Abstract
Objective
The aim of the current study was to identify typical alterations in resting state connectivity within different stages of the migraine cycle and to thus explore task-free mechanisms of headache attack generation in migraineurs.
Background
Recent evidence in migraine pathophysiology suggests that hours and even days before headache certain changes in brain activity take place, ultimately leading to an attack. Here, we investigate changes before headache onset using resting state functional magnetic resonance imaging (fMRI).
Methods
Nine episodic migraineurs underwent daily resting state functional magnetic resonance imaging for a minimum period of 30 consecutive days, leading to a cumulative number of 282 total days scanned. Thus, data from 15 spontaneous headache attacks were acquired. This allows analysing not only the ictal and the interictal phase of migraine but also the preictal phase. ROI-to-ROI (region of interest) and ROI-to-voxel connectivity was calculated over the migraine cycle.
Results
Within the ROI-to-ROI analysis, the right nucleus accumbens showed enhanced functional connectivity to the left amygdala, hippocampus and gyrus parahippocampalis in the preictal phase compared to the interictal phase. ROI-to-voxel connectivity of the right accumbens with the dorsal rostral pons was enhanced during the preictal phase compared to interictally. Regarding custom defined ROIs, the dorsal pons was ictally functionally more strongly coupled to the hypothalamic area than interictally.
Conclusions
This unique data set suggests that particularly connectivity changes in dopaminergic centres and between the dorsal pons and the hypothalamus are important within migraine attack generation and sustainment.
Introduction
Migraine is commonly seen as a cyclic disorder with certain bodily and sensory functional changes during different stages of the migraine cycle. Next to the common attack symptoms of heightened perception regarding light and noise, a high percentage of migraine patients experiences so-called premonitory symptoms including significant changes in appetite, fluid tolerance, sleep, fatigue and mood shortly before the beginning of migraine headaches (1–5). The premonitory phase of a migraine attack is then followed by the migraine headache (ictal phase) with its typical characteristics and accompanying symptoms as defined by the International Classification of Headache Disorders (6). Further in the cycle and after headache resolution, most patients experience a postdromal phase of various duration, characterized by alterations in mood, appetite and wakefulness (3,7) until bodily and sensory functions normalize and the patient enters the interictal phase. These clinical observations have led to the phase-model of migraine, as introduced and graphically characterized by Blau (7). Since then, studies differentiating between different migraine stages have become more frequent. Functional imaging studies have been able to show increased activity of certain brainstem areas during the ictal phase as compared to the interictal phase. The most prominent region with alternated function during the attack is the dorsal rostral pons, which also continued to be activated after pain release following sumatriptan administration (8–10). Preceding the attack, a number of regions have been reported to be hyperactive. Shortly before headache onset in nitroglycerin-triggered migraine attacks (11), the dorsal rostral pons already displays increased activation. Looking at the typical clinical presentation of the premonitory phase of migraine, the brain area commonly hypothesized to be involved is the hypothalamus (12), but the involvement of the dopaminergic system has also been reported (13,14). More precisely, the hypothalamus has been shown to be significantly more activated shortly before headache onset in nitroglycerin-triggered attacks (11) as well as in spontaneous attacks (15) when compared to the interictal stage. In fact, the hypothalamus shows specific changes in pain-dependent, that is, event-related functional connectivity during different stages of the migraine cycle (15). Changes in hypothalamic resting state functional connectivity have so far only been reported in a between-subject design comparing interictal migraineurs to healthy controls (16) and within a small within-subject study with eight preictal scans (17). A second region of interest during the emergence of a migraine attack are the spinal trigeminal nuclei, that show increased activity during the last 72 hours as well as 24 hours before headache onset as compared to the interictal state (10,18). For a complete picture, the only way to closely monitor functional connectivity changes during all phases of the migraine cycle is to scan patients consecutively over a longer time period. We investigated possible enhancement in functional connectivity across the whole brain during different stages of the migraine cycle in eight migraine patients who underwent daily resting state functional magnetic resonance imaging for 30 consecutive days.
Material and methods
Patients
Nine migraine patients underwent daily resting state fMRI for a minimum period of 30 days per patient, leading to a cumulative number of 282 total days scanned among these nine patients. For the current analysis, we only included patients with one or more clear-cut migraine attack preceded by consecutive pain-free days, which was the case in eight of these nine patients. In these eight patients, we analysed 15 headache attacks that were preceded by at least four pain-free days. This ensured three consecutive completely headache free scans (pre3, pre2, pre1) directly preceding headache onset (ict) and excluded possible postdromal effects of any previous attack. Thus the number of analysed attacks may differ from the number of observed attacks during the total scanning period. We purely chose a time criterion for phase definition, as premonitory symptoms, though assessed every day, are usually very inconsistent and variable over different patients. Thus a time criterion seemed much more feasible. Patients were asked to refrain from taking any acute migraine medication within the last 16 hours prior to each scan, but were allowed and encouraged to do so directly after the first ictal scan if necessary. In case of severe nausea, patients were offered dimenhydrinate as an antiemetic treatment prior to the scanning. This treatment was used in three attacks within one patient. As treatment effects could not therefore be excluded for the first pain-free scan after an attack, we did not specifically look at the postdromal phase. Patients were recruited via the Headache Outpatient Department of Hamburg, Germany, and via newspaper and online advertisement. Migraine diagnosis was confirmed by a trained headache expert. The study was approved by the local ethics committee of the chamber of physicians of Hamburg, Germany (number PV4522). Patients gave written informed consent to study participation.
fMRI data acquisition
Each daily session included acquisition of the resting-state functional connectivity and an anatomical T1-image on a 3 T MR Scanner (Trio, Siemens) with a 32-channel head coil. Resting state runs had a duration of 10 min (2000 ms TR, 30 ms TE, GRAPPA-accelerated) and comprised 300 volumes with 35 axial slices in parallel to the AC-PC line; covering the whole brain and brainstem (3 × 3 × 3mm voxel size, 1 mm gap, 74 × 74 matrix, 80° flip angle). Every day, subjects were reminded to keep their eyes closed and lie restfully but awake until the scanning session finished.
For analysis of rcfMRI, image pre-processing was performed using SPM12 (Wellcome Trust Center for Neuroimaging, London) and the conn toolbox (19,20). In more detail, the first five echo-planar images (EPIs) were discarded and motion displacement was estimated to obtain a mean image of all volumes. Segmentation of the T1 images produced individual masks of grey matter, white matter and cerebrospinal fluid (CSF). The T1-image was inclusively masked with grey and white matter masks (“skull strip”) and co-registered with the mean EPI image before all EPIs were realigned and resliced to the latter. Finally, all data were normalized to a DARTEL template in MNI space as provided by the CAT12 toolbox on the basis of the individual participants’ DARTEL flow fields and smoothed with a 6 mm full width at half maximum Gaussian isotropic kernel. For the next analysis steps, we additionally obtained warped but unsmoothed functional data as well as smoothed but warped functional data.
For the estimation of independent nuisance regressors, including motion, we performed independent pre-processing with FSL 5.0.4 (21) and ICA-AROMA (22). Here, the mean EPI image was co-registered with boundary-based registration (BBR) to the individual T1 image and warp fields from MNI standard space to individual T1 space were obtained by the application of linear and nonlinear registration with FLIRT and FNIRT, respectively. Warp fields, a brain mask, unwarped smoothed EPIs, and motion parameters from spm preprocessing were then processed using ICA-AROMA V.03beta (22) with automatic dimensionality estimation and partial component regression (nonaggressive), resulting in individual sets of nuisance regressors without changing the spm preprocessed data.
Finally, denoising and statistical analysis were conducted within the conn toolbox (V. 16a). We directly uploaded warped T1 images, grey matter, white matter, and CSF masks, as well as warped smoothed and unsmoothed (spm preprocessed) EPI data. Each day included in the analysed period (three pain-free preictal days, first ictal day) was defined as one session that was associated with one condition (pre3, pre2, pre1, ict), leaving all other conditions for that session empty. Of note, the same days were always assigned to the same condition, such that, for example, condition pre1 would label all days that directly preceded the day with the migraine attack. This sessions-per-subject approach ensured an adequate handling of statistical dependencies, as sessions within the same subject are statistically dependent, whereas statistical independence could be assumed for the between-subject data. After high-pass filtering (0.008), linear regression was set up linear to remove unwanted motion, physiological, and other artefactual effects from the BOLD signal before computing connectivity measures. For this purpose, we used theBOLD signal from the white matter and CSF masks (five PCA temporal components each), the six motion realignment parameters, automatically identified nuisance components from ICA-AROMA (23), and effects of conditions; that is, onset or ramping effects at the beginning of each session (including one derivative each).
ROI-to-ROI connectivity was estimated as standard bivariate correlation. Seeds of the analyses were 106 ROIs in both hemispheres, which included all Harvard-Oxford cortical and subcortical structures as provided by the conn toolbox, excluding cerebellar ROIs to reduce degrees of freedom. We additionally conducted exploratory analyses outside the whole brain analysis and defined three small ROIs within the brainstem and diencephalon corresponding to the dorsal rostral pons and the spinal trigeminal nuclei (both defined by a 2 mm sphere around the coordinates reported by Stankewitz et al. (10)), and the hypothalamus (defined by a 2 mm sphere around the coordinates reported by Schulte et al. (24) as seed regions of interest. Hence, 109 ROIs were used in total. For the main ROI with altered connectivity, we then performed ROI-to-voxel connectivity analysis to illustrate the connectivity results to a finer-grained extent. To contrast the different stages of the migraine cycle, the following contrasts were defined: pre3 < pre1, pre3 < ict.
The ROI to ROI analyses were FDR corrected for multiple comparisons. The applied FDR-correction adjusts the p-value for multiple comparisons (arising from multiple seed and target ROIs) as an adaptive threshold, using the Benjamini and Hochberg procedure (25).
For the ROI-to-voxel functional connectivity analyses, results were regarded significant at p < 0.05 FWE-corrected within regions of interest. For this we used two pre-defined regions of interest (each 8 mm spheres): Dorsal pons, as reported by Maniyar et al. (x = 6, y = −36, z = −27 (rounded to nearest voxel centre: x = 6, y = −36, z = −26) ) (11) and hypothalamus as reported by Moulton et al. (x = −6, y = −2, z = −8) (16).
Results
Patients
Of the nine patients originally scanned for this study, eight were included in the analysis, among these two males and six females (age 31.5 years ± 8.05 years). One patient had migraine with aura, whereas all the others had migraine without aura. Headache frequency ranged from two to six attacks on average among the eight patients analysed. None of the patients had side-locked attacks.
ROI-to-ROI connectivity
Exploring changes of whole networks across all possible ROI-to-ROI connections of Harvard-Oxford ROIs, we identified no change in number of nodes (i.e. number of suprathreshold connections) or overall connectivity strength (i.e. the sum of absolute T-values across ROIs connected above threshold) after correction for multiple comparisons. However, directly looking for the suggested pre-ictal hyperconnectivity (pre1–pre3), we were able to identify three pairs of ROIs showing significant increases in connectivity. Interestingly, all three pairs included the right nucleus accumbens; that is, during the last preictal scan (pre-1), functional connectivities of the right nucleus accumbens to the left amygdala, the left hippocampus and the left gyrus parahippocampalis were significantly higher than 3 days before headache onset (p < 0.05, FDR-corrected).
As the nucleus accumbens was included in all deviating ROI-to-ROI connectivities, we investigated its connectivity pattern with the more fine-grained ROI-to-voxel analysis with the nucleus accumbens serving as a seed region. This analysis additionally revealed increased functional connectivity to an area located medially within the dorsal rostral pons on pre-1 as compared to pre-3. Since pre-ictal activity has been described to endure during the attack, we also looked at increased ROI-to-voxel connectivity of the nucleus accumbens during the attack (ict > pre-3), which revealed a similar pattern (see Figure 1 for further detail). Of note, the seed region in this ROI-to-voxel analysis was selected on the same data basis and results may be distorted even though rigorous statistical testing was used during selection.
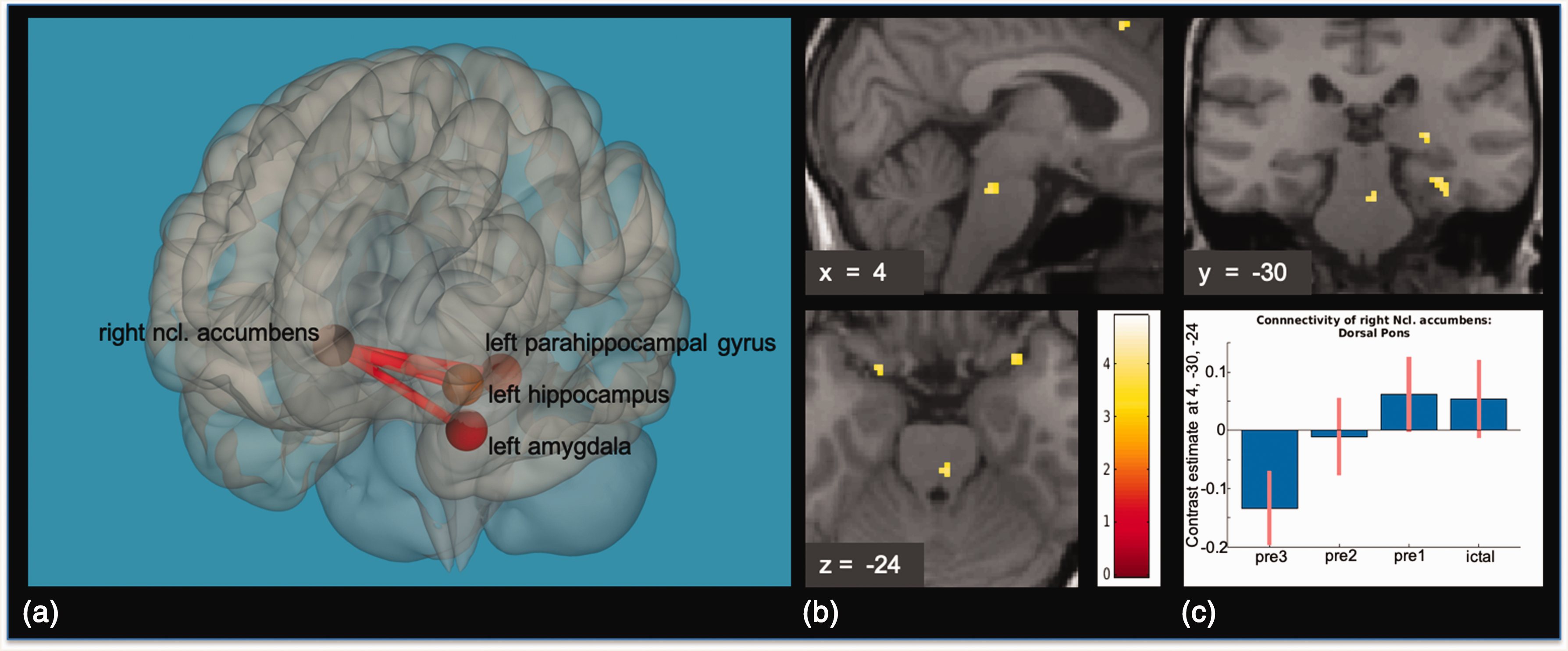
(a) Results of the ROI-to-ROI connectivity Analysis, pre3 < pre1: The right nucleus accumbens showed increased functional connectivity to the left amygdala, left hippocampus and left parahippocampal gyrus. Results are FDR-corrected, p < 0.05. (b) Results of the seed to voxel connectivity analysis with the right nucleus accumbens as a seed region, pre3 < pre1: On pre1, functional connectivity of the right nucleus accumbens to the dorsal rostral pons was higher as compared to on pre3. Results are small volume FWE-corrected, p < 0.05. Visualization threshold p < 0.001, k = 5, uncorrected. T-score-map overlaid on the normalized anatomical T1-image of one migraine patient. (c) Contrast estimates within the peak voxel of the dorsal rostral pons activation of the right nucleus accumbens seed to voxel connectivity analysis.
Regarding the connectivity analyses of our individually defined small ROIs of regions typically involved in migraine pathophysiology, only the area of the dorsal rostral pons, as defined by the peak voxel of the brainstem activation found in Stankewitz et al. (10) to the right hypothalamic area, was significantly higher on the first ictal day as compared to pre-3 (see Figure 2 for further detail). Table 1 shows an overview of peak voxels and T-values from the seed to voxel analysis of the dorsal pons and the right nucleus accumbens.
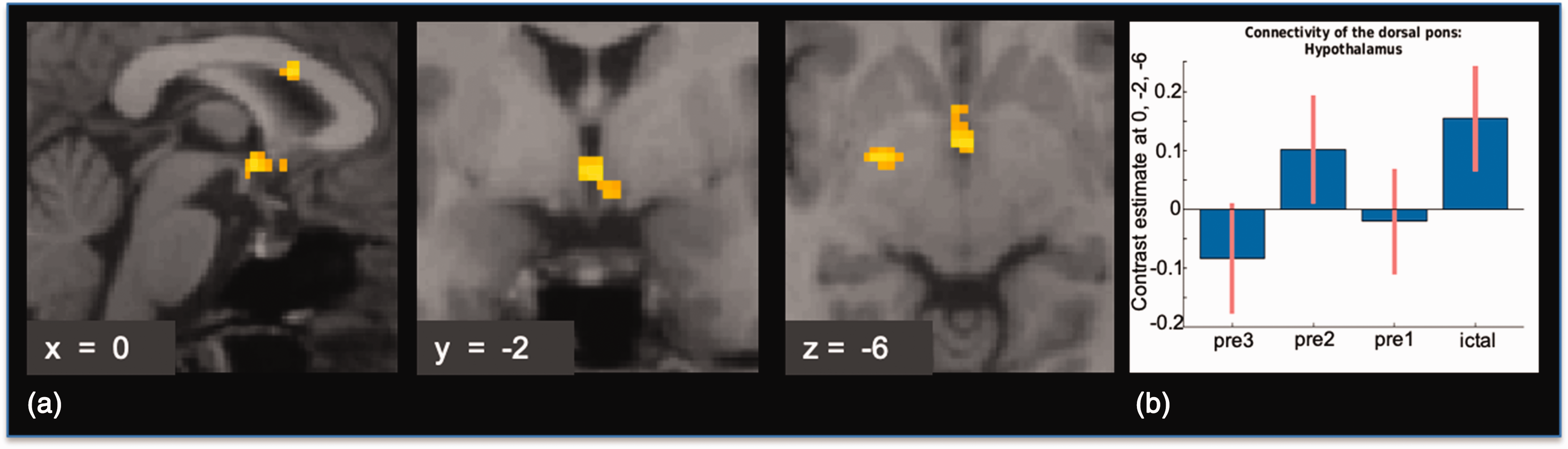
(a) Results of the seed to voxel connectivity analysis with the dorsal pons as a seed region, pre3 < ict: On the first ictal day, functional connectivity of the dorsal rostral pons to the left hypothalamic area was higher, as compared to on pre3. Results are small volume FWE-corrected, p < 0.05. Visualization threshold p < 0.005, k = 20, uncorrected. T-score-map overlaid on the normalized anatomical T1-image of one migraine patient. (b) Contrast estimates within the peak voxel of the hypothalamic activation of dorsal pons seed to voxel connectivity analysis.
Connectivity results of the seed to voxel analyses: Significant activations within the two predefined regions of interest.

Discussion
Scanning migraine patients on a daily basis for 4 weeks demonstrates that measures of resting state functional connectivity significantly change during different stages of the migraine cycle. Within the ROI-to-ROI connectivity analysis, three correlational links, all including the right nucleus accumbens, showed significantly altered connectivity to other ROIs, namely with the left amygdala, the left hippocampus and the left gyrus parahippocampalis, when comparing the last 24 hours before headache onset with the scan 3 days before headache onset. This indicates a predominant role of these brain regions within the preictal phase of migraine. Furthermore, in the seed to voxel analysis, the nucleus accumbens showed enhanced functional coupling with the dorsal rostral pons, a region which has been shown to increase activity shortly before and during migraine attacks (8,10,11,15,26,27). In the ictal phase, on the other hand, we found significantly higher functional coupling of the dorsal pons, as defined by previously published coordinates of this brain region (10) with the hypothalamus as compared to the scan 3 days before headache onset. While we were not able to replicate earlier findings of altered thalamic connectivity during induced migraine attacks in our spontaneous migraine attacks (27), these findings are in line with our previous data: In one migraine patient, the hypothalamus showed enhanced pain-dependent functional coupling to the dorsal rostral pons within the ictal state as compared to the interictal state (15). Whereas, however, our previous data used a task-dependent design and were thus only able to depict pain-dependent functional connectivity changes, the current data represent task free resting state changes within the migraine brain and are thus even more likely to represent a mechanism underlying cycling changes in migraine pathophysiology. Regarding other resting state studies in migraine, the hypothalamus has been shown to increase its connectivity to cortical resting state networks in chronic migraine (28). Due to the inclusion criteria for the attacks analysed within this study, we included 15 “trials” and thus have a relatively small sample size. It is, however, to our knowledge the largest one when specifically looking at studies focusing on the time preceding a headache attack in a longitudinal setting. Other relatively new studies focusing on the preictal phase using resting state fMRI use a very different design and result in a maximum of 10 “trials” investigated for the premonitory phase with an even smaller subset being truly longitudinal (17,18).
Given the wealth of data, it is undisputed that the dorsal pons plays a crucial role during spontaneous and NO-triggered migraine attacks (8–11,15,28,29) and is additionally even involved in the late premonitory phase of NO-triggered migraine attacks (11). The part of the dorsal pons that shows stronger functional coupling to the right nucleus accumbens during pre1 as compared to pre3 is in proximity to the hypothalamic region, which is active within the late premonitory phase of NO-triggered migraine attacks (11). Our data suggest that this region is not only generally more active shortly before the beginning of migraine headaches but that specific changes in connectivity in particular with the nucleus accumbens represent a pathophysiological mechanism for pontine activation in migraine. While the nucleus accumbens is not a prime candidate for migraine generation, it has been repeatedly reported to be involved in reward or anticipation-related aspects of pain (30–33), in shaping the affective and motivational value of pain (34) – maybe besides the nociceptive processing (35) – and shows pathological adaption during chronic pain in rodents (36). Even more importantly, it is of vast importance for dopamine-mediated analgesia or control of pain (37–39). Changing connectivity of the nucleus accumbens shortly before the onset of headache in migraineurs, especially with the dorsal rostral pons, might thus indicate functional changes in descending pain control. This effect might be mediated by dopamine: Dopaminergic input to the accumbens shell arises partly from the periaqueductal grey area and the dorsal raphe nucleus (40), which are both likely to be comprised in the observed dorsal pons area. A direct role of the dopaminergic system in migraine pathophysiology has long been debated (41,42): Dopamine agonists such as apomorphine could be shown to elicit symptoms like yawning and nausea, which are quite typical for the premonitory phase of migraine (14,43–46). Dopamine antagonists such as metoclopramide and haloperidol can be used as effective acute treatments of migraine (47–51). The current data might therefore corroborate a role of the dopaminergic system for migraine attack generation or insufficient attack inhibition with the changing connectivity of the nucleus accumbens as the key factor.
Of note, even the laterality of connectivity changes found in the current study are corroborated by findings from previous research: In interictal migraineurs, the volume of the right nucleus accumbens was decreased in comparison to healthy controls, whereas the volume of the left nucleus accumbens was not (52). In the same study, enhanced functional connectivity of the right accumbens to the parahippocampal gyrus was shown (52). Our data now indicate that these previously observed functional connections might also change during different stages of the migraine cycle. The fact that the dorsal pons shows enhanced functional connectivity with the hypothalamic region during migraine headaches further underlines the important function of both areas for migraine pathophysiology. The current data thus suggest that distinct changes in dopaminergic networks and their connectivity to the dorsal rostral pons might play an important role in migraine attack generation and that the functional coupling between the dorsal rostral pons and the hypothalamus is the basic mechanism for migraine pain sustainment.
Clinical implications
With this unique data, typical changes in functional connectivity involved in the preictal and ictal phase of migraine can be identified.
Connectivity changes in dopaminergic centers and between the dorsal pons and the hypothalamus are important within migraine attack generation and sustainment.
The found alterations of a specific network of brain regions in task-free resting state imaging imply that not only the transmitting and computing of trigeminal nociceptive input changes towards an attack, but indeed the state of the migraine brain determines migraine attack generation and migraine pain sustainment.
Footnotes
Acknowledgements
First and foremost, we thank the migraine patients who participated in this study. Furthermore, we would like to thank Dr. Jürgen Finsterbusch, leader of the MR physics group at our department and the radiographers Katrin Bergholz, Kathrin Wendt and Waldemar Schwarz for their continuous help during data acquisition.
Author contributions
Drafting of the study: AM and LHS. Data collection: LHS. Data analysis and/or interpretation: MMM, JH, LHS, AM. Drafting of the manuscript: LHS, MMM, AM.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Research Foundation, SFB936/A5 to AM and 7th Framework EU project EuroHeadPain (#602633) to AM.
