Abstract
Aim
The aim of this article is to investigate the pathophysiology underlying the alternation of the cognitive function and neuronal activity in spontaneous intracranial hypotension (SIH).
Methods
Fifteen patients with SIH underwent resting-state functional magnetic resonance imaging and working-memory (WM) test one day before and one month after a surgical operation. Alternation of the cognitive function and spontaneous neuronal activity measured as amplitude of the low-frequency fluctuations (ALFF) and the functional connectivity of the default-mode network (DMN) and frontoparietal networks (FPNs) were evaluated.
Results
WM performance significantly improved post-operatively. Whole-brain linear regression analysis of the ALFF revealed a positive correlation between cognitive performance change and ALFF change in the precuneus while a negative correlation was found in the bilateral orbitofrontal cortices (OFCs) and right medial frontal cortex (MFC). The ALFF changes normalised with the WM performance improvement post-operatively. The FPN activity in the right OFC was also increased pre-operatively. Partial correlation analysis revealed a significant correlation between WM performance and right OFC activity controlled for right FPN activity.
Conclusions
The abnormal activity of the OFCs and MFC that is not originating from the synchronous intrinsic network activity, together with the decreased activity of the central node of the DMN, could lead to cognitive impairment in SIH that is reversible through restoration of the cerebrospinal fluid.
Introduction
Spontaneous intracranial hypotension (SIH) or cerebrospinal fluid (CSF) hypovolaemia is a rare disorder mostly affecting young and middle-aged individuals (1). In SIH, CSF leakage in the spine leads to loss of dynamics of intracranial pressure or the buoyancy of the brain, resulting in various neurologic symptoms characterised by postural headache (1). Traction on intracranial structures is considered to directly cause headache and some visuoauditory symptoms such as diplopia, photophobia, tinnitus, and hypacusia. The impairments, notably headache, could also be attributed to subdural fluid collections, dural thickening, or engorgement of cerebral venous sinuses that occur to compensate for the volume reduction within the rigid skull, as is explained by the modern Monro-Kellie hypothesis (2).
Although frank dementia (3) or coma resulting from diencephalic or brainstem herniation in SIH is not frequent (4), subtle cognitive deficits often unrecognised before treatment are experientially known to be common (1). However, the pathophysiology of the neural subsystems underlying the impairment remains unknown. In contrast to hydrocephalus, in which raised intracranial pressure or ventricular dilatation has been shown to directly or indirectly, e.g. via hypoperfusion, lead to permanent cerebral injury (5), no specific neural pathology has been demonstrated in SIH (6). However, based on the clinical features resembling frontotemporal dementia affecting memory and executive function, as well as imaging characteristics of the sagging brain, mechanical compression of the basal structures of the brain has been hypothesised as a possible cause of the cognitive impairment (3,6). Examination of the patients with SIH was thus considered to provide an opportunity to study a distinctive reversible form of cognitive impairment.
High-order cognitive functions such as memory and executive function rely on coordination and interaction between the large-scale brain networks (7). Working memory is the ability to transiently store and simultaneously manipulate information to be used (8). Numerous studies have reported activation of the frontoparietal areas during the execution of n-back tasks, and that the synchronous interactions among these regions comprising frontoparietal networks (FPNs) support working-memory processes (9). Synchronised neuronal activity has been demonstrated not only during the task execution but also in the absence of tasks within both task-positive network and task-negative or the default-mode network (DMN) (10). Development of the resting-state functional magnetic resonance imaging (fMRI) enabled investigation of these large-scale intrinsic neuronal networks, and recent studies on healthy individuals have indicated that increased synchronisation within the DMN contributes to the facilitation or monitoring of cognitive performance (11). Furthermore, clinical studies on neurodegenerative and psychiatric diseases have consistently revealed dysfunction of the resting-state networks, notably the DMN, emphasising the importance of the large-scale brain networks’ function for cognition (12). While conventional task-based fMRI reveals changes in neuronal activity that is limited in small areas of activation for a specific task, resting-state fMRI can be used to study the whole-brain neural process, even for patients who cannot perform a task (12). In the present study we thus investigated changes in cognitive function and the neuronal activity to test the hypothesis that cognitive impairment in SIH is associated with disruption of normal spontaneous activity of the FPNs and/or DMN nodes.
Material and methods
Participants
From June 2013 to May 2014, 15 patients (eight men; mean age 44 ± 13 years) who fulfilled the diagnostic criteria for SIH by the Headache Classification Subcommittee of the International Headache Society (2004) (13) were prospectively included in the study. All patients underwent brain MRI and a conventional myelography for diagnosis and identification of the sites of leakage. A single epidural blood patch was performed on the day following the pre-operative evaluation. Informed consent was obtained from all participants before data acquisition. All procedures were in compliance with the Declaration of Helsinki, and the Sanno hospital ethics committee approved the study.
Data acquisition
Data were acquired one day before and one month following a blood patch procedure. MRI was performed with a single whole-body 1.5-T MR unit (Achieva, Philips Healthcare, Best, the Netherlands) by using an eight-channel phased-array head coil. In addition to high spatial resolution T1-weighted and T2-weighted imaging, 10-minute resting-state fMRI with single-shot T2*-weighted gradient-echo echo-planar imaging (repetition time/echo time 2000/30 ms, 90-degree flip angle, 64 × 64 matrix, 256 × 256 mm2 field of view, 4-mm-thick sections, no gap, 34 sections, and 304 frames) were acquired. All participants were instructed to lie still and remain awake with their eyes open.
Behavioural assessment including the Mini-Mental State Examination (MMSE), Headache Impact Test (HIT6) (14), and a working-memory (n-back) test were performed outside the scanner after imaging examinations. The n-back task comprised a one-back and a two-back test. Participants viewed a series of white numbers (0–9) presented in the centre of a black screen. Each number was presented for 1200 ms followed by 1000 ms of interstimulus interval. The target was any number that was identical to the one presented one or two trials back, respectively. Participants responded to the target (33% of 60 trials) by pressing a button on a laptop computer. Each response and response time was recorded and the performance was evaluated using the correct answer rate.
Data analysis
fMRI data were pre-processed and analysed using SPM 8 software (www.fil.ion.ucl.ac.uk/spm/software/spm8/) implemented in Matlab 7.13.0 (MathWorks, Sherborn, MA, USA). The first four volumes were discarded to allow for T1 equilibration effects. Data were realigned to the first volume, corrected for differences in acquisition time between sections, spatially normalised to standard Montreal Neurological Institute (MNI) stereotaxic coordinates using subject-specific 1 mm-iso-voxel T1-weighted images aligned to fMRI data, and spatially smoothed with a Gaussian kernel with 8-mm full width at half maximum. Following linear detrending, data were temporally band-pass filtered at 0.01–0.1 Hz. Possible artefacts from motion and physiological noise were modelled as nuisance regressors (rigid-body translations and rotations determined from the realignment stage and signal changes in the white matter and CSF) and removed with linear regression.
Behavioural performance and spontaneous brain activity
Spontaneous neuronal activity was measured as amplitude of low-frequency fluctuations (ALFF) by computing the integral of the signal amplitude in the selected frequency domain that was scaled for standardisation by dividing by the global mean ALFF value (15). In order to detect the areas whose ALFF change is significantly correlated with working-memory performance change after the therapeutic intervention, a whole-brain linear regression analysis of changes in ALFF was performed with the correct answer rate change of the two-back test as a regressor. All whole-brain statistics were corrected for multiple comparisons with a voxel-wise threshold of p < 0.001 and a minimum cluster size of 11 voxels to achieve a corrected map-wise false-positive probability of p < 0.05 as determined by 5000 iteration Monte Carlo simulation using the AlphaSim program included in AFNI (http://afni.nimh.nih.gov/afni).
While it is not considered to directly affect arterial blood flow, intracranial hypotension leads to dilatation of the venous systems, which can theoretically increase the amplitude of the blood-oxygenation-level dependent (BOLD) signal since the amplitude is dependent on fraction of venous blood within each voxel (16). However, the effect of vasodilation is expected to be cancelled out through scaling unless it affects the BOLD measurement heterogeneously across the brain. In order to assess the possibility, we examined the correlation between ALFF and CSF pressure. The effect of motion artefact was also examined. ALFF of each region was measured as an average within a 5-mm-diameter sphere centred on the peak of each region of interest (ROI).
Functional connectivity analysis
For the functional-connectivity analysis, FSL tools (www.fmrib.ox.ac.uk/fsl) were used to carry out dual regression analysis that allows for voxel-wise comparisons of resting functional connectivity (17). Pre-processed data were temporally concatenated across participants to create a single 4D dataset that was subsequently decomposed using probabilistic independent component analysis (ICA) (MELODIC Ver. 3.13) to identify large-scale patterns of functional connectivity, with the number of components constrained to 25 (18). Using group ICA spatial maps in a linear model fit against the separate fMRI data sets, matrices describing temporal dynamics for each component and individual were obtained (spatial regression) (17). Participant-specific spatial maps were subsequently estimated by applying the time-course matrices in a linear model fit against the associated fMRI data set (temporal regression) (17).
Based on the hypothesis as well as the results of the ALFF analysis showing involvement of the main nodes of the networks, namely, precuneus, bilateral orbitofrontal cortices (OFCs), and medial frontal cortex (MFC), the network analysis was focused on the DMN and FPNs. Correlation analysis was performed to test if cognitive performance was modulated with the regions’ connectivity with the network to which each node belongs; i.e. DMN in precuneus, right and left FPN (RFPN, LFPN) in right and left OFC (ROFC, LOFC), respectively, and RFPN in right MFC. In order to examine the contribution of network-level activity and to examine if the source of the abnormal neural activity affecting the cognitive performance is network level or local, linear partial correlations tests were performed. For all the analyses using ROIs, linear relationship was examined with Pearson’s correlation and statistical threshold was set at p < 0.05 (Bonferroni correction for multiple comparisons).
Results
Patient characteristics and behavioural performance
Synopsis of clinical findings.

0 = postural headache, 1 = neck stiffness, 2 = tinnitus, 3 = hypacusia, 4 = photophobia, 5 = nausea.
1 = evidence of low CSF pressure on MR; 2 = evidence of CSF leakage on conventional myelography or CT myelography.
Site of leakage: C = cervical; T = thoracic; L = lumbar spine; NA = not applicable (not identified in the spine).
Headache inventory score (pre-/post-op).
Presence (+) or absence (−) of subdural effusion/haematoma.
Evacuation = evacuation of the subdural haematoma.
Data are preoperative findings unless otherwise indicated.
M: male; F: female; CSF: cerebrospinal fluid; CT: computed tomography; HIT: Headache Impact Test; MMSE: Mini-Mental State Examination; SDH: subdural haematoma.
Two-back working-memory performance significantly improved after surgery (0.66 ± 0.29 vs. 0.79 ± 0.13; p = 0.047, t = 1.8), while one-back performance was at ceiling pre-operatively (0.99 ± 0.04 vs. 1 ± 0; p = 0.17, t = 1). The false alarm rate was small for two-back test (0.04 ± 0.04 vs. 0.02 ± 0.02; p = 0.14, t = 1.11) and zero for one-back test both pre- and post-operatively across patients. Averaged response time for correct answers did not significantly differ between the two conditions (two-back: 1020 ± 183 vs. 943 ± 126 ms; p = 0.07, t = 1.6; one-back: 826 ± 148 vs. 822 ± 136 ms; p = 0.46, t = –0.09). Percentage of correct answers for the two-back test was henceforward used as a cognitive index.
Working-memory performance and spontaneous brain activity
Whole-brain linear regression analysis revealed a significant positive correlation between changes in working-memory performance and ALFF of the precuneus/posterior cingulate cortex (MNI coordinates: −2, −48, 42) as well as a negative correlation between changes in cognitive performance and ALFF of the bilateral OFCs (Brodmann area, BA11; MNI coordinates, ROFC, 34, 60, −6; LOFC −38, 60, −14) and the right MFC (BA8/24; MNI coordinates, 10, 16, 54) (voxel-wise threshold of p < 0.001, map-wise threshold of p < 0.05), (precuneus, r = 0.89, p < 0.0001; ROFC, r = −0.83, p = 0.0001; MFC, r = −0.83, p = 0.0002; LOFC, r = −0.82, p = 0.0002) (Figure 1). Cognitive performance and ALFF were significantly correlated pre-operatively in all four regions (precuneus, r = 0.76, p = 0.001; ROFC, r = −0.89, p < 0.0001; MFC, r = −0.70, p = 0.004; LOFC, r = −0.94, p < 0.0001). However the correlations were attenuated post-operatively as a result of ALFF changes associated with cognitive task performance improvement in all (precuneus, r = 0.52, p = 0.049; ROFC, r = −0.40, p = 0.14; MFC, r = −0.05, p = 0.87; LOFC, r = −0.45, p = 0.089) (Figure 2).
Changes in working-memory task performance and spontaneous brain activity. Whole-brain linear regression analysis revealed significant positive correlation between working-memory performance change and amplitude of low-frequency fluctuations (ALFF) change in the precuneus/posterior cingulate cortex, while ALFF change of the bilateral orbitofrontal cortices (OFCs) and the right medial frontal cortex (MFC) (BA8/24) was negatively correlated with working-memory performance change (voxel-wise threshold of p < 0.001, map-wise threshold of p < 0.05). Cl = cluster size, R = right. Pre- and post-operative working-memory task performance and spontaneous brain activity. Cognitive performance and amplitude of low-frequency fluctuations (ALFF) were significantly correlated pre-operatively in all four regions (p < 0.05, Bonferroni correction), but the correlations were attenuated post-operatively as a result of ALFF changes associated with cognitive task performance improvement. R and p values for pre- and post-operative states are given in the upper and lower blocks, respectively. Filled and open circles denote pre- and post-operative data, respectively.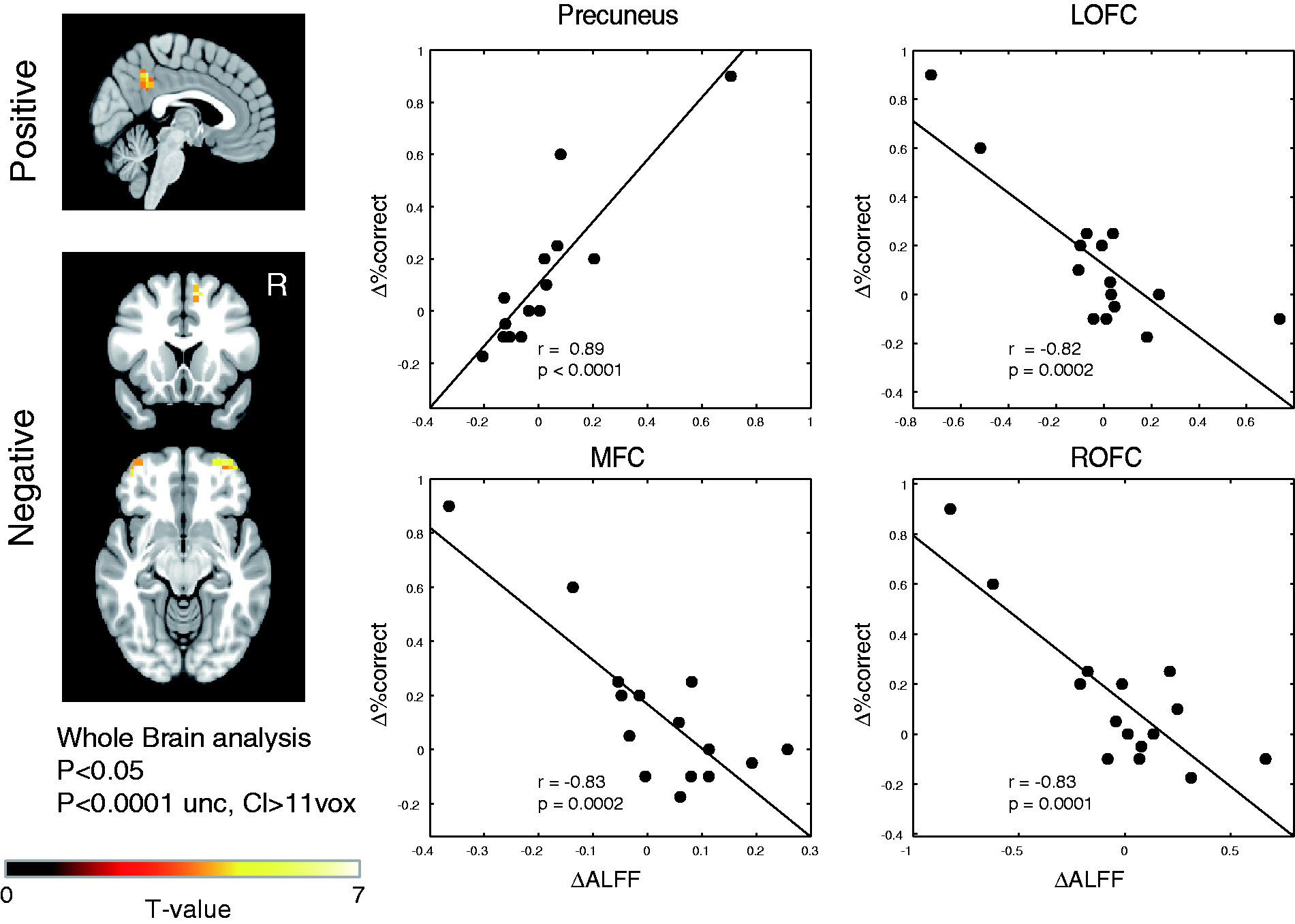

Whole-brain linear regression analysis revealed a significant positive correlation between ALFF and CSF pressure in the basal (interpeduncular and prepontine) cisterns, which might be due to low CSF fluctuations under hypovolaemia. However, no area with negative correlation indicating the presence of vascular confounds was found. Average head motion was neither different between pre- vs. post-operative state (p = 0.28, t = −1.13) nor correlated with task performance (pre-/post-operative: r = −0.0009, p = 0.99; r = 0.21, p = 0.46) or with the ALFF of the detected regions (pre-/post-operative: precuneus, r = 0.34, p = 0.22; r = −0.27, p = 0.32; ROFC, r = −0.17, p = 0.54; r = −0.64, p = 0.48; MFC, r = −0.008, p = 0.98; r = 0.24, p = 0.39; LOFC, r = 0.015, p = 0.96; r = −0.36, p = 0.19). Notably, increased ALFF of task-positive regions in severely impaired patients had a weak but negative correlation with head motion. These data strongly suggest that the correlation between task performance and fMRI measures is not an artefact of motion.
Functional connectivity analysis
The DMN was identified as the network composed of the posterior cingulate cortex, precuneus, medial prefrontal cortex, and temporoparietal junctions (angular gyri) (19) (Figure 3(a)). Bilateral FPNs were identified as the components composed of the anterior prefrontal cortex, dorsolateral prefrontal cortex, dorsomedial superior frontal cortex, and anterior inferior parietal lobule (20) (Figure 3(b), (c)).
Resting-state networks identified with independent component analysis (Z-score plot): (a) default mode network, (b) right frontoparietal network and (c) left frontoparietal network. R = right.
Correlation analysis revealed that changes in functional connectivity of the ROFC with the main nodes of the RFPN was significantly negatively correlated with working-memory performance change (r = −0.64, p = 0.011) (Figure 4(a)). While the RFPN synchronisation was significantly negatively correlated with task performance pre-operatively (r = −0.65, p = 0.009), the correlation was inverted and attenuated post-operatively (r = 0.02, p = 0.94) (Figure 4(b)). For the LFPN and DMN, no significant correlation with task performance change was found (LFPN, r = −0.03, p = 0.93; DMN, r = −0.02, p = 0.96). Partial correlation analysis between changes in working-memory performance and neural activity controlled for the RFPN activity in ROFC revealed that there was a significant correlation between performance change and local activity change (r = −0.71, p = 0.0046), while no significant correlation was found between performance change and RFPN activity change controlled for ALFF (r = −0.16, p = 0.58), indicating a local rather than network level origin of the abnormal neural activity. Significant partial correlation between performance and ALFF was also found pre-operatively (r = −0.89, p = 0.0006), but the correlation was attenuated post-operatively (r = −0.48, p = 0.085).
Changes of network activity vs. changes in cognitive task performance. Right frontoparietal network (RFPN) activity change within the right orbitofrontal cortex was negatively correlated with working-memory performance change (a). RFPN activity was significantly negatively correlated with task performance pre-operatively; however, the correlation was inverted and attenuated post-operatively (b). FC: functional connectivity; WM: working-memory. R and p values for pre- and post-operative states are given in the upper and lower blocks, respectively. Filled and open circles denote pre- and post-operative data, respectively.
Discussion
Significant improvement in two-back working-memory performance was shown in SIH patients who underwent surgical repair of the CSF leakage. The performance change was positively correlated with changes in local spontaneous neuronal activity of the precuneus that is the centre of the DMN (19,21). On the other hand, a negative correlation between cognitive performance and spontaneous neuronal activity was found in the bilateral OFCs and right MFC that are part of the task-positive regions commonly increasing activity during working-memory and decision-making tasks (22). This is in contrast to the previous findings in healthy individuals reporting a positive correlation between working-memory performance and ALFF of the task-positive regions in the superior parietal lobes (23) or no equivalent correlation (24). The abnormally increased ALFF of the OFCs and MFC in severely impaired patients all decreased in association with the cognitive performance improvement, and the abnormal correlations were attenuated post-operatively.
Functional connectivity analysis revealed that the ROFC activity synchronised within the RFPN was also relatively increased in severely impaired patients and decreased with the improvement of working-memory performance, leading to the attenuation of the correlation post-operatively, which might suggest that the abnormally increased activity was spreading over the network rather than being local. Alternatively, it might suggest that the abnormal increase in RFPN is the source of increased neural activity in ROFC. However, strong correlations were still found between cognitive performance and local neural activity even when we controlled the network-level activity, indicating the local rather than network origin of the abnormal neural activity.
Neuroimaging studies have shown increased activity of the frontal regions not only during the execution of a variety of cognitive tasks in aged individuals (25) but also in the resting state for patients with mild cognitive impairment (26,27). The paradoxical increase of the neuronal activity has been considered as reflecting functional compensation (28) or dedifferentiation of the function (29,30). Given the negative correlation with cognitive performance, the increased task-positive regions’ activity in the present study is less likely to be compensatory. Rather, the increased activity of the OFCs and MFC that is not considered originating from intrinsic synchronous network activity is in line with the dedifferentiation view that explains the increase of neuronal activation as a reflection of the decreased signal-to-noise ratio in cortical processing, resulting in reduced regional process-specificity and an increase in nonspecific cortical activation (25,30,31). Considering that the anterior basal part of the frontal lobes are particularly susceptible to mechanical damage because of its anatomical location (32) and hence is most frequently involved in traumatic brain injury, leading to the impairment of executive function (33,34), the increased spontaneous activity of the OFCs could be explained as the result of abnormal neuronal discharges under mechanical compression. Neurons are sensitive to mechanical manipulation, and even sublethal deformation predisposes neurons to excitotoxicity in vitro (35,36). Moreover, focal cortical compression has been shown to disrupt the excitatory-inhibitory balance and to reduce the electrically induced seizure threshold (37).
Furthermore, decreased activity of the precuneus in severely impaired patients was increased in association with the cognitive performance improvement, and the abnormal correlation was again attenuated post-operatively. The DMN was originally identified as the area that exhibit decreased activity during a wide variety of goal-directed behaviours in healthy individuals (19). Recent studies on epilepsy patients further revealed that generalised spike-and-wave activity or focal interictal epileptiform discharges of thalamocortical, temporal, frontal and posterior quadrant origins also suppress DMN activity (38,39). Therefore, the decreased activity of the DMN is not only seen during physiological activation of the task-positive regions but could also be associated with pathological excitation of a variety of regions, which supports the idea that the abnormal increase of the OFCs and MFC under hypovolaemia could lead to cognitive impairment in SIH via suppression of the central node of the DMN.
Few retrospective case reports have thus far described the clinical outcome of cognitive impairment in SIH patients. Hong et al. (3) reported a case of SIH exhibiting a frontotemporal dementia-like condition that was ameliorated after conservative treatment with prednisone. The single photon emission tomography showed normal cerebral perfusion and metabolism. On the other hand, lack of sustained cognitive improvement in response to various treatments was also reported in a series of presumed SIH patients (6). In the latter report, CSF leakage was not identified in most of the cases and the sagging of the brain remained the same after the treatments. In two of the patients evaluated, decreased metabolism of the frontotemporal basal regions was observed. As for the frontotemporal lobe metabolism, it might therefore be possible that the mechanical compression due to hypovolaemia leads to hypometabolism depending on the duration or the severity of the mechanical compression.
Limitations
A foremost limitation of this study stems from the modest size of the patient group that prevents the subgroup analysis. The study result might be influenced by the fact that a small number of patients who had large subdural haematoma were suffering from severe cognitive impairment while the others’ impairment was mild, which is due to the scarcity of severe neurological manifestation of this disease. However, even when we exclude the two most severely impaired patients, preoperative cognitive performance vs. ALFF correlation remained negative in OFCs and MFC, and the negative correlations were attenuated post-operatively. Another limitation is the potential bias of the working-memory performance measure. Although we tried to minimize the order effect by providing two sets of the task with exactly the same degree of difficulty (the series of the numbers were parallel shifted) in a counterbalanced design, it is difficult to completely exclude the influence of learning effect, which is inherent in any longitudinal study.
Conclusions
The present study provides the first evidence supporting the hypothesis that cognitive impairment in SIH is caused by abnormal activity of the OFCs and MFC that could be due to mechanical compression of the frontal basal region of the brain under hypovolaemia. The OFC and MFC activity that is not necessarily originating from synchronous intrinsic network activity of the FPNs, together with the decreased precuneus activity, could lead to cognitive impairment in SIH that is reversible through restoration of the CSF.
Article highlights
Longitudinal evaluation of changes in working memory performance and spontaneous neuronal activity of the whole brain reveals abnormal correlations indicating increased neuronal activity of the frontoparietal network nodes and decreased activity of the default mode network node in patients suffering from spontaneous intracranial hypotension. Pre-operative increased activity of the bilateral orbitofrontal cortices and right medial frontal cortex decreased with the improvement of the cognitive performance, while the decreased activity of the precuneus increased post-operatively. The enhanced activity that is not originating from synchronous intrinsic network activity of the frontoparietal network could be due to mechanical compression of the frontal basal region of the brain under hypovolaemia. The abnormal activity together with the decreased activity of the central node of the default mode network could lead to cognitive impairment in spontaneous intracranial hypotension that is reversible through restoration of the cerebrospinal fluid.
Footnotes
Funding
This work was supported in part by Grant-in-Aid for Young Scientists (B) 25861074 from Japan Society for the Promotion of Science to S.A.
Conflict of interest
None declared.
