Abstract
Introduction
Trigeminal neuralgia is an exemplary neuropathic pain condition characterized by paroxysmal electric-shock-like pain. However, up to 50% of patients also experiences concomitant continuous pain. In this neuroimaging study, we aimed to identify the specific anatomical features of trigeminal nerve root in patients with concomitant continuous pain.
Methods
We enrolled 73 patients with a definitive diagnosis of classical and idiopathic trigeminal neuralgia and 40 healthy participants. The diagnosis of trigeminal neuralgia was independently confirmed by two clinicians. Patients were grouped as patients with purely paroxysmal pain (45 patients) and patients also with concomitant continuous pain (28 patients). All participants underwent a structured clinical examination and a 3T MRI with sequences dedicated to the anatomical study of the trigeminal nerve root, including volumetric study. Images analysis was independently performed by two investigators, blinded to any clinical data.
Results
In most patients with concomitant continuous pain, this type of pain, described as burning, throbbing or aching, manifested at the disease onset. Demographic and clinical variables did not differ between the two groups of patients; the frequency of neurovascular compression and nerve dislocation were similar. Conversely, trigeminal nerve root atrophy was more severe in patients with concomitant continuous pain than in those with purely paroxysmal pain (
Conclusions
Our clinical and neuroimaging study found that in patients with trigeminal neuralgia, concomitant continuous pain was associated with trigeminal nerve root atrophy, therefore suggesting that this type of pain is likely related to axonal loss and abnormal activity in denervated trigeminal second-order neurons.
Introduction
Trigeminal neuralgia (TN) is an exemplary neuropathic pain condition characterized by recurrent unilateral paroxysmal electric-shock-like pain. The key mechanism underlying paroxysmal electric-shock-like pain in TN is the focal demyelination of primary trigeminal afferents at the trigeminal nerve root entry zone. Focal demyelination causes myelinated axons to become hyperexcitable and increases their susceptibility to ectopic excitation, ephaptic transmission, and high-frequency discharges (1,2). Increasing evidence suggests that rare variants in genes encoding voltage-gated ion channels may play a role in the pathogenesis of TN. Previous studies found gain-of-function mutations of Nav 1.6 and Nav1.8 in patients with TN (3,4). These gene mutations may increase hyperexcitability due to the focal demyelination. Recently, a cross-sectional study in patients with familial occurrence of TN identified rare variants in other voltage-gated channels and TRP channels that are part of the neuronal electrogenisome (4).
A significant proportion of patients with TN (24–49% of cases) (5,6) also suffers from concomitant continuous pain that manifests as ongoing background pain between paroxysmal attacks, which is commonly described as aching, throbbing, or burning (7). This condition has been defined in various ways, including as atypical TN or as TN type 2 (8,9). The International Headache Society classification (10) has now defined this condition as TN with concomitant continuous pain. However, evidence-based, case control studies on a large sample of patients are still needed to validate the clinical characteristics of TN with concomitant continuous pain, in order to improve our understanding of the mechanisms underlying pain in patients with TN and improve the way we diagnose and treat TN. As opposed to paroxysmal pain, the mechanisms underlying concomitant continuous pain are unclear, with serious implications for treatment. Several authors have reported that concomitant continuous pain improves less than paroxysmal pain after pharmacological treatment with sodium channel blockers or surgical treatment with microvascular decompression (1,6,11–14).
Although concomitant continuous pain in TN patients represents a relatively frequent and difficult-to-treat symptom, no study has directly investigated the mechanisms underlying this type of pain or the anatomical characteristics of trigeminal nerve root in patients with TN suffering from this condition. Therefore, in this clinical and neuroimaging study, we aimed to clarify the mechanisms underlying concomitant continuous pain by investigating the anatomical characteristics of the trigeminal nerve root in TN patients. To do so, we enrolled consecutive patients with a definite diagnosis of TN, identified patients with and without concomitant continuous pain using a dedicated questionnaire (4), and examined the trigeminal nerve root using 3 Tesla (3T) MRI.
Methods
We prospectively screened consecutive patients visiting the Center for Neuropathic Pain at Sapienza University from December 2012 to December 2016. The inclusion criterion was a definite diagnosis of TN. Diagnostic criteria were in line with the ICHD-3 2018 update, including 13.1.1.1 Classical TN and 13.1.1.3 Idiopathic TN, and the diagnostic grading for trigeminal neuralgia issued by the International Association for the Study of Pain (15). Exclusion criteria were a diagnosis of secondary TN, a diagnosis of orofacial pain other than TN, previous surgery for TN, cognitive disturbances, or communication barriers. The diagnosis of TN was independently confirmed by two clinicians (AT, GC).
Each patient underwent a detailed neurological examination including precise sensory profiling using bedside tools, as indicated by the European guidelines on neuropathic pain assessment (16). Touch was investigated with a piece of cotton wool and pinprick sensation with a wooden cocktail stick in all trigeminal divisions, and both negative (tactile and pricking hypoesthesia) and positive symptoms (allodynia and hyperalgesia) were assessed. In the patients with a definite diagnosis of TN, using a structured interview (4), we identified patients with purely paroxysmal pain and those who also had concomitant continuous pain. Clinical information was systematically collected during the first visit and verified by a second clinician in a subsequent clinical visit. During these two clinical examinations, scheduled at least once a month, pain management neuroimaging and neurophysiological assessment were performed. Then, patients were examined every 4–6 months.
In all patients, we investigated trigeminal reflex testing (17), including early and late blink reflex (R1 and R2) after electrical stimulation of the supraorbital nerve and early and late masseter inhibitory reflex (SP1 and SP2) after electrical stimulation of the infraorbital and mental nerve. Stimulation and recording adhered to the recommendations for clinical practice of the International Federation of Clinical Neurophysiology (18).
We also enrolled 40 healthy participants, matched for age and gender.
The institutional review board approved the research (ref: 874/12). Written informed consent was obtained from each participant.
Neuroimaging protocol
Patients and healthy participants underwent MRI acquired with a 3T magnet (Siemens Verio, Erlangen, Germany) equipped with a 12-channel head coil. Subjects laid supine with pillows under the head, foam wedges at the sides, and a retaining strap in order to minimize motion artefacts. The MRI scans, aimed at detecting neurovascular compression and trigeminal nerve root features, included 3D constructive interference in steady state images (CISS–TR 1000 ms, TE 132 ms, FOV 200 × 200 mm, slice thickness 0.5 mm, slice number 56, matrix 384 × 384) oriented in the axial plane so that the images included the trigeminal nerve root on both sides and guaranteed optimal definition of dark cranial nerves and vessels submerged in a bright cerebrospinal fluid background (19). To best distinguish the trigeminal nerve root from the vascular structures, we also acquired a 3D time-of-flight magnetic resonance angiography scan (TOF–TR 22 ms, TE 3.60 ms, FOV 181 × 200 mm, slice thickness 0.5 mm, matrix 696 × 768) that comprised the structures lying in the posterior fossa.
Image analysis
A neurovascular compression was identified in the presence of a vessel touching the nerve root without a visible cerebro-spinal fluid (CSF) layer between the two of them, and in the presence of morphological changes of the trigeminal nerve root such as indentation, flattening, dislocation or atrophy (assessed by a quantitative volumetric study) (20). Two expert neuroradiologists (FC and MF), blinded to any clinical data, completed a dedicated questionnaire investigating the presence of vascular compression, the site of compression and the presence of dislocation. In more detail, the site of compression was classified as “root entry zone” (REZ) or “non-REZ”, with REZ defined as the proximal 6-mm segment of the nerve adjacent to the pontine belly (20). Controversies were resolved by agreement. No attempt was made to identify the offending vessel, as doing so would have gone beyond the aim of the study.
In order to quantify atrophy, we performed a volumetric study of the trigeminal nerve root using the imaging software MIPAV (http://mipav.cit.nih.gov/). Sagittal and coronal image sets were reconstructed from the acquired axial 3D CISS images in order to obtain a triplanar workspace. In this space, we identified relevant scans as the ones encompassing the trigeminal root. In each of these slices, the trigeminal nerve root was manually delineated, using a mouse-driven cursor, from the point where the nerve emerges from the pons to an arbitrary cut made at the entrance of the nerve into Meckel’s cave. The volume of the nerve was then automatically calculated by 3D reconstruction software, as a summation of all the delineated voxels (21–23). Two clinicians (GDeS and GC), blinded to all clinical data, independently calculated the volume of both trigeminal nerve roots in each participant. We considered the mean trigeminal nerve root volume between the two observers.
For patients, atrophy was measured as a percentage value obtained by calculating the difference between the mean volume value of the asymptomatic nerve and symptomatic nerve, divided by the mean volume value of the asymptomatic nerve (trigeminal nerve root atrophy in patients =
To make this manual segmentation method uniform, the two observers underwent a training period in which they jointly measured trigeminal nerve root volumes in 20 random subjects. The data collected in this instructional phase were not considered in the statistical analysis.
Statistical analysis
Data distribution was assessed with the Shapiro-Wilk normality test. We used the Mann-Whitney or unpaired t-test to compare demographic, clinical, and nerve atrophy data between groups. For the comparison of categorical data, such as vascular compression, dislocation, and site of compression, we used Fisher’s exact test. We used the Bland-Altman method of comparison to assess interobserver agreement in the measurement of trigeminal nerve root volume (two clinicians, GDeS and GC, independently calculated the volume of the trigeminal nerve roots). The possible correlation of atrophy with age and disease duration was assessed using the Spearman r correlation coefficient. A
Results
Clinical data
We screened 101 patients with a definite diagnosis of TN and excluded 28 patients (five patients who had undergone surgery for TN, 12 with TN secondary to multiple sclerosis, three patients with a benign tumour of the posterior fossa, one patient with TN secondary to a superior cerebellar artery aneurysm, one patient with a megadolicobasilar artery, five patients with bilateral abnormal trigeminal reflex testing suggesting an idiopathic trigeminal neuropathy, and one patient with a previous Wallenberg syndrome). We have eventually included in the analysis 73 patients with classical and idiopathic TN, (49 females and 24 males; mean age 62.1 years, 95% CI 58.7–65.5).
In all enrolled patients, trigeminal reflex parameters fell within the normative ranges, without any significant asymmetry between the affected and the healthy side. In more detail, latency of the R1 component of the blink reflex ranged between 9.4 and 12 msec; latency of the SP1 component of the masseter inhibitory reflex, after stimulation of the infraorbital and mental nerve, ranged between 10 and 13 msec. Side asymmetry was less than 1.2 msec.
Patients and healthy participants did not differ in terms of age (
Twenty-eight patients (38%) reported concomitant continuous pain between the paroxysmal attacks; the remaining 45 patients suffered from purely paroxysmal pain.
Disease duration ranged from 2 months to 25 years (mean 5.3 years, 95% CI 3.7–6.9) in patients with purely paroxysmal pain, and from 6 months to 25 years (mean 7.7 years, 95% CI 5.3–10.1) in patients with concomitant continuous pain (
Of the 28 patients with concomitant continuous pain, 23 had this type of pain at the onset of TN; in the remaining five patients, concomitant continuous pain developed after a mean period of eight years (range 2–13 years). This type of pain, described as burning, throbbing or aching, was reported as being unrelenting day and night without pain-free intervals in four patients and lasting from minutes to 5 hours in the remaining patients. In all patients, concomitant continuous pain was distributed to the same divisions of paroxysmal pain. In one patient, pain was associated with an autonomic symptom (conjunctival injection).
Demographic data and clinical characteristics, including affected side and divisions, did not differ between patients with purely paroxysmal pain and those who also had concomitant continuous pain (Table 1).
Demographics and clinical variables in patients with purely paroxysmal pain and with concomitant continuous pain.
Five patients (two with trigeminal root atrophy and concomitant continuous pain) were refractory to first line pharmacological treatment with sodium channel blockers. In the remaining 68 patients, pharmacological treatment reduced pain by at least 30%.
MRI data
The frequency of vascular compression was significantly higher in patients (62 subjects, 85%) than in healthy participants (11 subjects, 28%) (
Anatomical characteristics of trigeminal root in healthy participants and patients with definite diagnosis of TN.

REZ: root entry zone.
Note: Site of compression and dislocation refer only to patients with vascular compression.
Bland-Altman method comparison of the trigeminal nerve root volume measures between the two observers showed a minor/negligible mean volume difference of 0.3 mm3 (95% CI −0.3–0.8).
Vascular compression was found in 89% of patients with purely paroxysmal pain and in 79% of patients with concomitant continuous pain. The frequency of idiopathic TN was therefore slightly lower in the group of patients with purely paroxysmal pain (five patients, corresponding to 11%) than in the group of patients with concomitant continuous pain (six patients, corresponding to 21%) (
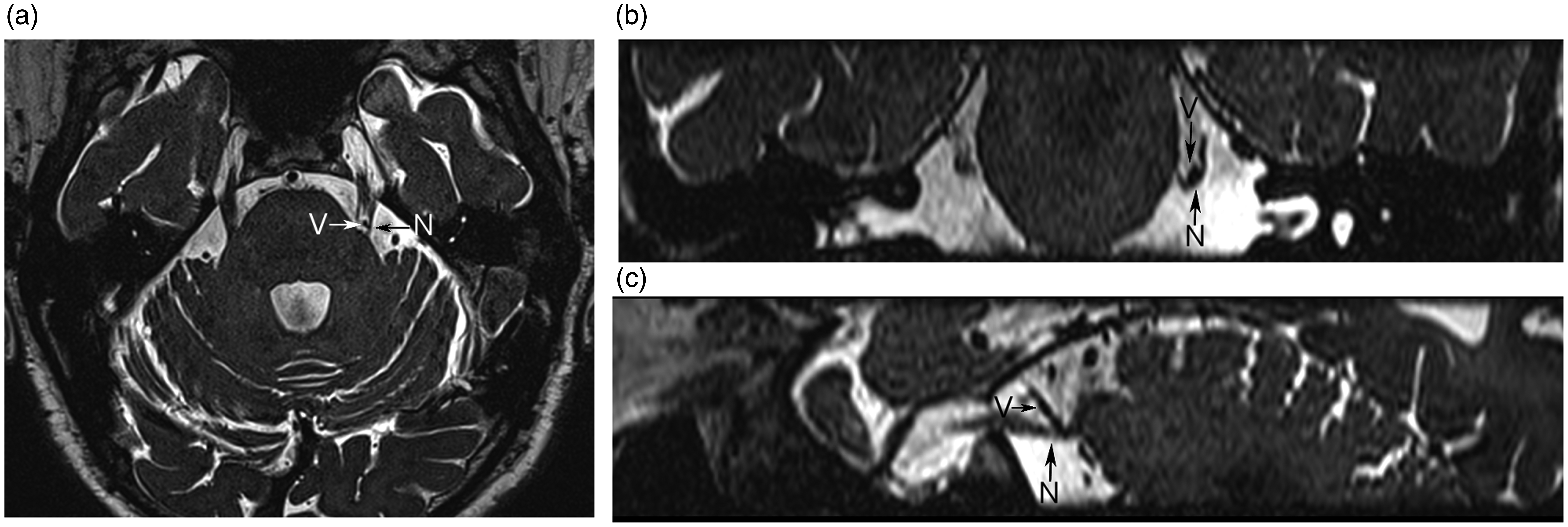
MRI scans of an exemplary TN patient with concomitant continuous pain. Left trigeminal nerve root (N) compressed by an offending vessel (V) at the root entry zone. The nerve shows visible atrophy. (a) acquired axial constructive interference in steady-state (CISS) image; (b) reconstructed image on frontal plane; (c) reconstructed image on sagittal plane.

MRI scans of an exemplary TN patient with purely paroxysmal pain. Left trigeminal nerve root (N) compressed by an offending vessel (V) at the root entry zone. The nerve appears distorted in the absence of atrophy. (a) acquired axial constructive interference in steady-state (CISS) image; (b) reconstructed image on frontal plane; (c) reconstructed image on sagittal plane.
Anatomical characteristics of trigeminal root in patients with purely paroxysmal pain and with concomitant continuous pain.

REZ: root entry zone.
Note: Site of compression and dislocation refer only to patients with vascular compression.
Trigeminal nerve root atrophy did not correlate with age (r = −0.06,
Discussion
This clinical and neuroimaging study in patients with definite TN provides previously unreported findings that concomitant continuous pain is distinctly associated with trigeminal nerve root atrophy. This peculiar anatomical characteristic may imply that axonal loss plays a role in the pathogenesis of concomitant continuous pain.
Clinical data
We found that 38% of our patients suffered from concomitant continuous pain between the paroxysmal attacks. This percentage is in line with previous publications reporting this type of pain in up to 49% of patients with classical TN (1,5,6).
Patients with concomitant continuous pain and those with purely paroxysmal pain did not differ in demographic data or clinical characteristics. In particular, we found that in both groups the right side was more frequently affected that the left side. This finding is inconsistent with a previous study reporting that the right side was more frequently affected than the left side only in patients with purely paroxysmal pain, while the right and left sides were similarly affected in patients with concomitant continuous pain (6). We do not have a simple explanation for these contrasting findings. However, in their study, Maarbierg and colleagues included several patients with concomitant continuous pain affecting the face bilaterally, while no patient with purely paroxysmal pain in that study had bilateral pain. This patients’ characteristic probably affected the difference in the right/left ratio between the two groups of patients. However, several studies have homogeneously reported that classical TN affects the right side more frequently (1,24). A previous study showed an involvement of the right side at nearly twice that of the left side (65.6% vs. 32.5%) (24). Accordingly, a similar right/left ratio in both types of classical TN (with and without concomitant continuous pain) seems reasonable. Admittedly, further studies in larger samples of patients are needed in order to confirm the predominant right-side involvement.
We found that disease duration did not differ between the two patient groups and concomitant continuous pain develops at TN onset in most patients. These findings support previous observations (6,25) and refute the common assumption that concomitant continuous pain develops in longstanding cases, thus arguing against the hypothesis that early surgery may prevent concomitant continuous pain development (26). Given that in a few patients concomitant continuous pain develops 2–8 years after paroxysmal pain, we cannot exclude, however, that different mechanisms may contribute.
MRI data
Although neurovascular conflict is frequently identified in healthy subjects, there is a strong association between TN and trigeminal nerve root morphological changes on the symptomatic side, including dislocation, distortion, flattening, and atrophy (15,20,27), abnormalities that are compatible with clinically-important vascular compression. In the study by Maarbjerg and colleagues, including 135 patients with classical TN, neurovascular contact causing displacement or atrophy of the trigeminal nerve was highly associated with the symptomatic side, thus suggesting that severe neurovascular contact is involved in the aetiology of classical TN (27). In line with these observations, vascular compression and nerve dislocation were more frequent in TN patients than in healthy participants (
The hypothesis that C-fibre loss may be the leading mechanism of concomitant continuous pain finds support from clinical studies demonstrating that objective sensory abnormalities possibly associated with axonal loss occurred more frequently in TN patients with concomitant continuous pain (6,36,37) than in those with purely paroxysmal pain. A recent study, however, using quantitative sensory testing (38) did not show significant differences in the frequency of abnormal sensory parameters (including warm detection threshold) on the symptomatic side between patients with and without concomitant continuous pain. Admittedly, future neurophysiological studies, directly testing unmyelinated C-fibre function in patients with TN suffering from concomitant continuous pain, may further clarify the role of unmyelinated nociceptive C-fibre damage in patients with concomitant continuous pain. Further studies are also needed to analyse the correlation between neuroimaging findings, drug response and surgical outcome. The assessment of drug response should include sodium channel blockers, and in particular antidepressants and gabapentinoids, effective in different ongoing pain conditions.
In our study, we enrolled patients with classical as well as idiopathic TN. In patients with idiopathic TN, dedicated MRI investigation failed to identify any neurovascular compression affecting primary trigeminal afferents. Presumably, genetic factors may concur to idiopathic TN. A recent study found rare variants in genes encoding voltage-gated ion channels and transient receptor potential channels in patients with classical and idiopathic TN (4). Accordingly, the evidence of trigeminal nerve root atrophy even in idiopathic TN further supports the theory that different factors other than neurovascular compression may affect the trigeminal nerve root, causing axonal loss and concurring to pain in patients with TN. Given the plausible multifactorial origin of TN, abnormal sodium channels may play a role in the context of a “multi-hit model” that can explain both adult onset and the focal nature of the disorder (39).
Conclusions
Our clinical and neuroimaging study in patients with a definite diagnosis of TN demonstrated an association between trigeminal nerve root atrophy and concomitant continuous pain. Trigeminal nerve root atrophy probably reflects axonal loss involving unmyelinated nociceptive C-fibres. Concomitant continuous pain may therefore arise from the abnormal activity of denervated nociceptive second-order neurons. These findings may ultimately contribute to improving the way we treat this peculiar type of pain in patients with TN.
Article highlights
Up to 50% of patients with trigeminal neuralgia suffer from concomitant continuous pain. Concomitant continuous pain is associated with trigeminal nerve root atrophy. Axonal loss may be the leading mechanism of concomitant continuous pain.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AT has received consulting fees or payment for lectures from Sigma Tau IFR, Angelini, Gruenenthal, Pfizer, Eliem Therapeutics. GC received consulting fees from Alfasigma, Angelini, Biogen, Gruenenthal. The other authors have no conflict to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
