Abstract
Background
Early observations by von Economo showed that the posterior part of the hypothalamus (PH) plays a prominent role in sleep-wake regulation. The PH is a candidate area involved in cluster headaches and other trigeminal autonomic cephalalgias (TACs) and is targeted for deep brain stimulation (DBS).
Case reports
Sleep studies in two men, 69- and 39-years-old, with pre-existing sleep disorders, before and after PH-DBS for pharamacoresistant cluster headache and SUNCT syndrome showed that PH-DBS led to a dramatic alteration of the patients’ sleep patterns. This coincided with an improvement of the predominantly diurnal TACs, suggesting a PH-DBS-induced change in sleep patterns. Hypnograms after DBS demonstrated disrupted sleep and a prolonged period of wakefulness after midnight in both patients, which was reproduced the second night.
Conclusions
PH-DBS, a promising treatment for severe refractory TACs, affects sleep quality and pre-existing sleep disorders. This needs to be considered when treating patients with PH-DBS.
Introduction
Since Constantin von Economo’s initial descriptions of encephalitis lethargica or Economo’s sleeping sickness, an encephalitis that specifically affects sleep regulation, the hypothalamus has been identified as the key player in determining sleep wake patterns (1,2). Early anatomical observations made by von Economo and experiments in animals identified that lesions within the posterior part of the hypothalamus, especially lesions at the junction between the midbrain and forebrain, manifest with severe hypersomnolence. In contrast, lesions of the anterior parts of the hypothalamus lead to severe insomnia (3).
Beside its prominent role in sleep-wake regulation, the posterior hypothalamus (PH) has been identified as a candidate area involved in cluster headaches and other trigeminal autonomic cephalalgias (TACs) such as short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT) syndrome (4,5). This pathophysiological insight is supported by functional neuroimaging techniques in humans showing activation of the PH grey matter in all the TACs (6,7). Deep brain stimulation of the ipsilateral PH region (PH DBS) has been used successfully in carefully selected cases with treatment-resistant TACs in the last 10 years (4).
Previous studies have shown that PH DBS in patients with chronic cluster headache improves sleep quality, possibly by controlling the frequency of headache attacks (8). In these reports, it is not clear how sleep is affected by the hypothalamic stimulation per se. Moreover, the effect of PH DBS on sleep in other TACs and on pre-existent sleep disorders is unclear. We present two patients undergoing treatment of pharmacoresistant trigeminal autonomic headaches (chronic cluster headache and SUNCT) with PH DBS; one patient also suffered from a pre-existing non-rapid eye movement (NREM) parasomnia, the other patient suffered from a suspected rapid eye movement (REM) behavioural disorder. We report changes in sleep following hypothalamic DBS. Both patients presented with a characteristic hypnogram after DBS treatment with decreased total sleep time and prolonged periods of wakefulness in the early morning hours.
Case 1
A 69-year-old with a 34-year history of pharmacoresistant left-sided cluster headache and a 22-year history of left-sided SUNCT syndrome underwent left PH DBS in 2010 (Figure 1(a–c)). He presented in the sleep clinic two years after implantation of the DBS with disrupted night sleep, reduced subjective sleep time and daytime sleepiness. The Epworth sleepiness scale score at that time was 15, indicative of significant daytime sleepiness. He reported vivid dreams for the last 20 years, some of which he had acted out, and in the past he awoke during night-time two or three times a week with immediate recall of these vivid dreams. He did not sleep-walk or get out of bed with these. This changed dramatically a few weeks following DBS implantation and stimulation, and he noticed a significant increase in the frequency of awakenings (three or four times every night) with more intense dreams; he would usually stay awake for a few hours per night, regularly around 2 a.m. Intriguingly, the headaches improved with the same delay as sleep became disrupted, suggesting that both effects could be attributed to hypothalamic region DBS. No medication changes were undertaken before and after the DBS implantation, except for a two-week trial of Duloxetine after DBS implantation resulting in a deterioration of his sleep pattern. There was a history of sleep paralysis with about two or three incidents per month which are unrelated to his vivid dreams and a previous history of sleep-talking. There was no history of hypnagogic hallucinations. A previous obstructive sleep apnoea screen revealed a borderline apnoea/hypopnoea index (9/h); provision of a continuous positive airway pressure (CPAP) mask made no significant change in the sleep pattern (fitted on the second night of polysomnography (PSG)). A two-night PSG after DBS implantation was performed to control for a first-night effect. This showed a short total sleep time (six hours, nine minutes the first and five hours, 43 minutes the second night) and poor sleep efficiency (58% first and 59% second night; normal ≥90%). There was poor sleep architecture due to very frequent awakenings with long wake periods in the middle of the night and in the early morning hours (both times around 2 a.m.), during which the patient stayed awake for ∼1.5 hours and this was reproduced the next night (Figure 1(e) and (f)); most awakenings were from REM sleep (17 awakenings each night). PSG was not supportive of NREM or REM parasomnias but these conditions could not be excluded.
MRI showing the site of DBS implantation and hypnograms pre- and post- DBS implantation. T2*-weighted images demonstrating lead location: axial MRI in patient 1 (panel (a)) and patient 2 (panel (g)) show the distal end of the DBS lead (white arrow) between the nucleus ruber and mammillothalamic tract in the left (a) and right (g) midbrain. Panel (b) and (c) (patient 1) and (h) and (i) (patient 2) show the trajectories of the DBS lead implanted via a frontal approach. Panels (e) and (f) show hypnograms of two consecutive nights in patient 1 after implantation of the DBS. Panel (j) shows a hypnogram of patient 2 prior to implantation of the DBS and in panels (k) and (l) are hypnograms of two consecutive nights after DBS implantation (patient 2). Note that both patients 1 and 2 have a prolonged awake period (around 2–3 a.m.) after DBS implantation, which is reproducible the second night. M: movement; W: wake; R: rapid eye movement (REM) sleep; 1,2,3,4: stages 1–4 non-REM (NREM) sleep; MRI: magnetic resonance imaging; DBS: deep brain stimulation.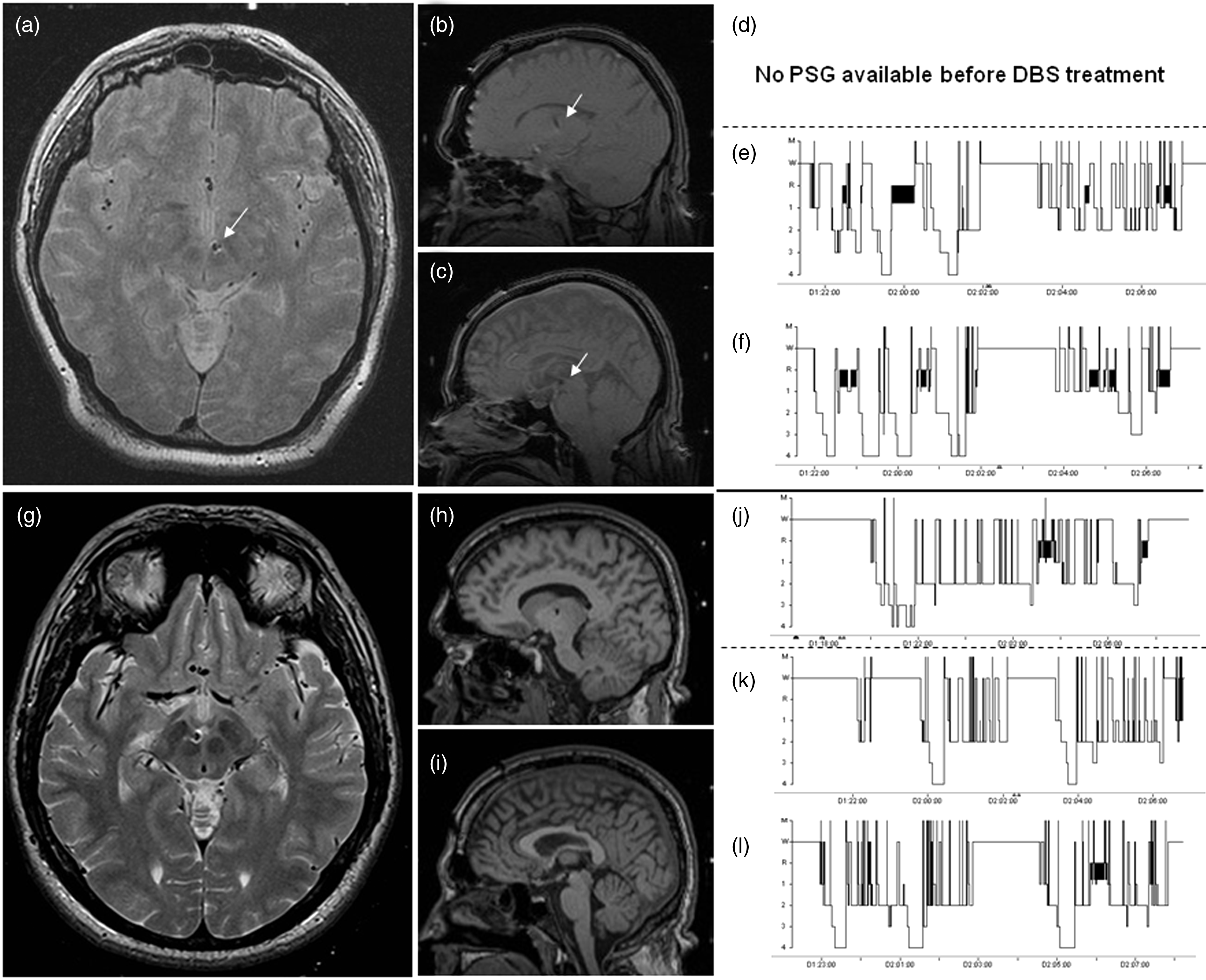
Case 2
This 39-year-old man was treated with right-sided PH DBS in 2012 for concomitant cluster headache and SUNCT syndrome (Figure 1(g–i)). One to two weeks after DBS implantation, headache frequency and pre-existent parasomnias improved dramatically. He had a history of sleep-talking and sleep-walking as a child; however, in the last 12 years this deteriorated with weekly episodes of sleep-walking, sleep-talking and occasionally violent attacks against his partner as part of the NREM behavioural disorder. Before DBS implantation he suffered from five episodes of NREM parasomnias per week supported by arousals out of slow wave sleep in a previous PSG in 2006 from another centre and also supported by a PSG performed in 2008 in our centre. In addition to that, the PSG in 2008 showed loss of REM atonia, suggesting that he suffers from both NREM parasomnia and REM parasomnia. This PSG showed that most of the awakenings were attributed to the NREM parasomnia. Following DBS he suffered only one episode per week. Cluster headaches and SUNCT syndrome improved after DBS implantation and the time course was similar to the improvement of the sleep-related disorder. Medication remained unchanged at the time the stimulator was implanted, and he was prescribed modafinil for daytime hypersomnolence and prazosin for the NREM and REM parasomnia. Of note, despite improvement in NREM parasomnia, sleepiness remained unchanged or even deteriorated and he continued to take frequent naps during the day; Epworth sleepiness scale score was 18 on admission and 15 in the past. There was a history of obstructive sleep apnoea, successfully treated with CPAP. A two-night PSG after DBS implantation showed a short total sleep time (four hours, 57 minutes the first and six hours. 40 minutes the second night) and poor sleep efficiency (47% first and 67% second night; normal ≥90%). Sleep architecture was disrupted with frequent awakenings. The patient wore his CPAP mask on both nights; SpO2 was measured the second night and showed a baseline saturation level of 96% with no significant desaturations. Interestingly, during both nights the PSG revealed a prolonged awake period for about 1.5 hours in early morning (each night around 2–3 a.m.; Figure 1(k) and (l)). In this patient a one-night PSG was performed four years prior to DBS implantation our sleep centre because of NREM parasomnia. This showed a normal total sleep time (eight hours, one minute) with poor sleep efficiency (74%; normal ≥90%; Figure 1(j)). Both PSGs (2008 and 2012) showed no overt features of non-REM parasomnia despite the strong history of NREM parasomnias, which is not unusual.
Discussion
The PH, the brain area stimulated in the two patients reported here, has long been recognised as a pivotal “wake” centre (2). Yet, sleep disturbances have not been reported after DBS of the PH. Here, we present two patients with pre-existing sleep problems in whom PH DBS led to a dramatic alteration of their sleep behaviour. This coincided with an improvement of the TACs (approximately one to two weeks after DBS implantation) and therefore suggests a DBS-induced change in sleep patterns. In our patients the DBS has not been switched off long enough to evaluate whether the effect of DBS on sleep and headaches is reversible. However, the change in sleep pattern coinciding with the improvement of headaches would argue for a PH-DBS-induced effect. Hypothalamic DBS is a promising treatment for severe refractory TACs with a 50%–75% responder rate in cluster headache patients (9,10); in the two patients reported here, cluster headaches and SUNCT headaches improved significantly after DBS. The target area for DBS for TACs is the midbrain tegmentum, an area located between the PH and the red nucleus (11,12) (see Figure 1(a–c) and (g–i)).
PH DBS involves a complex pattern of activation and deactivation of numerous brain areas as has been shown in a study using H2(15O)-positron-emission tomography, and activation has been reported at the stimulation site, i.e. in the ipsilateral hypothalamic area (13). The PH receives projections from the ventrolateral preoptic nucleus and when activated exerts strong wake-promoting effects through histamine neurons in the tuberomammillary nucleus of the PH (2,14,15). Recent evidence suggests that activation of this histaminergic PH pathway may be used as a strategy to treat narcolepsy as orexin receptor 2 expression in the PH reduced sleepiness in narcoleptic mice and led to prolonged periods of wakefulness (16). In keeping with that evidence, modulation of the PH region by DBS in our patients led to reduced sleep efficiency and prolonged period of wakefulness as demonstrated by the hypnograms (Figure 1(e), (f), (k) and (l)). Intriguingly, this prolonged period of wakefulness was reproduced the second night. In one patient the effect was perceived as a deterioration of his sleep condition with more frequent arousals. The aetiology of the condition remained undetermined by the PSG but by history was suggestive of REM parasomnia. In the second patient, a pre-existent NREM parasomnia appeared to improve after DBS. This may be related to the smaller amount of sleep and NREM sleep after DBS. This patient had pre-existent severe obstructive sleep apnoea, which was successfully treated with CPAP as was confirmed in the PSG indicating that frequent arousals are not due to oxygen desaturations. Both patients had poor sleep efficiency and a prolonged period of wakefulness suggesting that DBS of the hypothalamus had a wake-promoting effect in both patients. On the other hand, a case series of three patients reported by Vetrugno and colleagues showed that PSG recordings improved sleep efficiency that was related to an improvement of headache attacks (8). In contrast to our patients, they did not have a pre-existent sleep disorder and headache attacks occurred predominantly during sleep, making it difficult to disentangle a direct effect of hypothalamic stimulation on sleep from an indirect effect due to an effect on nocturnal headaches. In one patient a PSG prior to PH-DBS was not available and therefore we cannot exclude that ageing contributes to sleep deterioration although the temporal relation to DBS would be supportive of a mainly PH-DBS-induced effect.
Our findings provide insight into the role of the PH in sleep and wakefulness. These findings provide in vivo evidence and suggest that the PH may be a potential target area for DBS in disorders with overt sleepiness such as narcolepsy or idiopathic hypersomnia.
Clinical implications
Posterior hypothalamic deep brain stimulation (PH-DBS), used as a treatment for pharmacoresistant trigeminal autonomic cephalalgias (TACs), can result in changes of sleep-wake patterns. PH-DBS has a wake-promoting effect and can induce both deterioration and improvement of pre-existent sleep disorders in patients treated for pharmacoresistant TACs.
Footnotes
Funding
This work was undertaken at University of College London (UCL) and UCL Hospitals and was partly funded by the Department of Health National Institute for Health Research Biomedical Research Centres funding scheme. The Unit of Functional Neurosurgery, UCL Institute of Neurology, Queen Square, London, is supported by the Sainsbury Monument Trust and Parkinson’s Appeal for Deep Brain Stimulation.
Conflicts of interest
LZ occasionally receives honoraria for invited educational talks from Medtronic. MSM serves on the advisory board for St Jude Medical, and has received payment for the development of educational presentations from St Jude Medical and Medtronic.
