Abstract
Background
Acute therapies targeting calcitonin gene-related peptide (CGRP) for episodic migraine (EM) demonstrate efficacy in women, but evidence of efficacy in men remains to be established. By contrast, CGRP targeting therapies for migraine prevention are effective in both men and women with frequent EM or chronic migraine (CM). Preclinical studies have shown that supradural application of CGRP preferentially produces migraine-like pain behaviors in female rodents. We hypothesized that, in male mice, increased frequency of migraine-like pain may sensitize to nociceptive effects of CGRP and this might be associated with altered expression of CGRP in trigeminal ganglion (TG) neurons and/or in their dural projections.
Methods
CM was modeled in male and female mice by repeated administration of nitroglycerin (NTG). Medication overuse headache (MOH), a form of CM, was modeled by repeated daily administration of sumatriptan. Following resolution of transient cutaneous allodynia (CA) elicited by NTG or sumatriptan, mice received a sex specific subthreshold dose of supradural CGRP that does not elicit CA in naïve male or female mice, and CA was evaluated. CGRP-positive cell bodies in the ophthalmic V1 region of the trigeminal ganglion (TGV1) and CGRP-positive nerve fibers innervating the dura mater were assessed.
Results
Supradural administration of 1 pg of CGRP produced migraine-like pain behaviors in female, but not male, mice; a ten-fold lower dose was established as subthreshold in naïve female mice. Repeated NTG or sumatriptan produced transient CA in both female and male mice that resolved within 8–11 days after treatment cessation. Following resolution of CA, previously subthreshold doses of CGRP elicited CA in CM and MOH models in mice of both sexes, with no effects observed in vehicle treated controls. A higher number of CGRP-positive neurons in the TGV1 was found in naïve female compared to male mice. The number of CGRP-positive TGV1 neurons was increased in both sexes following repeated NTG. Similar nerve fiber density was observed in the dura mater of male and female mice and no differences were detected following repeated NTG.
Conclusions
As previously reported, CGRP produced female-selective migraine-like pain behaviors in naïve mice. Consistent with behavioral effects, female mice demonstrated a higher number of CGRP-positive cells in the TGV1. These findings appear relevant to clinical observations of female efficacy of CGRP-receptor antagonists for acute treatment in EM patients. In models of CM or MOH that are characterized by increased frequency of migraine-like pain, previously subthreshold doses of supradural CGRP now elicited migraine-like nociceptive behaviors in mice of both sexes. The increased pain responses were accompanied by increased number CGRP positive TGV1 cells in the NTG model in both female and male mice. These data suggest that increased frequency of migraine promotes physiological changes including increased expression of TGV1 CGRP along with sensitization to CGRP-induced nociception in both males and females. Thus, as EM transforms to CM, CGRP-dependent mechanisms may become increasingly important, consistent with observations of efficacy of CGRP targeting therapies for migraine prevention in both men and women. Our results also suggest the possibility of enhanced efficacy of CGRP-receptor antagonists for the acute treatment of migraine in men as migraine frequency increases.
This is a visual representation of the abstract.
Keywords
Introduction
Migraine is a highly prevalent, sexually dimorphic and progressive neurological disorder characterized by its multiphasic and multisymptomatic nature.1–4 It is the leading cause of disability worldwide among women5,6 and involves complex interactions between peripheral and central neuronal mechanisms, as well as non-neuronal factors.1–4 According to the International Classification of Headache Disorders, 3rd edition (ICHD-3), migraine encompasses several subtypes, including episodic migraine (EM) with aura, migraine without aura and chronic migraine (CM). 7 The overuse of some anti-migraine medications, including triptans, is known to produce a state of CM referred to as medication overuse headache (MOH). 8 Migraine has a complex and not fully understood pathophysiology,1–4 including uncertainty of mechanisms that lead to migraine chronification.
Calcitonin gene-related peptide (CGRP) has been recognized as a key factor in migraine pathophysiology.1–4,9 The clinical effectiveness of CGRP-targeting therapies, which include both small molecule antagonists and biologics that interfere with CGRP signaling,3,10–12 provides compelling evidence of a causal contribution of CGRP to migraine. Small molecule CGRP receptor antagonists (i.e. gepants) are used for acute treatment of migraine, as well as for migraine prevention.3,10–12 Monoclonal antibodies targeting the CGRP receptor or CGRP peptide are also used for prevention in patients with frequent EM and CM.3,10–12 Women exhibit greater migraine severity, persistence, co-morbidity profiles and a higher prevalence of the condition relative to men.13,14 Likewise, this female prevalence is evident in clinical settings and trials, where most participants are women. We recently conducted a subpopulation analysis of publicly available data from the US Food and Drug Administration (FDA) reviews of approved gepant drugs used in acute migraine therapy, as well as prevention outcomes for CGRP-targeting therapies including monoclonal antibodies (mAbs) of patients with EM and CM to assess potential sex differences in response rates. 15 The study concluded that anti-CGRP therapy is effective for acute migraine treatment in women, but the available data did not provide evidence of efficacy in men. On the other hand, CGRP mAbs therapies were effective for migraine prevention in men and women with frequent EM and in men and women with CM. 15 These analyses reveal potential sexual differences in the effects of CGRP-targeted therapies and underscore the need for a deeper understanding of CGRP neurobiology in both men and women,15–17 especially as frequency of migraine attack increases.
Preclinical studies have demonstrated that supradural administration of CGRP preferentially produces migraine-like pain in female rodents suggesting sexually dimorphic mechanisms promoting migraine. 18 This observation, coupled with the possible preferential effectiveness of CGRP therapies for acute migraine treatment in women, prompted the question of whether males might develop increased sensitivity to CGRP with increased frequency of migraine attacks. Additionally, it seemed possible that an increased frequency of migraine attacks might further promote increased sensitivity to CGRP in females. We therefore investigated the possibility of CGRP sexual dimorphism using animal models of CM and MOH.
Methods
Animals
All experimental procedures were approved by the Institutional Animal Care and Use Committee of the University of Arizona and were performed in accordance with the ARRIVE guidelines, the ethical guidelines of the International Association for the Study of Pain regulations on animal welfare and the National Institutes of Health guidelines for the care and use of laboratory animals. In total, 104 male and female 8–12-week-old C57BL/6J mice (Jackson Laboratories, Bar Harbor, ME, USA) were used in these studies. Eight and four mice per group were used for behavior and immunohistochemistry experiments, respectively. The estrous cycle of females was not monitored. Animals were housed four per cage under standard animal husbandry conditions of a temperature-, humidity- and light cycle-controlled environment, with free access to food and water in the University of Arizona animal facility. Group size requirements to obtain significance at the α = 0.05 and a statistical power of 0.9 were determined from previous experiments using power analysis. Mice were randomly divided into control and experimental groups. Experimenters were blinded to treatments.
Drugs
Nitroglycerin (NTG) was purchased from American Regent (Shirley, NY, USA), diluted in saline to 1 mg/mL and administered intraperitoneally (i.p.) at 10 mg/kg. Sumatriptan was obtained from Abmole Bioscience (Houston, TX, USA), diluted in saline and administered i.p. at 10 mg/kg. Controls received saline at 10 ml/kg. CGRP (Bachem, Torrance, CA, USA) was dissolved in phosphate-buffered saline (PBS) and injected onto the dura mater (supradural) at 0.1 or 1 pg/5 µl.
Cutaneous allodynia (CA) evaluation
Mice were placed in elevated individual Plexiglass chambers with mesh flooring and allowed to habituate for three days for two hours each day before evaluations. Following two hours of habituation, periorbital (cephalic) and hindpaw (extracephalic) tactile frequency of response was measured by 10 applications of the 0.4 g (3.61 N) von Frey filament to the periorbital region and 1 g (4.08 N) to the hindpaw region (Stoelting, Wood Dale, IL, USA) with sufficient pressure to cause the filament to display a slight arch. Swiping of the face (facial grooming) was considered to be a positive periorbital response. Sharp withdrawal, shaking and/or licking the paw were considered positive hindpaw responses. An increased frequency of response was considered as a translationally-relevant measure of CA. Frequency of response was calculated as ((number of positive responses/10) × 100).19,20
Models of CM
Chronic NTG model
To model CM-like pain, mice were administered NTG (10 mg/kg, i.p.) every other day for nine days (five treatments in total), with treatments performed at the same time each day (11:00 am), immediately after the behavior evaluation.21,22
MOH model
To mimic MOH, mice were administered sumatriptan (10 mg/kg, i.p.) once daily for nine consecutive days, with treatments performed at the same time each day (11:00 am), immediately after the behavior evaluation. 23
Supradural injection
Injectors were modified from commercially available cannulas (#C313I/SPC; Plastics One, Roanoke, VA, USA) by inserting a stopper to establish a length of 0.65–0.7 mm to maintain dura mater integrity. The injectors were connected to Tygon tubing (Cole Parmer Co., Vernon Hills, IL, USA), which was then attached to a 25-μl Hamilton syringe (Hamilton, Reno, Nevada, USA). Under brief isoflurane anesthesia, the injector was inserted through the sagittal and lambdoid suture junction for supradural delivery. 20
Immunohistochemistry
Mice were perfused with 4% paraformaldehyde, and then mouse dura mater-attached skulls and TGs were harvested, and post-fixed in paraformaldehyde for 4 h.
TGs
TGs were transferred to sucrose (15% then 30%) PBS solution at 4°C and then cut on a cryostat. TG sections (10 µm) with V1 area cell bodies were selected for staining, rehydrated in PBS. Selected TG slides underwent antigen retrieval (1X; cat #C9999; Sigma-Aldrich, St Louis, MO, USA) at 90°C for 10 min and then were blocked and permeabilized to suppress non-specific antibody binding for 1 h (0.1% BSA and 0.3% Triton X-100 in PBS). Sections were incubated with anti-CGRP (dilution 1:200; ab36001; Abcam, Cambridge, UK) overnight at 4°C. After washing, secondary antibodies donkey anti-goat AF488 (dilution 1:600; #705-546-147; Jackson ImmunoResearch, West Grove, PA, USA) and conjugated anti-neuronal nuclei (NeuN) AF555 (dilution 1:100; MAB377A5; Millipore Sigma, Burlington, MA, USA) were incubated for two hours. Slides were subsequently counterstained in 4′,6-diamidino-2-phenylindole (DAPI) and mounted with Prolong Glass (cat #P36980; Invitrogen, Waltham, MA, USA).
Dura mater
Following postfixation, dura mater was removed from the skull and stored at −20°C until staining. Free-floating samples of dura mater underwent antigen retrieval (1X; cat #C9999, Sigma-Aldrich) at 90°C for 10 min and then were blocked and permeabilized for 1 h (0.1% BSA and 0.3% Triton X-100 in PBS). Sections were incubated with primary antibodies, goat anti-CGRP (dilution 1:200; ab36001; Abcam), and mouse anti-b-tubulin III (Tubb3) (dilution 1:200; T8578; Millipore Sigma) overnight at 4°C. After washing, secondary antibodies donkey anti-goat AF488 (dilution 1:600; #705-546-147, Jackson ImmunoResearch) and donkey anti-mouse Cy3 (dilution 1:600; #705-546-147; Jackson ImmunoResearch) were incubated for 1 h. Slides were subsequently counterstained in DAPI and mounted with Prolong Gold Antifade media (cat #P36934; Invitrogen).
Confocal microscopy and analysis
The study specifically focused on the ophthalmic branch (V1) of the TG because of its critical role in innervating the cranial dura mater. 24 The ophthalmic V1 region of the trigeminal ganglion (TGV1) was identified from a longitudinal section using a Revolve microscope (Echo, San Diego, CA, USA). Confocal z-stacks from TGV1 and dura mater were acquired using a Fluoview FV1200 microscope (Olympus, Tokyo, Japan), with images captured using a 20×/0.8 objective. Consistent equipment and software settings were maintained throughout the duration of the experiments. The total number of neurons (NeuN+) and CGRP+ neurons were counted with IMARIS surface detection software (Oxford Instruments, Abingdon, UK): an automatic threshold based on IMARIS statistics was applied to 2D mean projections and then manually refined. Data were collected from n = 4 mice per experimental group with two to four replicates per mouse. Measurement of the innervation density of mouse dura mater was performed using Fiji (https://fiji.sc) with Otsu thresholding to determine area per mm2. CGRP+ and b-tubulin III (Tubb3)+ fibers were measured from a vascular and avascular location from each mouse (n = 4 mice per experimental group). Low magnification overviews were acquired using a Revolve microscope with a 4×/0.13 objective and stitched together using Fiji. Maximum intensity projections in figures were subjected to background subtraction (mean intensity of all image pixels).
Experimental design
CGRP was administered at 1 pg per 5 µl onto the dura mater of naïve female and male mice immediately after tactile frequency of response baseline. CA evaluation was performed at 10, 20, 40, 60, 120 and 180 min after supradural injections. TG and dura mater were collected in a different cohort of naïve female and male mice one week following the animal arrival at the University of Arizona animal care facility to assess the number of CGRP-positive cell bodies and CGRP-positive fibers using immunohistochemistry.
The effect of supradural administration of subthreshold CGRP on inducing CA was assessed using the dose of 0.1 pg per 5 µl for females and 1 pg per 5 µl for males. Subthreshold doses of CGRP were previously reported for both female and male mice.18,21 Tactile frequency of response was collected before (baseline) and at 10, 20, 40, 60, 120 and 180 min after supradural CGRP injection. On the following day, post-CGRP time course, tactile frequency of response was collected (baseline) and CM-like behavior was induced by repeated administration of either NTG or sumatriptan. Control mice received saline. CA was evaluated on indicated days post-NTG or post-sumatriptan onset. On days 18 post-NTG, or 21 post-sumatriptan, tactile frequency of response was collected as a baseline, and animals received the same previous supradural subthreshold dose of CGRP (i.e. 1 pg for males and 0.1 pg for females), followed by CA evaluation at 10, 20, 40, 60, 120 and 180 min after CGRP injection. TG and dura mater were collected the following day to assess the number of CGRP-positive cell bodies using immunohistochemistry. The timeline of each experimental procedure is shown in Figure 1.

Timeline of the experimental procedures. CGRP = calcitonin gene-related peptide; NTG = nitroglycerin; TGV1 = ophthalmic V1 region of the trigeminal ganglion. Created with Biorender.
Statistical analysis
Sample size was determined using the G*Power, version 3.1(Düsseldorf, Germany). p < 0.05 was considered statistically significant. Data are expressed as the mean ± SEM. Statistical analyses were performed using Prism, version 10 (GraphPad Software Inc., San Diego, CA, USA). Time course experiments for sensory thresholds were analyzed using two-way analysis of variance with repeated measurements followed by Tukeys's multiple comparisons test. The Shapiro–Wilk normality test was performed prior to the further statistical analysis in the immunohistochemistry data. All immunohistochemistry data passed the normality test. Subsequently, an unpaired two-tailed Student's t-test was applied. p < 0.05 was considered statistically significant. The statistical analysis, numbers of animals used (n), p values and F ratios are reported in the supplementary material (Table S1).
Results
Sexually dimorphic effect of supradural CGRP in eliciting migraine-like pain behavior
Administration of CGRP at 1 pg per 5 µl onto the dura mater induced periorbital (Figure 2) and hindpaw (see supplementary material, Figure S1) CA in female (Figure 2a; see also supplementary material, Figure S1a) but not male mice (Figure 2b; see also supplementary material, Figure S1b), as revealed by increased frequency of response to tactile stimulation after application of CGRP compared to vehicle.

Female, but not male, naïve mice display migraine-like pain behavior in response to 1 pg of supradural calcitonin gene-related peptide (CGRP) and exhibit higher number of CGRP-positive neurons in the ophthalmic V1 region of the trigeminal ganglion (TGV1) and neuronal fibers in the dura mater. Periorbital tactile frequency responses were collected at baseline (BL) and 10, 20, 40, 60, 120 and 180 min after supradural injection of CGRP at a dose of 1 pg in (a) female and (b) male naïve mice. Representative photomicrographs of longitudinal sections of (c) female and (d) male naïve mouse TGV1 immunostained for anti-CGRP (green), neuronal nuclei (NeuN) (magenta) and 4′,6-diamidino-2-phenylindole (DAPI) (blue). Scale bar = 50 µm. Representative confocal photomicrographs of sections of naïve (e) female and (f) male mouse dura mater immunostained with antibodies against CGRP (green), tubulin III (Tubb3) (magenta) and DAPI (blue). Scale bar = 1 mm. Quantification of CGRP positive neurons in (g) the TGV1 and (h) neuronal fibers in the dura mater of female and male naïve mice. Data are presented as the mean ± SEM and were analyzed using (a, b) two-way analysis of variance followed by Tukey's multiple comparison test for (n = 8 per group) or (g, h) an unpaired two-tailed Student's t-test (n = 4 per group with two to four replicates per mouse). *p < 0.05 for comparisons between groups. #p < 0.05 for comparisons to baseline within the CGRP-treated group. Data values for individual mice are shown as small symbols in (g) and (h). Details of the statistical analysis can be found in the supplementary material (Table S1).
Increased number of CGRP-positive TGV1 neurons in naïve female compared to male mice but similar CGRP-positive fibers in the dura mater
Longitudinal sections of mouse TGV1 immunostained with anti-CGRP (green) and pan-neuronal anti-NeuN (magenta) and counterstained with DAPI (blue) showed an increase number of CGRP immunoreactive neurons in female (Figure 2c) compared to male (Figure 2d) mice. We observed 42.4 and 28.8% CGRP-positive cells in female and male TGV1, respectively (Figure 2g).
Free floating dura mater was immunostained with anti-CGRP (green) and pan-neuronal βIII-tubulin (Tubb3) (magenta). The percentage of CGRP-positive fibers normalized to all fibers (Tubb3-positive) was not different between female (Figure 2e) and male (Figure 2f) mice, suggesting similar CRGP positive vascular and avascular innervations in both sexes (Figure 2h).
Increased CGRP sensitivity following induction of cm with repeated administration of NTG
The administration of a subthreshold dose of CGRP (i.e. 0.1 pg) onto the dura mater of female mice did not alter tactile sensory responses in the periorbital (Figure 3a) and hindpaw (see supplementary material, Figure S2a) regions. Similarly, supradural injection of subthreshold CGRP at a dose ten times higher in male mice (1 pg) also did not alter tactile sensory responses in the periorbital (Figure 3b; see also supplementary material, Figure S2b) regions. In the same mice, repeated administration of NTG elicited robust CA in the periorbital and hindpaw regions of female (Figure 3c; see also supplementary material, Figure S2c) and male (Figure 3d; see also supplementary material, Figure S2d) mice. The onset of CA began on day 5 after the first NTG treatment, with tactile thresholds returning to baseline levels by day 17 (i.e. eight days after NTG termination) (Figure 3c,d; see also supplementary material, Figure S2c,d). On day 18, the administration of respective subthreshold doses of CGRP onto the dura mater induced CA in the periorbital and hindpaw regions of female (Figure 3e; see also supplementary material, Figure S2e) and male mice (Figure 3f; see also supplementary material, Figure S2f) previously treated with repeated NTG. No CA was observed in animals that received repeated administration of vehicle.
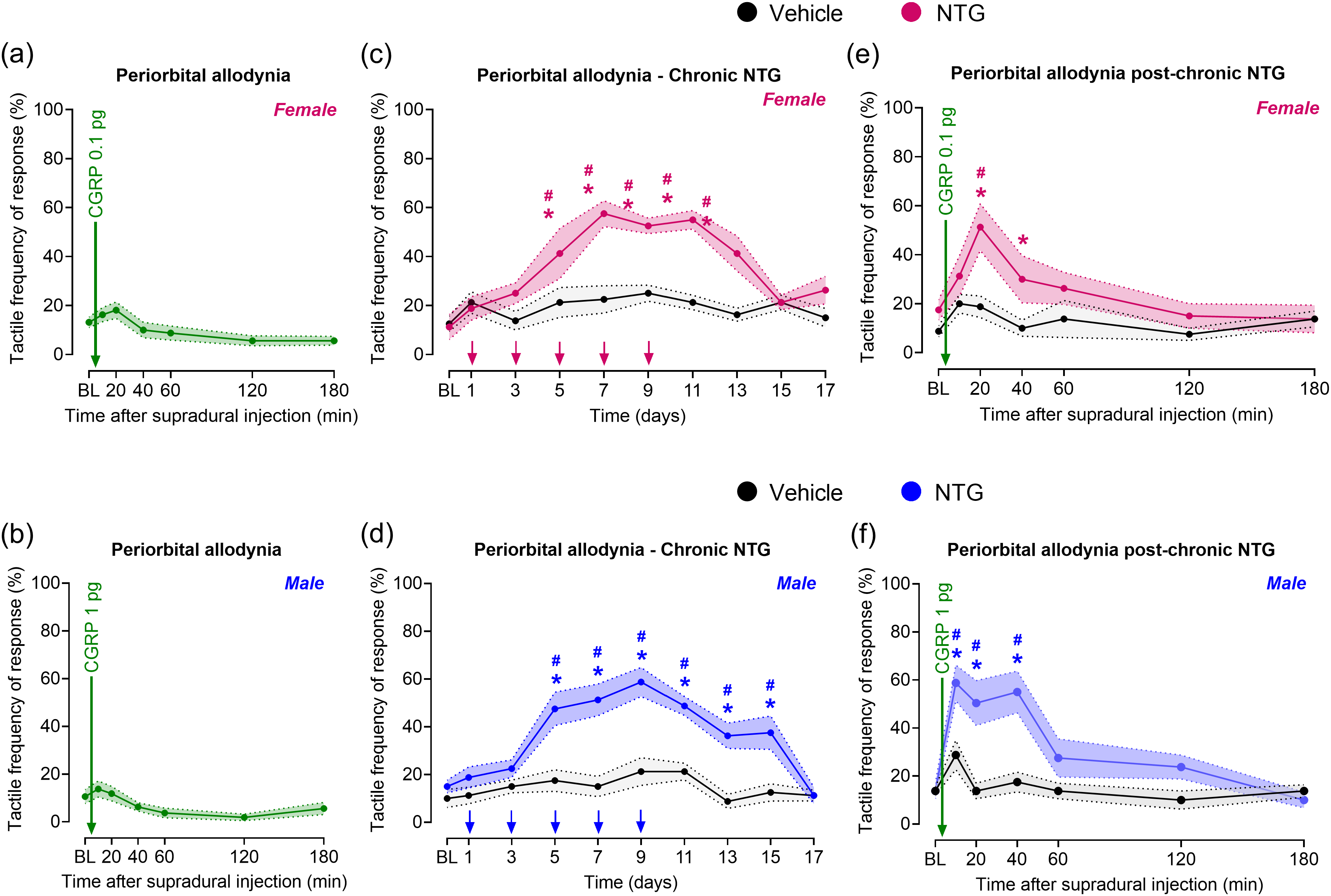
Repeated nitroglycerin (NTG) increases calcitonin gene-related peptide (CGRP) sensitivity to promote chronic migraine (cm)-like pain. Periorbital tactile frequency responses were collected at baseline (BL) and at indicated times after supradural injection of CGRP at a subthreshold dose of (a) 0.1 pg for females and (b) 1 pg for males. On the following day, BL periorbital tactile frequency responses were collected, and chronic migraine-like pain was induced in (c) female and (d) male mice by repeated administration of NTG (10 mg/kg, intraperitoneally) every other day for 9 days (5 treatments in total), as indicated by arrows. Control mice received saline. Periorbital tactile frequency responses were measured on days 1, 3, 5, 7, 9, 11, 13, 15 and 17 post-NTG onset. On treatment days, cutaneous allodynia was evaluated before NTG administration. On day 18 post-NTG onset (e) female and (f) male mice received a supradural injection of the respective subthreshold dose of CGRP, followed by periorbital tactile frequency response measurements. Data are presented as the mean ± SEM and were analyzed using two-way repeated measures analysis of variance followed by Tukey's multiple comparison test (n = 8 per group). *p < 0.05 for comparisons between groups. #p < 0.05 for comparisons to BL within the NTG-treated group. Details of the statistical analysis can be found in the supplementary material (Table S1).
Increased number of CGRP-positive TG cells in both female and male mice following repeated NTG
Immunostained TG tissues illustrate the neuronal immunoreactivity of CGRP in the TGV1 region of both female (Figure 4a) and male (Figure 4c) mice. The percentage of CGRP-positive neurons normalized to vehicle-treated groups confirms the increased number of CGRP-positive cell bodies in the TGV1 of female and male mice treated with repeated NTG compared to animals that received repeated vehicle control. In females, we observed an increase of 34% of CGRP-positive neurons in NTG-treated mice (Figure 4b); in males, the increase was 33% (Figure 4d).
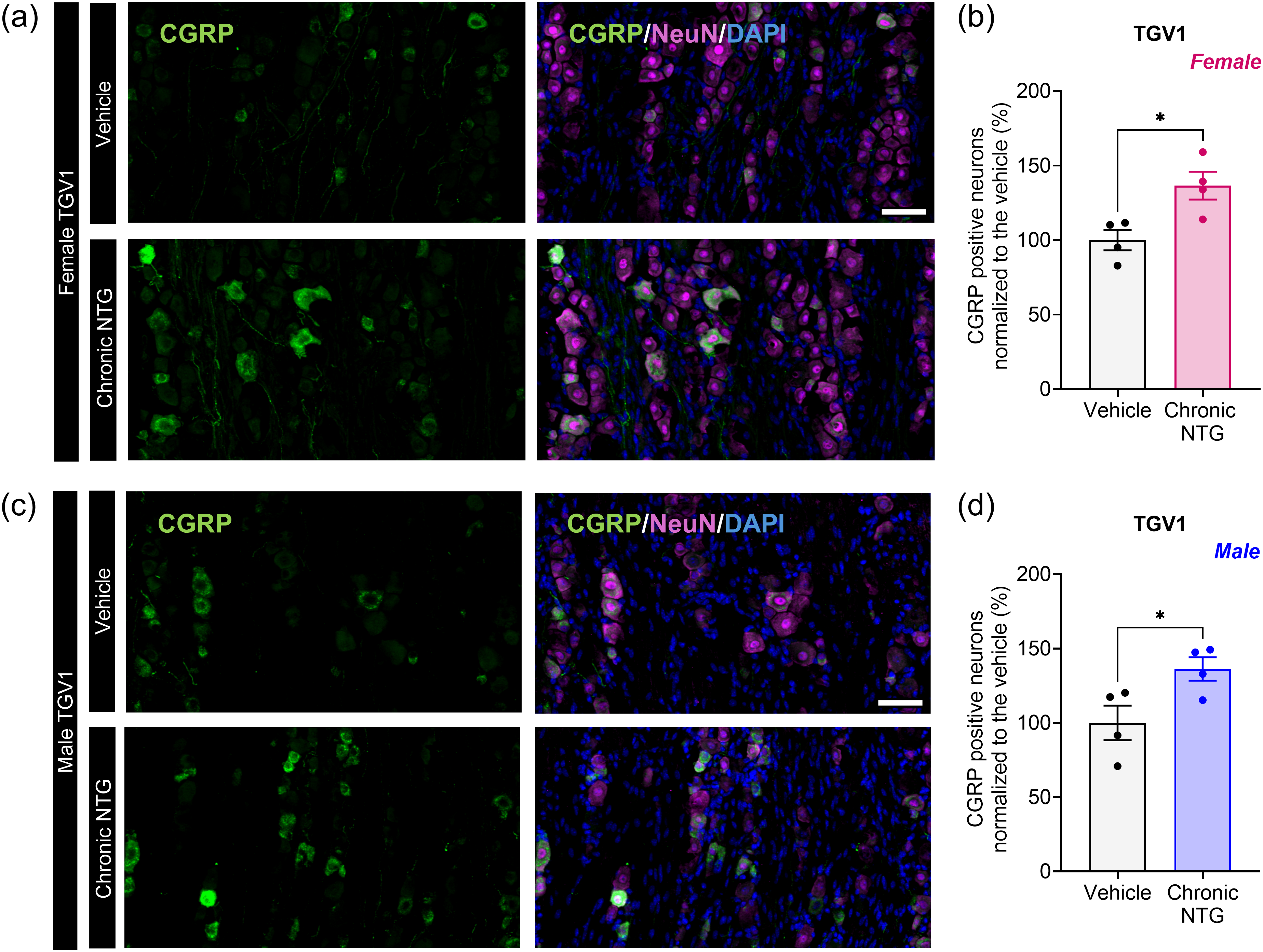
Repeated nitroglycerin (NTG) increased the number of calcitonin gene-related peptide (CGRP)-positive cell bodies in the ophthalmic V1 region of the trigeminal ganglion (TGV1) of female and male mice. Representative photomicrographs of longitudinal sections of repeated NTG- or vehicle-treated (a) female and (c) male mouse TGV1 immunostained for anti-CGRP (green), neuronal nuclei (NeuN) (magenta) and 4′,6-diamidino-2-phenylindole (DAPI) (blue). Scale bar = 50 µm. Quantification of CGRP positive neurons in the TGV1 of (b) female and (d) male mice. Data are presented as the mean ± SEM and were analyzed using (b, d) an unpaired two-tailed Student's t-test (n = 4 per group with two to four replicates per mouse)/ *p < 0.05 for comparisons between groups. Data values for individual mice are shown as small symbols in (b) and (d). Details of the statistical analysis can be found in the supplementary material (Table S1).
Next, we examined whole mount dura mater stained with CGRP and pan-neuronal Tubb3 of female and male mice treated with repeated NTG compared to animals that received repeated vehicle control. Repeated NTG did not increase the percent of CGRP positive fibers in the dura mater of both female (Figure 5a,b) and male (Figure 5c,d) mice.

Repeated nitroglycerin (NTG) did not modify the proportion of calcitonin gene-related peptide (CGRP)-positive neuronal fibers in the dura mater of female and male mice. Representative confocal photomicrographs of dura mater sections of (a) female and (c) male mouse treated with repeated NTG- or vehicle immunostained with antibodies against CGRP (green), tubulin III (Tubb3) (magenta) and 4′,6-diamidino-2-phenylindole (DAPI) (blue). Scale bar = 1 mm. Quantification of CGRP positive fibers as percent of Tubb3 fibers in the dura mater of repeated NTG- or vehicle-treated (b) female and (d) male mice. Data are presented as the mean ± SEM and were analyzed using (b, d) an unpaired two-tailed Student's t-test (n = 4 per group with two to four replicates per mouse). Data values for individual mice are shown as small symbols in (b) and (d). Details of the statistical analysis can be found in the supplementary material (Table S1).
Enhanced CGRP sensitivity following repeated sumatriptan administration to induce MOH in mice
We confirmed that administration of sex-dependent subthreshold doses of CGRP onto the dura mater did not alter tactile sensory responses in the periorbital and hindpaw regions of both female (Figure 6a; see also supplementary material, Figure S3a) and male mice (Figure 6b; see also supplementary material, Figure S3b) and then induced the MOH model in these mice . Repeated administration of sumatriptan elicited persistent CA in the periorbital and hindpaw regions of female (Figure 6c; see also supplementary material, Figure S3c) and male mice (Figure 6d; see also supplementary material, Figure S3d. The onset of CA began on day 5 after the first sumatriptan treatment, with tactile thresholds returning to baseline levels by day 20 (Figure 6c,d; see also supplementary material, Figure S3c,d). On day 21, the administration of subthreshold doses of CGRP onto the dura mater elicited CA in the periorbital and hindpaw regions of sumatriptan treated female (Figure 6e; see also supplementary material, Figure S3e) and male mice (Figure 3f; see also supplementary material, Figure S3f) but not in animals that received vehicle.

Repeated sumatriptan-induced medication overuse headache (MOH) enhances calcitonin gene-related peptide (CGRP) sensitivity to promote migraine-like pain behavior. Periorbital tactile frequency responses were collected at baseline (BL) and after supradural injection of CGRP at a subthreshold dose of (a) 0.1 pg for females and (b) 1 pg for males. On the following day, BL periorbital tactile frequency responses were collected, and chronic migraine-like pain was induced in (c) female and (d) male mice by repeated administration of sumatriptan (10 mg/kg, intraperitoneally) for 9 consecutive days, as indicated by the arrows. Control mice received saline. Periorbital tactile frequency responses were measured on indicated days post-sumatriptan onset. On treatment days, cutaneous allodynia was evaluated before sumatriptan administration. On day 21 post-sumatriptan onset, (e) female and (f) male mice received a supradural injection of the subthreshold dose of CGRP, followed by periorbital tactile frequency response measurements. Data are presented as the mean ± SEM and were analyzed using (c–f) two-way repeated measures analysis of variance followed by Tukey's multiple comparison test (n = 8 per group). *p < 0.05 for comparisons between groups. #p < 0.05 for comparisons to baseline within the sumatriptan-treated group. Details of the statistical analysis can be found in the supplementary material (Table S1).
Discussion
The present study addressed the possibility that CGRP may become increasingly important in promoting migraine as frequency of migraine attacks increases (i.e. as migraine becomes chronic). Our findings reveal that: (a) CGRP induces migraine-like pain preferentially in naïve female mice; (b) an increased number of CGRP-expressing neuronal cell bodies is observed in the TGV1 of naïve female mice when compared to males; (c) increased migraine-like frequency in models of CM and MOH sensitizes both female and male mice to previously subthreshold doses of supradural CGRP; and (d) the increased sensitivity to CGRP in the NTG model is accompanied by increased number of CGRP positive cell bodies in the TGV1 in both sexes. These data support the conclusion that increased frequency of migraine attacks is associated with increased sensitivity to CGRP and with higher TGV1 CGRP expression in both male and female animals and is consistent with clinical observations of efficacy of anti-CGRP therapies for prevention in patients of both sexes with increased migraine frequency. 15
Recent evidence from rodents, non-human primates and human post-mortem tissues has provided support for the conclusion of qualitative sex differences in pain mechanisms that may also be relevant to migraine.10,18,23,25–33 Women exhibit a higher prevalence and greater severity of migraine relative to men suggesting that underlying migraine pathophysiology may be sexually dimorphic.13,14,18 CGRP is a 37-amino acid peptide that can be expressed in both neuronal and non-neuronal cells9,34 and has been demonstrated to produce female-selective migraine-like nociception and pain behaviors.18,28 Comprehensive clinical and preclinical research has established a causal role of CGRP in the pathophysiology of migraine in some patients.35,36 Migraine attacks can be triggered by intravenous administration of CGRP in patients.9,35–37 Circulating levels of CGRP are elevated in body fluids such as the blood, saliva and cerebrospinal fluid of individuals with migraine during an attack.35,36,38,39 It should be noted that CGRP levels were measured predominantly in women with migraine (approximately a 4:1 ratio of females to males) and the outcomes of provocative studies of migraine with CGRP infusion have not been reported with analysis by patient sex. Several CGRP-targeted therapies have been approved by the FDA for the management of migraine. 15 Critically, in these studies, the effectiveness of CGRP therapies was assessed primarily in patient cohorts that were overwhelmingly female. In addition, female sex hormones, including their influence on CGRP levels, have long been recognized as key factors contributing to migraine across all stages of a woman's life. By not controlling for the estrous cycle, the study captures the natural variability observed in the human population, thereby enhancing its translatability.
Although preclinical studies have established that female rodents respond to lower doses of CGRP and have longer-lasting effects compared to males,18,40–43 higher doses of CGRP produce nociception in male rodents. In agreement with these studies, we demonstrated that supradural CGRP induced migraine-like pain behavior at lower doses in naïve female mice. This observation suggests that compared to males, naïve female animals are essentially “sensitized” to CGRP at baseline possibly as a consequence of increased trigeminal CGRP expression or signaling.18,40–43 CGRP is expressed in the TG, specifically in small to medium-sized neurons9,44–48 and reports from multiple laboratories suggest expression in approximately 30–50% of neuronal cell bodies.9,44,47 Whether sexual dimorphism exists in CGRP expression in rodents remains uncertain, however, because most previous preclinical studies regarding the expression of CGRP either failed to report the sex of the animals or performed the studies in only one sex. In the present study, naïve female mice exhibited a higher number of CGRP-positive neurons in the TG compared to males, possibly contributing to the increased sensitivity of female animals to CGRP-related nociceptive behaviors. Although we did not detect sex differences in CGRP-positive dural sensory innervation, the limitations of focusing analysis solely on area-based measurements should be noted and do not eliminate the possibility of differences within smaller microvascular structures in the dural stroma.
Although the efficacy of CGRP receptor antagonists for the treatment of acute migraine in men remains to be established, preventive therapies targeting CGRP or its receptor are clinically efficacious in both men and women with frequent EM and in men and women with CM.15–17 These observations raise the possibility that the role of CGRP in promoting migraine may change as the frequency of migraine increases from EM to frequent EM to CM patients. The definition of CM has evolved over the past two decades, gradually replacing earlier terms such as “chronic daily headache” and “transformed migraine”.49,50 CM is now diagnosed when patients experience headache on 15 or more days per month of which eight are migraine days over a period of three months. 7 More recent interpretations of CM may now include patients experiencing less than 15 headache days per month (e.g. eight headache days) likely more accurately reflecting changes in underlying biology.51,52 Migraine, however, is a neurological disorder that occurs for many decades over the lifespan of patients.2,3,53 The International Association for the Study of Pain (IASP) defined chronic pain as pain that persists or recurs for more than three months. 54 Patients with EM would therefore be classified as chronic pain patients by the IASP definition that focuses on duration of pain conditions. By contrast, the ICHD3 definition of migraine chronicity is based not on duration of the disease but on frequency of attacks. It is well established that increased frequency of migraine attacks is among the strongest predictors of migraine chronification. 55 Migraine has been described as a sensory threshold disorder 56 where each attack increases vulnerability to the next attack by lowering thresholds to activation of meningeal nociceptors to normally subthreshold stimuli. Each migraine attack may additionally promote and sustain a state of central sensitization that amplifies nociceptive inputs resulting in migraine from normally subthreshold stimuli. Peripheral and central mechanisms of nociceptor amplification may therefore both contribute to migraine chronification.
Frequent migraine-like pain can be modeled in preclinical studies by increasing the frequency of migraine-like pain attacks with provocative agents such as repeated administration of NTG representative of CM or by establishing a state resembling medication overuse headache, commonly seen in those with CM, with repeated doses of some medications used to treat migraine such as opioids or specific anti-migraine medications such as triptans.22,23,57–61 Increased CGRP levels in the blood have been reported after NTG infusion in patients, as well as by prolonged sumatriptan exposure.61,62 In the present study, we used the NTG model of CM and the sumatriptan-induced model of MOH in both female and male mice. We did not observe sex differences in the development or maintenance of migraine-like pain behavior in either model.22,23,61
The present study demonstrated that models of CM can elicit a state of latent sensitization, as evidenced by the priming effect of female and male mice to subthreshold CGRP applied onto the dura mater. Although the subthreshold dose of CGRP is 10 times lower in females than in males, following sensitization, the magnitude and duration of effects of subthreshold CGRP-induced CA is similar in both male and female animals. We also observed that the NTG model of CM was associated with an increased number of CGRP cell bodies in the TG of both sexes. This finding is consistent with our previous studies that demonstrated that repeated sumatriptan produced an increase in the number of TG neuronal cell bodies retrogradely labeled from the dura mater in male rats. 63 These findings highlight a significant role of CGRP signaling and expression in both sexes with increased frequency of migraine attack and are consistent with the clinical observation regarding efficacy of CGRP-targeted therapy in patients with frequent EM or CM. 15 The precise contribution of CGRP to migraine as frequency changes remains to be established. In this regard, it should be noted that the efficacy of preventive CGRP-targeting therapies in randomized controlled trials appears to be less in CM compared to patients with EM. 64 It seems likely that, as the frequency of migraine increases (i.e., becomes chronic), other CGRP-independent or parallel mechanisms such as loss of conditioned pain modulation 65 and contributions of other neurotransmitter or neuropeptide systems may contribute to the observed effects resulting in blunted effects of CGRP-targeting therapies in both women and men.
In summary, the present study suggests that female animals are more sensitive than males to CGRP signaling and additionally implies that women may be more sensitive to migraine-associated effects of CGRP than men. This conclusion would be consistent with the demonstrated clinical efficacy of CGRP-targeting therapies for acute migraine in women with EM. Whether CGRP-independent mechanisms may play a more prominent role in promoting migraine in males with EM requires further investigation. By contrast, sensitization to CGRP occurs as migraine become more frequent consistent with the equal effectiveness of these therapies in both sexes with frequent EM or CM. The increasing contribution of CGRP to migraine with increased frequency of attacks also suggests that men with frequent EM or CM might respond favorably to anti-CGRP therapies for acute management of migraine or for management of breakthrough migraine even when on preventive therapies.
Article Highlights
In line with previous reports, in naïve mice, supradural CGRP promotes migraine-like pain behavior preferentially in females. Naïve female mice show significantly increased number of CGRP-expressing neurons in the ophthalmic region of the trigeminal ganglion (TGV1) compared to males; no difference in CGRP dural innervation fiber density was observed. Induction of CM with repeated NTG or of MOH with sumatriptan increases the sensitivity to migraine-like pain elicited by previously subthreshold doses of supradural CGRP in both female and male mice. Following the induction of CM, mice of both sexes exhibit an increased number of CGRP-expressing neuronal cell bodies in the TGV1; sex differences in CGRP-positive dural sensory innervation were not detected. Mice of both sexes become sensitized to CGRP with increasing migraine frequency consistent with clinical observations of efficacy of CGRP-targeting therapies for migraine prevention in both men and women frequent EM or CM. Sensitization to CGRP-induced migraine-like nociception suggests the possibility of increased efficacy of CGRP targeting therapies in management of acute migraine in men with more frequent migraine.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251317446 - Supplemental material for Chronification of migraine sensitizes to CGRP in male and female mice
Supplemental material, sj-docx-1-cep-10.1177_03331024251317446 for Chronification of migraine sensitizes to CGRP in male and female mice by Gege Guzman, Caroline M. Kopruszinski, Kara R. Barber, Robson C. Lillo Vizin, David W. Dodick, Edita Navratilova and Frank Porreca in Cephalalgia
Footnotes
Acknowledgments
We kindly thank Dr Xu Yue and Veronica Hode for technical support.
Author contributions
CMK, EN and FP conceived and designed the study. GG, CMK, KRB and RCLV participated in data acquisition and analysis. CMK, GG, KRB, RCLV and FP drafted the manuscript. RCLV prepared the figures and figure legends. GG, CMK, KRB, RCLV, DWD, EN and FP read, revised and approved the final version of the manuscript submitted for publication.
Data availability
Data are available upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by a grant from the National Institutes of Health (NIH) to FP (R01NS129552).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
