Abstract
Introduction
Although it is widely accepted that facial pain paroxysms triggered by innocuous stimuli constitute a hallmark sign of trigeminal neuralgia, very few studies to date have systematically investigated the role of the triggers involved. In the recently published diagnostic classification, triggered pain is an essential criterion for the diagnosis of trigeminal neuralgia but no study to date has been designed to address this issue directly. In this study, we set out to determine, in patients with trigeminal neuralgia, how frequently triggers are present, which manoeuvres activate them and where cutaneous and mucosal trigger zones are located.
Methods
Clinical characteristics focusing on trigger factors were collected from 140 patients with trigeminal neuralgia, in a cross-sectional study design.
Results
Provocation of paroxysmal pain by various trigger manoeuvres was reported by 136 of the 140 patients. The most frequent manoeuvres were gentle touching of the face (79%) and talking (54%). Trigger zones were predominantly reported in the perioral and nasal region.
Conclusion
This study confirms that in trigeminal neuralgia, paroxysmal pain is associated with triggers in virtually all patients and supports the use of triggers as an essential diagnostic feature of trigeminal neuralgia.
Introduction
Trigeminal neuralgia (TN) is a unique neuropathic pain condition characterized by unilateral paroxysmal pain, usually described as stabbing or electric shock-like, and restricted to the distribution of one or more divisions of the trigeminal territory (1,2). In the beta version of the 3rd edition of the International Classification of Headache Disorders (ICHD-3 beta), the most used classification of headache and facial pain disorders, the diagnostic criteria of classical TN include provocation of paroxysmal pain from innocuous stimuli, but not as an essential condition; that is, TN can be diagnosed without a trigger if three other pain characteristics are present. By contrast, the novel diagnostic grading system issued by the Special Interest Group on Neuropathic Pain of the International Association for the Study of Pain makes the presence of a trigger an essential criterion, without which the clinical diagnosis of TN cannot be established (2). The view was taken on the basis of a literature review that suggested the presence of trigger zones in a high percentage of patients diagnosed with TN (2). In none of the quoted papers, however, was the frequency or characterization of trigger zones the main purpose. It is therefore imperative that, given the dominance of triggers in the new classification, their frequency and characteristics are defined as accurately as possible. Not only is this critical for the diagnosis of an individual patient, but also to support meaningful research on the pathophysiology and treatment of TN.
Provocation of paroxysmal facial pain by innocuous stimuli is very rare, except for TN. A very high percentage of triggers in patients who report all other pain qualities suggestive of TN, i.e. intense, short-lived pains of abrupt onset and cessation, coming in multiple paroxysms, would strengthen the concept of TN as a unique pain, and an explanation for its pathophysiology would have to include generation of trigger zones. A low percentage would in turn suggest that TN can present with and without triggers and requiring separate pathophysiological explanations. In this study, we aimed to determine the frequency and nature of triggers as they pertain to patients with TN whose diagnosis is compatible with the ICHD-3 beta criteria.
Methods
In this cross-sectional study, we prospectively screened consecutive patients attending the Centre for Neuropathic Pain at Sapienza University from January 2015 to December 2016. Inclusion criteria were a diagnosis of TN according to the ICHD-3 beta criteria (1), including 13.1. Classical trigeminal neuralgia, 13.1.2.4 Painful trigeminal neuropathy attributed to MS plaque and 13.1.2.5 Painful trigeminal neuropathy attributed to space-occupying lesion (in this classification, both latter diagnostic categories refer to patients who report facial pain with the characteristics of those in classical TN, and are also called “secondary TN” (2,3)). Exclusion criteria were cognitive disturbances and diagnosis of another orofacial pain condition. We also excluded four patients with secondary TN from the present series because they were shown to have a benign tumour at the cerebellopontine angle verified by MRI, and were promptly referred for surgery; therefore, they were not available for the present study. Two patients were excluded because, even though they presented with one-sided paroxysmal attacks and had normal MRI scans, the trigeminal reflex testing showed bilateral abnormalities in the mouth area that were typical of trigeminal neuropathy (2). The total number of patients prospectively enrolled was 70.
Each patient underwent a precise sensory profiling using bedside tools as indicated by the European guidelines on neuropathic pain assessment (4). Touch was investigated with a piece of cotton wool and pinprick sensation with a wooden cocktail stick, in all trigeminal divisions. Both negative (tactile and pricking hypoesthesia) and positive symptoms (allodynia and hyperalgesia) were assessed. Each patient also underwent trigeminal reflex testing (5). Neurophysiological testing of the trigeminal reflexes included the early and late blink reflex (R1 and R2) after electrical stimulation of the supraorbital nerve and early and late masseter inhibitory reflex (SP1 and SP2) after electrical stimulation of the infraorbital and mental nerve. Stimulation and recording were carried out with surface electrodes, adhering to the recommendations for clinical practice of the International Federation of Clinical Neurophysiology (6). Patients also underwent 3T MRI, with specifications optimized for identification of the cause of TN (7,8).
Three staff members were involved in the clinical examination and two in the neurophysiological testing. The diagnosis of TN was confirmed by two clinicians. Clinical characteristics were systematically collected using a dedicated questionnaire, focusing on triggers. The first section addressed demographic data and clinical characteristics, including age at onset, duration of disease, side and anatomical localization of pain, number and duration of paroxysms, pain intensity and the presence of concomitant continuous pain. Pain remission periods and possible nightly awakenings were also collected through dedicated items. The second section was meant to collect details about the trigger manoeuvres and zones, grouped into intra and extraoral. Patients provided a thorough description of all trigger manoeuvres, encompassing also possible unusual triggers, and drew both the trigger zones and the evoked paroxysmal pain distribution on a facial map. The overlap profiling of the trigger zones was carried out with dedicated software that provided representation and sum of the trigger areas on a standard 3D model of the face and mouth. The frequency distribution of each trigger zone was computed at pixel level by counting the number of times each pixel of the model fell within each trigger area.
Besides the prospective patient enrolment, we also included retrospective patients. A staff nurse selected from our database the names and medical records of the most recent 70 outpatients with a diagnosis of TN (according to ICHD-3 beta), who had attended our centre up to the point of commencement of the above prospective study (i.e. December 2014). A further staff member analysed the records and the diagnostic investigations, to confirm the diagnosis and classify the patients in classical or secondary TN. The principal investigator subsequently examined the individual records for information on pain and triggers.
All patients included in the prospective or retrospective series suffered from paroxysmal attacks of pain affecting one or more divisions of the trigeminal nerve, regardless of the presence of concomitant continuous pain (2,3).
For all patients included, we used as main outcome variables the side, distribution, and time course of pain, as well as manoeuvres triggering the paroxysms and distribution of trigger zones. For the prospective group, we also collected information about the mean severity of paroxysms in the last month, as assessed with a numerical rating scale (NRS) ranging from 0 (no pain) to 10 (worst possible pain), and represented the trigger zones on the 3D face model.
Statistical analysis
Descriptive statistics only were used for evaluation of the frequency of triggers. For comparisons between the prospective and retrospective groups we used the Mann-Whitney test, given that the main demographic and clinical data did not have a normal distribution. For comparisons of categorical data, we used Fisher’s exact test and Chi-squared tests.
Results
Demographics, age at onset, duration of disease, side and anatomical localization of pain.
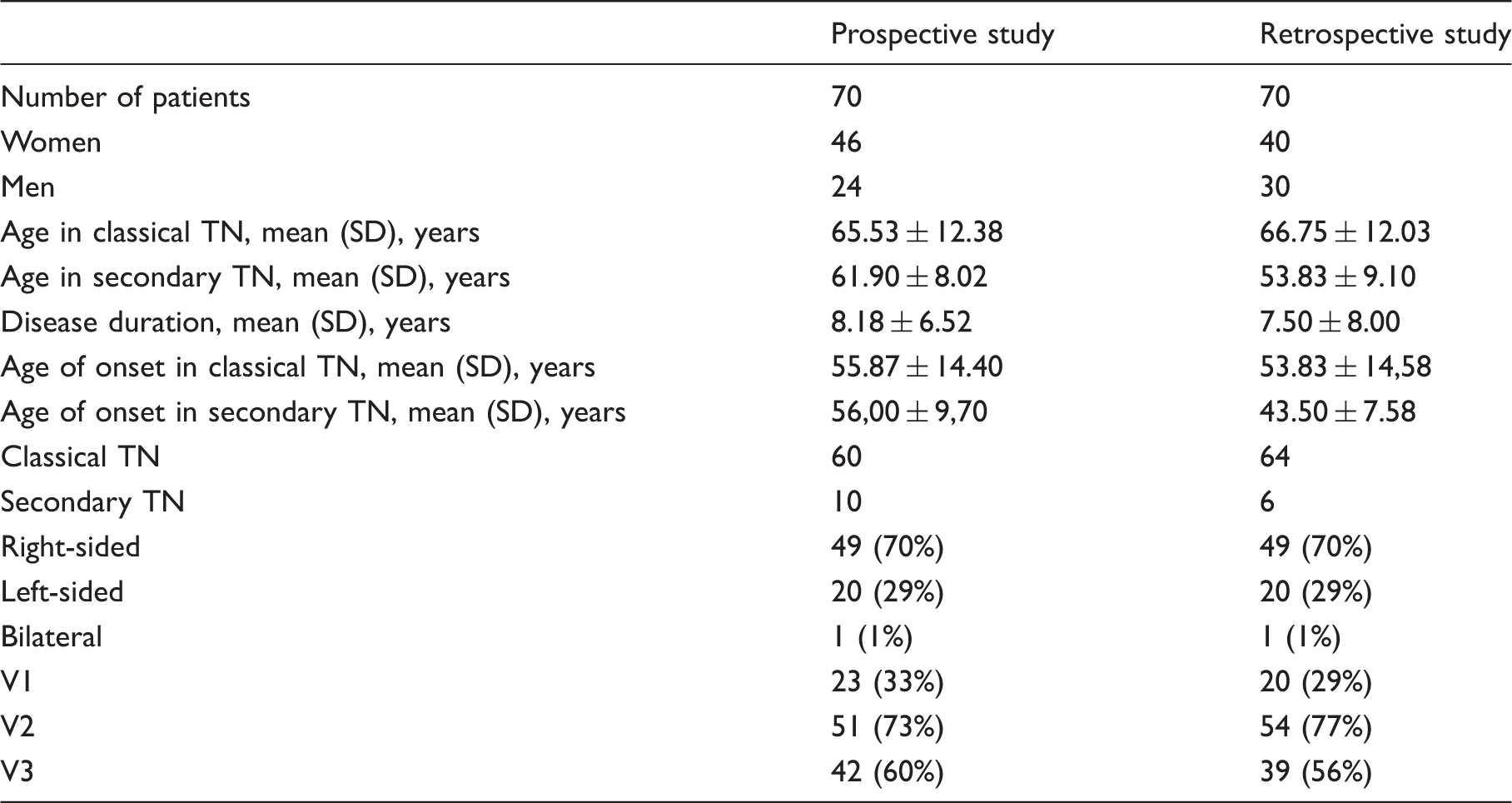
In all 140 patients combined, pain was more often located on the right (70%) than left side (29%), and the second (V2) and the third (V3) trigeminal divisions were more frequently affected than the first trigeminal division (V1) (Table 1). Patients described paroxysmal pain as a very abrupt, short-lasting pain, stabbing or similar to an electric shock in quality. Although most patients reported only daytime pain, 27 patients in the prospective group (39%) reported paroxysmal pain also during the night. The intensity of paroxysmal pain was 8.5 ± 1.6 on NRS (0–10). The duration of paroxysms was less than one second in four patients, from one to two minutes in nine patients and had a mean duration of seven seconds in the remaining patients. The mean number of paroxysms was 11 a day. Fifty three percent of patients experienced pain remission periods of widely variable duration (median six months, range 1–48) during which external stimuli failed to provoke any attacks.
Thirty-three patients (24%; 28 with classical and five with secondary TN) experienced concomitant continuous pain in the same division affected by paroxysms. Continuous pain was described as dull, burning or tingling. Only in five patients was this kind of pain also evoked by trigger manoeuvres, including talking, eating, drinking, swallowing and gently touching the face. The mean pain intensity of continuous pain was 7.4 ± 2.5 in the prospective group. Concomitant pain was reported as being unrelenting day and night without pain-free intervals in 12 patients and lasting from five minutes to two hours in the remaining patients.
Whereas there was no difference in age at onset of TN between prospectively and retrospectively recruited patients, we found that patients with secondary TN had a lower age at onset compared to those with classical TN (median 51, 95% CI 46–57; median 61, 95% CI 57–62; Histogram of age of onset. X-axis: Age of onset. Y-axis: Number of patients.
Trigger analysis
Frequency of trigger manoeuvres in trigeminal neuralgia.

Frequency of trigger zones in trigeminal neuralgia.
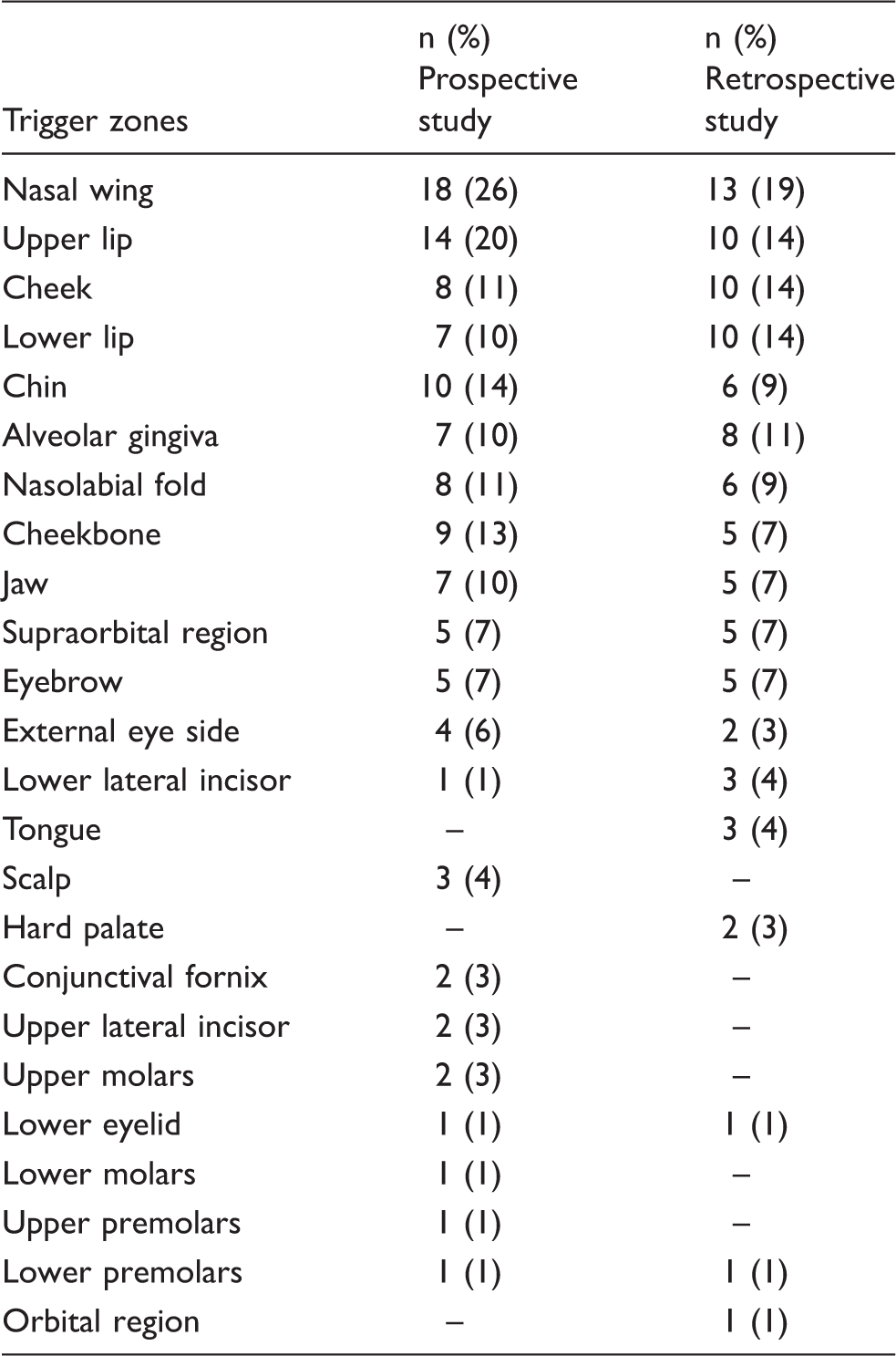

Trigger zone distribution. Data from 70 prospectively-enrolled patients with classical or secondary TN. Upper panel: Extra-oral territories. Lower panel: Intra-oral territories. Left column: Trigger-zone contours. Right column: Trigger-zone overlap profiling. The number of superimpositions ranges from 2 (cyan) to 15 (dark orange). The number of trigger zones in the intra-oral territory is smaller in comparison with the number of patients reporting talking or chewing as the main trigger manoeuvres, because of the patients’ difficulty in identifying a circumscribed trigger zone region within the mouth.
Of the four patients apparently without trigger manoeuvres (three in the prospective and one in the retrospective group), one suffered from multiple sclerosis-related TN. Of the three patients with classical TN, although being unaware of any specific trigger manoeuvre, one did report that to avoid the pain paroxysms she had to keep her face perfectly still, thus suggesting that a facial movement was the trigger.
Discussion
In this clinical study with a large sample of patients, we showed that in virtually all TN patients (97%) paroxysmal pain is associated with a trigger.
The frequency of triggers we found is relatively higher (96% in the prospective study group) than that reported in a previous study (91%) (9). This difference probably reflects the dedicated questionnaire we used in our study for collecting information on triggers. We also hypothesize that in the few patients with no apparent trigger, the pain attacks are evoked by muscle movements they are unaware of, such as eye-blinking or facial mimicry. The observation that, besides spontaneous paroxysmal pain, patients always invariably report triggered pain further supports the use of triggers as a criterion for clinically established TN, and with that for probable neuropathic pain (2,10).
As shown on our 3D face model, trigger zones were most frequently located in the nasal and perioral region. Although four of our patients felt the evoked paroxysmal pain in a trigeminal division different from that of the trigger zone, most often evoked paroxysms and trigger zones are located in the same trigeminal division. This finding, in line with previous studies (11), is consistent with the mechanism of cross-excitation via ephaptic transmission from adjacent unaffected fibres within the trigeminal root (12,13,14).
The trigger zones were variable in size, and while some were no more than a pinpoint, most were much larger. Similar variability was mentioned in a previous study (15). Our method of zone drawing has the advantage that it allows the patient to indicate the size directly on the facial map without relying on the estimate by an examiner. We hypothesize that the evoked paroxysmal pain might in some patients require a sequential activation of mechanoreceptors, resulting in an enlarged trigger area. This hypothesis might also explain the “towel’s sign”, that is, some patients soon learn to dry their face by slowly pressing, rather than brushing, the affected side to avoid the painful paroxysms.
Unexpectedly, we found that a few patients (4%) reported that consuming hot or cold food/water could provoke paroxysms. This finding goes against the common notion that only innocuous mechanical stimuli can evoke the paroxysmal pain (15,16). However, while the specific qualification of a thermal component was volunteered by the patients, the muscular activity in the lips, tongue and pharynx during eating or drinking is certainly sufficient to act as a trigger, contrasting the argument that heat or cold alone could do so.
Unlike the common belief that TN does not awake patients from sleep, we found that many patients (38% of our sample in the prospective group) have nightly painful awakenings. This finding is, however, in line with the only other studies that used a dedicated questionnaire for patients and their partners about painful awakenings in TN (9,17). Given that all our patients with nightly painful awakenings had extra-oral triggers, we suggest that an innocuous contact of the patient’s skin with the sheet or the pillow acted as a triggering factor. We probably only found patients with nightly awakenings in the prospective group because we included a specific question about this in the questionnaire. It is worth pointing out that nightly attacks are less common in TN than in cluster headache, where they are a prominent feature.
Besides the paroxysmal pain, several patients (24% of our sample) reported concomitant continuous pain. All these patients, by definition, felt this type of pain in the same territory as the paroxysmal pain. These findings, consistent with previous studies (18,19,20), support the idea that continuous pain is a relatively common symptom in TN. Central mechanisms (21,22) and progressive root damage due to compression (23) have been proposed as possible mechanisms underlying this type of pain.
We found that age at onset of pain in patients with secondary TN was significantly lower than that of patients with classical TN, confirming the current long-held view. However, there was substantial age overlap between the two populations, preventing this aspect being used as an indicator of secondary TN, in line with previous guidelines on TN (5).
The demographic and clinical variables in our cohort of patients are similar to those reported in previous studies (9,24). TN is more frequent in women and more frequently affects the right side of the face, in the V2 and V3 divisions, probably because of the somatotopic distribution of sensory fibres in the trigeminal root (25).
The main limitation of the study is reliance on both retrospective and prospective data. However, the demographics, clinical features, frequency of triggers and the nature of trigger manoeuvres were very similar between the two groups. The information extracted from the medical records was facilitated by a longstanding interest of the study centre in TN and the established clinical practice of detailed collection of all clinical data.
We anticipate future studies field-testing the sensitivity and specificity of triggers in correctly diagnosing TN versus other orofacial pain conditions. But, as there is no gold-standard diagnostic test in TN, the problem of relying on an independent clinical diagnosis alone remains.
Conclusion
In nearly all patients with the diagnosis of TN based on ICHD-3 beta, a trigger capable of provoking a paroxysm can be identified. Trigger zones are seen most commonly in the nasal and perioral area, and are variable in size. In all the trigger manoeuvres listed by the patients a mechanical component (touch or muscle movement) is present. These findings will be of assistance in future studies on the pathophysiological mechanisms of TN.
Ethics
The present study was approved by the Ethics Committee of Policlinico Umberto I, Sapienza University of Rome (ref: 874/12).
Article highlights
Facial pain paroxysms triggered by innocuous stimuli constitute a hallmark sign of trigeminal neuralgia (TN). In virtually all TN patients, paroxysmal pain is associated with a trigger. The most frequent manoeuvres are gentle touching of the face and talking. Trigger zones are predominantly reported in the perioral and nasal region and are variable in size.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
