Abstract
Background
There are various studies on experimentally provoked ‘ice-cream headache’ or ‘headache attributed to ingestion or inhalation of a cold stimulus’ (HICS) using different provocation protocols. The aim of this study was to compare two provocation protocols.
Methods
Ice cubes pressed to the palate and fast ingestion of ice water were used to provoke HICS and clinical features were compared.
Results
The ice-water stimulus provoked HICS significantly more often than the ice-cube stimulus (9/77 vs. 39/77). Ice-water-provoked HICS had a significantly shorter latency (median 15 s, range 4–97 s vs. median 68 s, range 27–96 s). There was no difference in pain localisation. Character after ice-cube stimulation was predominantly described as pressing and after ice-water stimulation as stabbing. A second HICS followed in 10/39 (26%) of the headaches provoked by ice water. Lacrimation occurred significantly more often in volunteers with than in those without HICS.
Discussion
HICS provoked by ice water was more frequent, had a shorter latency, different pain character and higher pain intensity than HICS provoked by ice cubes. The finding of two subsequent HICS attacks in the same volunteers supports the notion that two types of HICS exist. Lacrimation during HICS indicates involvement of the trigeminal-autonomic reflex.
Keywords
Background
According to the current International Classification of Headache Diseases (ICHD-3 beta), ice-cream headache is now called ‘headache attributed to ingestion or inhalation of cold stimulus’ (HICS). It is characterised by a short-lasting frontal or temporal pain, which may be intense, induced in susceptible people by passage of cold material (solid, liquid or gaseous) over the palate and/or posterior pharyngeal wall (1). HICS is one of the most common primary headache disorders with a prevalence ranging from 38.3% to 79% in children and 5.9% to 74% in adults (2–8). Various studies have investigated HICS by different experimental approaches (3,4,8–13). Three studies used ice cubes of different sizes to provoke HICS (8–10). Other studies used ice cream (3,11,13) and ice water ingested through a straw (4,12) and ingested without a straw (13). So far, no study has compared the effects of different cold stimuli. In particular, it has not been established which method induces HICS most effectively and whether the clinical features of HICS depend on the cold stimulus used. As previously mentioned, ‘an important question remains to be answered: would other kinds of cold stimuli trigger different forms of HICS?’ (10). In the present study two experimental protocols (ice cubes and ice water) to provoke HICS attacks were compared and evaluated.
Participants
Seventy-seven healthy volunteers (47 females, mean age 27 ± 7 years, range 18–60) were included in this study. Forty-four of 77 (57%) volunteers reported a positive history of HICS. There was no significant difference of HICS prevalence between females and males (17/30 males and 27/47 females). Exclusion criteria were other forms of headache disorders, cardiovascular diseases, brain tumours, epilepsy, pregnancy and history of syncopes. Volunteers were recruited via postings on the university blackboard and via mailing lists of local medical students. The study was conducted between June and July 2015. The study was approved by the local ethics committee. Participants gave informed consent before taking part.
Methods
In this cross-sectional analytical observational study, two protocols for provoking HICS were tested.
Ice-cube stimulation (according to Selekler et al. (8)): Volunteers were asked to press a trapezoid ice cube to the hard palate (contact area to the tongue 30 mm × 25 mm and to the palate 25 mm × 20 mm) for 90 seconds (s) from closing the mouth until removing the ice cube. Ice cubes were taken directly out of the freezer with a temperature of –16℃ (3.2°F). Ice-water stimulation: Volunteers were asked to drink 200 ml of ice water with a temperature of 0℃ (32°F) as fast as possible.
During both cold stimulations volunteers were asked to raise their thumb when the headache occurred and lower it when it disappeared. Subsequently an examiner who was blinded to HICS history of the volunteers inquired as to the clinical features of the provoked headaches. Data on latency from the beginning of the cold stimulation to HICS occurrence, duration of HICS, pain quality, pain intensity and lacrimation were collected.
Ice cream was not used because taste and texture might influence the induction of HICS. In addition, localisation of the cold stimulus is difficult to standardise with ice cream. Ice water ingested through a straw was not used because placement of the straw at the palate cannot be standardised (4).
For statistical analysis of latencies and duration, Student’s
Latency and duration data are reported as median and range. All other data are reported as mean ± SD.
Results
Ice-cube protocol
HICS characteristics in response to ice cube and ice water.
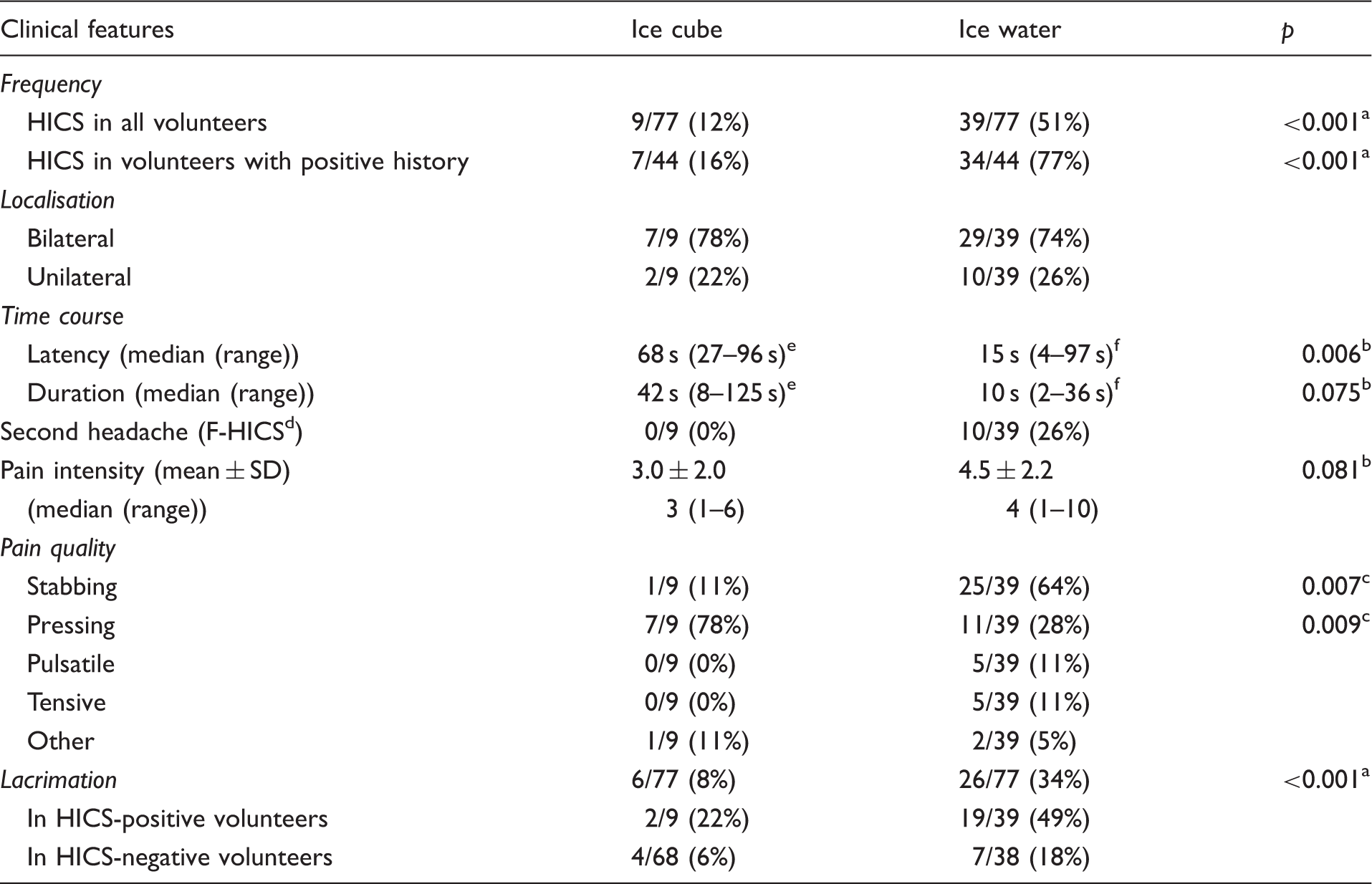
χ2-test; bStudent’s
HICS started 68 s (27–96 s) after the beginning of the ice-cube stimulation and the duration was 42 s (8–125 s). Pain intensity was 3.0 ± 2.0 (range 1–6) of 10 on a numerical pain scale. Most volunteers experienced HICS in response to ice-cube intake as a pressing bilateral pain in the forehead and temple (Figure 1(a) and Table 1).
Localisation of HICS in response to ice-cube stimulation in eight volunteers (a) and in response to ice-water stimulation in 39 volunteers (b). Frequencies were grouped into three different grey intensities (<20%; 21%–40%; >41%). HICS: headache attributed to ingestion or inhalation of cold stimulus.
Ice-water protocol
Ice-water-provoked HICS in 39/77 (51%) volunteers. In volunteers with a positive history of HICS, the ice-water-induced HICS in 34/44 (77%). Thus, in five of 33 (15%) volunteers, HICS was provoked for the first time in this protocol (Table 1).
HICS started 15 s (4–97 s) after beginning of the ice-water intake and the duration was 10 s (2–36 s) (Figure 2). Pain intensity was 4.5 ± 2.2 (range 1–10) of 10 on a numerical pain scale. Most volunteers experienced HICS in response to ice-water intake as a stabbing bilateral pain in the forehead and temple (Figure 1(b) and Table 1). HICS occurrence was not different between females and males (26/47 females and 13/30 males, Latency (a) and duration (b) of HICS in 36 volunteers after ice-water stimulus. Times were grouped into 10-second (a) and five-second (b) intervals. HICS: headache attributed to ingestion or inhalation of cold stimulus.
Following HICS
A second HICS followed in 10/39 (26%) volunteers after ice-water intake but not after the ice-cube stimulus. It started 43 s (16–200 s) after ice-water intake and the duration was 12 s (3–26 s). The time interval between the end of the first headache and onset of the second headache was 11 s (2–141 s) (Figure 3).
Latency and duration of HICS in 10 volunteers with a second headache (F-HICS) after ice-water intake. Each horizontal line represents one volunteer. HICS: headache attributed to ingestion or inhalation of cold stimulus; F-HICS: following headache; s: seconds.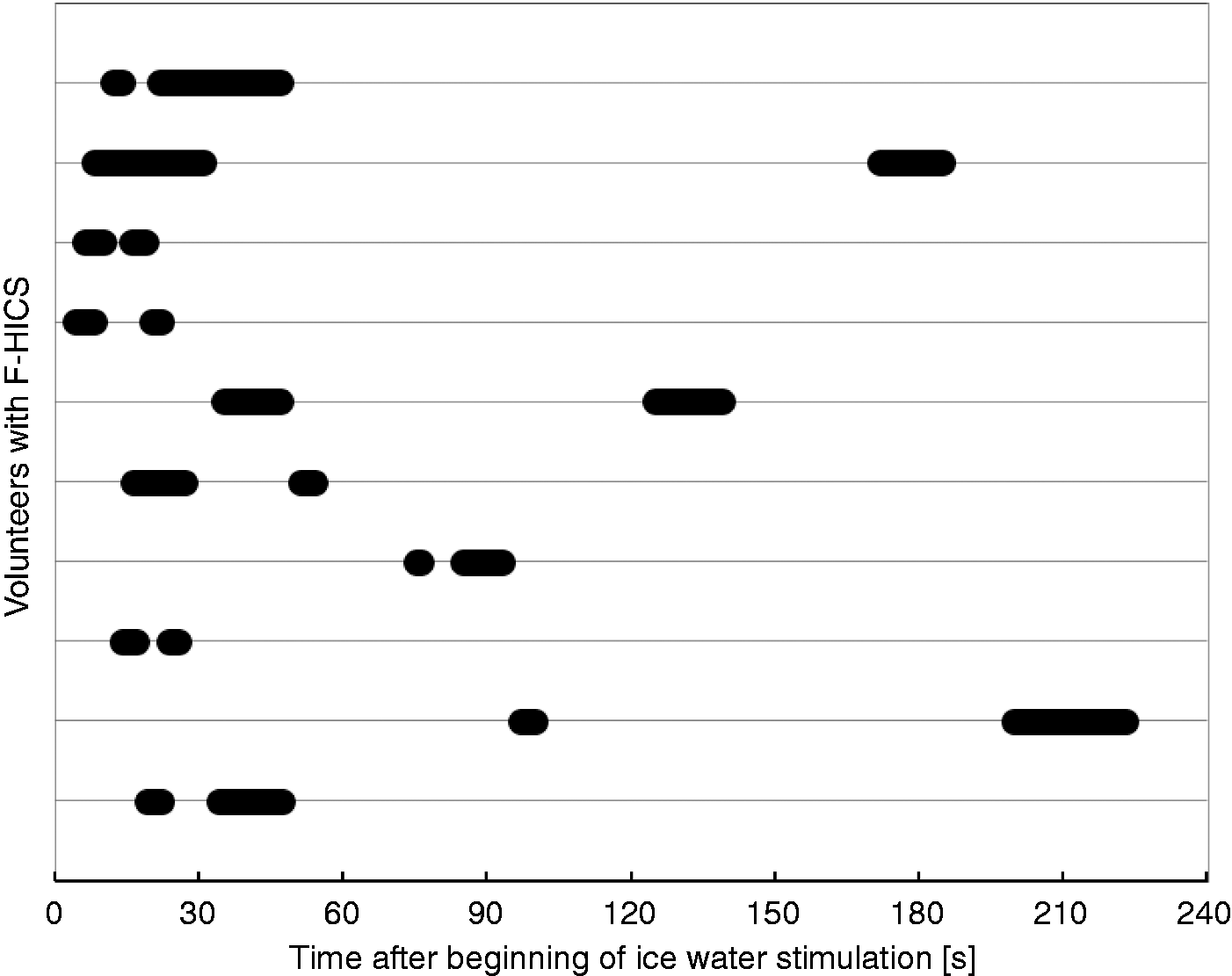
Ice-cube-provoked HICS compared to ice-water-provoked HICS
The ice-water stimulus provoked HICS significantly more often than the ice-cube stimulus (odds ratio (OR) 7.75, 95% confidence interval (CI) 3.39–17.72,
Discussion
The ice-cube stimulus and the ice-water stimulus provoked different forms of HICS. HICS provoked by ice water had a shorter latency, different pain character and higher pain intensity than HICS provoked by ice cubes. The ice-water stimulus provoked HICS significantly more often than the ice-cube stimulus. Although temperature is the main stimulus to provoke HICS, a further drop in temperature from ice water to ice cubes (difference 16℃; 28.8°F) does not increase the frequency of HICS induction. The area exposed to a given stimulus, spatial extent, spatial summation and a high intake speed, achieved by drinking ice water, seem to be more important to effectively provoke HICS. Contrary to other reports a longer cold exposure as achieved by the ice-cube stimulus did not lead to an increased frequency of provoked HICS in this study (8,11). While the ice cube is capable of cooling down only a small surface on the tongue and palate, ice water cools down the entire oral cavity and pharyngeal wall in a short period of time. Thus, ice water stimulates not only the trigeminal nerve but also other nerves of the palate and pharynx such as the glossopharyngeal and the vagal nerve. A previous study showed that intake speed of the cold stimulus is relevant for HICS induction (3). This is consistent with the clinical observation in a questionnaire-based survey that only 4% of students experienced HICS during slow in contrast to rapid ice cream intake (14). Furthermore, it has been hypothesised that intense cooling of the pharynx by drinking ice water cools down the blood in the internal carotid artery due to the topographic proximity. The cooled down blood flow might lead to HICS by vasoconstriction in subsequent arteries (13). Cooling of the oesophagus and stomach with crushed ice did not lead to headache in a previous study (15).
It could be speculated that the different latencies might be explained by the different time profiles and localisation of the cold stimuli. The ice cube initially cools down only a small surface on the tongue and palate. While melting the ice cube might start to cool down a larger area at the palate and pharynx. The longer latency to the onset of HICS might be explained by the time-dependent melting process of the ice cube. The latency with ice cubes in this study is in line with the study by De Oliveira and Valença using ice cubes (10).
Another explanation might be that there are two types of HICS as proposed by Bird et al. (11). In that study on HICS provoked by ice cream, there was one group (80% of volunteers) with an early onset (12.5 s) and short duration (21 s) and another group (20% of volunteers) with a later onset (102 s) and longer duration (236 s). The late-onset HICS was most common after swallowing ice cream. Therefore, it has been speculated that the early- and late-onset HICS were due to different areas and mechanism of cold stimulation (e.g. palate and teeth via trigeminal nerve vs. less-direct mechanisms through vasospasm or muscle contraction) (11).
The present findings of two subsequent HICS in the same volunteers after ice-water stimulation support the notion that indeed there might exist two different types of HICS. The term F-HICS (following headache) is proposed in analogy to F wave in neurophysiology. In analogy to the hypotheses of Bird et al. (11), the first headache attack might be due to the trigeminal stimulation of the palate and the F-HICS might correspond to vasospasm or muscle contraction. Lacrimation during ice-water-induced HICS indicates involvement of the trigeminal autonomic reflex.
In the present study median HICS duration in the ice-cube protocol tended to be longer than in the ice-water protocol (42 s vs. 10 s). Duration of HICS during the ice-water protocol was less than 30 s in 35/36 (97%) volunteers. This is consistent with data from two large questionnaire studies in which 72.3% and 77% reported HICS duration of less than 30 s in previous HICS attacks (2,14). Therefore, HICS duration provoked by ice water seems to be closer to the natural course of HICS than when provoked by ice cubes.
In the present study on volunteers without any other primary headache disorder, HICS was provoked in 12% of volunteers during ice-cube stimulation. This is consistent with the study by De Oliveira and Valença in which 17% of volunteers without any other headache disorder developed HICS after ice-cube stimulation (10). However, in patients with migraine or tension-type headache, HICS was provoked in up to 74% of patients by ice cubes (8,10). This indicates that headache patients react differently to an oral cold stimulus than healthy volunteers. Therefore, the homogeneous group of volunteers without any other headache disorder in the present study allows a clear characterisation of HICS-provocation protocols without confounding effects.
A limitation of this study is the acquisition of volunteers. Due to the acquisition method young volunteers and volunteers with a positive history of HICS might have been more attracted to participating in this study. Therefore, the frequency of volunteers with provoked HICS during the study protocols may not be confused with HICS prevalence in the general public. However, investigating the prevalence of HICS was not the focus of this study.
Article highlights
Ice water is more effective in triggering ‘headache attributed to ingestion or inhalation of a cold stimulus’ (HICS) than ice cubes. HICS provoked by ice water had a shorter latency, different pain character and higher pain intensity than HICS provoked by ice cubes. Twenty-six per cent of the volunteers with provoked HICS during the ice-water protocol experienced a following headache after the first one resolved (F-HICS). Lacrimation during HICS indicates involvement of the trigeminal-autonomic reflex in HICS.
Footnotes
Acknowledgements
The authors would like to thank all volunteers who took part in the study and Prof Dr Andreas Wienke and Dr Charly Gaul for critical comments on this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
