, indomethacin, piroxicam, ketoprofen, celecoxib, tolfenamic acid and their combinations with NSAIDs.
Include: tramadol, pethidine and ketobemidone.
Include: caffeine, methyldopa and clonidine.
Include: lisinopril, candesartan, gabapentin and clonazepam.
Non-narcotic analgesics, the most frequently used migraine agents during pregnancy, were used by 54.1% of all women with migraine during this period; triptans were used by 25.4% and opioid analgesics by 6.9% of pregnant women. Paracetamol, the most commonly used migraine agent, was used by 39.7% of women during the first 5 months of pregnancy, whereas sumatriptan, the most frequently used triptan, was used by 10.5% of all women with migraine during this period.
There was almost no change in the extent or type of pharmacotherapy used by women prior to and during pregnancy among those who reported having migraine both prior to and during pregnancy. Ergotamine combination preparations continued being used in this group of migraineurs even though this drug is contraindicated during pregnancy (Table 2).
Maternal characteristics associated with migraine pharmacotherapy
Both lifestyle and comorbidity were found to be associated with migraine pharmacotherapy during pregnancy. Table 3 shows the results of logistic regression analyses performed on maternal characteristics that were found to be significantly (P < 0.001) associated with migraine agent therapy during pregnancy after adjustment for age, parity, pregestational BMI, sleep duration and various comorbidities.
Factors associated with migraine agent use during pregnancy (significant associations are shown in bold)
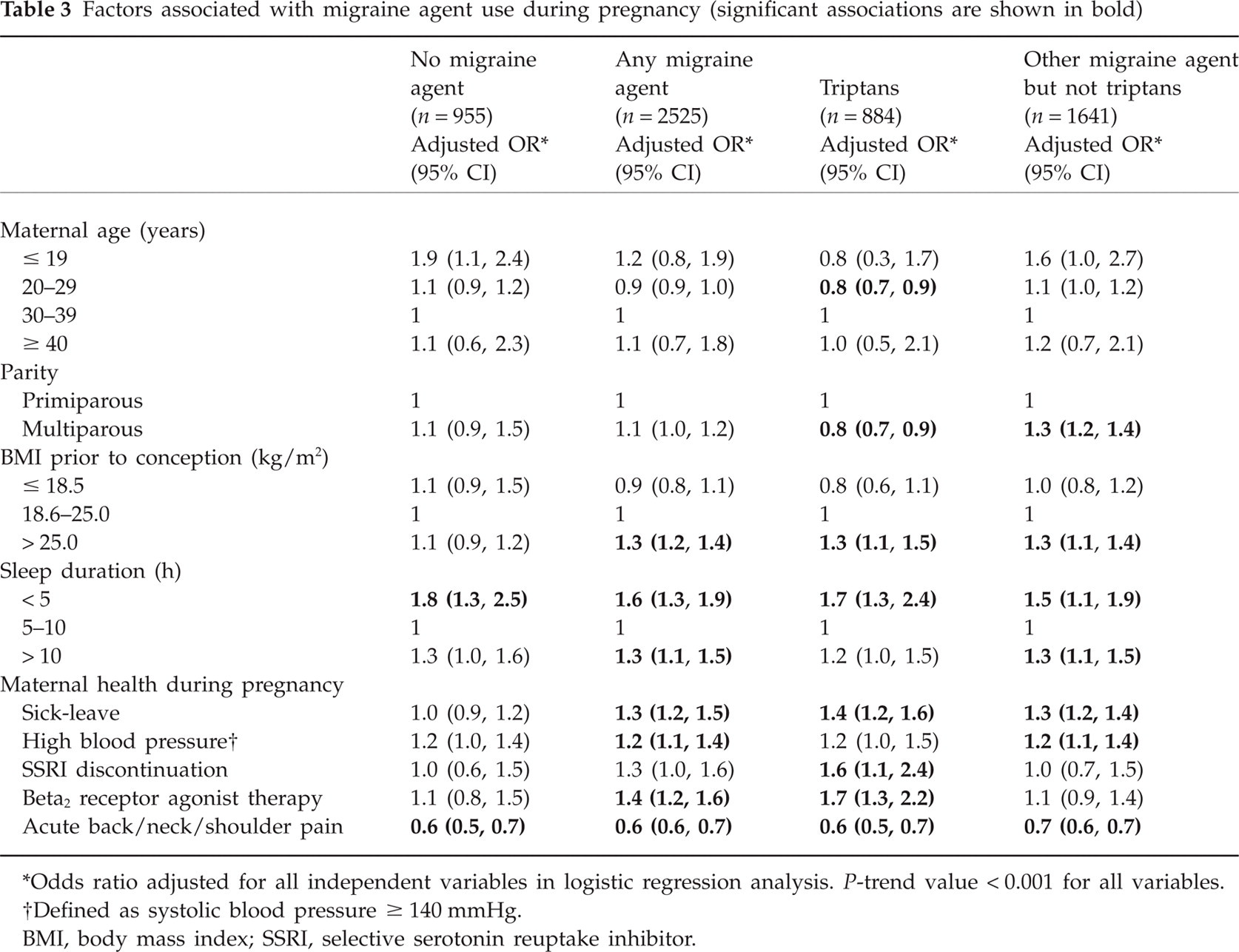
Odds ratio adjusted for all independent variables in logistic regression analysis. P-trend value < 0.001 for all variables.
Defined as systolic blood pressure ≥ 140 mmHg.
BMI, body mass index; SSRI, selective serotonin reuptake inhibitor.
Sleep duration < 5 h per night, pregestational BMI > 25.0 kg/m2 and being on sick-leave were the factors associated with all types of migraine pharmacotherapy during pregnancy. Acute musculoskeletal pain of the back, neck and/or shoulder was negatively associated with migraine pharmacotherapy during this period (Table 3). Young maternal age and multiparity were negatively associated with triptan use, whereas discontinuation of SSRI therapy and β2-receptor agonist use were both associated with triptan therapy (Table 3). Of the women who used triptan therapy during pregnancy, 2.2% had depression, all of whom discontinued SSRI therapy. Of the women who used other migraine agents, 3.0% had depression, 67.4% of whom discontinued SSRI therapy.
Discussion
Of all the 60 435 pregnant women in the cohort, 6580 (10.9%) reported having migraine. This finding is consistent with results from other studies that were also based on the prevalence of self-reported migraine (1–3, 22).
More than 70% of women who had migraine during pregnancy reported using migraine agents during this period. This indicates the necessity and importance of migraine pharmacotherapy during pregnancy.
Women who reported having migraine both prior to and during pregnancy had the same pattern of drug use during pregnancy when compared with their use prior to pregnancy. This is of interest, since the use of NSAIDs in particular is not generally recommended at the beginning of the first trimester (23). Furthermore, the women continued using ergotamine combination preparations, which are contraindicated during pregnancy. This may have been due to the fact that they did not yet know they were pregnant at the time they had used the ergotamine combination preparations or were unaware of the possible adverse effects of this drug on pregnancy outcome (24).
Migraine agent use among women who reported having migraine during pregnancy only was lower than that among women who reported having migraine prior to and during pregnancy. The most marked difference was found in triptan and opioid analgesic agent use. The most likely explanation for this is a general reluctance to prescribe and use migraine agents with limited safety documentation during pregnancy, and the fact that many prophylactic migraine agents are contraindicated during pregnancy (25). It is also possible that many physicians and patients alike prefer the ‘wait-and-see’ approach and choose to treat migraine symptoms acutely with non-narcotic analgesics, especially paracetamol with its good safety profile, and hope the migraine symptoms will soon resolve.
More than half of the women who used triptans during pregnancy reported using triptans other than sumatriptan. This fact is interesting, since sumatriptan use during pregnancy is the most documented of all the triptans and is therefore recommended by most experts when the use of triptans during pregnancy is considered necessary (24). The most likely explanation is the reluctance of the prescriber and/or patient to change an already established and effective therapy when a triptan other than sumatriptan had been used prior to pregnancy.
Maternal characteristics associated with migraine and migraine agent use during pregnancy
Several sociodemographic factors were found to be associated with both migraine and migraine agent therapy during pregnancy. Some factors were associated with the use of all types of migraine pharmacotherapy during the first trimester, whereas others were specifically associated with triptan use.
Several studies have shown a link between migraine and lack of sleep or sleep of poor quality (26–30). This may possibly be attributable to serotonin fluctuations (19, 20, 31, 32). Sleep duration of < 5 h per night was also found to be associated with both migraine and all types of migraine pharmacotherapy in this study (Tables 1 and 3). It is possible that migraine itself may lead to suboptimal sleep quality, since sleep lasting > 10 h was also associated with migraine pharmacotherapy (Table 3). Significant associations were found between pregestational BMI > 25.0 kg/m2 and both migraine itself and all types of migraine pharmacotherapy (Tables 1 and 3). It is known from previous studies that an association between obesity and migraine exists (15, 32–34). A similar association between sick-leave, migraine, and all types of migraine pharmacotherapy was found (Tables 1 and 3), indicating that women on sick-leave perhaps suffered from more severe forms of migraine.
On the other hand, acute musculoskeletal pain of the back, neck and/or shoulder was found to be negatively associated with migraine (Table 1) and also with less migraine pharmacotherapy during pregnancy (Table 3). It is possible that pregnant women who reported having acute musculoskeletal pain of the back, neck and/or shoulder also concomitantly used non-narcotic analgesics, thus decreasing their need for specific migraine agents.
Younger maternal age and multiparity were both found to be negatively associated with triptan use. These women may have had a positive experience with non-triptan agents or spontaneously improved migraine symptoms during their previous pregnancies, which may have influenced their choice of migraine pharmacotherapy during the current pregnancy.
SSRI discontinuation was also associated with triptan therapy during pregnancy (Table 3). As an increased risk of serotonergic adverse effects has been described in patients concomitantly treated with SSRIs and triptans (34), it is likely that this association is to a large extent due to physicians choosing to discontinue the SSRIs before establishing triptan therapy to avoid possible drug interaction. This is shown by the fact that all triptan users discontinued their SSRI therapy. However, migraine coexisting with depression and the subsequent use of SSRI therapy has been shown to be more severe (18, 33, 35), and this may have led to the necessity of triptan therapy. SSRI discontinuation may also have been associated with exacerbation of migraine due to serotonin level fluctuations. The possibility that this finding is due to chance only cannot be excluded. No other studies analysing the association between SSRI discontinuation and migraine have been conducted so far.
Another factor that may have been associated with triptan therapy during pregnancy was β2-receptor agonist therapy (Table 3). Due to their smooth muscle relaxant activity and possible vasodilatory effect, the fact that β-receptor antagonists are used as prophylactic agents in migraine and also the fact that β2-receptor agonists have been associated with headache side-effects, the use of β2-receptor agonists was considered as a possible exacerbating factor for migraine. The association found between asthma and migraine is in accordance with previous findings (36); however, no previous investigations exist of the effect of β2-receptor agonist therapy on migraine.
Further studies are needed to confirm the possible associations between SSRI discontinuation or β2-receptor agonist therapy and migraine before taking the above findings into clinical consideration.
High blood pressure was found to be associated with both migraine and migraine agent use during pregnancy, except for triptan therapy (Tables 1 and 3). It is unclear whether migraine is precipitated by high blood pressure, whether migraine increases blood pressure or whether both processes occur simultaneously and independently of each other. Previous studies have shown a significant association between migraine during pregnancy and the occurrence of pre-eclampsia and increased diastolic blood pressure (37–40) Abnormal vascular reactivity occurring during migrainous episodes has been suggested as a possible cause (39).
Study strengths and limitations
This study, consisting of > 60 000 pregnant women, represents the largest general study population on migraine during pregnancy. The vast spectrum of health-related and sociodemographic data enabled subanalyses to be performed on the associations between maternal characteristics and migraine pharmacotherapy while controlling for potential confounders. The study has also several limitations. First, it is based upon self-reported migraine and migraine pharmacotherapy. The migraine diagnosis has not been validated, and the prevalence data must therefore be considered with caution. There might also be underreporting of drug use due to limited space of recording in the questionnaire. However, there is no reason to believe this reporting to be differential among the women. Despite the low participation rate, the differences between participants in the Norwegian Mother and Child Cohort Study and in the general population of pregnant women are only minor (21). Thus, the estimates of the various associations are most likely to be valid also in the general population of pregnant women.
Conclusion
Even though migraine symptoms often improve during pregnancy, many women still require migraine pharmacotherapy during this period. Drug therapy during the first months of pregnancy is particularly challenging, as this is the period when the fetus is most vulnerable. Most pregnant women use acute attack migraine agents such as paracetamol, but drugs that are not recommended during pregnancy or are insufficiently documented are also being used. Both lifestyle and comorbidity may influence the need for and choice of migraine pharmacotherapy during pregnancy. As drug use for migraine is so common during pregnancy, it is essential to provide these women with adequate information about safe and effective pharmacotherapy.
