Abstract
This prospective, open-label study aimed to evaluate the efficacy of pregabalin treatment in patients suffering from trigeminal neuralgia with and without concomitant facial pain. Fifty-three patients with trigeminal neuralgia (14 with concomitant chronic facial pain) received pregabalin (PGB) 150-600 mg daily and were prospectively followed for 1 year. The primary outcome was number of patients pain free or with reduction of pain intensity by > 50% and of attack frequency by > 50% after 8 weeks. Secondary outcome was sustained pain relief after 1 year. Thirty-nine patients (74%) improved after 8 weeks with a mean dose of 269.8 mg/day (range 150-600 mg/day) PGB: 13 (25%) experienced complete pain relief and 26 (49%) reported pain reduction > 50%, whereas 14 (26%) did not improve. Patients without concomitant facial pain showed better response rates (32 of 39, 82%) compared with patients with concomitant chronic facial pain (7 of 14, 50%, P = 0.020). Concomitant chronic facial pain appears to be a clinical predictor of poor treatment outcome. PGB appears to be effective in the treatment of trigeminal neuralgia.
Introduction
Trigeminal neuralgia (TN) usually leads to paroxysms of short-lasting but very severe pain. Between attacks patients are usually asymptomatic, but a constant dull background pain may persist in some cases (1). The incidence of TN is 4.3 per 100 000 persons per year, with a slightly higher incidence for women compared with men (2). The preferred treatment for TN consists of anticonvulsant drugs, muscle relaxants and neuroleptic agents (3). Large-scale placebo-controlled clinical trials have been few. The most intensively investigated drug is carbamazepine (4), but gabapentin (GBP) (5) and lamotrigine (6) have also shown adequate efficacy in small-sample, prospective, randomized, placebo-controlled trials. One of the disadvantages of GBP is its saturable absorption rate within the usual dosing range. Pregabalin (PGB) maintains the biological activity of GBP, but provides improved pharmacokinetic properties. PGB exerts its pharmacodynamic effect by modulating voltage-gated calcium channels and thus reduces release of excitatory neurotransmitters. It is completely absorbed, not bound to plasma proteins, not metabolized, and eliminated unchanged through the kidneys (7). Clinical trials have shown that PGB is effective in relieving neuropathic pain associated with postherpetic neuralgia and diabetic peripheral neuropathy (8–14). The aim of this study was to assess prospectively the efficacy of PGB in patients suffering from TN with and without concomitant chronic facial pain.
Methods
Patients were prospectively recruited from our headache clinic, a tertiary headache centre, from August 2004 to May 2006. All participants gave their written informed consent according to the Declaration of Helsinki. The study protocol was approved by the Ethics Committee of the University of Duisburg-Essen.
We investigated 53 patients with TN according to the current version of the International Classification of Headache Disorders (ICHD-II) (1). Mean age was 62.7 ± 10.9 years (range 43–82 years). Thirty of the patients were women (57%). Forty-seven patients (89%) had idiopathic TN and six (11%) had symptomatic TN (five due to demyelinating lesions associated with multiple sclerosis, one due to neurinoma at the root entry zone). Thirty-nine patients had TN without concomitant chronic facial pain, whereas 14 patients (26%) reported chronic dull background facial pain with superimposed, typical neuralgic, paroxysmal TN attacks. Of these 14, six patients had additional hypoaesthesia and hypoalgesia in the affected trigeminal branch on clinical examination. All symptomatic TN patients had episodic attacks without persistent background pain.
Exclusion criteria were additional severe medical or psychiatric illnesses. Women could not be lactating and were required to have a negative pregnancy test result and to use appropriate contraception if of childbearing potential. No patient was receiving tricyclic antidepressants, opioids or serotonin/norepinephrine reuptake inhibitors. Phenothiazines, antiarrhythmic agents and pteridine as well as the combination of antihistamines and macrolide antibiotics were prohibited during the study owing to their proarrhythmic activity. Aspirin (up to 300 mg/day) as secondary prophylaxis for myocardial infarction or cerebral ischaemia as well as paracetamol (up to 3 g/day) were allowed.
All patients were examined by a board-certified neurologist and received ECG as well as diagnostic magnetic resonance imaging. Extensive laboratory blood work was obtained from all patients, including haematology, chemistries, endocrinology and urine.
Previous antineuropathic medications to treat TN were discontinued as soon as PGB treatment started. The dose of PGB was titrated from 75 to 600 mg daily with a maximum dose increase of 75 mg/week for the first 2 weeks and 150 mg/week after this initial adaptation period. Additional antineuropathic pain medication was permitted after 8 weeks and considered in patients without complete pain relief under 600 mg/day PGB and the wish to intensify conservative therapy. Carbamazepine 600–1200 mg/day or lamotrigine 50–200 mg/day were administered as add-on therapy (Fig. 1).
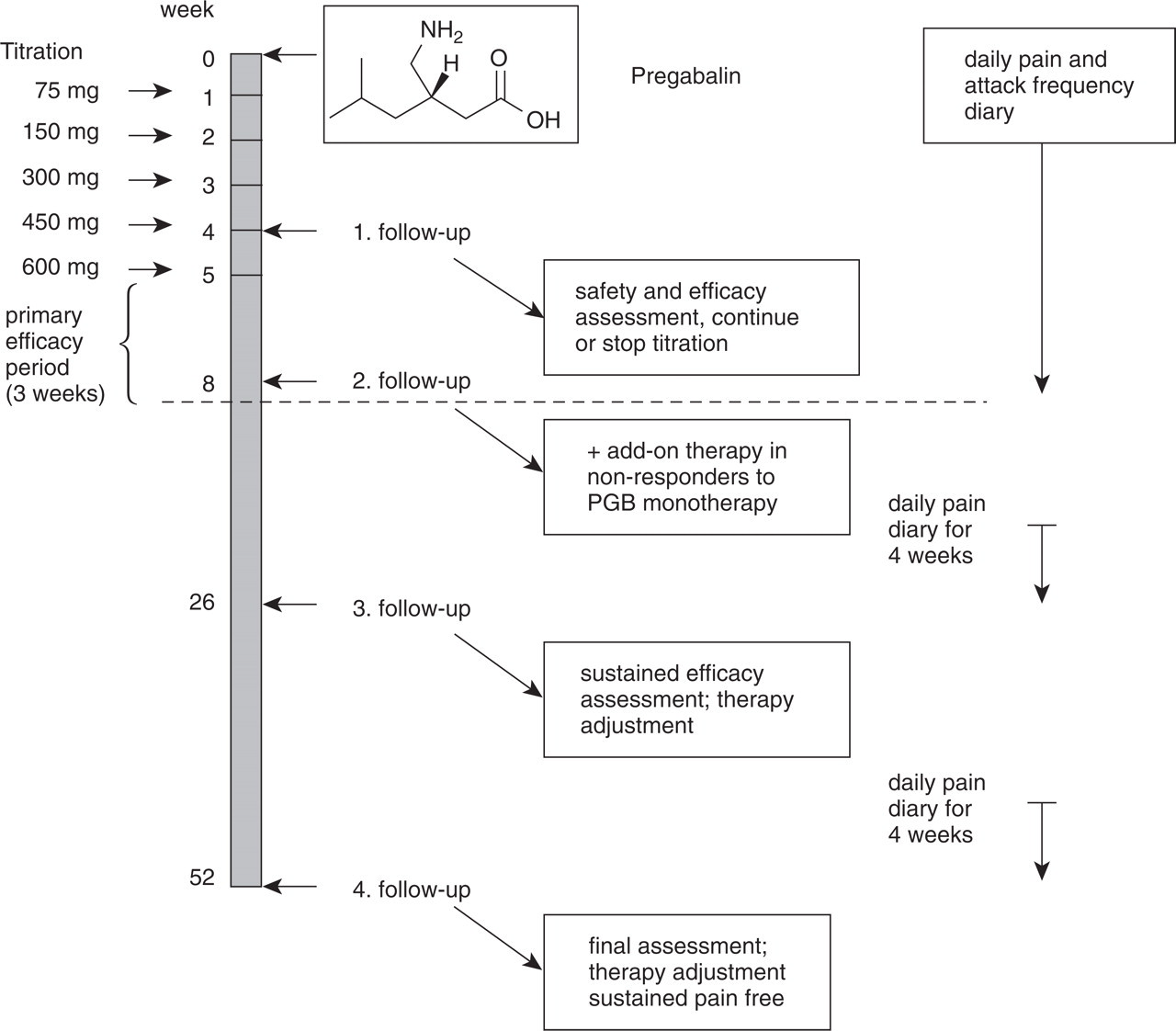
Study time course and pregabalin titration regimen.
Follow-up visits were conducted after 4 weeks (visit 1), after 8 weeks (visit 2), after 6 months (visit 3) and after 1 year (visit 4) (Fig. 1). Patients were asked to complete a standardized pain diary including pain severity on a verbal rating scale (VRS; 0 = no pain to 10 = worst possible pain) and attack frequency assessment on a daily basis in the first 8 weeks. Four weeks prior to visits 3 and 4 patients were asked by telephone to begin a daily pain diary again until the scheduled visit. Reported pain severity and attack frequency reflected the mean pain diary entries for each patient within the 4-week period prior to the next visit. Side-effects were assessed with each visit.
The primary outcome parameter was number of patients free of pain or with pain reduction > 50% and attack frequency reduction > 50% after 8 weeks. These patients were considered treatment responders. The secondary outcome parameter was sustained pain relief after 1 year.
Analysis of variance for repeated measures was calculated to determine the statistical significance of pain reduction and attack frequency reduction at 4 and 8 weeks of PGB treatment. The between-subject factor was TIME (baseline vs. 4 weeks vs. 8 weeks). Post-hoc pairwise comparisons were performed using a T-test with Bonferroni correction. A χ2 test was also used to evaluate whether TN patients with and without concomitant facial pain showed differences in treatment response. All statistics were calculated with SPSS 14.0 (SPSS Inc., Chicago, IL, USA). Level of significance was set at P < 0.05.
Results
Patient demographics and baseline characteristics are summarized in Table 1. After 8 weeks' PGB treatment, 39 (74%) patients responded to treatment with a mean dose of 269.8 mg/day (range 150–600 mg/day): 13 (25%) experienced complete pain relief and 26 (49%) reported pain reduction > 50%. Fourteen (26%) patients did not respond to PGB (Fig. 2).
Demographics and patient characteristics

TNcp, Trigeminal neuralgia with concomitant chronic facial pain; VRS, verbal rating scale; V1, ophthalmic nerve; V2, maxillary nerve; V3, mandibular nerve.

Pain intensity reduction and attack frequency reduction over time with Pregabalin monotherapy including standard error of the mean (SEM). Statistical significance was set at P < 0.05 (∗∗). Non-responders and non-pain-free patients received add-on medication after 8 weeks.
The majority, 11 of the 13 patients experiencing complete pain relief at 8 weeks of treatment, stayed pain free after 1 year. The remaining two reported recurrence of mild TN attacks after 7 and 9 months, respectively, but refused additional medication. Twenty-one of 26 patients with pain reduction > 50% received additional medication and were pain free at the 6 months visit. Eighteen of them received carbamazepine 852.3 ± 275.1 mg/day (range 600–1200 mg/day) and three patients received lamotrigine 85.3 ± 60.3 mg/day (range 50–200 mg/day) as add-on medication until reaching a pain-free state. The majority of patients not responding at all to PGB treatment (n = 14) discontinued PGB within the first 8 weeks (n = 10), whereas the others (n = 4) continued PGB as add-on therapy with a lower dose (300 mg/day). The remaining non-responders received either carbamazepine 920.3 ± 253.4 mg/day (seven of 10 patients) or lamotrigine 127.4 ± 61.2 mg/day (three of 10 patients). Three patients out of this group receiving carbamazepine remained refractory to all treatment attempts and were lost to follow-up at the 6 months visit.
Patients with concomitant chronic facial pain had significantly lower response rates with add-on medication (carbamazepine and lamotrigine) at the end of the 1-year follow-up period (nine of 14; 64%) compared with patients without concomitant facial pain (35 of 39; 90%) (χ2 = 4.7, P = 0.030). Pain reduction and attack frequency reduction showed a parallel response to PGB treatment (Fig. 3). No difference was found between the response of patients receiving carbamazepine and that of lamotrigine treatment as add-on medication, suggesting that neither of the two is superior to the other as add-on medication. Two patients in the non-responder group and one patient in the > 50% pain reduction group received microvascular decompression surgery with sustained pain relief without additional medication at final 1-year evaluation.
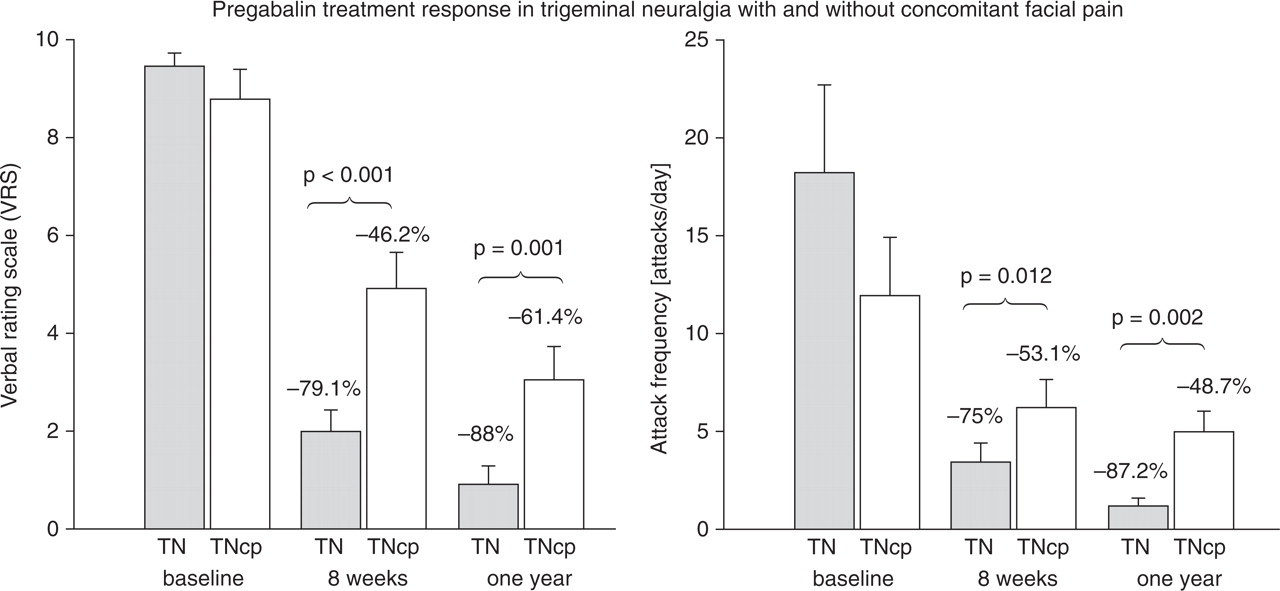
Comparison of pregabalin treatment response in patients suffering from trigeminal neuralgia with (TNcp) and without concomitant facial pain (TN) in percent reduction (%) after 8 weeks and after 1 year divided for pain reduction (left) and attack frequency reduction (right). Patients without concomitant facial pain show a better response to treatment compared with those with concomitant chronic facial pain for pain and attack frequency reduction after 8 weeks as well as after 1 year. Add-on medication was permitted after 8 weeks in non-responders. Level of significance of reported
Discussion
This prospective, open-label clinical study suggests that PGB might be effective in the treatment of patients suffering from TN. Results are in line with treatment results reported with other anticonvulsant drugs such as gabapentin (5), lamotrigine (6) and carbamazepine (15, 16). PGB has some advantages over other treatment options: fewer side-effects, more rapid titration potential, shorter onset of action and can be administered twice daily (bid). An interesting finding is that concomitant chronic facial pain could be a predictor of poor treatment response. These patients seem to show a reduced treatment response compared with those without concomitant pain. This phenomenon does not seem to be related to PGB treatment alone, but also concerns other anticonvulsants such as carbamazepine or lamotrigine. It has not been adequately considered in the evaluation of past treatment efficacy studies on TN. Symptomatic and episodic TN patients showed similar treatment responses to PGB.
Current models regarding the pathophysiology of TN explain the pain as originating either from defects in the trigeminal nerve near the root entry zone in the brainstem, the trigeminal ganglion or from changes in central connectivity. Pain arises through focal injury to trigeminal neurons, leading to ectopic spontaneous firing and increased mechanosensitivity, with possible excitability spreading to neighbouring neurons, with possible additional ephaptic impulse transmission from non-nociceptive to nociceptive afferents, and/or functional derangement of wide-dynamic-range neurons in the spinal trigeminal nucleus (mainly subnucleus caudalis, partly subnucleus interpolaris) (17–22). Patients with concomitant chronic facial pain show evidence of central sensitization indicating overactivation of supraspinal (thalamic or cortical) sensory transmission (23). This may represent a possible adaptive mechanism for the development of chronic pain and might explain the poor treatment response rate of these patients. Patterns of chronicity may involve a complex and wide network of cortical and subcortical structures with different modulatory nociceptive functions. Whether or how these complex functions are influenced by anticonvulsant drugs such as PGB remains subject to further research. It seems that the reversal of central sensitization is difficult to achieve with PGB treatment alone.
Membrane-stabilizing drugs such as anticonvulsants are thought to control paroxysmal pain by suppressing ectopic transmission by blocking Na+ channels. This is true for carbamazepine and lamotrigine, whereas gabapentin and pregabalin interact with the α2δ subunit of voltage dependent Ca2+ channels and increase the cerebral concentration and rate of synthesis of γ-aminobutyric acid (GABA) (7, 24–26). The neurotransmitter GABA is thought to suppress central pain. Although PGB has proven effective in relieving neuropathic pain, its exact method of action remains uncertain. A recent review has summarized several randomized, placebo-controlled clinical trials that proved efficacy of PGB in the treatment of diabetic neuropathy, with a total of 1068 patients receiving PGB 300–600 mg/day showing significantly greater improvement in mean pain scores compared with placebo. Patients with postherpetic neuralgia with PGB doses of 450–600 mg/day had significantly greater improvement in pain relief and pain-related sleep interference than placebo recipients (27). No data on PGB for the treatment of TN have yet been provided, but a few reports on GBP exist (28–31). Considering the similar mode of action of GBP and PGB, a comparable effect on the reduction of neuropathic pain can be expected. In direct comparison PGB seems to have some substantial advantages compared with GBP. In the above-mentioned retrospective study, nearly half of 92 patients who were receiving GBP reported relief or considerable reduction in paroxysmal pain with a mean dose of 930 mg/day ranging from 100 to 2400 mg/day divided into three applications per day (28). We observed initial pain relief with PGB of 25% and reduction of pain > 50% of nearly 50% of patients with a mean dose of 269.8 mg/day (range 150–600 mg/day), leading to a response rate to treatment with PGB of almost two-thirds of patients with doses comparable to other studies on neuropathic pain. PGB can be administered bid and thus provides more convenience for the patient, possibly leading to higher compliance.
As the incidence of TN increases with age (32), age-related physiological changes that alter pharmacokinetics such as reduced hepatic and renal function, blood flow decline, less predictable drug–protein binding and interactions with multiple other medications required due to concomitant illness come more and more into focus. PGB and GBP are neither metabolized nor bound to serum protein, and interactions with other substances are exceedingly rare. Approximately 6–10% of patients cannot tolerate carbamazepine (33, 34). Multiple pharmacological interactions and a narrow therapeutic window of tolerability further limit the use of carbamazepine, especially in the older patient. Even though surgery is available in TN patients non-responsive to medication, the limitations of current pharmacological therapies for neuropathic pain may require the use of complicated multidrug treatment. PGB may also prove to be useful as add-on therapy to a currently used drug or drugs that are unable to provide complete pain relief (14). Treatment attempts with PGB should be limited to 8 weeks when no sufficient pain relief is observed, as most patients evaluated showed no significant change of response to PGB after that period.
The strength of this study is the relatively high number of patients with TN that were recruited and the prospective design with an observational period of 1 year. Little knowledge has been gathered on the treatment of TN with newer anticonvulsants such as PGB, and subgroup analyses such as the response of patients with concomitant chronic facial pain are lacking. Some limitations have to be addressed. The most pressing question is whether the lack of a placebo-controlled comparison group may have overvalued the antineuropathic effect of PGB. Ethical concern about an untreated control group considering the severity of associated pain in TN has to be carefully evaluated (6), as confounding effects due to placebo response have been reported to be nearly negligible in patients suffering from TN, compared with other, more sustained forms of neuropathic pain (33, 35–37). A non-inferiority trial is difficult to realize in a rare disorder such as TN. Larger scale, placebo-controlled studies would be desirable, but are difficult to realize due to the mentioned obstacles. One additional, probable limitation was the selection bias typical of a tertiary referral centre with the majority of patients being difficult to treat and refractory to previous treatment attempts. A patient population more medically and surgically naive might have provided higher response rates to PGB.
The results of this 1-year follow-up, prospective, open-label clinical observation are encouraging. PGB is easy to use in the clinical setting due to its bid administration without the need for complex titration regimens, but providing fast and sustained pain reduction. Concomitant chronic facial pain in TN appears to be a clinical predictor of poor treatment outcome.
