Abstract
Objective
Studies suggest that pain thresholds may be altered before and during migraine headaches, but it is still debated if a central or peripheral dysfunction is responsible for the onset of pain in migraine. The present blinded longitudinal study explores alterations in thermal pain thresholds and suprathreshold heat pain scores before, during, and after headache.
Methods
We measured pain thresholds to cold and heat, and pain scores to 30 seconds of suprathreshold heat four times in 49 migraineurs and once in 31 controls. Sessions in migraineurs were categorized by migraine diaries as interictal, preictal (≤one day before attack), ictal or postictal (≤one day after attack).
Results
Trigeminal cold pain thresholds were decreased (p = 0.014) and pain scores increased (p = 0.031) in the ictal compared to the interictal phase. Initial pain scores were decreased (p < 0.029), and the temporal profile showed less adaptation (p < 0.020) in the preictal compared to the interictal phase. Hand cold pain thresholds were decreased in interictal migraineurs compared to controls (p < 0.019).
Conclusion
Preictal heat hypoalgesia and reduced adaptation was followed by ictal trigeminal cold suballodynia and heat hyperalgesia. Our results support that cyclic alterations of pain perception occur late in the prodromal phase before headache. Further longitudinal investigation of how pain physiology changes within the migraine cycle is important to gain a more complete understanding of the pathogenic mechanisms behind the migraine attack.
Introduction
Altered pain perception may be of importance for migraine pathophysiology. Several studies have shown decreased experimental pain thresholds and increased pain scores in migraineurs in the headache-free interval (interictal phase) compared to healthy controls (1–13). About 60% of migraineurs report cutaneous allodynia during headache (the ictal phase) (14–17). This is comparable to the proportion with headache-related allodynia found in an experimental study (18).
Various symptoms may precede the headache, such as yawning, mood change, lethargy, neck symptoms and light sensitivity (19–23). However, little is known about the central mechanisms and sequence of events that initiates these warning/premonitory symptoms. Several symptoms may also outlast the headache (postdromal symptoms) (19,22,24,25). The premonitory and postdromal symptoms, as well as imaging (26–28) and neurophysiological (29–38) findings indicate that migraine is driven by cyclic central nervous system alterations that precede and outlast the ictal phase.
Several studies have shown increased responses to experimental pain during the ictal phase compared to the interictal phase (11,39–43). Although the alteration in pain perception is most pronounced during migraine attacks, subtler changes may be present before and after the headache (preictal and postictal phase, respectively). Few have investigated pain-related physiological changes across migraine phases. A longitudinal study demonstrated decreased thermal pain thresholds preictally compared to interictally (36). An association between heat pain thresholds and hours to the next attack (10), and an association between activation in the spinal trigeminal nuclei by nociceptive stimuli and the time to the next attack (44), have also been reported. Exploring pain perception in the preictal and postictal phases could contribute to a better understanding of the pathophysiology (45).
Experimental tonic pain may resemble clinical pain better than pain thresholds (46), and the temporal profile may reflect both peripheral and central mechanisms (47,48). Furthermore, in order to elucidate migraine mechanisms, intraindividual changes to tonic painful stimulation during the different migraine phases may be more relevant than comparing migraineurs in the interictal phase to healthy controls. This has not been investigated earlier.
Longitudinal studies are preferred when estimating changes in pain perception between the different phases (57). We have earlier reported preictal heat suballodynia, that is, a pain threshold decrease within the normal range (see Weissman-Fogel et al. (12) for a discussion of the term), in migraine patients (36). However, the number of migraineurs with both interictal-ictal and interictal-postictal paired measurements was too low to be analyzed in our previous study published in 2008 (36).
The present blinded longitudinal study included a larger number of migraineurs with both interictal-ictal and interictal-postictal paired measurements. We test the hypothesis that pain thresholds decrease and pain scores increase both the day before, during and the day after the ictal phase compared to the interictal phase, indicating that suballodynia and/or hyperalgesia precedes and outlasts the headache during migraine attacks. Secondly, we test the hypothesis that migraineurs in the interictal phase have lower pain thresholds and increased suprathreshold pain scores than headache-free controls.
Methods
We measured thermal pain thresholds once a week for four weeks in migraineurs (mean ± SD: 6.7 ± 1.9 days between sessions) in the period between June and December 2012. The migraineurs completed a headache diary for four weeks before, during and four weeks after the examinations in order to determine how the examinations were related to the migraine attacks (i.e. interictal, preictal, ictal or postictal). Thermal pain thresholds and scores were measured once in headache-free controls.
Subjects
Fifty migraineurs and 31 headache-free controls were recruited by advertising in the local newspaper, on the local hospital’s webpage (St. Olavs Hospital, Trondheim University Hospital; www.stolav.no/seksjon-engelsk) and on the Intranet within our university (NTNU, Norwegian University of Science and Technology; www.ntnu.edu).
Controls had to have headache less than once a month. If they had any occasional headache, we asked if they had consulted a physician regarding headache, if the headache was experienced as painful, and if they used abortive medication for their headache. They were excluded if they confirmed more than one of these three questions. Forty control subjects were screened over the telephone, two did not meet the criteria and seven dropped out. Thus, a total of 31 controls completed one examination each.
Migraineurs were evaluated by neurologists according to the ICHD-II criteria for migraine with or without aura (49). Included subjects had an attack frequency between two and six per month and had no more than ten days with migraine attacks per month. They could use symptomatic, but not prophylactic migraine treatment. Exclusion criteria were coexisting tension-type headache greater than or equal to seven days per month in migraineurs, neurological or psychiatric diseases, sleep disorders, active infectious diseases, connective tissue diseases, metabolic, endocrine or neuromuscular diseases, other clinically-relevant painful conditions including recent injuries, malignancy, previous craniotomy or cervical spine surgery, heart disease, cardiopulmonary or cerebrovascular diseases, pregnancy, medication for acute or chronic pain, antipsychotics, antidepressants, anticonvulsants or other drugs that may influence neuronal, vascular or muscular function, alcohol or drug abuse, ferromagnetic implants and prophylactic allergy treatment.
One migraineur withdrew consent after the first examination and was not included in the analysis. Three migraineurs attended only once, twice and three out of four times respectively. Forty-nine migraineurs completed a total of 190 examinations (Figure 1). Table 1 shows demographic and clinical data. (50)
Flow chart for the migraineurs in the study. The number of subjects who dropped out due to personal reasons is shown at the bottom. Demographic and clinical data after exclusions. Migraine days/month: 0: < 1/month, 1: 1–3/month, 2: 4–7/month, 3: 8–14/month, 4: > 14/month. Migraine intensity: 1: Mild, 2: Moderate, 3: Severe, 4: Extreme. Average duration of an attack with or without use of symptomatic medication. MwoA: migraine without aura. MA+MwoA: some attacks with and some without aura (both diagnoses according to ICHD-III (50)). MA: Migraine with aura (in 100% of attacks). NA: Not applicable.

Investigators were blinded to diagnosis on the subjects’ first visit, and to migraine phase on the subsequent visits. Co-workers undertook inclusion, coordination and follow-up of participants, and participating subjects were specifically told not to reveal to which group they belonged to the investigators. The Regional Committees for Medical and Health Research Ethics approved the protocol, and all subjects gave their written informed consent. Migraineurs and controls received the equivalent of $125 and $30 respectively, to cover expenses.
Procedure
All sessions on one subject were at the same time of day. The method of limits was used to measure thermal pain thresholds (51). Recordings were performed on SOMEDIC SenseLab equipment (Somedic Sales AB, Stockholm). The right hand (thenar eminence overlying the abductor pollicis brevis muscle) and right side of the forehead (frontal region above the eyebrows aligned with the inner canthus) were stimulated with a hand-held rectangular 25 × 50 mm Peltier element thermode (Somedic Sales AB, Stockholm). Target start temperature was 32℃, and the actual start temperature was recorded by the system. The stimulation range was 5–50℃ and the slope was 1℃/s. Cold pain threshold (CPT) and heat pain threshold (HPT) were measured four times consecutively with four to six seconds’ random inter-stimuli intervals. The order was constant; CPT before HPT and hand before forehead. Participants were instructed to press a button when the stimulus was perceived as painful. An introductory round was carried out at the beginning of each the day, consisting of two measurements of both thresholds on the hand.
Temporal profiles of suprathreshold heat pain scores were obtained during 30 seconds’ continuous suprathreshold heat pain stimulation on the right forearm and temple. The individual determined tonic temperature that scored six on a verbal numerical rating scale (NRS), ranging from 0 = no pain to 10 = unbearable pain, was set as the test stimulus (52). We used the same equipment and thermode as when testing thresholds, controlled by the Exposure30 software by SOMEDIC. Start temperature was set at 32℃, slope 1℃/s. To determine a temperature level for the test stimulus, subjects were first exposed to stimuli of seven seconds’ duration at 45℃. They verbally reported pain scores using NRS continuously throughout stimulation. The highest pain score reported determined the temperature for the next test stimulus. We increased the temperature if NRS was less than six and decreased it if NRS was more than six. At least three stimuli were applied on both sites with a minimum of a one-minute inter-stimulus interval on the same site. The temperature perceived as the NRS score closest to six was chosen for the test stimulus. Two temperatures were determined, one for the temple and one for the forearm. The main suprathreshold heat pain test procedure consisted of one continuous stimulation per site with 30 seconds’ duration. Verbal NRS scores were reported continuously. Subjects were instructed to update their pain score verbally whenever the experienced pain changed. The last reported NRS score at 0, 10, 20 and 30 seconds was stored for analysis, where 0 seconds represents the time the thermode reached the test stimulus temperature. The same individually determined temperatures were used for the next three examination days.
Data analysis
Thresholds were defined as the difference from the measured start temperature (dCPT = start − CPT and dHPT = HPT − start). Outlier detection software was applied, removing single dCPT and dHPT responses with magnitudes of more than three times or less than one third of the mean of the three associated responses from the same examination day. Examinations were classified by the headache diary as interictal (more than one day before attack onset or one day after the attack ended), preictal (less than one day before attack onset), ictal (migraine headache during examination) and postictal (less than one day after the attack ended). A secondary set of analyses were also performed with a three-day limit. Eleven of the 190 examinations were unclassifiable and excluded from data analysis, mainly because they had attacks both the day before and the day after examination. The distribution of phases is shown in Figure 2.
Bar graph showing the distribution of phase combinations among migraineurs. The labels on the y-axis represents the number of exams in each phase (interictal, preictal, ictal and postictal, respectively). Hence, for example, 2,1,0,1 means two interictal, one preictal, zero ictal and one postictal recording. The number of subjects with a particular combination of phases are represented by the size of the corresponding bar and labeled on the x-axis. Drop-outs account for six missing tests, while 11 tests were excluded as unclassifiable.
STATA (StataCorp LP, version 13.1) was used to run separate multilevel models (53) for each response variable (dCPT, dHPT and suprathreshold heat pain scores). Inclusion of fixed effects was determined by the research questions. The first three models compared migraineurs’ within-subject change by migraine phase and site. In addition, to explore adaptation and sensitization effects, we included pain rating-time to explore possible differences within each time-point of the continuous suprathreshold heat pain stimulation protocol. Secondly, in three models we compared between-group responses from controls and migraineurs in the interictal phase.
The lower limit of the thermal threshold equipment was 5℃, i.e., dCPT = 27. A substantial number of dCPT-measurements reached this limit. We knew that these dCPT were above 27, but not by how much, and they were thus defined as censored (54). The distribution of censored responses was skewed, e.g. more in controls than interictal migraineurs. One may underestimate a possible difference between the groups if the censored variables are not properly accounted for. Analysis of dCPT was done by modeling both the change of non-censored responses between phases and the probability of reaching the limit, while accounting for dependencies in the data; see the Supplementary Appendix for details.
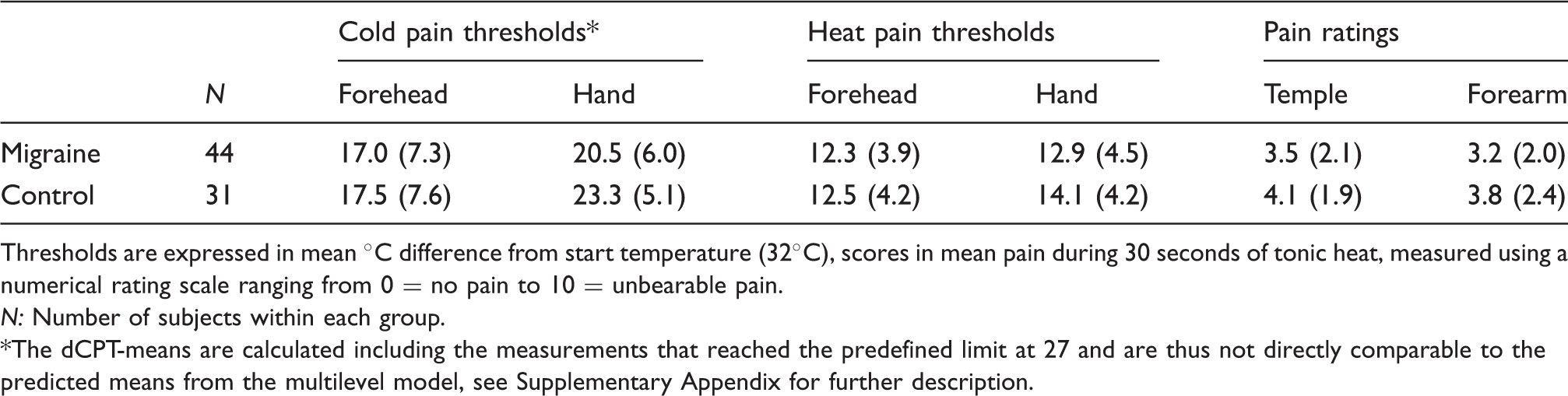
Observed mean (SD) thermal pain thresholds and pain scores by migraine phase and stimulation site.

Thresholds are expressed in mean ℃ difference from start temperature (32℃), scores in mean pain during 30 seconds of tonic heat, measured using a numerical rating scale ranging from 0 = no pain to 10 = unbearable pain.
N: Number of subjects with at least one recording at the respective phase.
n: Total number of measurements by phase.
The dCPT-means are calculated including the measurements that reached the predefined limit at 27 and are thus not directly comparable to the predicted means from the multilevel model, see Supplementary Appendix for further description.
Mean (SD) thermal pain thresholds and pain scores in interictal migraineurs and controls.
Thresholds are expressed in mean ℃ difference from start temperature (32℃), scores in mean pain during 30 seconds of tonic heat, measured using a numerical rating scale ranging from 0 = no pain to 10 = unbearable pain.
N: Number of subjects within each group.
The dCPT-means are calculated including the measurements that reached the predefined limit at 27 and are thus not directly comparable to the predicted means from the multilevel model, see Supplementary Appendix for further description.
As additional secondary sub-analyses, we extended the models with selected factors and covariates that might have had an effect on the results. Aura and headache lateralization were tested as factors. Differences in summation of pain thresholds between phases and groups were tested by including a linear covariate of test repeats.
To test if there was a linear relationship between pain thresholds and scores and time to the next attack, three additional multilevel models were conducted. They were specified the same way as the three main models, except the dummy-coded variable “phase” was exchanged with the continuous variable “days to next attack”. Interictal recordings were first analyzed, while preictal and interictal recordings were included in a second set of analyses.
With 30 controls and 50 migraine subjects, the power to detect a low medium-sized effect equal to 0.65 SD (55) based on a two-sample t-test was calculated at 80%. As we estimated having approximately 20 pairs for intraindividual phase-related comparisons, power (based on paired t-tests) to detect a similar medium-sized effect (0.65 SD) was calculated at 83%.
Results
Migraineurs by phase
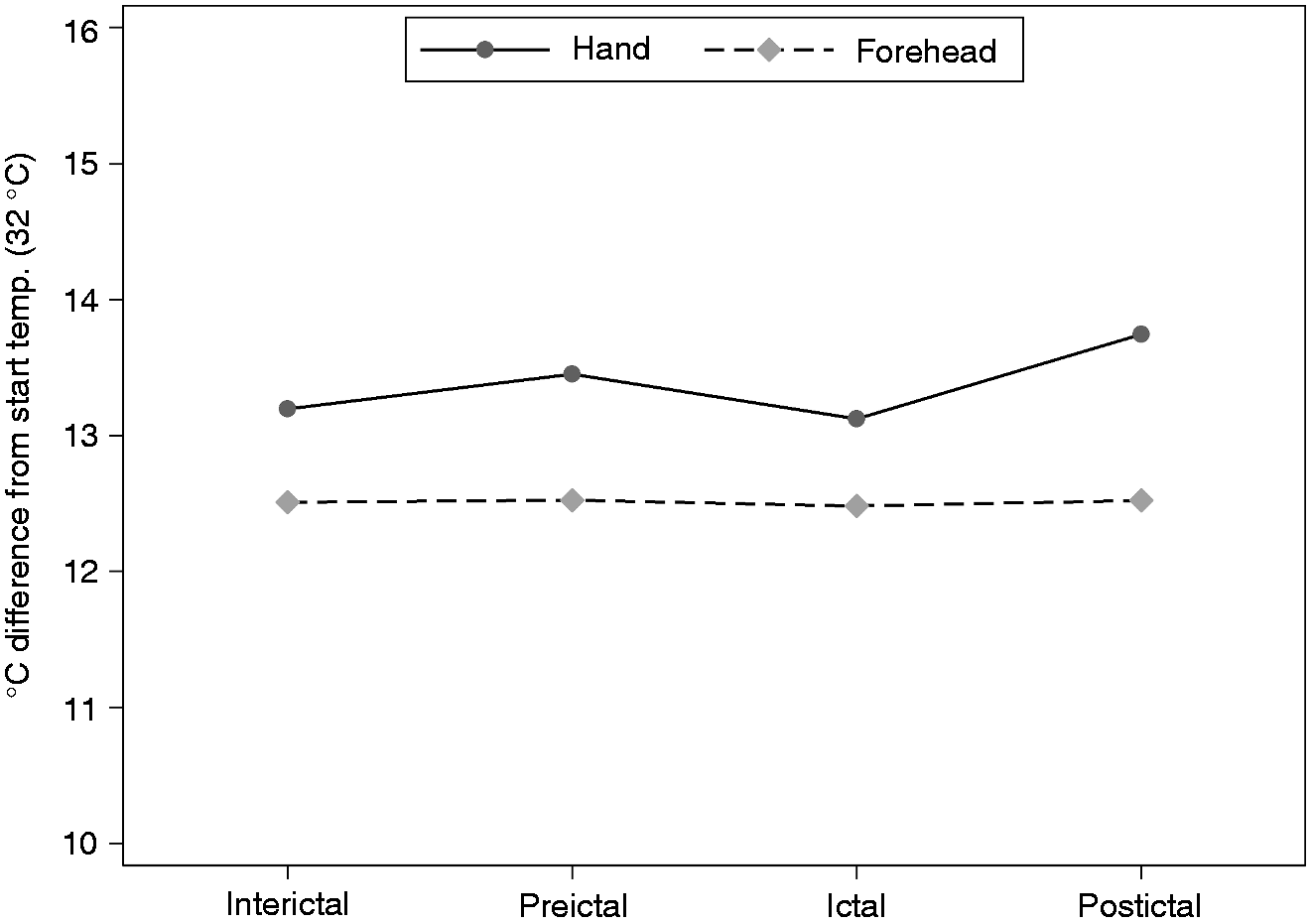
Table 2 shows descriptive means of dCPT, dHPT and pain scores by phase and site. Forehead dCPT decreased by 2.2 (95% CI: 0.5, 4.0) ℃ (p = 0.014) in the ictal phase compared to the interictal phase (Figure 3). The interictal-ictal forehead dCPT change was significantly larger than the interictal-ictal change at the hand (p = 0.013). Neither preictal nor postictal dCPT changed compared to interictal dCPT. Post-hoc analysis of contrasts showed that ictal forehead dCPT was significantly decreased compared to both preictal (p = 0.043) and postictal (p = 0.037) dCPT. These findings were interpreted as ictal forehead suballodynia. There were no significant hand or forehead dHPT differences between phases (p > 0.10, Figure 4).
Cold pain thresholds. Graphical display of estimated margins from the main multilevel model comparing the effects of phase and site on cold pain thresholds. Ictal forehead thresholds were significantly decreased compared to interictal forehead thresholds. The decrease was within the normal range, thus interpreted as trigeminal suballodynia in the ictal phase. Heat pain thresholds. Graphical display of estimated margins from the main multilevel model comparing the effects of phase and site on heat pain thresholds. There were no significant differences between phases.
Overall pain scores to the continuous suprathreshold heat pain stimulation at the temple were 0.6 (95% CI: 0.1, 1.2) points higher ictally compared to interictally (p = 0.031). When looking at the pain scores separately for each time point, lower scores were found preictally for the first time point. Temple pain scores at 0 seconds were 0.8 (0.2, 1.4) and forearm scores 0.7 (0.1, 1.3) points lower in the preictal compared to the interictal phase (p < 0.029, Figure 5). Less adaptation was found preictally compared to interictally, as pain scores at both sites decreased from 0 to 20 and 30 seconds in the interictal phase (p < 0.001), while preictal pain scores decreased significantly less (p < 0.020).
Pain scores during continuous suprathreshold heat pain stimulation. Graphical display of estimated margins from the main multilevel model comparing the effects of phase, site and time on pain scores. The x-axis represents the four time-points at which pain scores were recorded during 30 seconds of tonic heat. The overall pain scores at the temple were increased ictally compared to interictally, interpreted as trigeminal hyperalgesia in the ictal phase. At time point 0s, preictal pain scores were decreased at both sites compared to interictal pain scores, interpreted as initial preictal hypoalgesia. There were interictal decreases in pain scores from 0s to 20s and 0s to 30s at both sites, interpreted as interictal adaptation of pain scores. The preictal pain scores decreased significantly less, interpreted as preictal lack of adaptation.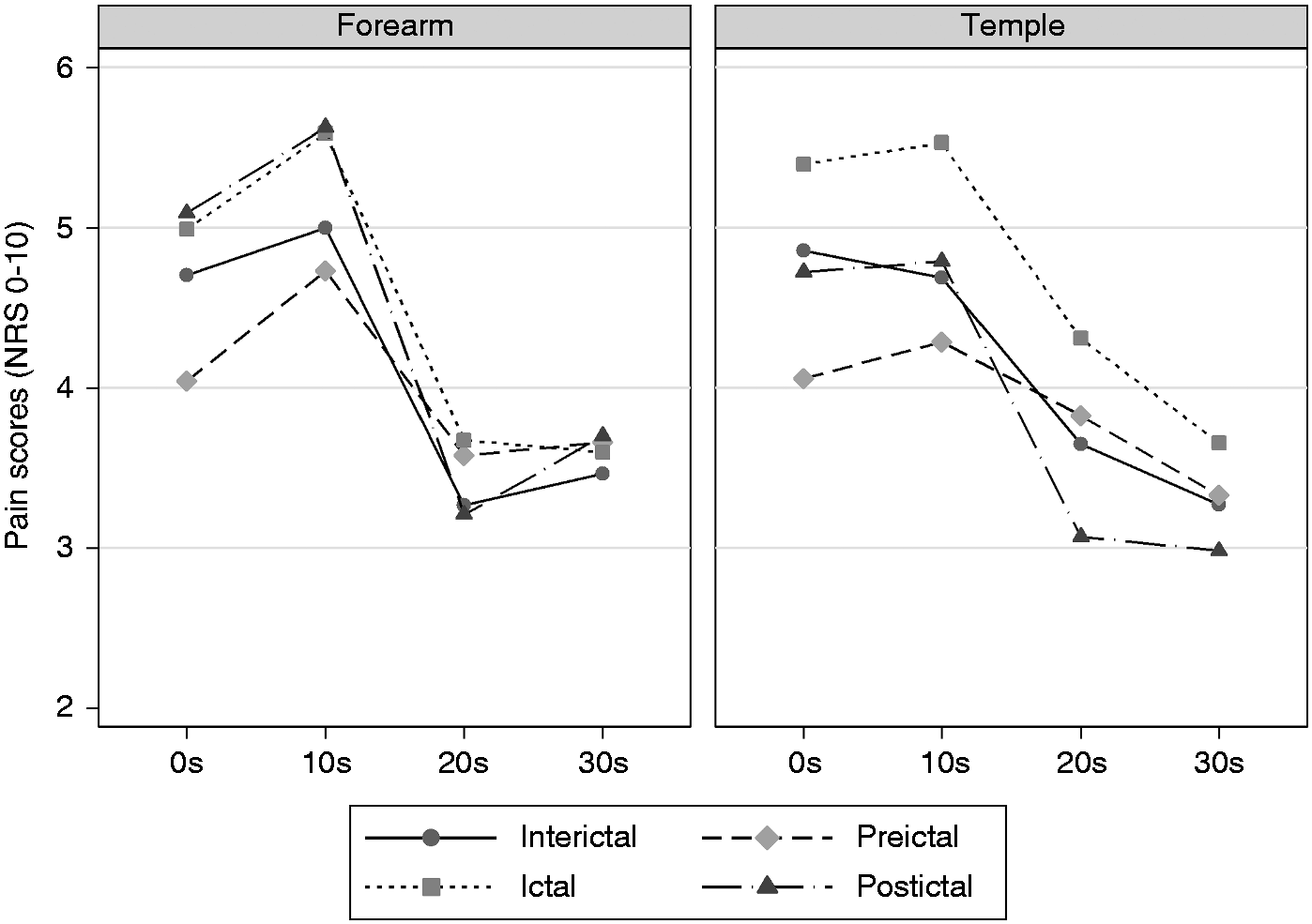
Neither dCPT, dHPT nor pain-score results were significantly altered by controlling for aura or headache laterality. Both dCPT and dHPT showed a significant linear summation of pain during the four stimuli (p < 0.001). However, the summation did not differ between phases (p > 0.079) and did not alter the original results.
Days to the next attack did not affect dCPT and dHPT, either for the interictal group or the combined interictal and preictal group (p > 0.34). For the interictal subgroup, a daily increase in pain score towards the next attack was estimated to 0.08 (0.01, 0.15) (p = 0.033) on the temple and 0.09 (0.02, 0.16) (p = 0.008) on the forearm. However, when preictal recordings were added, the significant association disappeared. Adaptation of pain scores from 0 to 20 and 30 seconds remained significant in both analyses (p < 0.004).
For dCPT and dHPT, changing the definition of the preictal and postictal phases from a one-day limit to a three-day limit did not change the original results. However, preictal pain scores at 0 seconds and the adaptation from 0 to 20 and 30 seconds were then no longer significantly different between the interictal and preictal phase (p > 0.79).
Interictal migraineurs and controls
Table 3 shows descriptive means of dCPT, dHPT and pain scores by group and site. Hand dCPT was decreased by 4.4 (0.7, 8.1) ℃ (p < 0.019) in interictal migraineurs compared to controls. Forehead dCPT was not different between groups (p = 0.76). Neither dHPT nor pain scores differed significantly between groups (p > 0.11). Pain scores during continuous suprathreshold heat pain stimulation decreased in both groups from 0 to 20 and 30 seconds (p < 0.001). Test stimulus temperature means (±SD) were also not significantly different between migraineurs and controls (temple: 46.7 ± 1.9 vs. 46.9 ± 2.1℃, p = 0.69, forearm: 45.9 ± 1.8 vs. 46.5 ± 2.1℃, p = 0.22).
Discussion
We observed trigeminal cold suballodynia and heat hyperalgesia during the ictal phase. Pain thresholds did not change from the interictal to the preictal or postictal phase. This finding indicates that the initial cortical processes responsible for the prodromal symptoms are not associated with substantial sensitization of extracranial thermal nociceptors, at least not until the actual headache phase is rather close.
In line with the previously reported ictal thermal allodynia (18), preictal heat and cold suballodynia (36), increased nociceptive activity in the spinal trigeminal nuclei (44) and decreased HPT towards the next attack (10), one would expect that pain thresholds would gradually decrease and pain scores increase from the interictal to the preictal and subsequently to the ictal phase. Schwedt et al. (10) found an association between decreased forehead HPT and the proximity to the next attack, in accordance with Sand et al. (36). Another small study did not find significant differences in pressure and thermal pain thresholds between interictal, preictal and postictal migraineurs (1), but the latter study did not possess sufficient statistical power to disprove the concept. Pain thresholds did not change from the interictal to the preictal phase in the present study, and we could accordingly not confirm our previous result regarding preictal thermal suballodynia (36). However, both dHPT and dCPT means were lower in the ictal compared to the interictal phase (Table 2), suggesting that an interictal-preictal-ictal gradient can exist. Although pain thresholds were not affected linearly by days to next attack when interictal and preictal patients were combined and analyzed over a 15-day time range, it is still possible that preictal thermal suballodynia evolves closer to the attack, e.g. within some hours, in many episodic migraine patients.
The present results may also suggest that preictal abnormalities in heat pain processing may be more consistently expressed as subtle suprathreshold pain score differences. Surprisingly, preictal pain scores demonstrated hypoalgesia compared to interictal scores, which was the opposite of what we expected. However, the pain scores at 0 seconds were no longer lowered preictally when changing the definition of the preictal phase from one to three days before the attack. In fact, the subanalysis with the linear effect on days to next attack showed increasing pain scores closer to the attack when the data from the preictal phase were excluded. Thus, migraineurs had increasing hyperalgesia towards the next attack and hyperalgesia during headache, as expected. However, this general pattern was interrupted for a limited time-window preceding headache, interpreted as preictal hypoalgesia. These results suggest that significant central events affect processing of pain on the day before headache.
Stankewitz et al. (44) found lower fMRI-activation in response to trigeminal pain in the spinal trigeminal nuclei in interictal and ictal migraine subjects compared to controls, while activation was normal in the preictal group within 72 hours before the next attack. However, pain scores were unaltered between phases (44). A recent study scanned one migraineur daily for 30 days to analyze fMRI-activation by phase, in response to trigeminal pain (27). The migraine patient experienced three attacks during the period, and results showed that hypothalamic activity increased towards each migraine attack. Further, functional coupling analysis showed increased coupling between the hypothalamus and the spinal trigeminal nuclei preictally (with a 24h limit), whereas during the ictal phase, coupling to the trigeminal nuclei was significantly decreased (although increased between the hypothalamus and the dorsal rostral pons) (27). These results, combined with the preictal hypoalgesia observed in our study, may suggest that fMRI-activation of the trigeminal nuclei reflect increased descending modulation preictally (26). Preictal hypoalgesia was present both in the face and in the arm in the present study, supporting that preictal pain scores are altered by central rather than peripheral mechanisms.
The observed temporal profile of pain scores during continuous suprathreshold heat pain stimulation in the present study is at variance with some (52,56,57), but not all previous studies (58–62). Migraineurs demonstrated lower initial pain and significantly less adaptation in the preictal compared to the interictal phase. A-delta fibers may be important for the initial rise and fall in pain scores observed in the first 15 seconds of the continuous suprathreshold heat pain stimulation (60,63,64). Our observed lower pain scores could have reflected a blunted preictal A-delta nociceptive response, but since a central mechanism is most probable, we interpret this finding as a blunted preictal saliency perception.
The decreased hand dCPT in migraineurs between attacks compared to controls may reflect a state of slight chronic sensitization of pain pathways, possibly due to frequent pain experiences (43), as pain thresholds may decrease in relation to increased attack frequency (65–67). Cortical pain modulation seems to be disturbed in migraine (68). Altered sensory modulation in general is also reflected by phono- and photophobia, prodromal symptoms (19,23), and migraine triggers such as cognitive stress (69) in susceptible subjects (70). However, enhanced interictal sensitization was of moderate magnitude in our present study, as only hand CPT was affected, indicating that pain thresholds and pain scores may be largely unaltered interictally. In accordance with a previous study (12), pain scores to tonic suprathreshold heat did not differ between interictal migraineurs and controls. Overall, thermal pain sensitivity changes in migraine may be easier to observe in the cold than the heat domain.
Studies comparing experimental pain in migraineurs and controls have shown variable results; either hypersensitivity (1–13) or no differences (1,5,9,12,36,67,68,71–76), but never hyposensitivity. Some subgroups may be more hypersensitive than others; for instance, migraineurs with non-sleep related migraine attacks had lower CPT and HPT than controls (77), while less slow wave sleep was associated with higher pressure pain thresholds (1). Disease severity may also be of importance, as headache history duration may modulate CPT (36), while chronic migraineurs (>15 days/month) were more sensitive to pain compared to episodic migraineurs in one study (66), but not in another (9). Headache frequency correlated with temporal summation of electrical and mechanical stimulation (12) and pressure thresholds (67), although there are contradictory findings (4). Thermal pain thresholds did not correlate with headache frequency, allodynia symptom severity, anxiety scores or depression scores (10). Migraine is divided into subgroups of subjects with and without aura, but these groups did not differ in the present study and do not seem to differ systematically by pain thresholds in previous studies (5,36). Thus, since a multitude of factors may influence sensitivity in individual patients, this heterogeneity may explain why results regarding pain thresholds and other sensitivity measures vary between studies.
Strengths and limitations
By prospectively measuring pain thresholds and scores four times within each patient, we obtained a substantial number of subjects measured at different phases. Blinding of the investigators during recording and analysis adds further strength to the study (78). We used robust and flexible multilevel statistical models, enabling us to analyze all the data without prior mean calculations and listwise deletions, optimize the model fit and to properly account for the substantial and uneven censoring of dCPT between groups. An alternative study design, such as asking patients to present for a test session during attack, would increase the number of ictal recordings and thus power for an interictal-ictal comparison, although it would be more difficult to control factors such as time of day, blinding of phase, and anticipation. But more importantly, we chose random recordings with diary-based classification to be able to investigate the preictal phase.
To obtain reproducible results, we applied a standardized procedure (79); the room was quiet with constant lightning (no windows), pre-written instructions were read to all subjects, the test was always done in the same manner and the same examiner did all the testing. The repeatability of thermal pain thresholds has proven to be satisfactory, although CPT may be a less robust measure due to relatively large standard deviations (80–82).
The comparisons of interictal migraineurs and controls could have been biased by habituation/sensitization effects, because we included all the exams of migraineurs in the interictal phase. However, the conclusions did not change when rerunning the analyses with only exams from the first day (results not reported).
We tested the pain thresholds and scores systematically on the right side, regardless of the side on which the migraineurs most commonly experienced headache. This may be a drawback, since allodynia ipsilateral to the headache may occur before contralateral allodynia (83). However, a previous study demonstrated no significant difference between the symptomatic and non-symptomatic side for the interictal-preictal differences (36), and inclusion of headache laterality in the subanalyses did not affect the results. Migraineurs were allowed to take abortive medications. However, it is unlikely that the medication has an effect on other phases than the ictal phase due to short half-life, and the effect is likely to be increased pain thresholds and decreased scores, the opposite of what we found in the ictal phase. Six of the migraineurs reported prodromal allodynia by questionnaire. We did not collect information on self-reported clinical interictal or ictal allodynia, an explanatory variable that could be of importance.
Repetitive painful stimuli evoke pain amplification characterized by increased responses in the dorsal horn and in descending modulation of pain (84). The central mechanisms to pain amplification may be common for both phasic and tonic pain (52). We obtained temporal profiles during 30 seconds of suprathreshold heat stimulation. Future studies should extend the stimulation period in order to analyze pain intensification during the second minute of tonic heat stimulation (57,62) and further elucidate variations in central pain modulation between phases.
Conclusion
The present longitudinal study is unique in recording experimental pain from patients on four different occasions, aiming to perform intraindividual analysis of the most clinically relevant pain-physiology parameters (reflecting hypo- hyperalgesia/allodynia/ temporal summation) by migraine phase from a sufficiently large sample. We found trigeminal cold suballodynia and heat hyperalgesia during the ictal phase of migraine headache, and heat hypoalgesia and reduced adaptation to tonic suprathreshold heat pain preictally in both trigeminal and peripheral sites. Our findings suggest that central modulation of pain depends on migraine phase. Although the ictal phase is characterized by increased trigeminal pain sensitivity, different (and subtle) changes were found in the preictal phase, possibly due to increased descending pain modulation, affecting mainly suprathreshold pain scores. Our results support the theory that migraine is a cyclic disorder of the central nervous system related to global alterations of brain excitability and homeostasis. Studies with an emphasis on the preictal phase, preferably longitudinally with high temporal resolution and with parallel paraclinical recordings using fMRI, etc., are needed to further elucidate migraine pathogenesis.
Article highlights
This blinded longitudinal study investigated within-subject fluctuations of thermal pain sensitivity by migraine phase. We found heat hypoalgesia on the day before headache, as suprathreshold pain scores were decreased. We found cold suballodynia and hyperalgesia during headache, as cold pain thresholds were decreased and suprathreshold pain scores were increased. Cyclic central changes in pain physiology seem to emerge during the preictal phase, possibly related to headache-initiating mechanisms.
Footnotes
Acknowledgements
We are most grateful for invaluable assistance from Lena Hoem Nordhaug, Jo Willy Gråwe, Morten Engstrøm, Gøril Bruvik Gravdahl and Marit Stjern.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from NTNU, Norwegian University of Science and Technology.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
